Abstract
Objective
To develop a predictive model of intravenous calcium supplementation needs in patients with secondary hyperparathyroidism (SHPT) after total parathyroidectomy (t-PTX).
Methods
A retrospective analysis was conducted on data from 550 SHPT patients treated at the Department of Hepatobiliary Pancreatic Spleen Thyroid Surgery, General Hospital of Northern Theater Command, from January 2015 to December 2022. Various biochemical indices of the patients were evaluated, including sex, serum ferritin (SF), alkaline phosphatase (AKP), and free thyroxine (FT4). Statistical tests, such as t-tests, were performed to identify significant risk factors associated with the need for postoperative intravenous calcium supplementation.
Results
Sex (t = −1.988, p = 0.049), SF (t = 2.155, p = 0.033), AKP (t = 4.501, p = 0.000), and FT4 (t = −3.128, p = 0.002) were identified as risk factors associated with the need for postoperative intravenous calcium supplementation. The developed predictive model (intravenous calcium supplementation = 1235.561–158.547*gender + 0.141*SF + 0.442*AKP−494.055*FT4; male: 0, female: 1) was effective and passed the F test (F = 22.034, p = 0.000).
Conclusions
The developed predictive model provides a reliable tool for estimating the amount of intravenous calcium supplementation needed by SHPT patients following t-PTX. By incorporating sex, SF, AKP, and FT4, clinicians can make informed decisions and tailor individualized calcium supplementation schemes. This evidence-based approach may contribute to improved patient outcomes and optimize postoperative care for SHPT patients undergoing t-PTX. Further validation and application of the model are warranted to enhance its clinical utility.
Keywords
Introduction
Secondary hyperparathyroidism (SHPT) is a condition in which individuals present with elevated levels of parathyroid hormone (PTH) secondary to other causes, with chronic kidney disease being the most common cause of this condition. The long-term elevation of PTH can result in an imbalanced calcium: phosphorus ratio and abnormal bone metabolism. 1 Total parathyroidectomy (t-PTX) is generally recognized as the most effective treatment for this condition 2 but can result in potentially severe postoperative hypocalcemia that can be difficult to accurately predict and treat. As such, treating postoperative hypocalcemia is integral to the safe treatment of SHPT patients. While researchers have developed increasingly robust models to predict the incidence of postoperative hypocalcemia, 3 no corresponding models have been designed to predict the magnitude of postoperative intravenous calcium supplementation required by affected patients. All studies must be reported according to the relevant Equator network guideline.
To address this gap in the literature and improve the quality of patient care, the present retrospective study was conducted analyzing clinical data from 550 SHPT patients treated in the Department of Hepatobiliary Pancreatic Spleen Thyroid Surgery of the General Hospital of Northern Theater Command from January 2015 to December 2022. The goal of this study was to develop and validate a model capable of predicting the level of calcium (Ca) supplementation required to treat hypocalcemia following t-PTX in SHPT patients in order to ensure that patients can maintain appropriate blood calcium levels following discharge via oral calcium supplementation, thereby improving their overall prognosis and quality of life. There was a preprint posted on 09 May 2022.
Materials and methods
Research participants
Those SHPT patients meeting the indications for surgery were recruited from the Hepatobiliary Surgery Department of the Northern Theater General Hospital from January 2015 to December 2022. Surgical indications were based upon expert consensus regarding surgical clinical practice of hyperparathyroidism secondary to chronic renal failure 4 : ① patients with clear clinical manifestations for whom drug treatment has proven ineffective; ② patients with hypercalcemia and hyperphosphatemia that do not effectively respond to treatment; ③ patients with persistent PTH levels above 800 pg/ml; and ④ patients with ultrasonographic findings revealing a minimum of one hyperplastic parathyroid gland >1 cm in diameter or radionuclide scans revealing a high-density lesion. Surgical treatment was recommended for patients that may any of the four criteria outlined above. Our study was a retrospective study and was in accordance with the WMA Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Participants (2024 version). This research had been approved by the Ethics Committee of the General Hospital of the Northern Theater Command (Approval No. Lunshen Y[2020] 075). All the patient's details have been de-identified in our research.
