Abstract
Keywords
Introduction
Owing to its rarity, debilitating nature, and a lack of definitive treatment in certain cases, trigeminal neuralgia remains a topic of ongoing research. The global prevalence of this condition ranges between 29.5 and 108.8 per 100,000 population, and in Turkey it ranges between 52.1 and 98.5 per 100,000 population.1,2 The incidence of trigeminal neuralgia increases with age, and is higher in women than in men, with an approximate female-to-male ratio of 2:1. 3 Based on its etiology, trigeminal neuralgia is categorized into classical, secondary, and idiopathic types. Despite the significant advancements in imaging modalities, a considerable number of cases are still diagnosed as idiopathic trigeminal neuralgia in daily clinical practice, with the underlying etiology remaining undetermined.
In a recently developed model of trigeminal neuropathic pain (TNP), the etiopathogenesis of idiopathic trigeminal neuralgia was inferred to potentially originate from the cervical region, whereas the hypothesis that idiopathic trigeminal neuralgia may be linked to the upper cervical area was evaluated through the induction of mechanical allodynia on the ipsilateral side through compression of the upper cervical dorsal horn. 4 Molecular studies have identified potential abnormalities in cation channels associated with trigeminal neuralgia and other pain syndromes. Dysfunctional ion channels may lead to neuronal hyperexcitability and consequent pain.5–7 Furthermore, the involvement of chloride, calcium, potassium, and transient receptor potential cation channels in trigeminal neuralgia has been investigated.8–12
Voltage-gated sodium channels (Nav), comprising nine distinct α subunits, play a fundamental role in the generation and propagation of electrical signals in neurons. 13 Considerable research has demonstrated the involvement of the following genes in the pathophysiology of neuropathic pain: SCN3 gene, which encodes Nav1.3; and SCN9, SCN10, and SCN11, which encode Nav1.7, Nav1.8, and Nav1.9, respectively. Clinical studies have shown the aberrant expression of Nav subtypes in the peripheral axons or neuromas of patients with trigeminal neuralgia and neuropathy.14,15 Mutations in the genes associated with the Nav1.3, Nav1.7, Nav1.8, and Nav1.9 subtypes have been identified in patients with trigeminal neuralgia.16–18 Although Nav1.1 and Nav1.6 mutations are not commonly implicated in human pain syndromes, gain-of-function mutations in Nav1.1 (encoded by SCN1A) have been associated with familial hemiplegic migraine, while a similar pathogenic mechanism involving Nav1.6 (encoded by SCN8A) has been reported in a case of trigeminal neuralgia.19–20
Voltage-gated calcium channels are essential components of excitable cells that facilitate neuronal communication by converting action potentials into calcium ion flows that trigger the release of neurotransmitters. Voltage-gated calcium channels dysfunction within sensory systems can lead to the sensation of pain. An association between neuropathic pain and the CACNA1C gene, which encodes the α1C-subunit of CaV1.2 has been reported,21–23 while a CACNA1A (which encodes CaV.2.1) mutation, which possibly modulates channel-gating kinetics, has been linked to trigeminal neuralgia. This indicates that changes in Cav2.1-mediated synaptic transmission within the trigeminal system could mediate mechanisms associated with pain processing. 23 In another TNP animal model induced by chronic constriction injury of the infraorbital nerve (IoN-CCI), the expression of the CACNA1C ion channels was considerably increased in the trigeminal ganglion and trigeminal spinal nucleus on the seventh day. 24
Potassium channels (Kv) have been found in peripheral neurons, such as the dorsal root ganglia (DRG), and are associated with pain pathways. The expression of Kv1.1 and Kv1.2 channels, which are encoded by the KCNA1 and KCNA2 genes, respectively, have been linked to neuropathic pain, whereas various animal models have been employed to investigate their involvement in pain pathophysiology. 25
Although numerous animal models have been established, no model has thus far fully met the criteria for a trigeminal neuralgia model,26–29 and the challenges associated with obtaining a clinical sample of trigeminal neuralgia warrant the development of a straightforward, dependable, and consistent animal model with a high success rate in order to elucidate the underlying pathophysiological mechanisms. The present study sought to investigate alterations in the expression of voltage-gated Na, K, and Ca ion channels in the dorsal horn, trigeminal ganglion, and infraorbital nerve. This investigation aimed to ascertain the validity of the “cervical dorsal horn compression model” as an animal model of TNP and its potential as an underlying etiology of idiopathic trigeminal neuralgia.
Materials and methods
Animals
This study employed a total of 18, 4-month-old healthy, male New Zealand White rabbits weighing 2.5 to 3.5 kg.
Experimental procedure
The rabbits were individually housed in polypropylene cages for at least 7 days before the experiment. Throughout this period, they had ad libitum access to food and water and were kept under a natural light–dark cycle. The ambient temperature was consistently maintained between 22°C and 24°C. Tissue samples were obtained on the eighth day following sacrifice. The study adhered to ARRIVE reporting guidelines. 30 Adequate care was provided to all animals. Guide for the Care and Use of Laboratory Animals, 8th Edition was followed. 31 Maximum effort was exerted to minimize the number of animals used and to decrease their suffering.
