Abstract
The cost-effective, eco-friendly, and easily available adsorbent prepared using atmospheric pressure plasma (APP)-induced epichlorohydrin-grafting onto alkali-treated pineapple leaf fiber (APF) and immobilization with polyethyleneimine (PEI) was investigated and applied for p-nitrophenol (PNP) removal. The results from main characterization methods such as morphological observation, Fourier transform infrared analysis, and x-ray photoelectron spectroscopy revealed the surface changes and confirmed the success of the surface modifications. According to the experimental data, APP pre-treated adsorbent following by PEI immobilization (t-APF-e-PEI) exhibited fast and efficient adsorption with the adsorption capacity up to 128 mg/g, whereas the adsorption capacities for APF-e-PEI and APF were 115 and 21 mg/g, respectively, at 30 °C. The adsorption process was exothermic whereas the adsorption behavior conformed to the pseudo-first-order kinetics, intra-particle diffusion, and Langmuir isotherm models. Electrostatic ion–dipole interaction between protonated amines of PEI and negative-charged sites of PNP was proposed as the main mechanism. The prepared adsorbents exhibited good adsorption efficiency with possessing high adsorption capacity for repeated uses. The modified adsorbent provided from agricultural waste had a great potential as low cost, eco-friendly, and easily available adsorbent material to adsorb PNP from aqueous solutions.
Introduction
p-Nitrophenol (PNP) is a phenolic compound that has a nitro group at the opposite position of the hydroxyl group on benzene ring. This phenolic compound tends to remain in the environment for long time and its accumulation has a toxic impact on both animals and humans. 1 The contamination in water resource mainly arises from the discharging of PNP from various industrial, municipal and agriculture activities. In terms of toxicity, PNP irritates the eyes, skin, and respiratory tract resulting in inflammation. Prolonged contact with skin may cause allergic response and it causes abdominal pain and vomiting when ingested. Moreover, PNP is responsible for methemoglobinemia, potentially causing cyanosis, confusion, and unconsciousness. 2 So far, there are a number of methods used to minimize PNP pollution from wastewater, such as chemical oxidation, 3 biodegradation, 4 catalytic reduction, 5 liquid membrane separation, 6 and adsorption.7,8 Among these removal techniques, adsorption is considered to be the most convenient due to its operating flexibility, design simplicity and cost-effectiveness. Many kinds of materials with different removal efficiencies have been employed as the adsorbents.1,7–11 However, the high cost of some effective adsorbents limited this method's large-scale application. Thus, the search for and development of low cost and efficient adsorbents still receive much attention.
Natural cellulose fibers obtained from agricultural waste materials are one of the interesting bio-materials widely used for wastewater treatments due to their attractive advantages such as biodegradability, low cost, eco-friendly, regenerable, and ease of availability.12–15 Pineapple has become one of the most produced tropical fruits across the globe with global production of ∼28.6 million tons in 2021. 16 With such extensive cultivation, enormous amount of wastes is left on the ground or even burned after pineapple harvest. Moreover, a significant amount of carbon dioxide (CO2) released from burning of organic compounds certainly contributes to atmospheric pollution and climate change. 17 This is a well-recognized issue that necessitates a better approach. It is, therefore, necessary to process such wastes to produce value-added products such as green reinforcing material for polymer composites, raw fiber material for textiles production, and potential adsorbents for removal of pollutants in wastewater.18–20 It has been known that pineapple leaf fiber (PF) has superior mechanical properties due to its high cellulose content (70–80%) and relatively low microfibrillar angle.21,22 In fact, the cellulose-based adsorbent fibers can be alternative choice for the existing synthetic adsorbents and have high potential for industrial applications due to its flexibly of modification and convenience of utilization and separation, compared with the nanosized materials. However, although natural fiber contains hydroxyl groups, which can mainly act as binding sites for pollutants facilitating the adsorption of a wide range of contaminants, the relatively low efficiency of its adsorption is frequently encountered. Various chemical modifications using active functional groups or monomers by attaching to the respective hydroxyl groups or cellulose backbone for PF have been attempted.23–26 For instance, a number of modification methods using succinic anhydride/pyridine and methyl sulfoxide, 23 ethylenediaminetetraacetic acid and carboxymethyl groups, 24 aminosilane, 25 AcOH/H2O2, 27 polyacrylic acid-grafting, 28 alkali treatment, 29 cationic surfactant (hexadecyltrimethylammonium bromide, HDTMA-Br), 30 and diethylenetriamine 31 has been employed for PF in order to improve its adsorption performance. However, multi-step modification, toxic chemical consumption, physical stability of the modified adsorbent, weak cellulose-attached functional groups binding, and relatively low adsorption capacity are the main issues needed to be considered and developed.
Polyethyleneimine (PEI) is one of the interesting modifying agents for surface substrate due to it possesses intrinsic amine-rich structure, enabling to effectively chelate for various adsorbates. PEI has been used for fiber surface modification using the simple coating method but the weak binding with adsorbent substrate tends to encounter, resulting in the de-bonding of PEI. 32 This change leads to the decrease of adsorption performance of the adsorbents. A number of attempts has been done to immobilize PEI on hydroxyl groups of cellulose using various reactions 33 including carbamate linkages formation.34,35 Although efficient adsorption was gained from using PEI immobilization, the PEI-carbamate linkages were proceeded under multi-steps and using organic solvent such as dichloromethane including several other relatively high-cost chemicals. Therefore, the simpler, relatively more cost-effective, eco-friendly, and flexible methods for surface modification have been interested as the novel alternative routes. Epichlorohydrin (ECH) is one of interesting crosslinking agents with relatively low cost, easily soluble in ethanol, and easy to react. The crosslinking is achieved by releasing of chlorine atom and rupturing of the epoxide ring under alkaline condition, and the covalent bindings with PEI are formed at carbon atoms of ECH.36,37 Reducing types of chemicals, avoiding organic solvent use, relatively lower cost of chemicals, and ease of process are profitable when compared to carbamate-linked process. 34
Atmospheric pressure plasma (APP) is one of the eco-friendly methods, frequently employed to generate the active surface and/or induce grafting of active groups on the adsorbent surface. 38 “Plasma” is a combination of several particles like atoms, free electrons, ions, radicals, and molecules. The chemical and physical alteration on the surface of material is established by the connection of these plasma species with the solid surface. The adsorbents when treated with plasma do not alter the bulk characteristics, it only modifies the surface properties of the material. 39 APP is an easy-to-use and economical surface treatment technique consisting of four main categories, namely, dielectric barrier discharge, corona, atmospheric pressure glow discharge, and atmospheric pressure plasma jet (APPJ). 40 Especially, APPJ can generate plasma with a high concentration of free electrons and directly attacks the substrate through an opening in the quartz tube, yielding the improvements in surface roughness, bonding strength thermal insulation, etc.39–41 In addition, the plasma treatment methods can achieve the purpose of selectively grafting and crosslinking monomers with different functional groups.42,43 So far, surface modification of adsorbent using PEI for PNP removal has been rarely reported. To the author's knowledges, the PEI/SiO2 composite has been prepared as the adsorbent for PNP removal. 44 Therefore, APP-assisted ECH-grafting and PEI immobilization onto PF surface as the novel and effective method is of interest to use for modification of the adsorbents.
In this work, APP treatment on pineapple fiber surface to enhance ECH-grafting ability and PEI immobilization was investigated. The performance of PNP adsorption for the modified adsorbents in comparison with the unmodified one was explored. The adsorption kinetics and isotherms for each adsorption system were elucidated and compared.