Inclusion and exclusion criteria
Patients with maintenance hemodialysis or maintenance peritoneal dialysis who underwent TPTX were included as study subjects from 1 January 2015 to 31 December 2022. The inclusion criteria were as follows: ① t-PTX was performed in our hospital; ② Maintenance hemodialysis or peritoneal dialysis patients, dialysis time is greater than or equal to 3 months; ③ The iPTH was > 800 pg/ml in the last 6 months; ④ hypercalcemia or hyperphosphatemia still existed after treatment with calcitriol, paricalciferol, sevilam, and lanthanum carbonate; ⑤ accompanied with severe clinical symptoms, such as bone pain, unbearable skin itching, or muscle weakness affecting daily activities; ⑥ Imaging examination (neck color ultrasound) showed enlargement of at least one parathyroid gland (>1 cm in diameter) and abundant blood flow signals could be detected. The exclusion criteria were as follows: ① diagnosis as malignant tumors, such as parathyroid carcinoma; ② cannot tolerate surgery due to serious heart and lung disease; ③ in a state of severe infection or inflammation; ④ patients with a history of radiotherapy or surgery in the neck; ⑤ patients with recurrence after parathyroidectomy; ⑥ unwilling to participate in the study; and ⑦ incomplete data collection, such as lack of serum alkaline phosphatase (AKP). This study was reviewed by the Medical Research Ethics Committee of our hospital, and all patients signed informed consent.
The operation procedure
The patient was positioned in a supine posture with bilateral shoulder elevation. A low cervical anterior curvilinear incision was created, followed by dissection of the skin flap, incision of the linea alba of the neck, and exposure of the thyroid gland. The left and right thyroid lobes were dissected, respectively, with careful preservation of the inferior thyroid pole vessels and middle thyroid veins. Subsequent exploration of the parathyroid glands was performed, and their morphological classification was determined. All hyperplastic parathyroid glands were completely resected and labeled in a clockwise sequence: the right inferior, right superior, left superior, and left inferior parathyroid glands were designated as parathyroid gland ①, ②, ③, and ④, respectively. Ten minutes after t-PTX, blood samples were collected and sent for PTH detection. Concurrently, the resected parathyroid gland specimens were submitted for frozen section pathological examination. In cases where frozen section pathology failed to confirm parathyroid hyperplasia or the PTH level did not decrease to the preset threshold, the surgical exploration range was further expanded. This extended exploration included the extracarotid sheath region, the area superior to the thyroid superior pole, central compartment lymph nodes, sternothyroid ligament, and thymus. Any suspicious hyperplastic parathyroid glands identified during this extended exploration were resected, and additional specimens were sent for PTH redetection and repeated frozen section pathological examination. After achieving thorough hemostasis in the surgical field, a drainage tube was placed, and the surgical incision was closed in a layered manner.
Patient data collection
Patient age, gender, years of dialysis, years of SHPT, years of bone pain, bone pain scores, hematological findings, liver function, kidney function, thyroid function, postoperative intravenous calcium supplementation, and other hypocalcemia-relevant indicators were recorded.1,3,5–8 Hypocalcemia was defined as a postoperative blood calcium level <2 mmol/L, as per the quality of prognosis of kidney disease (K / DOQI) guidelines. 9
Calcium supplementation plan
At present, there is no unified standard for the specific calcium supplementation plan for hypocalcemia after t-PTX. Through years of research, our department has summarized the standardized stepwise intravenous calcium supplementation plan as follows: ① 10% calcium gluconate injection and 0.9% normal saline are configured with calcium in a ratio of 1:1; ② The external jugular vein was used as a calcium supplementation channel, and the rate was 20–50 ml/h; ③ Monitor blood calcium on the postoperative day without intravenous calcium supplementation, except for those with clinical symptoms; ④ The dose of calcium supplementation was adjusted according to the blood calcium results of the first 6–8 h after operation and next morning after surgery, and the blood calcium results were 1.8–2.0 mmol/L, mildly hypocalcemia, and 100 ml of 10% calcium gluconate injection was intravenously supplemented 24 h; The blood calcium result was 1.6–1.8 mmol/L, moderate hypocalcemia, 200 ml of 10% calcium gluconate injection intravenously supplemented 24 h; When the blood calcium result was less than 1.6 mmol/L, severe hypocalcemia, 300 ml of 10% calcium gluconate injection was supplemented intravenously within 24 h; ⑤ The follow-up calcium supplementation plan was determined by the blood calcium value in the morning of the next day; ⑥ In the case of no intravenous calcium supplementation, the blood calcium >2.0 mmol/L in the morning for two consecutive days can be discharged.
