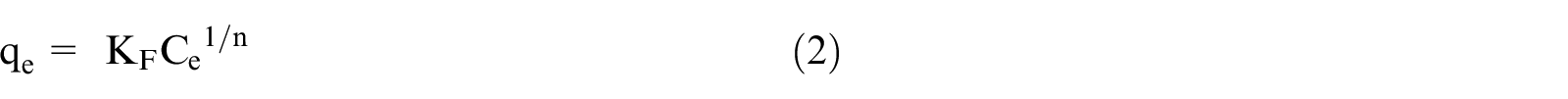Abstract
The chemical modifications of lignin-based superplasticizers have attracted extensive attentions during recent years. The comprehending of the structure-activity relationship of lignin-based superplasticizer is important to promote the modification and application research of lignin resources. However, lignin features complex and variable molecular structure, which is not conducive to study on structure-activity relationship of lignin-based superplasticizer as well as development and application of new lignin-based superplasticizer. However, the related research work can be simplified by selecting small molecular compound with appropriate molecular structure as the lignin model compound. This article intends to study the structure-activity relationship of lignin-based superplasticizer by using dihydroeugenol as the lignin model compound. Through the substitution of lignin by dihydroeugenol during the synthesis process, a model compound lignin-based superplasticizer (DAFS) was synthesized. The adsorption and dispersion properties of this superplasticizer and reference sample (LAFS) were investigated by fluidity test, Zeta-potential measurement, Total organic carbon analysis and others. The results suggest that the adsorption behavior of both DAFS and LAFS conformed to the Langmuir isotherms and Pseudo-second order kinetic. In cement paste, added with 1 g/L of LAFS and DAFS, Zeta potential were reduced from
Introduction
It is well-known that with the addition of water, the rapid dissolution of clinkers will transfer the cement paste to a thermodynamically unstable state, large amounts of different ions such as Ca2+, Si4+, Al3+, and SO42− will dissolve from the clinkers due to the initial high degree of undersaturation. The ions were then captured by the large surface of cement particles, resulted in different charged properties of particles, and this will further result in the flocculation of cement which is very harmful to the fluidity. In order to solve this problem, superplasticizer, usually anionic surfactant, was added to the system. It can directly interact with the positive ions on the surface of cement and change the charge state to disaggregate the agglomerated particles, therefore the cement particles were well dispersed and the hydration kinetics, the rheological and mechanical properties of cement paste were altered.1–2
As the main component of waste liquid in paper pulping, lignin has a complex and highly branched molecular structure which is favor for chemical modification.3–4 Some researchers have obtained lignin-based superplasticizer with good water-reducing and dispersing properties by the sulfonation of lignin.5–6 However, it is uneasy to characterize and analyze the structure-activity relationship of lignin-based superplasticizer due to the complicated molecular structure of lignin, and this problem will postpone the progress in modification research and the application of lignin resources. 7 Here, we try to use some small molecular chemical materials which have the similar chemical structures of lignin as the lignin model compound, and this can help to investigate the relationship between structural feature and activity in modified lignin-based superplasticizer.
In this paper, we selected dihydroeugenol as the model compound to replace lignin in the preparation of modified superplasticizer named DAFS. Moreover, a lignin-based superplasticizer named LAFS as a reference sample was also synthesized by the same procedure, the adsorption and dispersion mechanisms of those products were investigated from the total organic carbon analyzer and cement paste fluidity test. The main aim of this work is to verify that the usage of dihydroeugenol as a lignin model compound to synthesize lignin-based superplasticizer is reasonable and conductive to clarify the structure-activity relationship.
Materials and methods
Materials
The soluble lignin sodium phenolate was obtained from the straw pulp liquor (pH = 12.6) provided by Hubei Jianli Dafeng Paper Industry Co., Ltd, china. It was then converted to an insoluble state by using dilute sulfuric acid. 8 After that, this insoluble product was repeatedly washed with pure water and ground, a final powdery acid-precipitated lignin (LAP) was obtained by filtering and drying at a vacuum environment for 24h.