Methodology
Materials and chemicals
The PF was prepared using milling method. The fiber preparation process was previously reported elsewhere. 35 The separated PF has 6 mm long, diameter range of 3–80 μm with cellulose, hemicellulose, and lignin contents of 70.98, 19.80, and 1.98%, respectively. Analytical grade chemicals used in this work consisted of sodium hydroxide (NaOH, CAS number: 1310-73-2, Carlo Erba Reagents), ECH (CAS number: 106-89-8, Sigma-Aldrich, ≥99%), hydrochloric acid (HCl, CAS number: 7647-01-0, Quality Reagent Chemical), glacial acetic acid (CAS number: 64-19-7, QRëC™, Quality Reagent Chemical), branched PEI (CAS number: 9002-98-6, Acros Organics, 50 wt% aqueous solution, Mn∼60,000, Italmar Co, Ltd), ethanol (EtOH, CAS number: 64-17-5, Merck), and PNP (CAS number: 100-02-07, Sigma-Aldrich).
Preparation of adsorbents
Fiber surface cleaning
Prior to surface modification, the PF was surface cleaned by soaking in alkali solution (5% NaOH). The mixture was stirred in a thermostatic shaker (Thermo Fisher Scientific, Newington, USA) at 150 rpm for 30 min and soaked for 6 h at 30 °C. The PF was withdrawn and neutralized by washing with distilled water. The fiber was then air and vacuum (80 °C) dried for 24 and 12 h, respectively. The sample code for the alkali-cleaned PF was abbreviated as APF.
Atmospheric pressure plasma treatment
APPJ made by the laboratory of plasma research unit (department of physics, Mahasarakham university, Thailand) was employed to modify the surface of APF. It is a proprietary designed with a movable single jet head operating at a resonant frequency of 80 kHz. Pure argon with a constant flow rate of 4 slm was used as the working gas to generate the plasma. The processing power was supplied at 40 W for a treatment time of 60 s. The distance between the plasma source and the samples surface was 5 mm. After treatment, the samples were kept in the zipper storage bag for further use. The plasma-treated APF gained in this step is designated as t-APF.
Grafting of epichlorohydrin and modification with polyethyleneimine
APF or t-APF of ∼1.00 g, 15 mL of ECH, 6 g of NaOH, 10 mL of EtOH, and 100 mL of distilled water were mixed in a 250 mL Erlenmeyer flask. The mixture was shaken in a thermostatic shaker for 5 h at 50 °C. After that, the fiber was withdrawn, put into 100 mL of distilled water containing of 1.5 g of PEI, and shaken for 4 h at 30 °C. Next, the modified adsorbent was withdrawn, neutralized by distilled water, and dried in an oven for 5 h at 60 °C. The fibers grafted with ECH and functionalized with PEI are designated as APF-e-PEI or t-APF-e-PEI, respectively. A proposed reaction mechanism for surface modification is shown in Figure 1. The chemical reaction proposed here was drawn based on cellulose component as a represent reactant which possesses highest content ratio. Moreover, the hydroxyl groups at C2 in glucose unit were additionally proposed as represent active sites for t-APF adsorbent.

Proposed reaction for atmospheric plasma-assisted PEI immobilization on cellulose fiber using ECH crosslinker. PEI: polyethyleneimine.
Characterization of adsorbents
The morphology of APF and t-APF-e-PEI before and after PNP adsorption was observed using a digital camera (Canon, EOS-500D, Tokyo, Japan) and scanning electron microscopy (SEM, SNE-4500M, Seoul, South Korea). The thermal stability of the adsorbents was analyzed by thermogravimetric (TG) analysis (SDT Q600, Luken's drive, New Castle, DE). The TG test was carried out under non-isothermal heating from ambient temperature to 600 °C at a heating rate of 10 °C/min in nitrogen atmosphere. Attenuated total reflection-Fourier transform infrared (ATR-FTIR) spectroscopy (Spectrum GX-1, Perkin Elmer Co, Ltd, UK), powder X-ray diffraction (XRD) Bruker D8 Advance diffractometer equipped with a Cu anode (Cu Kα source of the wavelength of 1.5406 Å) between 5 and 60° (2θ) and x-ray photoelectron spectroscopy (XPS; AXIS ULTRADLD, Kratos analytical, Manchester, UK) were employed for characterization of the surface changes of the adsorbents.
Batch adsorption experiments
PNP solutions with various concentrations were prepared by dissolving PNP powder in deionized water. The batch adsorption studies were conducted in 25 mL of PNP solution with ∼ 1.0 mg of adsorbents at various pH from pH = 3 to pH = 10. The batch mixture was shaken at 80 rpm using a thermostatic shaker at the selected temperature (30, 45, and 60 °C). After reaching the selected contact time, the adsorbent was rapidly filtered off. The absorbance of the residual PNP solution was examined using UV-spectrophotometry (Perkin Elmer, UK) at a maximum wavelength (λmax) of 318 nm. The content of PNP adsorbed on the adsorbents in the unit of mg/g can be calculated by using the following equations:
Regeneration test
The PNP-loaded fiber of 5.0 mg was eluted using 50 mL of 0.1 M NaOH. The mixture was agitated in a thermostatic shaker bath at 80 rpm for 2 h at 30 °C. The desorbed material was withdrawn, washed with distilled water, and then oven-dried at 60 °C for 12 h. The desorbed adsorbent was regenerated five cycles under the same adsorption condition.
Results and discussion
Morphology
The optical and SEM images of the adsorbents are shown in Figure 2. The surface characteristics of APF and t-APF (Figure 2I-(a) and (b)), observed from optical images are not clearly different. The surface of t-APF is notably roughened as seen from SEM image (Figure 2II-(b)), mainly arising from striking of plasma species on the substrate surface. However, after grafting with ECH and modifying with PEI, t-APF-e-PEI exhibits more surface roughness (Figure 2II-(d)) when compared with APF-e-PEI (Figure 2II-(c)). The change in surface roughness may arise from the surface etching contributed from the pre-treatment by plasma. From optical images, the adsorbents before PNP adsorption (APF and t-APF) show a slight and soft shade of the yellow color whereas the respective increase in yellow color intensity is observed for AF/PNP and t-AF/PNP. Interestingly for both APF-e-PEI/PNP and t-APF-e-PEI/PNP, very high intense color is observed (Figure 2I-(g) and (h)) indicating high PNP adsorption performance of the adsorbents. However, surface roughness of the latter two samples, revealed from SEM images, is not clearly different.

Optical (column I) and SEM images (column II) of APF (a), t-APF (b), APF-e-PEI (c), t-APF-e-PEI (d), AF/PNP (e), t-AF/PNP (f), APF-e-PEI/PNP (g) and t-APF-e-PEI/PNP (h). APF: alkali-treated pineapple leaf fiber; PEI: polyethyleneimine; PNP: p-nitrophenol; SEM: scanning electron microscopy.
Fourier transform infrared spectra
The FTIR spectra for the adsorbents before and after PNP adsorption are compared in Figure 3(A) and (B), respectively. Although characteristic peaks of cellulose fiber for the whole range of wavenumber (400–4000 cm−1) is not shown here, the peaks at ∼3332 and ∼2902 cm−1 corresponding to the respective O-H stretching of hydroxyl group and C-H stretching are observed for all samples. Generally, plasma treatment results in dramatic changes not only in surface morphology but also in structure of substrates. Especially, after APP treatment, nitrogen or oxygen groups were formed immediately when modified sample comes in touch with air. However, in some cases, these changes cannot be readily distinguished by direct comparison from individual FTIR spectrum due to weak signals of functional groups. In order to clearly distinguish the difference of FTIR spectra, the spectra of t-APF subtracted by that of APF is carried out and additionally presented in Figure 3(A). It is seen that formation of amide (–CONH–) and nitrosyl (–NO) from aerial nitrogen could be observed at ∼1645 and ∼1540 cm−1, respectively. 45 Moreover, stretching of C=O and C–O is also observed at ∼1730cm−1 and ∼1166 cm−1, respectively. The FTIR spectra compared among all PNP-adsorbed samples and PNP are also shown in Figure 3(B). APF/PNP, APF-e-PEI/PNP, and t-APF-e-PEI/PNP show the peak at 1568 cm−1 corresponding to C-N bending of PNP. Especially for t-APF-e-PEI/PNP, the C-N band is clearly observed, indicating that surface modification using APP-assisted ECH-grafting along with PEI immobilization further improves the adsorption of PNP onto adsorbent surface. One may partly arise from the generation of active free radicals on glucose units causing from the APP-assisted treatment, such as the free radical generated at hydroxyl groups of C2 and/or C3, etc. Therefore, the APP employed in this study is not only efficient in improved surface roughness but also introducing chemical etching which generates the oxygen-containing groups or other reactive groups to promote the modification efficiency, chemical etching nature of oxygen. This issue will be extensively discussed in the section of XPS results.