Management of postoperative blood calcium
① Strictly monitor the patient's vital signs after surgery and check for obvious signs of low calcium, such as facial numbness, hand and foot convulsions, and positive cheek reactions. ② Pay close attention to the laboratory examination results of patients, and receive routine oral medication for preventive treatment, 0.25 µg calcitriol twice a day and 800 IU calcium carbonate and vitamin D3 three times a day, for all patients after surgery. ③ High-calcium dialysis was performed within 24 h after surgery, with a calcium concentration of 1.75 mmol/L in the dialysate. ④ After stopping intravenous calcium supplementation, if the blood calcium level exceeds 2 mmol/L in the morning for 2 consecutive days, the patient can be discharged. ⑤ Strengthen discharge guidance and correctly guide patients’ understanding of hypocalcemia. Certain symptoms that cannot be articulated, such as peripheral nerve numbness, diarrhea, and other gastrointestinal symptoms similar to those of the gastrointestinal tract, decreased blood pressure, and muscle cramps, may be thought to be associated with postdialysis complications. Encourage patients to have regular blood calcium checks.
Statistical analysis
SPSS 21.0 was used for all statistical analyses. Initially, the Kolmogorov–Smirnov test was used to assess whether data conformed to a normal distribution. Normally and non-normally distributed continuous data were then presented as `x ± s and median M (P25, P75), respectively. The relationships between the amount of intravenous calcium supplementation and other variables were assessed through Pearson correlation analyses, with those variables exhibiting significance (p < 0.05) in these initial analyses being incorporated into a multiple linear regression analyses to identify variables independently associated with the amount of intravenous calcium supplementation (dependent variable). A predictive model was further established based on the results of this analysis, the homogeneity of the determination coefficient (R2), and variance. Perform residual analysis and collinearity diagnosis on the prediction model to ensure that there is a linear relationship between the independent and dependent variables in multiple linear regression analysis, and that each variable is independent of each other without collinearity. The standardized residual is distributed normally.
Results
Baseline data of patients
In total, 288 males and 262 females (52.4% and 47.6%) were included in this study cohort. For a summary of key demographic and clinical variables pertaining to these patients, including intravenous calcium supplementation, age, years of hyperparathyroidism, years of bone pain, preoperative bone pain scores, key preoperative clinical variables (red blood cell count, hemoglobin [HGB], blood β2 microglobulin, serum ferritin (SF), PTH, 25 hydroxyvitamin D, alanine aminotransferase, AKP, albumin, creatinine, phosphocreatine kinase, phosphocreatine kinase isoenzyme [CKMB], glucose quantification, CA, phosphorus, free triiodothyronine [FT3], free thyroxine [FT4], and thyroid-stimulating hormone), and intraoperative PTH levels (see Table 1).
Clinical and demographic characteristics of SHPT patients.

Univariate analysis of factors associated with patient intravenous calcium supplementation amount
Serum ferritin, PTH, AKP, CKMB, FT3, and intraoperative PTH were all found to be significantly positively correlated with the amount of intravenous calcium supplementation, while gender, age, HGB, CA, and FT4 were significantly negatively correlated with the amount of intravenous calcium supplementation. For further details, see Table 2.
Univariate analysis of factors associated with patient intravenous calcium supplementation amount.