The structure of lignin basically contained three fragments: p-hydroxyphenyl (H), guaiacyl (G), and syringyl (S), 9 and Guaiacol is the smallest union that constitutes the guaiacyl structure fragment of lignin. The hydrogen atoms located at o-position or p-position in phenolic hydroxyl group of guaiacol are very active, so that the hydroxymethylation reaction can be easily happened on those position. Dihydroeugenol is the derivative of guaiacol, the hydrogen atoms at p-position inphenolic hydroxyl group are usually substituted by a propane group, as a result, the methylolation reaction can only take place at the ortho position, and this molecular configuration is very similar to that of lignin (comparison showed in Figure 1). Base on above, it is more suitable to use dihydroeugenol as a lignin model compound than guaiacol.

Schematic diagram of Lignin and the model compound.
The infrared spectrum (IR) of LAP was shown in Figure 2. The result shows that the peak at 3394 cm−1 is the absorption of phenolic hydroxyl group of lignin, and peaks at 2924 and 2850 cm−1 are the absorption of methylene groups. The characteristic peaks of benzene ring were shown at 1600, 1512, and 833 cm−1, while that of methyl group was appeared at 1460 cm−1.

Infrared spectra of the acid-precipitated Lignin.
The 1H NMR spectrum of LAP was shown in Figure 3. The weak peak at 8.63 ppm corresponds to the H atoms of phenolic hydroxyl group, and the peak presented at 6.55–6.73 ppm is the character of H atoms in aromatic ring. The strong peak at 3.74 ppm corresponds to methoxy H atom, and peaks at 1.49 and 0.86 ppm relate to the methylene and methyl groups, respectively. Overall the IR and 1H NMR test results confirmed that the LAP contained dihydroeugenol structural fragment, therefore, dihydroeugenol can be selected as a lignin model compound for subsequent studies.
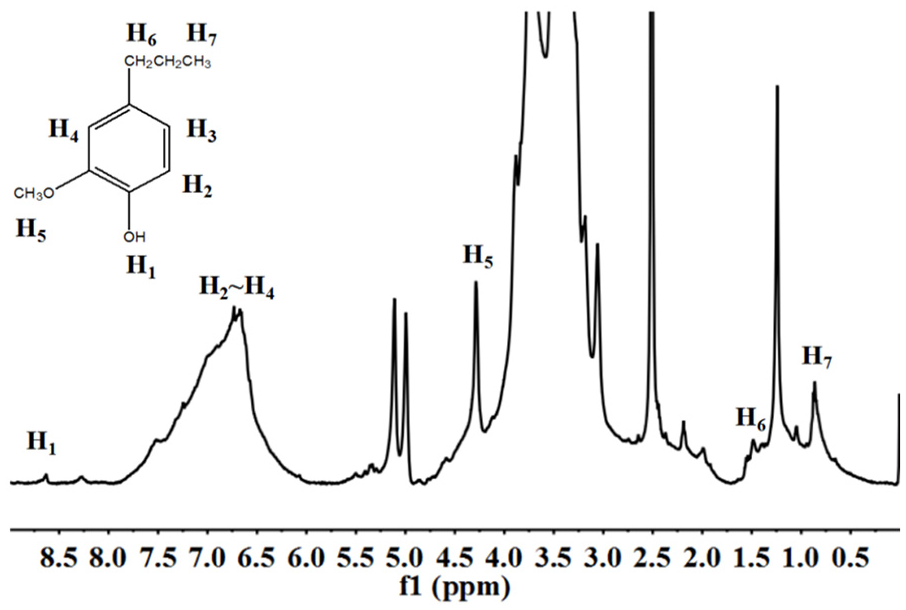
1H NMR spectrum of the acid-precipitated Lignin.
The reagents such as dihydroeugenol, acetone, formaldehyde, sodium metabisulfite, sodium hydrogen sulfite, and sodium hydroxide are all analytically pure. Dihydroeugenol was purchased from Beijing Beida Zhengyuan Technology Co., Ltd. and others were all purchased from Sinopharm Chemical Reagent Co., Ltd.