Based on a procedure described by Türk Börü et al., 4 the rabbits were examined by a veterinarian blinded to subgroup allocation, and divided into three groups (spinal cord compression group (n:6), sham group (n:6), and control group (n:6)); the anesthesia and monitoring techniques were undertaken, and the surgical procedure was performed by the same surgical team. 4
Anesthesia and monitoring
Xylazine hydrochloride (Xylazinbio,%2, 50 ml Vial, Bioveta, Bioveta, a.s., Komenskeho Czech Repablic) and ketamine hydrochloride (Ketasol, %10, 10 ml vial; İnterhas Medical and Chemical Products Industry and Inc. Ankara-Turkey) were used for inducing anesthesia isoflurane (2%) was used for maintenance of anesthesia by mask method (COMEN AX-400-V automatic ventilatory anesthesia machine with double voparisator, MVM Medical, Istanbul-Turkey). The mechanical ventilation rate was set between 20 and 25 breaths per minute. All rabbits received oxygen supplementation. Blood pressure was measured (Mindray, uMEC15 bedside multichannel monitory¸ Mindray Medical Equipment Corp. China). The mean arterial blood pressure was kept around 80 mmHg. Body temperature was monitored and regulated within the range of 38.5–39.5°C using rectal probes connected to the same monitor, along with heated blankets. None of the animals experienced hypothermia. Electrical cardiac activity, recorded via electrocardiogram (ECG), and oxygen saturation (SpO₂) were continuously tracked using a bedside monitor.
Surgical procedure
The rabbits were positioned in the prone position on the operating table. The dorsal cervical area was shaved with a clipper, and the surgical field was prepared under sterile conditions. A midline incision was made along the cervical segments to expose the C2-C5 laminae. The ligamentum flavum between the C3–4 laminae was removed, creating a small opening near the facet. A left-sided dorsal hemilaminectomy was then performed at the C3 vertebral level following the standard procedure. A PTCA angioplasty balloon catheter (Balloon angioplasty catheters, 2.0 mm×26 mm×120 cm, Extender Drug Eluting Balloon catheter, ®INVAMED, Health Drug Industry, Ankara-Turkey) was utilized in the procedure. The catheter was inserted into the extra-dural and sublaminar region of the spinal cord at the C3 level, with its dorsal side facing upwards, and inflated to a pressure of 1 atm. After maintaining inflation for one minute, the balloon was deflated, and the catheter was withdrawn to induce SCC. The paravertebral fascia and soft tissue were subsequently closed using simple interrupted absorbable sutures (Vicryl Rapide, Polyglactin 910, 4-0, Ethicon Corp., USA), while the midline skin was sutured with simple interrupted nonabsorbable sutures (Prolene, Polypropylene, 3–0, Tıpkimya Medical, Chemical Industry, İstanbul, Turkey). No interventions were performed in the control group. In the SG group, dorsal hemilaminectomy was omitted, and the surgical site was closed without compression.
Mechanical allodynia
Mechanical allodynia evaluation was conducted using calibrated von Frey nylon monofilaments (North Coast Medical Inc., Morgan Hill, CA, USA). Assessments were performed preoperatively and continued daily up to the eighth postoperative day, within a quiet room between 1:00 and 4:00 p.m. To minimize distress, each rabbit was gently restrained with a soft cloth while placed on a nonslip surface, allowing free movement of both forelimbs and hind limbs. Sensory testing was applied bilaterally on the facial area. Stimulation began with the 0.07 g monofilament and proceeded incrementally up to 8 g until a response was observed. In cases where no reaction occurred with lower intensities, filaments of 4, 6, and 8 g were utilized to reduce both examination time and potential animal stress. 32 Stimulation was focused on the vibrissal pad region, with each filament applied five times at 5 to 10 second intervals. The most pronounced response for each stimulus was recorded. A composite “response score” was calculated by averaging the scores from the three most reactive filaments. 33 Nocifensive behavior was graded as follows: 0—no response; 1—exploratory recognition of the filament; 2—slow facial withdrawal; 3—rapid withdrawal with paw lifting; 4—more than three facial wipes with the forepaw; and 5—three or fewer wipes of the facial region. 34
Sacrification and sample collection
Rabbits in all groups were sacrificed by intraperitoneal injection of thiopental sodium at a dose of 150 mg/kg for the purpose of tissue sampling on day 8 at the end of the experiment. Subsequent to this, the rabbits were placed in sternoabdominal position and underwent dorsal laminectomy at the C1–6 level. Spinal cord tissue samples were collected at the C3 level. The craniotomy was then performed in accordance with the established technique. The trigeminal ganglion was meticulously dissected, and a tissue sample was collected for further analysis. The ION was then subjected to a detailed dissection, meticulously executed in accordance with the established technique, and a tissue sample was collected for further analysis.
Quantitative polymerase chain reaction
Tissue samples were collected and promptly placed into cryotubes, rapidly frozen in liquid nitrogen, and stored at −80°C until further analysis. Total RNA was extracted by using the GeneJet RNA Purification Kit (Thermo K0732, USA) in accordance with the manufacturer's instructions. The RNA concentration was determined using the Qubit RNA HS Assay Kit (Thermo Fisher, Q32855, USA) with a Qubit 2.0 Fluorometer (Invitrogen, USA), whereas the RNA quality was assessed with the Multiskan Go (Thermo Scientific, USA). To eliminate any potential DNA contamination, the isolated RNA samples were treated with RNase-free DNase I (Thermo Fisher Scientific, EN0525). Subsequently, single-strand cDNA synthesis was conducted using the Maxima First Strand cDNA Synthesis Kit (Thermo Fisher Scientific, K1672) in accordance with the recommended protocol.