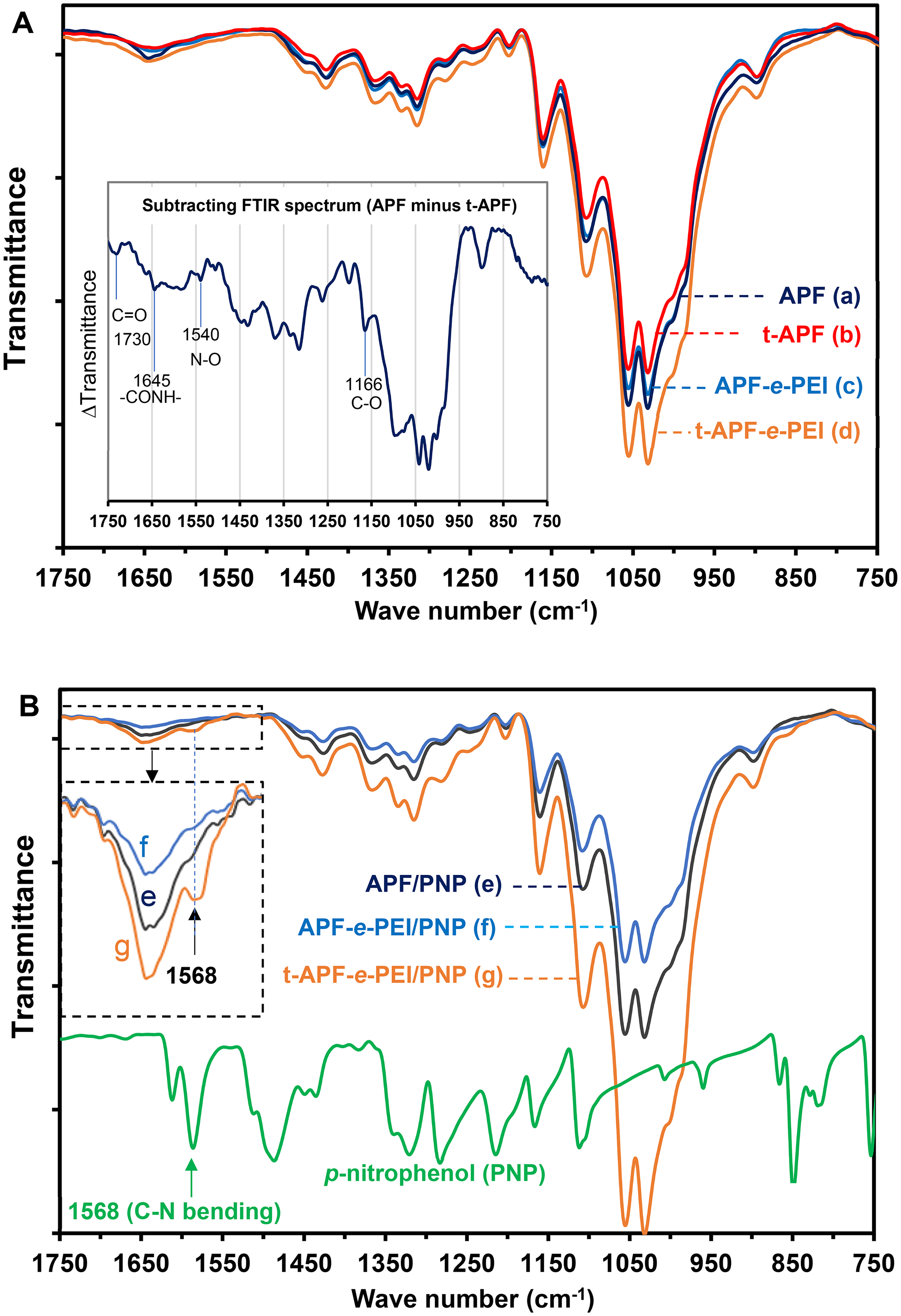
FTIR spectra of the adsorbents before adsorption (A) for APF (a), t-APF (b), APF-e-PEI (c), t-APF-e-PEI (d) and after adsorption (B) for APF/PNP (e), APF-e-PEI/PNP (f) and t-APF-e-PEI/PNP (g) (The subtracting FTIR spectrum = Transmittance of APF minus that of t-APF). APF: alkali-treated pineapple leaf fiber; PEI: polyethyleneimine; PNP: p-nitrophenol; FTIR: Fourier transform infrared.
Thermal stability
To assess the thermal stability of the modified adsorbents in comparison with the unmodified one, TG method was conducted and the TG curves are presented in Figure 4(a). The major stage of weight loss all adsorbents mostly appears at the heating temperature higher than 300 °C and corresponds to the degradation of hemicellulose (220–315 °C), cellulose (∼370 °C), and lignin (280–500 °C). 46 APF and t-APF thermally degrade at nearly the same onset-temperature (∼330 °C), whereas APF-e-PEI and APF-e-PEI show the onset-temperature of about 310 and 300 °C, respectively. The slight decrease in thermal stability of PEI-immobilized samples compared to APF and t-APF may be due to the increase of possible reactive sites of lower thermally stable PEI. 34 However, the thermal stability of all adsorbents is nearly the same in the temperature range lower than 250 °C which is profitable for further application of the modified absorbents with maintaining thermal stability as good as the unmodified one. Note that, very low content of char residue for t-APF-e-PEI is remained at temperature higher than 440 °C. This may partly arise from APP treatment and PEI immobilization-induced reactive sites, resulting in lowering the thermal stability of the adsorbent.

TG curves (a) and XRD patterns (b) for PNP and adsorbents. PNP: p-nitrophenol; XRD: X-ray diffraction.
X-ray diffraction patterns
In this work, the XRD investigation was used for evaluation of the change in amorphous and crystalline characteristics of the adsorbents before and after adsorption. The XRD patterns for all adsorbents in comparison with PNP are shown in Figure 4(b). XRD patterns of all sample show reflections mainly at 2θ of ∼16.2°, ∼23.0°, and ∼34.8°, respectively, represent the (100), (200), and (004), crystallographic planes of a typical cellulose I structure. 47 APP-assisted modification with PEI immobilization seems to has little effect on the crystalline properties of the adsorbents. Similarly, the crystalline characteristics of the PNP-adsorbed materials are not clearly different from that of the un-adsorbed ones. This indicates that the washing (mercerization), modifications, and adsorption processes strongly affect the amorphous regions of cellulose, resulting in less effect on crystalline regions.48,49
X-ray photoelectron spectroscopy analysis
The XPS method is for analysis of the element constituting the sample surface, its composition, and chemical bonding state which is useful for monitoring the surface changes of the adsorbents. The survey XPS spectra and its essential data such as element concentration and the peak position of binding energies (BE) for all adsorbents are presented in Figure 5 and Table 1, respectively. C1s and O1s peaks of APF are found at 285 (68.71% C) and 532 eV (30.33% O), respectively. Even the peak is not clearly observed in XPS spectra, the low signals of Al 2p and S 2p with very concentration are detected in several samples as presented in Table 1. This may arise from the silicones-containing vacuum greases used as vacuum seals. The silicones or other impurities may be turned out from vacuum seals that were introduced in the XPS chamber. It is seen that, in comparison with APF, the concentration of O 1s and C 1s for t-APF increases and decreases, respectively. For the same meaning, O/C atomic ratio is increased for t-APF which mainly contributes from oxidation reaction during APP treatment. Note that, for PEI-modified adsorbents (APF-e-PEI and t-APF-e-PEI), the peak of N 1s is additionally observed at 399 eV (Figure 5(c) and (d)), confirming the successful PEI immobilization onto the fiber surface. In case of APF/PNP, very small peak of N 1s with very low atomic concentration (0.95%) is observed at the same BE position (399 eV) assigning to N in nitro-groups of PNP. Very low signal of N 1s for PNP is probably related to the low adsorption performance of the unmodified adsorbent. However, the N 1s peak is clearly observed for PNP-loaded modified materials (APF-e-PEI/PNP and t-APF-e-PEI/PNP) similar to those of APF-e-PEI and t-APF-e-PEI.