Cement clinkers were obtained from Hubei Yangxin Washi cement Co., Ltd, and gypsum was provided by Wuhan Iron and Steel Corporation. In order to avoid heterogeneous problem on the testing results, the mix of clinkers and gypsum were grinded for 5 h with a mass ratio of 95:5. 10 The specific surface area of this resulting cement was 3420 cm2/g and the mineral compositions of C3S, C2S, C3A and C4AF were 61.3%, 17.1%, 6.8%, and 8.8%, respectively.
Synthesis of superplasticizers
The specific synthesis of Superplasticizers is according to the method showed in papers.11,12 A sulfonated acetone formaldehyde superplasticizer AFS was prepared by the condensation reaction while the lignin-based superplasticizer DAFS and LAFS were prepared base on the model compound with or without replacement (substituted lignin by dihydroeugenol). 13
Molecular structure characterization
The structural information of superplasticizers is collected by Fourier transform infrared spectroscopy (FT-IR) and 1H nuclear magnetic resonance (1H NMR). The samples were uniformly mixed and ground with KBr powder at a mass ratio of 1:100, then they were pressed into a transparent sheet for FT-IR testing at a wavelength range of 400–4000 cm−1. A portion of superplasticizers were dissolved using DMSO before 1 H NMR characterization.
Adsorption measurement
The adsorption amounts of the superplasticizer samples on the cement particles were quantitatively measured by the depletion method. In a typical experiment, 3 mL of DI water contained different amounts of superplasticizer and 0.3 g of cement were added to a 10 mL centrifuge tube, the tube was then placed and shaken in a wobbler (LH-ZD20 drug wobbler, Yangzhou Longhu Medical Instrument Factory) for 1h, after that, the resulting suspension was centrifuged for 10 min at 4000 rpm to obtain the filtrate. A total organic carbon analyzer (liquiTOC, Elementar Analysensysteme Company, Hanau, Germany) was implemented to quantify the organic carbon content of the filtrate, which was immediately diluted with deionized water and tested by a combustion at 890°C. The adsorbed amount of superplasticizer was calculated by the difference of concentration before and after the addition of superplasticizer. This procedure was repeated three times, and take the average value as the valid data.
The adsorption isotherm between solid and liquid interface is usually described by Langmuir 14 or Freundlich 15 models, shown in equations (1) and (2), respectively. The equation (2) is usually replaced by equation (3). Among these equations, qe represents the equilibrium adsorbed amount (mg g−1), Ce is the concentration of superplasticizer (g L−1) in the solution at equilibrium state. q∞ corresponds to the maximum adsorbed amount (mg g−1). KL and KF are the constants for Langmuir and Freundlich models, while n is the nonlinear factor.
In general, the adsorption kinetics of liquid phase can be described by a pseudo-first-order (4) 16 and pseudo-second-order 17 equations, as shown in equations (4) and (5), qe and qt represent the equilibrium adsorbed amount and the adsorbed amount at time t (mg g−1), respectively, and K1 and K2 are the pseudo-first and pseudo-second order adsorption rate constants, respectively.
Zeta potential test on cement paste
The zeta potential of cement particles was measured at 25°C by using a Zetasizer (Nano ZS, ZEN 3600, Malvern Instruments Ltd, UK). Normally, Samples were prepared by mixing 0.8 g of cement with 8 g of superplasticizers to form aqueous solution. In order to analyze the impact of superplasticizers on the zeta potential of the cement particles, a reference sample of cement paste without the addition of superplasticizer was also prepared and measured. Each test was repeated three times, and the mean value was calculated.
Cement paste fluidity measurement
The fluidity of cement pastes with different dosages of superplasticizers were tested at the water to cement ratio (W/C) of 0.29, according to the Chinese Standard GB/T 8077-2012. In brief, puts the cement paste with superplasticizers into a Vicat cone (top diameter of 36 mm, bottom of 60 mm and height of 60 mm) and tests the degree of slump at 30 s after lifting the cone vertically. The detail preparation and testing procedures were described in the literature. 11 The dosage of superplasticizer was accounted by the effective solid content.