Primers were designed using FastPCR 6.0 software. 35 Gradient polymerase chain reaction (PCR) was conducted to determine the optimal melting temperatures (Tm) for each primer. Table 1 includes the details of primer pairs, Tm, and cDNA sizes for the genes used in real-time PCR reactions. The optimal cDNA dilution for all genes was determined to be 1:50. Real-time PCR analysis was undertaken using RealQ Plus 2× Master Mix Green (Ampliqon, A323402, Denmark). Each 25-µL PCR reaction mixture contained 2 µL cDNA (diluted 1:50), 1× RealQ Plus Master Mix, and 5 µM each of forward and reverse primers. The QuantStudio 5 real-time PCR system (Thermo Fisher Scientific, USA) was set for an initial denaturation at 95°C for 15 minutes, followed by 40 cycles of 30 s, at 95°C, and 1 minutes, at 61°C. Two negative controls, using ultrapure water instead of cDNA, were included in each plate. Melting curve analysis was subsequently performed for each gene and sample to confirm the absence of DNA contamination and primer dimerization. The fold changes in mRNA expression levels of the target genes were calculated using the 2−ΔΔCt method with the REST (2009) software (Relative Expression Software Tool V. 2.0.13), based on the cycle threshold (Ct) values of the amplification obtained curves. The GAPDH gene (housekeeping gene) served as an endogenous control, and the expression levels of the target genes were normalized to that of the GAPDH gene in each sample.
List of the genes, primers, GenBank accession numbers, Tm values, and mRNA sizes used in the real-time polymerase chain reaction analysis.
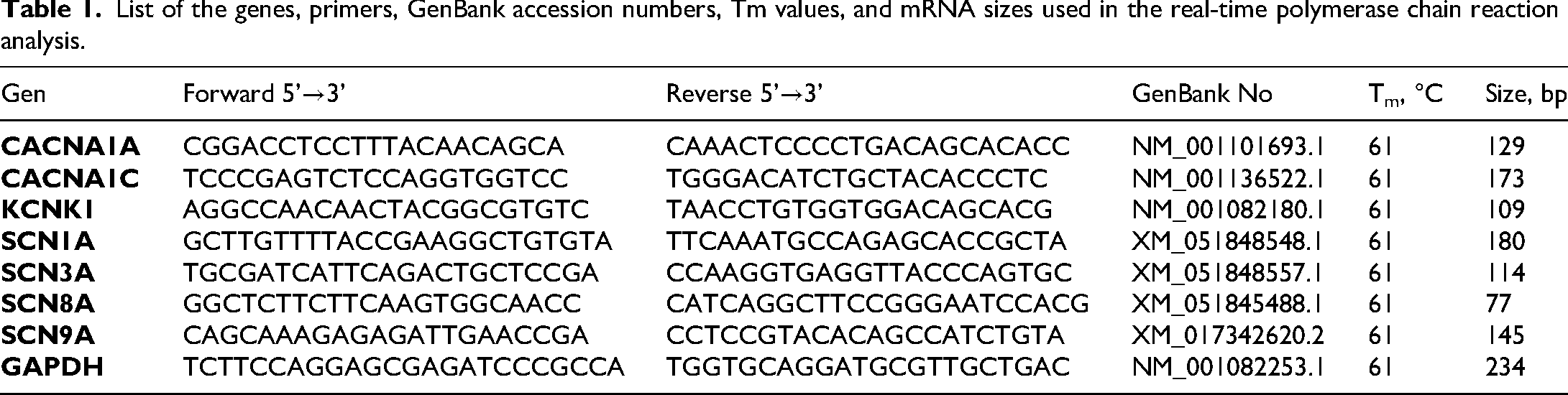
All analyses were conducted in triplicate for each sample, and the mean value of these three measurements was used for statistical analyses. Details of the genes, primers, GenBank accession numbers, Tm values, and mRNA sizes used in the real-time PCR analysis are provided in Table 1.
Statistical analysis
For the analysis of mechanical allodynia, an independent Samples t-test was used to compare the groups (left side—ipsilateral and right side—contralateral) for each day. Additionally, a repeated measures analysis of variance (ANOVA) was applied to evaluate time points (preoperative and postoperative days 1–8) within each group. The normality of the data distribution was assessed using the Shapiro-Wilk test, while variance homogeneity was tested with Levene's test. In cases where a significant difference was detected in the repeated measures ANOVA analysis, Bonferroni post hoc tests were conducted to further analyze the differences between the groups. Results were reported as mean ± standard deviation (SD), and a significance level of p < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS 21.0 (IBM, Windows version) software.
Results
Mechanical allodynia
In the spinal cord compression group, the mechanical allodynia score increased significantly on the ipsilateral side on the third day and maintained an increasing trend until the eighth day. There was no significant difference between the two sides in the sham and control groups (Figure 1).

Mechanical allodynia scores. An independent samples t-test was employed to facilitate a comparison between the two sides (left and right) on a daily basis. The level of statistical significance was set at 0.05.