Survey XPS spectra of APF (a), t-APF (b), APF-e-PEI (c), t-APF-e-PEI (d), APF/PNP (e), APF-e-PEI/PNP (f) and t-APF-e-PEI/PNP (g). APF: alkali-treated pineapple leaf fiber; PEI: polyethyleneimine; PNP: p-nitrophenol; XPS; x-ray photoelectron spectroscopy.
XPS data of the adsorbents.

The low signals of Al 2p and S 2p revealed in several samples may arise from the impurities turned out from vacuum seals in the XPS chamber.
APF: alkali-treated pineapple leaf fiber; PEI: polyethyleneimine; PNP: p-nitrophenol; XPS: x-ray photoelectron spectroscopy; XPS: x-ray photoelectron spectroscopy.
To discuss in more deep details, high-resolution XPS spectra for C 1s, N 1s, and O 1s of all adsorbents are depicted in Figure 6. Four main characteristic peaks of C 1s at 285 (C-C), 286.6 (C-O, C-H), 288.2 (O-C-O, C=O), and 289.3 eV (O-C=O) are observed for APF (Figure 6I-III(a)), whereas O 1s spectra reveal main three types of oxygen bonds at around 531.8, 533.0, and 534.4 eV corresponding to O-H, C-O, and C=O, respectively.34,35 The main peaks of C 1s and O 1s revealed for APF are mostly similar to those of APP-treated sample (t-APF). Interestingly, the BE peak of O 1s for t-APF is additionally observed at 535.3 eV (Figure 6I-(b)) assigning to free H2O molecule and the additional C 1s peak of carbonate anion [CO3]2− is also observed at 290.2 eV (Figure 6III-(b)). 50 Generally, the release or deposition of molecules or chemical species during plasma treatment mainly arises from a number of phenomena, such as oxidation, dehydrogenation, chain scission, ring opening, and reorientation which is mostly dependent on the structures of substrate and the treatment conditions. From the XPS data reported in Table 1, the respective decrease and increase of atomic concentration for C 1s and O 1s of t-APF adsorbent mainly contributes from the occurrence of C–O, C–OH and O=C–O bonds. For instance, the additional peak of O-C=O of O 1s, revealed for t-APF at 535.3 eV (Figure 6I-(b)), agrees well with the plasma-treated cellulose reported by a number of research groups.45,51 This means that, during plasma treatment, despite the occurrence of the polar groups, the cleavage of ether linkage followed by oxidation also takes place which generates the -COOH (O-C=O) groups. Moreover, the APP treatment using argon gas under atmospheric condition is efficient in introducing oxygen-containing resulting from oxidation reaction. 52 These generated reactive O-containing groups are expected to additionally react with ECH and hence, enhancing the ECH-grafting dose onto the fiber prior to binding with PEI as the last step of modification. However, the active ions or charged species simultaneously produced during plasma treatment tends to possessing low stability and has short life time period.

High-resolution XPS spectra for O 1s (column I), N 1s (column II) and C 1s orbitals (column III) of APF (row a), t-APF (row b), APF-e-PEI (row c), t-APF-e-PEI (row d), APF/PNP (row e), APF-e-PEI/PNP (row f) and t-APF-e-PEI/PNP (row g). APF: alkali-treated pineapple leaf fiber; PEI: polyethyleneimine; PNP: p-nitrophenol; XPS: x-ray photoelectron spectroscopy.
The XPS signals of APF-e-PEI are shown in Figure 6I-(c), II-(c) and III-(c) for O 1s, N 1s, and C 1s, respectively. The main BE peaks for O 1s of PEI-immobilized adsorbents mostly shift to the lower energy probably due to the occurrence of linkages of PEI-active oxygen atoms of the PEI-modified adsorbents. The deconvoluted spectra for N 1s appeared at 399.3 (=N-), 400.6 (-NH2 or -NH), and 402.4 (N-O) eV are observed for both APF-e-PEI and t-APF-e-PEI (Figure 6II-(c) and II-(d)). In case of PNP-adsorbed samples, N 1s signals are found at ∼398–404 eV. For APF/PNP (Figure 6II-(e)), low intensity of N 1s signal at ∼400 eV contributed from PNP is found, relating to the low atomic concentration (Table 1) previously mentioned. For APF-e-PEI/PNP and t-APF-e-PEI /PNP (Figure 6(f) and (g)), the corresponding deconvoluted peaks of O 1s, N 1s, and C 1s are not clearly different. This is indicated that, after PEI is immobilized on the t-APF or APF using ECH crosslinker, the further PNP adsorption process for both adsorbents similarly occurs at the amine groups in PEI. This means that the PNP adsorption exhibits the same mechanism for APF-e-PEI/PNP and t-APF-e-PEI/PNP systems. Note that, the intensity ratios for -OH (at ∼531.8 eV) to C-O (at ∼532.8 eV) of APF-e-PEI/PNP and t-APF-e-PEI/PNP clearly decrease compared with that of APF/PNP. This means that most of -OH active sites for the modified adsorbents are used for binding with PEI and hence, less -OH sites are remained. As seen from Figure 6I-(f) and I-(g), the intensity ratio of -OH/C=O (intensity of -OH at ∼531.8 eV divided by intensity of C=O at ∼534.2 eV) is higher in APF-e-PEI/PNP than in t-APF-e-PEI/PNP. This change is similar to the case of APF compared with t-APF (Figure 6I-(a) and I-(b)). For APP-assisted treatment, one of the main chemical species generated on the fiber surface is C=O groups 45 whereas hydroxyl groups are mostly reacted with PEI according to the proposed mechanism illustrated in Figure 1. Therefore, the intensity ratio of -OH/C=O is reduced for the APP-treated samples.
Effect of pH
The effects of initial solution pH (from 2 to 10) on the adsorption capacity of the selected adsorbents, APF and t-APF-e-PEI were investigated and compared in Figure 7. The performance of adsorption progressively increases with increasing pH from 2 to 5 and gradually descends from pH 6 to 10. From the obtained results, the maximum adsorption capacity is observed at solution pH = 5. Under acidic condition, amine groups will be protonated to form positive charge sites such as -NH3+, resulting in intensified the electrostatic attraction between the adsorbent surface with a positive charge and the negative-charged PNP molecules.1,34 The protonation of amines decreases at higher pH while the concentration of hydroxyl ions increases. This change leads to the competition of hydroxy anions with negative-charged PNP and hence decrease in adsorption capacity. Thus, the PNP solution adjusted at pH = 5 was used as the optimum condition for adsorption studies throughout this work.