Test of rheological properties
The rheological properties of cement with superplasticizers was tested by the R/S rheometer (Plus RHEOMETER, Bookfield Company), and the cement paste was prepared according to the fluidity test method. The test procedure is as follows: the shear rate increases linearly from 0 to 200 s−1 within 3 min, and decreases to 0 linearly within 3 min.
Results and Discussion
Structural characterization of superplasticizer
The molecular structure of DAFS and sulfonated acetone formaldehyde condensate AFS were determined by the FT- IR and 1H NMR. The infrared spectrum of AFS was shown in Figure 4, the result indicates that peak at 2931 cm−1 is the adsorption of methylene group, peak at 1643 cm−1 is the characteristic adsorption of carbonyl group, the stretching vibration peaks of S–O and S=O from sulfonic acid group were appeared at 1188 cm−1 and 1041 cm−1, respectively. And peak at 525 cm−1 represent the stretching absorption of ether bond. Comparatively, the above characteristic peaks are also presented in the structure of DAFS, the difference is that the adsorption peaks of benzene rings are additionally appear at 1585, 1493, 1454, and 741 cm−1 in the IR spectrum of DAFS, and this suggest that DAFS contains both benzene ring structural fragment and AFS fragments.

Infrared spectra of the superplasticizers.
Back to the IR spectrum of acid-precipitated lignin (Figure 2), the absorption peak of benzene ring was exhibited at 833 cm−1, which corresponds to the characteristic adsorption of two adjacent hydrogen atoms. Comparatively, there is no absorption peak at the same position in the IR spectrum of DAFS, instead, a new methylene absorption peak occurs at 849 cm−1, this is because the adjacent hydrogen atom of the phenolic hydroxyl group is replaced by methylene group during the hydroxymethylation process.
The structure of DAFS was further analyzed by nuclear magnetic resonance (1H NMR). As shown in Figure 5, new absorption peaks of methylene H atom (–CH2–) at 1.55 ppm and aromatic ring H atom at 6.55–6.73 ppm were exhibited in DAFS compared with AFS. According to the IR and 1H NMR test results, it is confirmed that DAFS was successfully synthesized by using dihydroeugenol and other raw materials.

1H NMR spectrum of the superplasticizers.
Cement adsorption characteristics
Adsorption isotherms
In general, Langmuir and Freundlich isotherms can well explain the relationship between the adsorbed amounts and the concentrations of solute. The Langmuir isotherm mainly describes the monolayer adsorption, it assumed that the solid surface is homogeneities, all the adsorptive vacancies are independent and possess equal adsorption ability. In contrast, the Freundlich isotherm considers the solid surface was heterogeneous and an active adsorption centre exists.
Refer to the equations (1) to (3), the relationships between the adsorbed amounts of LAFS and DAFS on the surfaces of cement particles and the concentrations of solution were investigated. Figures 6 and 7 were the fitted curves of adsorption amount versus concentration based on those two isotherms. For Langmuir model, the adsorption correlation coefficients R2 of LAFS and DAFS are 0.941 and 0.985, respectively, while that for Freundlich model are 0.768 and 0.772, respectively. This result suggests that the adsorption behavior of the LAFS and DAFS superplasticizers conformed to the Langmuir model instead of the Freundlich model. Consider the assumed premise of the Langmuir model, the adsorption of both LAFS and DAFS on the surfaces of the cement particles must fit the monomolecular layer adsorption.

Curves of isothermal adsorption: (a) Langmuir model and (b) Freundlich model.

Curves of isothermal adsorption: (a) Langmuir model and (b) Freundlich model.
Adsorption kinetics
The pseudo-first/second-order equations were used to describe the change of adsorbed amount of solute by time. The pseudo-first-order kinetic model is established on the assumption that the adsorption is controlled by a diffusion step, while the adsorption process of pseudo-second-order kinetic model is regulated by a chemisorption mechanism.