The mechanical allodynia scores are shown in Table 2.
Mechanical allodynia scores.
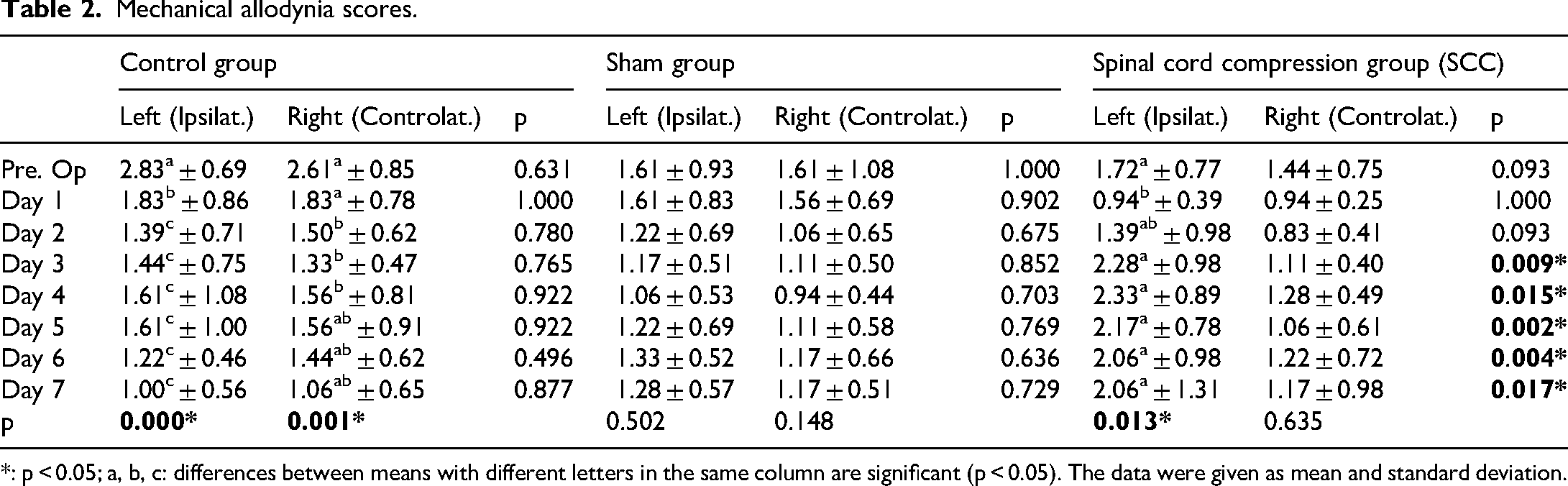
*: p < 0.05; a, b, c: differences between means with different letters in the same column are significant (p < 0.05). The data were given as mean and standard deviation.
Expression of SCN1A, SCN3A, SCN8A, and SCN9A in the spinal dorsal horn
Compared to the control group, the SCC group demonstrated decreased mRNA expression of SCN1A, a voltage-gated sodium channel, within the spinal dorsal horn. Conversely, increased expression of SCN3A, SCN8A, and SCN9A was observed. However, compared to the control group, only the increase in SCN9A expression in the SCC group reached statistical significance (p < 0.001).
Expression of CACNA1A, CACNA1C, and KCNK1 in the spinal dorsal horn
Compared to the control group, the SCC group exhibited a notable elevation in the mRNA expression levels of CACNA1A, CACNA1C, and KCNK1. A statistically significant augmentation in the expression of CACNA1C and KCNK1 was further identified in the SCC group (p < 0.001) compared with the control group.
Expression of SCN1A, SCN3A, SCN8A, and SCN9A in the trigeminal ganglion
Compared to the control group, the SCC group revealed an upregulation in the mRNA expression level of SCN1A, SCN3A, SCN8A, and SCN9A in the trigeminal ganglion; however, only the increase in SCN9A expression was statistically significant (p < 0.001).
Expression of CACNA1A, CACNA1C, and KCNK1 in the trigeminal ganglion
A downregulation of the CACNA1A gene expression was observed in the trigeminal ganglion of the SCC group compared to the control group, albeit without statistical significance. The expression levels of CACNA1C and KCNK1 were higher in the SCC group than in the control group; however, only the increase in KCNK1 expression reached statistical significance compared to the control group.
Expression of SCN1A, SCN3A, SCN8A, and SCN9A in the infraorbital nerve
A significant upregulation of the expression of mRNAs for SCN1A, SCN3A, SCN8A, and SCN9A was observed in the SCC group compared to the control group (p < 0.001), for all voltage-gated sodium channel mRNA expressions (Figure 2).

Relative fold changes in the mRNA expression of voltage-gated sodium channels (Nav) in the infraorbital nerve compared to the control group (***p < 0.001). The fold changes in the mean mRNA expression levels of the target genes were calculated using the 2−ΔΔCt method with the REST (2009) software (Relative Expression Software Tool V. 2.0.13), based on the cycle threshold (Ct) values of the amplification obtained curves. SCC, spinal cord compression group; SG, sham group.
Expression of CACNA1A, CACNA1C, and KCNK1 in the infraorbital nerve
A notable elevation in mRNA expression levels of CACNA1A, CACNA1C, and KCNK1 was observed in the SCC group relative to the control group. The augmentation in the expression of all three genes was statistically significant (p < 0.001) (Figure 3).