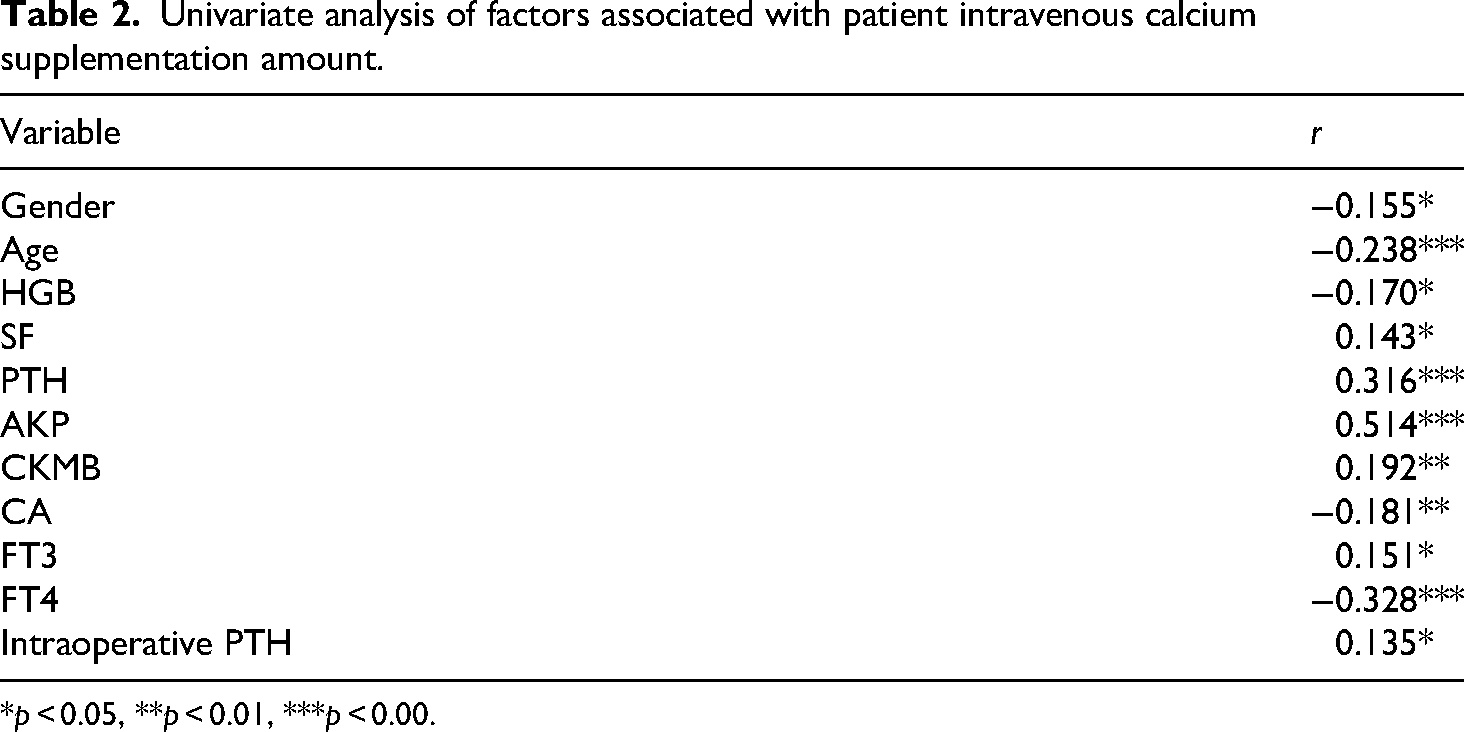
*p < 0.05, **p < 0.01, ***p < 0.00.
Multivariate analysis of factors associated with patient intravenous calcium supplementation amount
Next, gender, age, HGB, SF, PTH, AKP, CKMB, CA, FT3, FT4, and intraoperative PTH levels were selected as independent variables, while the amount of intravenous calcium supplementation was selected as a dependent variable for multiple stepwise regression analysis. The resultant predictive model incorporated gender (male: 0, female: 1), SF, AKP, and FT4, and had the formula shown below:
This model revealed that both gender and FT4 levels negatively impact the amount of intravenous calcium administered, whereas SF and AKP levels positively impact the amount of intravenous calcium administered. The R2 value for this model was 0.410, indicating that these four variables can explain 41.0% of the variation in the amount of intravenous calcium supplementation. Importantly, this model passed the F test, thus demonstrating it to be effective (F = 22.034, p = 0.000). For further details, see Table 3.
Multiple stepwise regression analysis results of factors associated with patient intravenous calcium supplementation amount.

Dependent variable: intravenous calcium supplementation units.
Residual analysis and collinearity diagnosis
A multicollinearity test for this model further indicated that all VIF values in this model were less than 5, thus indicating the absence of any collinearity issues. In addition, the D-W value was close to 2, consistent with an absence of any autocorrelation or correlation among sample data, further supporting the integrity of the constructed model. The residual analysis is shown in Figure 1. The standardized residual can meet the requirements of a normal distribution, and the model and data can be matched. The calculation results are statistically significant, and the independent variable can accurately predict the total amount of intravenous calcium supplementation. The expected and observed cumulative probabilities are shown in Figure 2.

Residual analysis. Combined with the fact that all VIF (variance inflation factor) values are less than 5, this indicates that there is no multicollinearity in the model, and the results are statistically significant.

Expected and observed cumulative probabilities plot. The scattered points are closely distributed around the diagonal line, which indicates that the observed data in the model are in good agreement with the expected distribution and conform to the assumptions.