According to equations (4) and (5), the adsorbed amounts over time were fitted as shown in Figures 8 and 9. It is clear that the adsorption kinetics of both DAFS and LAFS have a poor correlation with the pseudo-first-order model. Comparatively, the pseudo-second-order model is more suitable to explain the adsorption kinetics, this result suggested that the interaction between DAFS/LAFS and cement particles were mainly conducted by sharing electron pairs or transferring electrons, and the adsorption process was controlled by chemical adsorption mechanism rather than diffusion step. Overall, the adsorption isotherms and adsorption kinetics testing results indicate the similar adsorption properties of LAFS and DAFS.
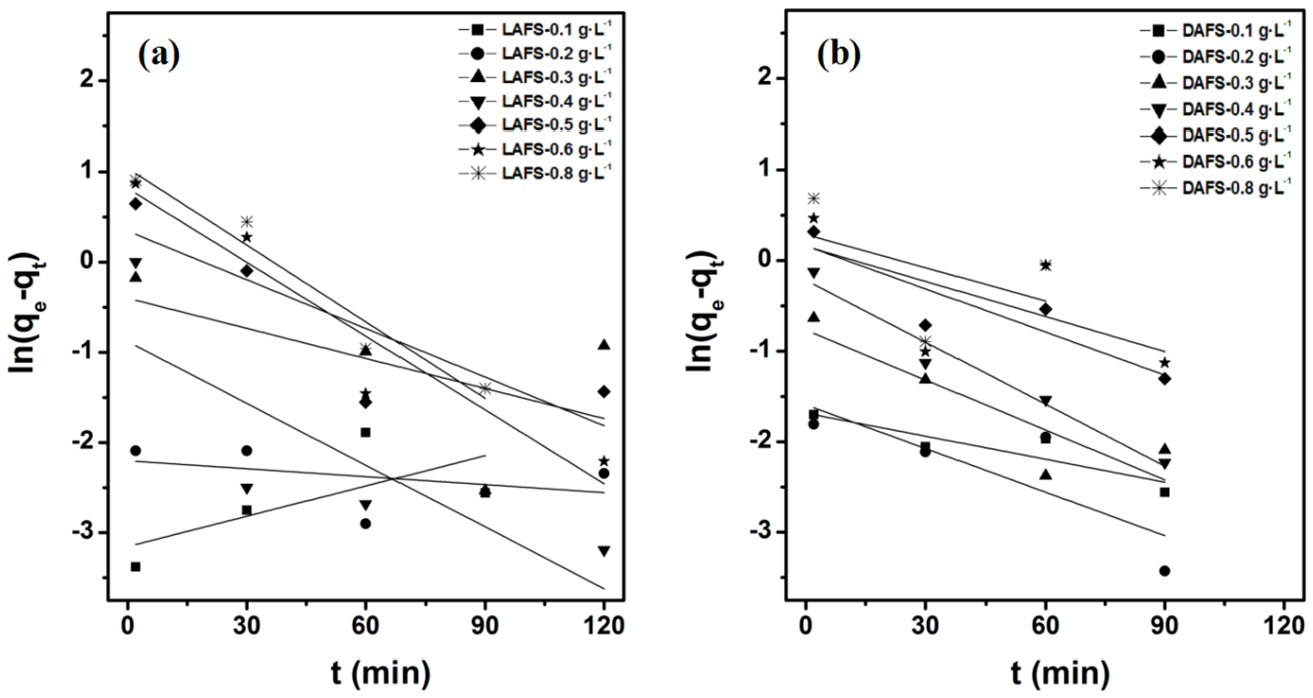
Fitted curve of Pseudo-first order adsorption kinetic: (a) LAFS and (b) DAFS.

Fitted curve of Pseudo-second order adsorption kinetic: (a) LAFS and (b) DAFS.