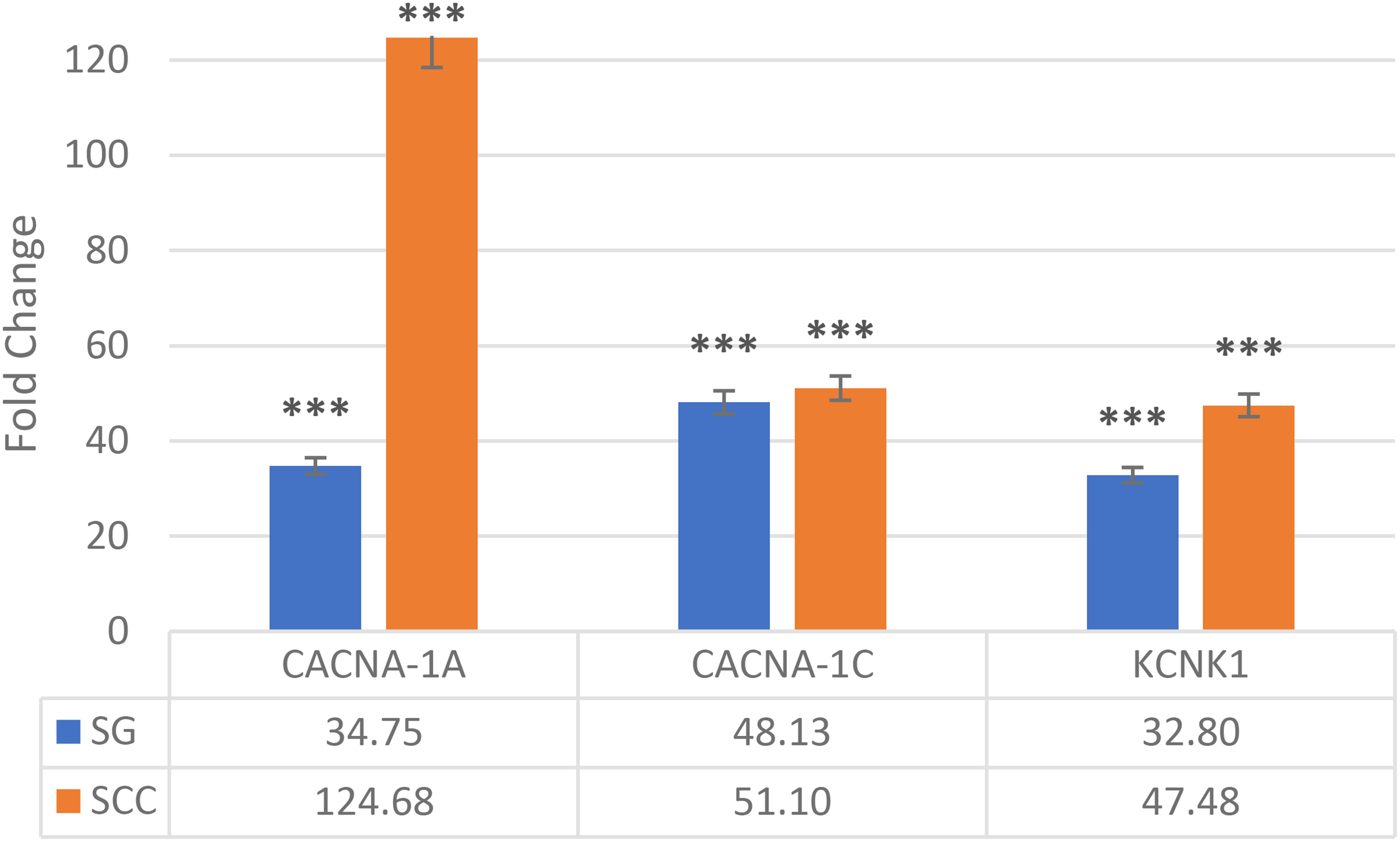
Relative fold changes in the mRNA expression of calcium and potassium ion channels in the infraorbital nerve compared to the control group (***p < 0.001). The fold changes in the mean mRNA expression levels of the target genes were calculated using the 2−ΔΔCt method with the REST (2009) software (Relative Expression Software Tool V. 2.0.13), based on the cycle threshold (Ct) values of the amplification obtained curves. SCC, spinal cord compression group; SG, sham group.
Comparison of the results in the spinal cord compression and sham groups
In the cervical spinal dorsal horn, the expression of CACNA1A and CACNA1C were decreased in the SCC group compared with the sham group, whereas KCNK1 expression increased (Table 3). Furthermore, SCN1A, SCN3A, SCN8A, and SCN9A expression decreased, although the difference was not statistically significant.
Relative fold changes in mRNA expression of genes in the ipsilateral spinal dorsal horn, trigeminal ganglion, and infraorbital nerve in the SCC group compared to the sham group.