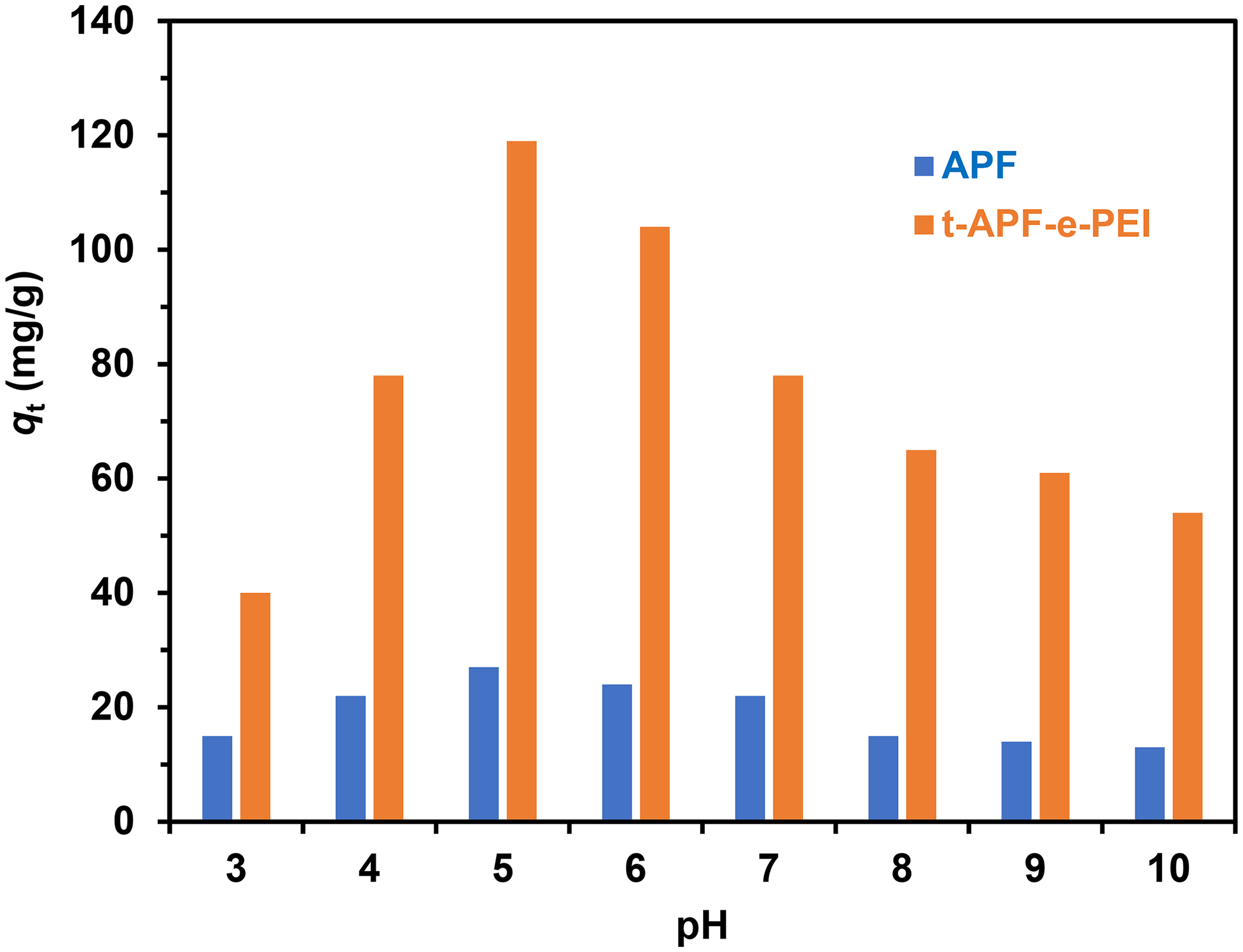
Effect of solution initial pH on equilibrium adsorption capacity of APF and t-APF-e-PEI adsorbents (C0 = 160 mg/L, contact time = 120 min, temperature = 30 °C). APF; alkali-treated pineapple leaf fiber; PEI: polyethyleneimine.
Effect of temperatures and contact times
The adsorption performance of PNP on the adsorbents at various temperatures and contact times are shown in Figure 8. PNP is rapidly adsorbed onto all adsorbents starting from 0 to 10 min and then the adsorption rate gradually decreases before approaching equilibrium state at contact time higher than 30 min. Increase in temperature lowers the adsorption capacity indicating exothermic process. Meanwhile, the rate of desorption was increased with an elevated temperature leading to an increase of diffusive mass transfer and solubility of PNP in water. Oppositely, the strength of adsorptive forces between adsorbent sites and adsorbate is decreased. Consequently, the accessibility and affinity between the adsorbent and PNP declined, resulting in removal less efficient.1,53 APF and t-APF exhibit relatively low adsorption performance (qe∼20–30 mg/g), whereas APF-e-PEI and t-APF-e-PEI show relatively higher adsorption performance with qe of ∼80–130 mg/g. The experimental adsorption capacity of t-APF-e-PEI is slightly higher than that of APF-e-PEI depending on adsorption temperature. As discussed in the previous section of XPS analysis, the APP-assisted treatment generates the oxygen-containing reactive groups on the fiber surface by oxidation process. The chemical species are in the forms of radicals, ions or active oxygen-containing functional groups which can elevate the ECH-grafting ability, resulting in subsequent enhancing the PEI immobilization. However, short life time for a number of active species after plasma treatment tends to limit the ECH-grafting and consequently lowering the ability of PEI immobilization.
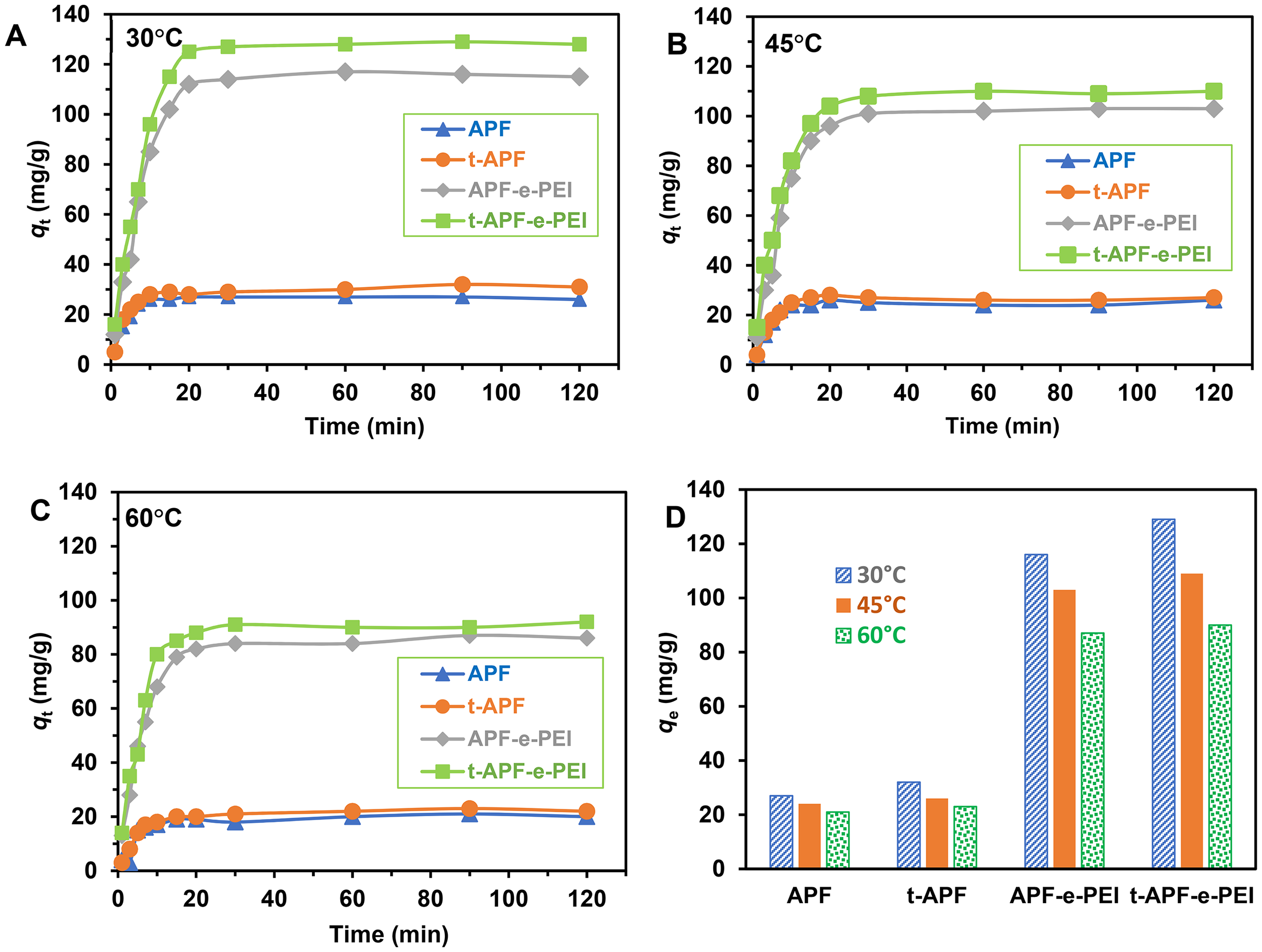
Adsorption capacity versus contact time for various adsorbents under adsorption conditions of C0 = 160 mg/L, pH = 5 at 30 (a), 45 (b) and 60 °C (c) and comparison of equilibrium adsorption capacity at various temperatures (d).