Discussion
Hypocalcemia and intravenous calcium supplementation after t-PTX in SHPT patients
Hypocalcemia is a common complication that can develop within 24–48 h after t-PTX, resulting in clinical symptoms including numbness and convulsions of the hands and feet, arrhythmia, epilepsy, a positive cheek reaction, severe asphyxia, and even cardiac arrest. 10 At present, there is a large amount of research on the prediction of hypocalcemia, but there is little research on postoperative intravenous calcium supplementation. Clinicians should pay more attention to how to standardize the treatment during the perioperative period and prevent serious complications caused by hypocalcemia. The treatment of hypocalcemia is a long-term and repetitive process, and its duration and mechanism are closely related to bone transformation. In severe cases, it can last for 6–12 months. Long-term calcium supplementation in the hospital not only reduces clinical work efficiency but also increases the patient's economic burden. However, only 29.82% of affected patients exhibit clinical symptoms. The primary causes of hypocalcemia following t-PTX include a sudden drop in PTH levels following parathyroidectomy, resulting in the transfer of large quantities of calcium ions from the blood into the bone and a concomitant rapid drop in serum calcium concentrations. 11 In the present study, hypocalcemia developed in 72.7% of patients, consistent with the 29–80% incidence rate reported previously.6,7,12–14 However, there are no unified standards available to treat postoperative hypocalcemia in SHPT patients at present. Based upon several years of clinical experience, our department generally treats this condition through calcium supplementation via the intravenous infusion of 100 ml normal saline + 100 ml 10% calcium gluconate at a flow rate of 20–50 ml/h. This infusion is performed once, twice, or thrice daily when the blood calcium level is 1.8–2.0, 1.6–1.8, and <1.6 mmol/L, respectively. Such intravenous calcium supplementation was terminated when the blood calcium level was >2.0 mmol/L on the mornings of two consecutive days, and the patients did not exhibit any symptoms of perceived hypocalcemia. The total amount of intravenous calcium supplementation in the present study was 0–3600 ml, in line with previous reports of high levels of intravenous calcium supplementation during hospitalization (0–2800 ml), with an average supplementation volume of 557.86 ± 376.20 ml. Differences among studies may be attributable to differences in sample size, operative approach, preoperative dialysis, or patient management. Our department conducted postoperative follow-up assessments for patients and found that 82.47% were able to maintain appropriate blood calcium levels by taking oral calcium tablets containing vitamin D3, while 5 patients required high-concentration intravenous calcium supplementation. These findings highlight the clinical significance of operative approach selection and intravenous calcium supplementation.
Construction of a model to predict the volume of intravenous calcium supplementation in SHPT patients following t-PTX
An extensive literature search revealed that several models to predict postoperative hypocalcemia following t-PTX have been developed to date6, yet no corresponding predictive tools are available to estimate the amount of postoperative intravenous calcium supplementation required by these patients. 15 In this study, the final predictive model incorporated independent variables including gender, SF, AKP, and FT4 that were associated with the need for postoperative intravenous calcium supplementation, in contrast to variables included in other studies such as age, HGB, CA, AKP, and PTH. 8 The results of this study are similar to the prediction model constructed in the study by Ming Cheng et al., where the formula is: calcium requirement for 1 week after surgery = 33.798–8.929 × immediate postoperative calcium + 0.190 × C-reactive protein – 0.125 × age + 0.002 × preoperative intact PTH + 0.003 × preoperative AKP (R² = 0.8). The study results by Liesbeth Viaene et al. 16 indicated that preoperative PTH and serum calcium are factors influencing the amount of intravenous calcium supplementation. In contrast, the study by Byung Heon Kang et al. 17 identified preoperative AKP, PTH, and phosphorus levels at 48 h postoperatively as the influencing factors for the amount of intravenous calcium supplementation. These findings differ from those of the present study, and the reasons may be related to factors such as differences in sample size and perioperative management.
The developed model revealed a negative relationship between gender and the amount of intravenous calcium supplementation such that the amount of intravenous calcium administered to men was, on average, higher than that administered to women. This may be attributable to the higher number of men in this study relative to women, and to the fact that the average population age was 48 years. At this age, the rate of osteoporosis among women is roughly twice that among men, and the lower levels of postoperative blood calcium in women may suggest that men require higher levels of blood calcium to maintain sufficient calcium levels within the bone. However, this will need to be tested in future studies.