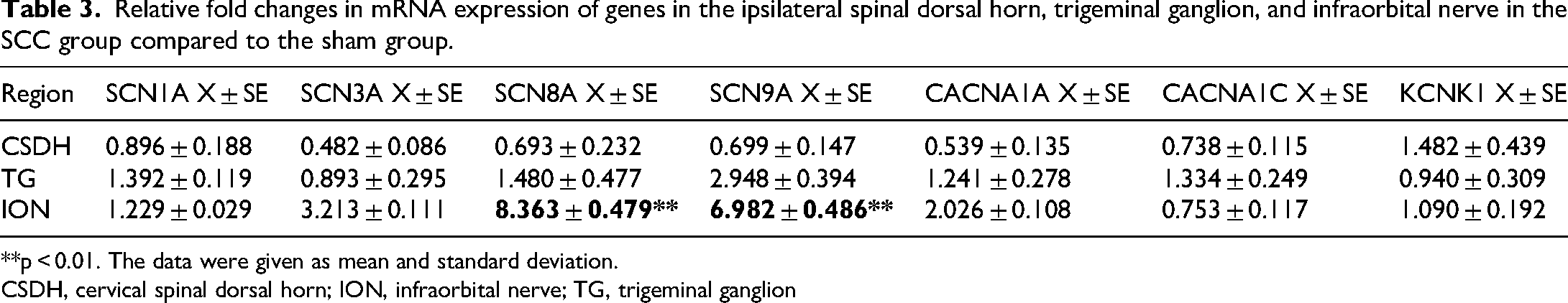
**p < 0.01. The data were given as mean and standard deviation.
CSDH, cervical spinal dorsal horn; ION, infraorbital nerve; TG, trigeminal ganglion
In the trigeminal ganglion, the expression of KCNK1 and SCN3A decreased, whereas the expression of CACNA1A, CACNA1C, SCN1A, SCN8A, and SCN9A increased; however, none of these changes attained statistical significance.
In the infraorbital nerve, except CACNA1C, the expression of all ion channels, including KCNK1, CACNA1A, SCN1A, SCN3A, SCN8A, and SCN9A, increased although significantly increased expression was noted only in SCN8A and SCN9A compared to the sham group. These results indicate that the increased expression of SCN8A and SCN9A in the infraorbital nerve particularly may indicate a potential role of Nav1.6 and Nav1.7 channels in the pathophysiology of trigeminal neuralgia.
Discussion
In this study, we used a previously developed rabbit model of TNP to investigate the relationship of the expression of Na, K, and Ca ion channels in the cervical spinal dorsal horn, trigeminal ganglion, and infraorbital nerve with TNP. 4 Mild compression of the spinal dorsal horn at the C3 level induced mechanical allodynia in the vibrissae pad within the ipsilateral trigeminal nerve-innervated area, from the third day, while replication of a previously developed TNP model was achieved.
In our previous study, mild compression of the cervical spinal dorsal horn at the C3 level on the ipsilateral side induced TNP in the SCC group, and increased levels of astrocytic (GFAP) and microglial (Cd11b) markers, as identified through immunohistochemistry in the ipsilateral spinal dorsal horn, trigeminal ganglion, and infraorbital nerve; additionally, apoptosis was detected via TUNEL labeling. 4 Neuroinflammation and ultrastructural modifications in the compressed cervical dorsal horn, ipsilateral trigeminal ganglion, and infraorbital nerve were confirmed by immunohistochemistry and transmission electron microscopy. Moreover, retrograde sensitization of the peripheral nerve led to the spread of inflammation the infraorbital nerve, and contributed to the ultrastructural changes. 4 Activation of microglia after spinal cord injury via the TLR4/MyD88/NF-κB inflammatory pathway was shown to leads to the release of pro-inflammatory cytokines that activate and sensitize spinal nociceptive neurons, thereby exacerbating neuropathic pain. 36 Microglial involvement following spinal cord compression is primarily mediated by the release of pro-inflammatory cytokines and chemokines. Upon activation, microglia initiates the secretion of these cytokines, triggering an inflammatory response. This inflammatory environment can subsequently stimulate other cellular components, such as astrocytes, further exacerbating neuropathic pain 37
In the pathological context of spinal cord injury, astrocytes become activated, leading to a significant upregulation of GFAP expression, a key indicator of astrocyte hyperplasia. The activation of Cx43 half-channels in reactive astrocytes facilitates the release of ATP, glutamate, CCL2, and other signaling molecules. These molecules interact with neuronal purinergic P2X/Y receptors, NMDA receptors, or CCR2, resulting in neuronal hyperactivation. Additionally, ATP stimulates reactive microglial proliferation by acting on P2X/Y receptors in microglia, further intensifying abnormal pain signaling. 38 In general, microglia, astrocytes, and relative proinflammatory factors such as IL-1β, interact with each other closely and participate in both development and maintenance of neuropathic pain. 39
The results of this study revealed that the mRNA expression levels of CACNA1C, KCNK1, and SCN9A were significantly higher in all regions (cervical spinal dorsal horn, trigeminal ganglion, and infraorbital nerve) in the SCC group compared to the control group. Although not statistically significant, CACNA1A expression was found to be decreased in the cervical spinal dorsal horn and increased two-fold in the infraorbital nerve. Moreover, SCN3A expression decreased in the cervical spinal dorsal horn and trigeminal ganglion, but increased in the infraorbital nerve compared to the sham group, although this increase was not statistically significant. The expression levels of SCN8A and SCN9A were higher in the infraorbital nerve in the SCC group than in the sham group. The elevated expression of Nav1.3, Nav1.6, and Nav1.7, particularly in the infraorbital nerve, indicates that these sodium channels play a role in TNP. Thus, we inferred that Nav channels, particularly Nav1.6 and Nav1.7, play a pivotal role in the pathogenesis of TNP. However, CaV2.1, CaV1.2, and K2P1.1 channels may also be involved. The absence of statistical significance in the comparison of ion channel expressions between the sham and SCC groups may indicate that the surgical intervention itself, rather than the spinal cord compression alone, is the primary factor influencing gene expression. However, this may also be due to the limited number of samples. Molecular studies have identified potential abnormalities in the expression or function of cation channels that are associated with TNP and other pain syndromes. 40