Figure 9(a) shows the overview images of the residual PNP solution and the adsorbed fibers. The obtained results confirm the important roles of both APP-assisted treatment and PEI immobilization to enhance the adsorption performance. In comparison with APF and t-APF systems, intense yellow color indicating high amount of PNP adsorbed on APF-e-PEI and t-APF-e-PEI is observed. The possible proposed between PEI-immobilized fiber and PNP schematically presented in Figure 9(b). Normally, the pristine cellulose possesses hydroxyl groups as the main reactive sites, leading to negative-charge nature. Thus, under acidic condition, the binding of the hydroxyl groups of the neat cellulose with PNP would mainly occur by electrostatic interaction. However, the adsorption efficiency is not as good as that for PEI-modified adsorbents due to lower reactive performance of hydroxyl groups compared with amine-rich structure. Under acidic condition, the amine groups would be protonated to form positive charge sites (-NH3+/−NH2+) and these positive sites can form an electrostatic attraction to negative sites (O−) of PNP. Moreover, the additional interactions may arise from hydrogen bonding between the adsorbent-free hydrogen and oxygen within PNP structure and π-π interaction between PNP aromatic rings. 54 Note that, the protonation generally decreases with increasing pH, leading to the increase of hydroxyl ions. This condition results in the competition of hydroxyl ions and the negative-charged sites of PNP molecules, leading to reducing the adsorption performance. Moreover, physical adsorption in the porous surface of the adsorbent is also proposed during adsorption.
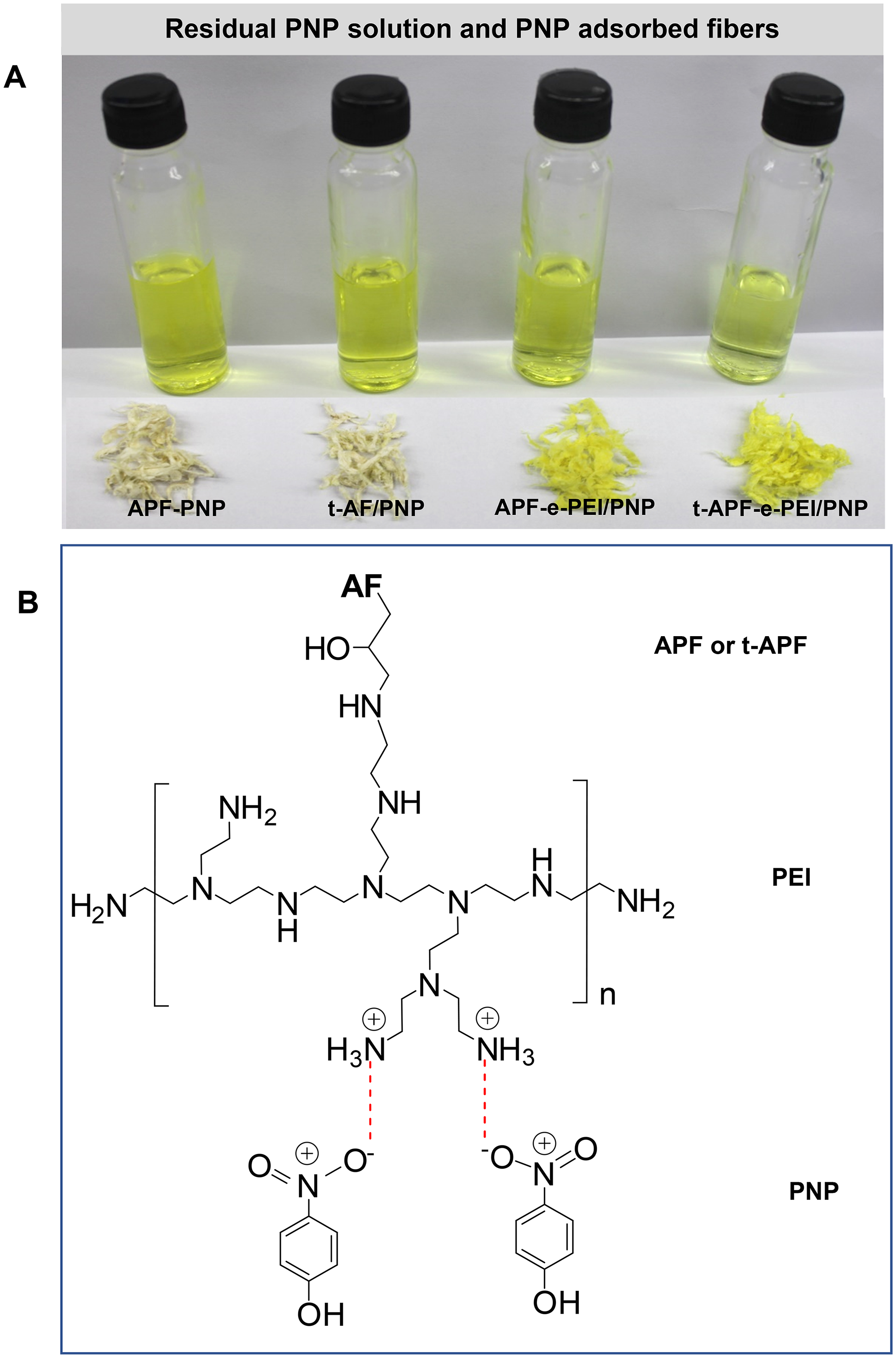
Overview small-scale images of residual PNP solution and the adsorbents after adsorption (a) and proposed main mechanism of PNP adsorption (b) under adsorption conditions of C0 = 160 mg/L, pH = 5, contact time (t) = 120 min and temperature = 30 °C. PNP: p-nitrophenol.