The developed model further indicated that ferritin levels had a positive impact on the amount of intravenous calcium supplementation, with higher SF levels corresponding to higher amounts of postoperative calcium administration. Serum ferritin is the primary iron storage mechanism in the body and thus the main index used to evaluate iron deficiency or overload. It can also be used to evaluate malnutrition, calcium/phosphorus metabolism disorders, and the presence of microinflammatory states. 18 In prior research, elevated SF levels have frequently been observed in dialysis patients and independently associated with mortality in this patient population, and a high SF environment can impact phosphorus excretion and utilization. Higher levels of phosphorus in the blood can, in turn, inhibit calcitriol formation and lower blood calcium levels. 19 Lien et al. 20 reported that increases in iron ion concentrations were associated with reductions in calcium ion levels within osteoblasts. As iron ions are closely tied to the metabolism of calcium ions, they can thus impact bone metabolism. Kim et al. 21 determined that when SF levels are appropriately controlled, this can alleviate bone metabolism and microinflammation related to iron deficiency, thus improving patient outcomes. This evidence all supports the incorporation of SF levels in our postoperative intravenous calcium supplementation model.
In the developed model, AKP was found to positively impact the amount of postoperative intravenous calcium supplementation in this patient population, in line with prior results. 7 Serum AKP is primarily secreted by the liver and by osteoblasts, and functions as a key enzyme that promotes bone matrix mineralization. KDIGO guidelines recommend the use of AKP levels as a metric to gauge the severity of bone mineral metabolic abnormalities in chronic kidney disease patients. 22 Ge et al. 23 found AKP to be associated with osteoblast activity and to reflect the severity of postoperative hungry bone syndrome. Yang et al. 24 further determined that AKP can reflect bone reconstructive activity, serving as a valuable index to guide postoperative calcium supplement dosing. Tsai et al. 25 analyzed 62 patients who had undergone t-PTX and determined that higher AKP values were associated with a greater risk of postoperative hypocalcemia, more serious symptoms, and the need for a larger supplemental calcium dose.
The regression coefficient in the developed model indicates that GT4 levels negatively impact the amount of postoperative intravenous calcium supplementation. Laowalert et al. 26 previously demonstrated that FT4 can impact bone metabolism, altering the absorption and formation of bone tissue. Liangos et al. 27 found that patients suffering from chronic renal failure often exhibit normal or reduced FT4 levels owing to impaired pituitary thyroid functionality. As renal function deteriorates, pronounced FT4 abnormalities can develop. Abnormal FT4 metabolism can also be associated with high PTH levels and hypogonadism, with high PTH inhibiting the production of T3 from T4. As such, we can infer that FT4 may impact bone and calcium metabolism to some degree. However, research on this topic is limited, and further research will thus be critical to test this hypothesis, with the present large-scale study serving as a valuable foundation for future studies.
Our study has a large sample size, and the data are representative. The constructed prediction model can well reflect the real situation of patients with SHPT, and based on the results, an appropriate calcium supplementation method can be selected for patients before surgery. However, this study still has certain limitations: all data in this study were obtained from a single center, which may lead to certain biases. In the future, collaboration with other medical centers can be carried out to further verify the effectiveness of the model.
Conclusions
In summary, the present study successfully established a predictive model capable of estimating the amount of postoperative intravenous calcium supplementation required for SHPT patients undergoing t-PTX. The prospective assessment of this parameter using this model has the potential to guide clinical decision-making pertaining to the route of postoperative calcium supplement (subclavian vein, central vein catheterization, external jugular vein, etc.) and any other preoperative or postoperative interventions that may be appropriate. Overall, these findings thus provide a robust theoretical foundation for the development of a standardized calcium supplementation strategy that can reduce the incidence of complications and improve patient outcomes.
Footnotes
Ethical considerations
We confirm that all the research meets ethical guidelines and adheres to the legal requirements of the study country. Our study was in accordance with the WMA Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Participants (2024 version).
Consent to publish
I confirm that any participants (or their guardians if unable to give informed consent, or next of kin, if deceased) who may be identifiable through the manuscript (such as a case report), have been given an opportunity to review the final manuscript and have provided written consent to publish.
Author contributions
BT conducted the main research, JLY collected the patient's data, BSJ wrote the original manuscript. GMC supervised the whole research. GS conducted the statistical analysis. FXD and ZW formulated the research concept and designed the study. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Joint Plan of Liaoning Provincial Science and Technology Plan (2024-MSLH-531).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