Adsorption and dispersion mechanism
Zeta potential
As cement contacted with water, positive ions such as Ca2+, Mg2+, Na+, K+, and also negative ions like SiO43-, AlO33− were dissolved rapidly from cement clinkers, those ions were then captured by the surface of cement particles. During the dissolution, some voids (formed because of the escape of cations) on the surface were filled by the OH− to establish an anionic layer, which reabsorbed the cations from the liquid phase. Attributed to this continue charge displacement, an electric double layer near the surface was finally form at the equilibrium state. As shown in Figure 10, the inner layer (IL, well known as the Stern layer) of the electric double layer consists of positive ions and water dipoles, the positive ions connected to the surface of cement particle are closely packed with each other. Comparatively, the negative ions in the outer layer (OL), known as the Gouy layer, have a dispersive arrangement. 16 The discrepant distribution of charges in the electric double layers will cause a potential difference, when the ions in Stern layer moved with the movement of cement particles due to the hysteresis activity, a slipping plane between the IL and OL will be created. Normally, the zeta potential of a particle is exactly the potential difference between the slipping plane and the bulk of the solution. 17 When Superplasticizer is added into the cement paste, it will be adsorbed on the surface of cement particles and significantly reduced the zeta potential of the surface, consequently a strong repulsive force between cement particles exists, result in a well dispersion of each particles. 18

Electric double layer of cement particles: (a) pure cement and (b) with the presence of superplasticizer.
Figure 11 shows the zeta potential of cement paste contained LAFS and DAFS. It is clear that without any chemical admixtures, the blank cement paste possess a zeta potential of +3.5 mV, this positive value is due to the relatively quick dissolution of C3A to produce [Ca2Al(OH)6]+ which was rapidly absorbed on the surface of cement particles. 19 When superplasticizer is present, zeta potential decreases dramatically by the increasing concentration of LAFS and DAFS, added with 1 g/L of LAFS and DAFS, zeta potential of cement paste were reduced to −15.2 and −18.7 mV, respectively. However, when the concentration of superplasticizer exceeds 1 g/L, the absolute value of zeta potential tends to slowly increase and gradually become stable. This can be explained by the fact that superplasticizer can be rapidly absorbed on the surface of cement particles, and as the dosage increases, more superplasticizer molecules were captured, so the zeta potential decrease quickly at first. As the adsorption amount is close to saturation, the excess superplasticizer is no longer adsorbed, so the zeta potential change little.

Zeta potential of cement pastes.
Consider the adsorption and dispersion mechanism of naphthalene sulfonate formaldehyde condensate superplasticizer (NSF) and other polycondensation type superplasticizers which conformed to the traditional DLVO theory (cement particles are mainly dispersed by electrostatic repulsion), the zeta potential of cement paste should be lower than −15 mV to ensure the stable dispersion of cement particles.20–21 However, this is not the limitation for polycarboxylate superplasticizer (PCE), whose dispersive ability is much stronger than that of naphthalene, the zeta potential of cement paste added with PCE was significantly lower than that of naphthalene-based superplasticizer. 22 Studies have shown that the dispersion mechanism of cement particles by PCE was largely depend on the steric hindrance effect.1,23–25 The different adsorption and dispersion mechanisms of the above two types of superplasticizers result in completely different dispersive properties and also impact on cement hydration.
From the above analysis and Zeta potential results, it can be confirmed that LAFS and DAFS have the same dispersion mechanism represented primarily by the electrostatic repulsion.
Cement paste fluidity
The impact of lignin-based polycondensation type superplasticizers LAFS and DAFS modified by using dihydroeugenol on the fluidity and rheology of cement paste were investigated. As shown in Figure 12, with the same dosage, cement paste contained DAFS exhibit approximately the same flow performance with that contained LAFS, the result indicates that the usage of dihydroeugenol instead of lignin to synthesize the superplasticizer do not significantly affect the dispersive property. This test is further confirmed that dihydroeugenol is suitable to replace lignin to study the structure-activity relationship of lignin-based superplasticizer.