Effect of initial PNP concentration
The effect of initial PNP concentrations (C0) on adsorption capacity of PNP on APF-e-PEI and t-APF-e-PEI is comparatively shown in Figure 10. The corresponding optical images for the adsorbed fibers at different initial concentrations of PNP are also are also shown here. With increasing initial PNP concentrations, the adsorption performance of the adsorbent progressively increases, indicating the remaining of unoccupied active sites on the adsorbent surface. One explanation for this is that high concentration supplies a driving force to migrate of PNP molecules towards active adsorption sites on the adsorbent surface. 55 Based on the results obtained, the equilibrium adsorption can be approached at PNP concentration higher than 200 mg/L. Moreover, high intensity of yellow color causing from high amount PNP adsorbed on the fibers is observed at high C0 which confirms the adsorbate concentration dependence of adsorption performance.
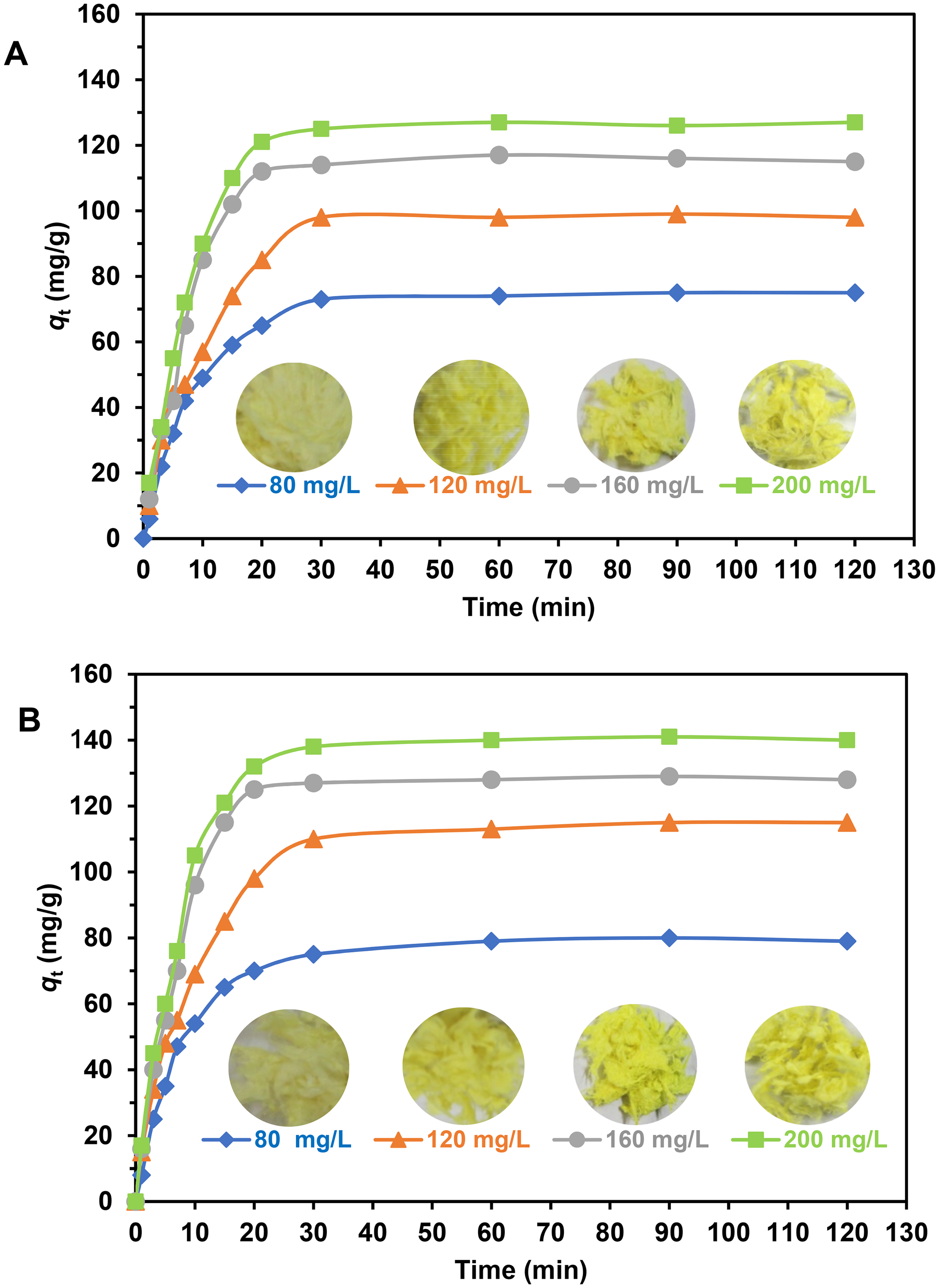
Effect of initial PNP concentration on adsorption capacity of APF-e-PEI (a) and t-APF-e-PEI (b) at 30 °C. APF: alkali-treated pineapple leaf fiber; PEI: polyethyleneimine; PNP: p-nitrophenol.
Adsorption kinetics
Adsorption kinetics before reaching the equilibrium state is one of essential issues to provide the kinetic parameters and the information adsorption mechanism. The kinetic parameters such as rate constant (k), reaction order (n) including the information of adsorption mechanism are obtained after investigation. Generally, the pseudo-first-order, pseudo-second-order, and intra-particle diffusion models as the simple methods were widely employed to describe the adsorption behavior. The three kinetic models can be expressed as follows
15
:
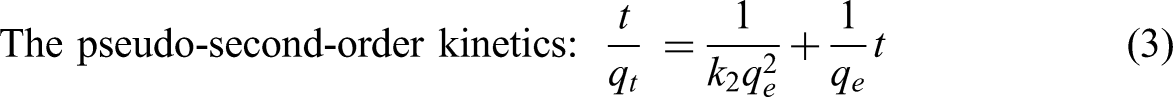
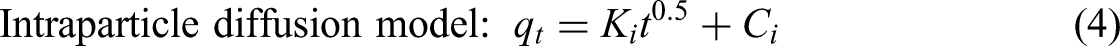

Kinetic and isotherm plots for pseudo-first-order (a), pseudo-second-order (b) and intra-particle adsorption (c), Langmuir (d) and Freundlich (e) models for the selected adsorption system at 30 °C.
Kinetic parameters for the adsorption of PNP onto various adsorbents at 30 °C.

alkali-treated pineapple leaf fiber; PEI: polyethyleneimine; PNP: p-nitrophenol.
Adsorption isotherms
Investigation of isotherms is an important topic for describing the interaction behavior of the adsorption process. In this work, Langmuir
58
and Freundlich
59
isotherms have been employed. Langmuir is a simple isotherm widely used to explain the adsorption behavior based on the basic assumptions of monolayer adsorption process and homogenous sites within the adsorbent. The linear relation forms of the Langmuir isotherm are generally expressed as following equation:
Freundlich isotherm, based on assuming a heterogeneous surface and a nonuniform distribution energy over the surface without a saturation of adsorption sites, is also studied. The linear equation of Freundlich isotherm can be expressed as follows: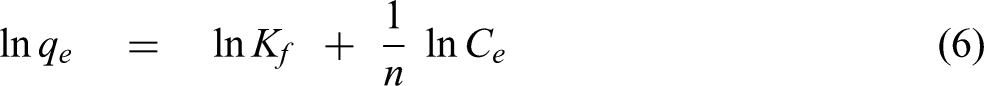
Isotherm data for APF-e-PEI and t-APF-e-PEI adsorption systems at 30 °C.

APF: alkali-treated pineapple leaf fiber; PEI: polyethyleneimine.
Reusability test
It is important that any adsorbents being utilized to offer a large adsorption capacity to be practically useful and effectiveness for various applications. Its reliable use is primarily attributed to its ability for regeneration that can notably decrease both the cost of adsorbent preparation and the solid waste accumulation. Regeneration of APF-e-PEI compared with t-APF-e-PEI adsorbents using batch experiments was investigated by repeating five cycles. NaOH solution was employed as an efficient eluent to assess the performance for each repeating cycle. The regeneration data for each adsorbent at each recycle is shown in Figure 12. Clearly, the adsorption capacity of both samples progressively decreases with increasing repeating time due to the reduction of active sites on the adsorbents arising from repeating elution and washing processes. Interestingly, the adsorption capacity of t-APF-e-PEI is higher than that of APF-e-PEI for the first regeneration cycle and nearly the same with increasing repeating time of reuse. However, the adsorbents still exhibit high performance of adsorption after repeating five times (qe > 60 mg/g).