Role of Na channels
The voltage-gated sodium channel (Nav) family is crucial for the initiation and propagation of action potentials. These channels are intricate heteromultimeric protein structures, in which the α-subunits form the core functional pore and are accompanied by one or more auxiliary β-subunits. The α-subunits are encoded by a set of 10 distinct genes, which orchestrate their expression across various excitable cell types. 41 Nav1.6 is expressed in small DRG neurons that give rise to unmyelinated C-fibers; consequently, the absence of this channel in DRG neurons leads to a reduction in C-fiber conduction velocity, which underscores its involvement in transmitting noxious stimuli. Increased sodium channel density has been associated with a higher probability of action potential generation in demyelinated axons. 42
The Nav1.1, Nav1.2, Nav1.3, and Nav1.6 channels are primarily expressed in the central nervous system, whereas Nav1.7, Nav1.8, and Nav1.9 are primarily present in the peripheral nervous system. Various Nav channel subtypes, including Nav1.3, Nav1.6, Nav1.7, Nav1.8, and Nav1.9, play significant roles in pain sensation.43–46 The association between Nav1.6 with TNP is not fully understood. To date, only one patient with trigeminal neuralgia has been reported to have an Nav1.6 mutation. Tanaka et al. identified an Nav1.6 mutation (Met136Val) in a patient with trigeminal neuralgia, which caused an average 1.6-fold increase in resurgent current in trigeminal ganglion neurons. This increase in resurgent current may provide a physiological basis for the enhanced firing of trigeminal ganglion neurons. 16
Previous animal model studies have highlighted the role of Nav1.6 in neuropathic pain. Localized knockdown of Nav1.6 in the DRG significantly diminished pain behaviors, reduced sympathetic sprouting, and decreased the spontaneous activity of sensory neurons in the spinal nerve ligation model. These effects were evident when Nav1.6 was silenced in the DRG, effectively blocking both mechanical pain and abnormal spontaneous activity in myelinated neurons caused by DRG inflammation.47,48 Among mature neurons, Nav1.6 is primarily situated at the axon initial segment the unmyelinated proximal region where action potentials originate and at the nodes of Ranvier, which are essential for the propagation of action potentials along the axon. Nav1.6 can produce substantial, prolonged sodium currents, a type of sodium channel activity that profoundly affects the physiological behavior of excitable cells.49–51
In another study employing a TNP animal model induced by infraorbital nerve injury, an increase in Nav1.3, Nav1.7, and Nav1.8 mRNA levels was observed in the ipsilateral infraorbital nerve, while a decrease was noted in the trigeminal ganglion. 14 Xu et al. 52 investigated the roles of Nav1.3, Nav1.7, Nav1.8, and Nav1.9 in TNP using the IoN-CCI model in rats. Significantly decreased expression of Nav1.7, Nav1.8, and Nav1.9 was observed in rats subjected to IoN-CCI surgery as compared with the sham group. In contrast, our study demonstrated an increase in Nav1.7 expression in both the trigeminal ganglion and infraorbital nerve compared to the control group. Given that our TNP model involves compression of the dorsal horn of the spinal cord, differences in the expression of Nav1.7 may be observed depending on whether the injury site is peripheral or central.
In a TNP model involving IoN-CCI, the proportion of Nav1.3-positive cells increased significantly by postoperative day 7, peaking at day 14. 21 In our study, this increase was more than three-fold higher compared to the sham group on day 8, and it is plausible that this elevation could have been even more pronounced had the study duration been longer. Siqueira et al. 53 conducted a case-control study to examine the expression of Nav1.3, Nav1.7, and Nav1.8 in gingival tissues from 10 patients with trigeminal neuralgia, compared to controls. Their findings revealed that Nav1.7 was downregulated, whereas Nav1.3 was upregulated in these patients.