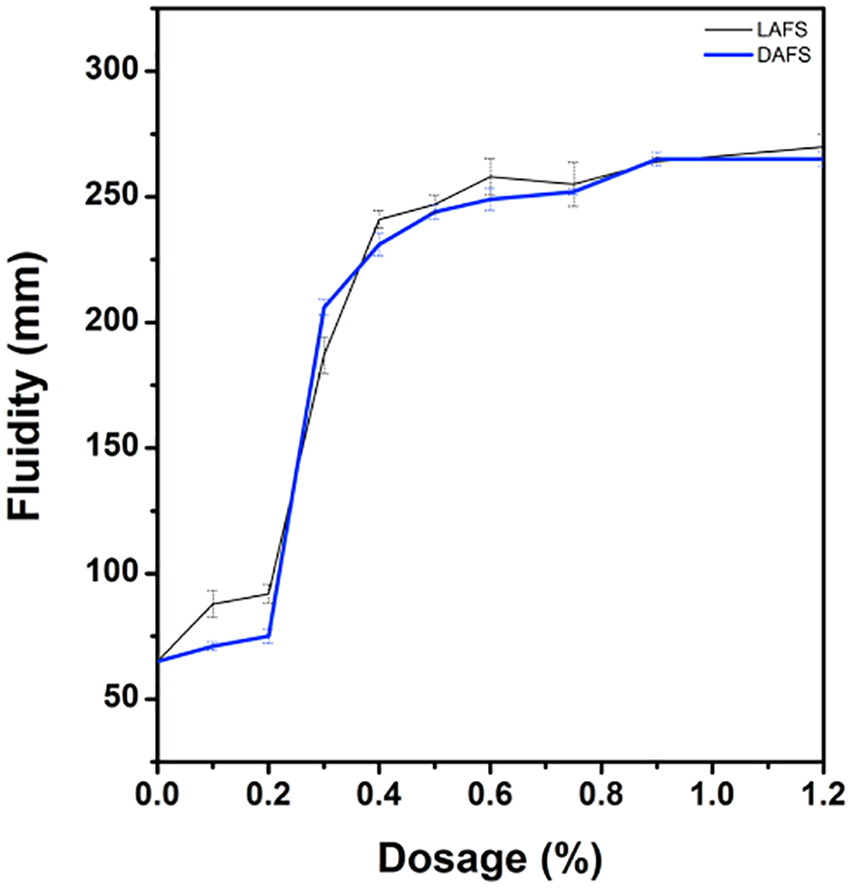
Fluidity of cement pastes.
From Figure 13, we can see that, although there is no obvious difference in fluidity, there are certain differences in the rheological properties of different types of cement pastes. The rheological properties of cement paste with DAFS are not sensitive to the shear rate, and the viscosity curves of the rising section and the falling section obviously overlap. But the cement paste with LAFS has a shear thinning phenomenon, and the viscosity curve of the falling section is located below the rising section. Based on this, the model will be further optimized in future work.

Rheology of cement pastes. (a) is shear stress curve and (b) is viscosity curve.
Conclusion
Due to the complex and variable molecular structure of Lignin, it is difficult to investigate the structure-activity relationship of modified lignin products. In the paper, we selected dihydroeugenol which possess the similar structure of guaiacyl fragment as a lignin model compound to replace lignin when prepared the polycondensate superplasticizer DAFS and also LAFS (prepared by lignin) as the reference, the difference in adsorption and dispersion properties of these two superplasticizers were investigated and the following conclusions were drawn:
Infrared spectroscopy and nuclear magnetic resonance (NMR) tests confirmed that the lignin obtained by acid precipitation contained a dihydroeugenol structure. The model product of lignin-based superplasticizer DAFS can be successfully synthesized by using dihydroeugenol instead of lignin, and the structure of DAFS contains both benzene ring structural fragment and AFS fragments.
The adsorption correlation coefficients R2 of LAFS and DAFS for Langmuir model are 0.941 and 0.985, while that for Freundlich model are 0.768 and 0.772, respectively. This suggests that the adsorption behavior of the LAFS and DAFS superplasticizers conformed to the Langmuir model instead of the Freundlich model. And the pseudo-second-order model was found to be more suitable to explain the adsorption kinetics of LAFS and DAFS.
In cement system, with the same dosage, LAFS and DAFS show the similar impact on Zeta potential and fluidity of cement paste, all the results suggest that dihydroeugenol is suitable to replace lignin on exploring the structure-activity relationship of lignin-based superplasticizer. At the same time, the model also needs further optimization to achieve the similarity of rheological property.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.