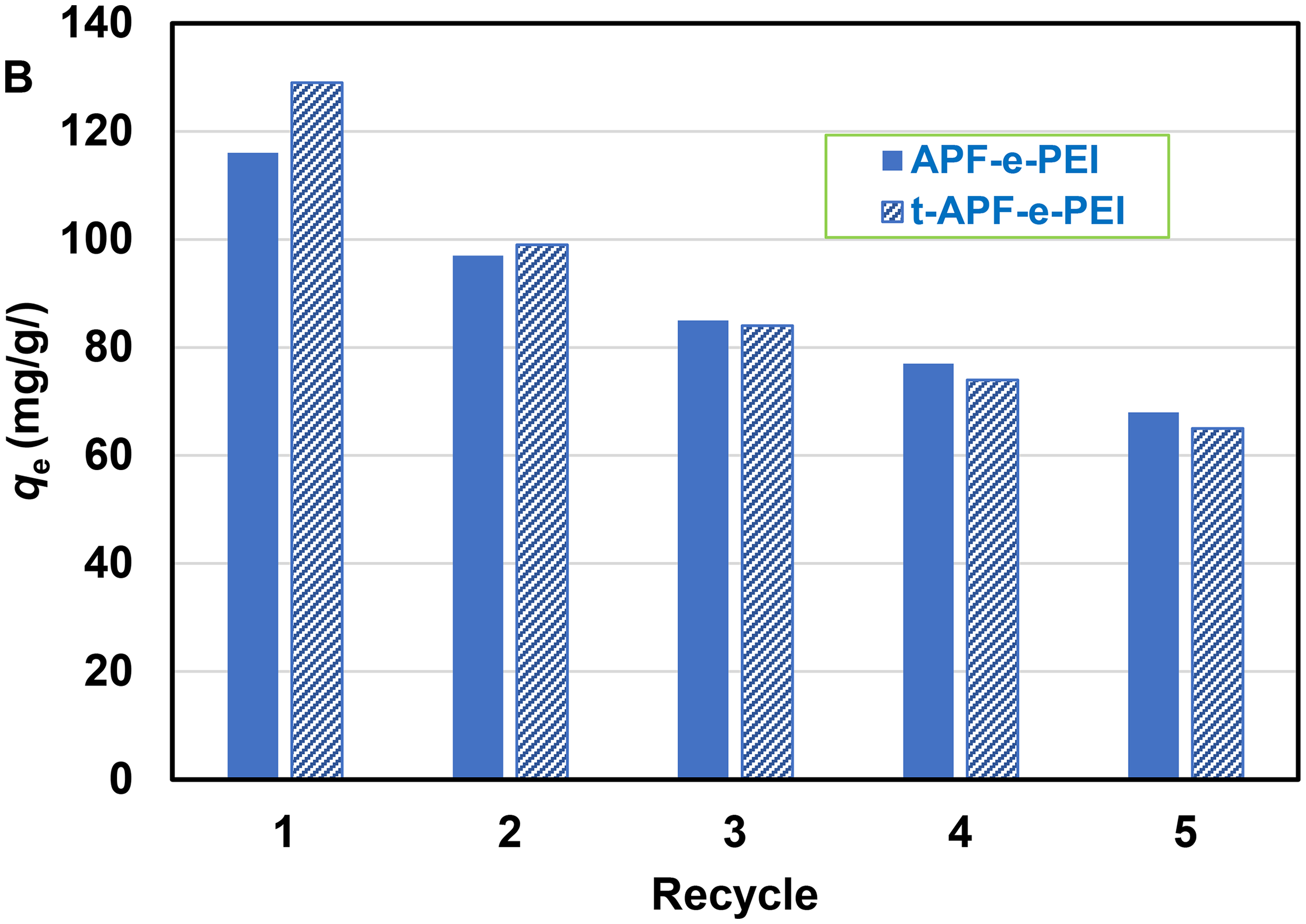
Reusability of APF-e-PEI and t-APF-e-PEI adsorbents. APF: alkali-treated pineapple leaf fiber; PEI: polyethyleneimine.
Comparison of performance and adsorption behavior
To explore the adsorption potential level in overview, comparison of the experimental adsorption capacity among other relevant adsorbents is presented in Table 4. It is found that, not only for the rapid adsorption process, good adsorption performance is gained form the bio-adsorbents prepared in the present work. Although the adsorption performance t-APF-e-PEI is better than those for various kinds of absorbents, its adsorption capacity is lower than those for a number of adsorbent-based polymer resins and nanomaterials, for example, graphene nanosheets, 8 chloromethyl polystyrene resin supported task-specific ionic liquid, 9 clay and clay-containing composites,10,11 and activated carbon/MOFs composite. 60 This mainly arises from a lower surface area of the microfiber, compared to the nanosized adsorbents. Interestingly, utilization of bio-adsorbents with relatively larger size, provided from agricultural wastes, is profitable not only in terms of cost-effectiveness and eco-friendly awareness but also comfortable preparation, utilization, and separation.
Comparison of PNP adsorption performance among various adsorbents.

APF: alkali-treated pineapple leaf fiber; PEI: polyethyleneimine; PNP: p-nitrophenol; ECH: epichlorohydrin.
Conclusion
The adsorbent based on PF was successfully prepared using APP-assisted ECH-grafting and modification with PEI (t-APF-e-PEI). The adsorbent was then applied for the PNP removal in comparison with APF-e-PEI and APF adsorbents. Characterization of adsorbents using various analytical methods such as SEM, FTIR, and XPS confirmed the surface changes after surface modification and adsorption. Kinetic and isotherm analyses showed that all adsorption systems fitted well on the pseudo-first-order model and Langmuir isotherm. According to the experimental data, APF-e-PEI and t-APF-e-PEI exhibited maximum adsorption capacity of 115 and 128 mg/g at 30 °C, respectively, and the adsorption performance decreased with increasing temperature (exothermic process). Regeneration of the adsorbents could be accomplished with high adsorption capacity of PNP (qe > 80 mg/g) after reuse three times. The APP-assisted ECH-grafting with PEI immobilization was the efficient method for surface modification and further improved adsorption performance of the natural fiber-based adsorbents. The results obtained have a high potential to practically apply for PNP removal from aqueous solutions.
Highlights
APP-assisted ECH-grafting and PEI immobilization were first investigated onto pineapple fiber. The modified fiber was applied as bio-adsorbent for PNP removal. The novel surface modification effectively resulted in further improved adsorption performance of the adsorbent.
Footnotes
Acknowledgements
This research project was financially supported by Thailand Science Research and Innovation (TSRI), Mahasarakham University (FF-2025; Project Code: 206291; Contract no. 6817054). The authors wish to express their profound gratitude and sincere appreciation to the Center of Excellence for Innovation in Chemistry (PERCH-CIC) and Ministry of Higher Education, Science, Research, and Innovation. Moreover, the authors also would like to thank Asst. Prof. Dr Artit Chingsungnoen, laboratory of plasma research unit, department of physics, Mahasarakham university, Thailand, for encouragement of APP surface treatment.
Ethical considerations
We confirm that this work is original and has not been published elsewhere nor is it currently under consideration for publication elsewhere.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by all authors. The first draft of the manuscript was written by SS and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was financially supported by Thailand Science Research and Innovation (TSRI), Mahasarakham University (FF-2025; Project Code: 206291; Contract no. 6817054).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The article contains the data that substantiates the results of this investigation.