In the TNP model induced in rats with IoN-CCI, approximately 73% of trigeminal ganglion neurons expressed the Nav1.7 channel, which appears to be highly expressed in both peptidergic C and Aδ neurons. Administration of CRMP2-Ubc9 inhibitors via the nasal route reduced the activity of Nav1.7 and mitigated TNP. Additionally, Nav1.7 was shown to play a significant role in TNP within the trigeminal ganglion. In our study, Nav1.7 expression was relatively higher in the cervical dorsal horn, trigeminal ganglion, and infraorbital nerve as compared to the control group. Previous studies on Nav1.7 gene knockout have indicated that its expression may be crucial in both somatosensory and sympathetic neurons in the context of neuropathic pain.54,55 The presence of Nav1.7 channels at nociceptive terminals, where generator potentials occur in response to nerve ending stimulation, suggests that Nav1.7 may amplify generator potentials, acting as a threshold channel that sets gain in nociception. Additionally, the biophysical properties of Nav1.7 may contribute to the initiation of action potentials in response to depolarization of sensory neurons caused by noxious stimuli.56,57
Overall, these results suggest that Na channels may play a role in the pathogenesis of trigeminal neuralgia. It has been hypothesized that mutations in this gene may result in a reduction of the thresholds and an increase in the frequency of action potential firing in sensory neurons. It is postulated that other gain-of-function Nav1.7 mutations that cause defective fast inactivation may be associated with burning pain, a symptom of paroxysmal extreme pain disorder, which is typified by pain precipitated by mechanical stimulation. A functional mutation in the SCN9A gene, which encodes Nav1.7, may potentially contribute to extreme paroxysmal pain disorders and affect the mandibular region. However, the role of Nav1.7 in neuropathic pain remains uncertain, and study findings are inconsistent.58–60
Role of K channels
In this study, we recorded that the expression of KCNK1/K2P1.1 was higher in the cervical spinal dorsal horn, trigeminal ganglion, and infraorbital nerve of experimental animals compared to controls. KCNK1 has already been reported to be associated with trigeminal neuralgia.61–65
Potassium channels are thought to regulate the excitability of spinal neurons, potentially playing a role in pain hypersensitivity. KCNK1 belongs to the two-pore domain potassium K2P1.1 channel family and is believed to be the first identified member. It is particularly abundant in the peripheral and central nervous systems, including the DRG, trigeminal nerve, and spinal cord. 65
Role of Ca channels
While the present study observed an increase in CACNA1A (Cav2.1 P/Q-type) expression in the spinal dorsal horn, a decrease in the trigeminal ganglion, however, statistically significant increase was recorded in the infraorbital nerve, in comparison to the control group., it is crucial to highlight recent research investigating the potential involvement of calcium channel gene mutations in patients with trigeminal neuralgia.66–70 Specifically, mutations in the CACNA1A may modify the gating properties of calcium channels, indicating that alterations in Cav2.1-dependent synaptic communication within the trigeminal system might play a role in the pathogenesis of trigeminal neuralgia. 68
CACNA1A plays a role in regulating trigeminal synaptic transmission by influencing neurotransmitter release. A gain-of-function mutation in this gene has been linked to trigeminal neuralgia in one case, 23 while Di Stefano et al. 8 previously identified a variant of CACNA1A in a patient with trigeminal neuralgia.
Our study illustrated that the CACNA1C is upregulated in the cervical spinal dorsal horn, trigeminal ganglion and infraorbital nerve, compare to controls. Similarly, one prior study recorded that the expression level of the ion channels CACNA1C was considerably increased in the trigeminal ganglion and trigeminal spinal nucleus in the TNP model developed by chronic constriction injury. 27
Summary of discussion
In light of these findings, it is reasonable to hypothesize that the mRNA expression of SCN8A and SCN9A, which encode sodium channels (Nav1.6 and Nav1.7), particularly in the infraorbital nerve, plays a primary role in the TNP model that has been developed. It further indicates that it may have an effect on calcium ion channels, especially CACNA1C and KCNK1 gene which encoded CaV1.2 and Kv1.1. It would thus be beneficial to explore further the potential contribution of pathophysiological changes in ion channels to neuropathic pain, as current analgesics have been found to cause numerous side effects and may not be fully effective in treating TNP.
The limitations of this study include the short duration of the experiment and the relatively small sample, which may have influenced the results and could have potentially impaired the generalizability of the study's findings. Additionally, the absence of a sample size calculation indicates a potential limitation in the statistical significance of the results, given the limited number of samples. Finally, it should be noted that the protein expression levels of these genes were not evaluated. In subsequent studies, the examination of the protein expression levels of these genes will serve to strengthen the study's results.
Conclusion
In this study, we demonstrate that compression of the cervical spinal dorsal horn alters the gene expression of ion channels, in addition to the neuroinflammation induced by astrocyte and microglia activation, as previously demonstrated in our study. The upregulation of SCN8A and SCN9A in the infraorbital nerve in comparison with the sham group indicates that Nav1.6 and Nav1.7, which are encoded by these genes, may play a significant role in TNP. Further research in this area will provide additional insights and further clarify the subject.
Footnotes
Acknowledgements
The author would like to thank their colleagues from their university who provided insight and expertise that greatly assisted the research. They would also like to thank editing service for assistance with proofreading reading and language editing.
ORCID iDs
Ethical considerations
Ethical approval was obtained from Afyon Kocatepe University Local Ethics Committee for Animal Experiments (27/02/2023, ref 18/49533702).
Authors’ contributions
ZKS, ÜTB was involved in writing—review & editing, writing—original draft, visualization, supervision, project administration, methodology, investigation, formal analysis, and conceptualization; ME in writing—review & editing, writing—original draft, visualization, project administration, investigation, formal analysis, and data curation; CB in writing—review & editing, writing—original draft, supervision, investigation, formal analysis, and data curation; FGO and HA in writing—review & editing, writing—original draft, visualization, project administration, methodology, investigation, and data curation; and YK and Fİ in writing—review & editing, writing—original draft, supervision, project administration, investigation, formal analysis, and conceptualization.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author CB.
