Abstract
Objectives
Cardiac syndrome Y (CSY) phenomenon is characterized by the gradual opacification of the coronary vasculature at the distal level. The leuko-glycemic index (LGI) is a blood glucose and white blood cell count index. This study investigates the relationship between CSY and the leuko-glycemic index.
Methods
A case-control, retrospective study was conducted. Consecutively selected individuals with complaints of angina were included in the study. Subjects with normal coronary flow (n = 132) and coronary slow flow (n = 136) were classified, after confirmation of coronary angiography results.
Results
The age ranges were recorded as 57 (48–64) for the NCF group vs. 52 (46–58) for the CSY group, P = .010. The mean hematocrit (Hct), Hemoglobin (Hg), White blood cells (WBC), and LGI were significantly higher in the CSY group than in the normal coronary flow group (P < .001). Accordingly, CSY was positively correlated with Hct, Hg, and WBC (P < .001). ROC curve analysis indicated that a cut-off value of ≥9.28 for the LGI predicted CSF with a sensitivity of 78% and a specificity of 78.1% [Area under the curve (AUC): 0.626 and 95% CI: 0.559–0.693].
Conclusions
In this study, we show for the first time that elevated LGI levels can independently predict CSF formation in the subclinical process.
Introduction
Cardiac syndrome Y is an angiographic phenomenon defined by the delayed transit of contrast in the absence of obstructive coronary artery disease. Previous research has found that the prevalence of CSY ranges between 1% and 7%. This phenomenon may generate angina-like chest pain during rest or exercise.1–3 It may be an early indicator of atherosclerosis, although the underlying causes of CSY are unknown, its diagnosis and treatment are critical due to its association with cardiovascular complications such as recurrent angina, unnecessary hospitalization and intervention, and fatal arrhythmias.4,5 Furthermore, this behavior is linked to a number of pathophysiological illnesses such as small vessel disease, endothelial dysfunction, inflammatory diseases, and neurohormonal imbalance.6,7 CSY is defined as the identification of Thrombolysis in Myocardial Infarction (TIMI) grade 2 flow in angiographically normal or near normal coronary arteries without ischemia provoking activities. 8 TIMI frame count is another CSF diagnostic technique. The first frame in this procedure is when the contrast agent enters the coronary artery. The final frame is the one in which the contrast agent reaches the distal end of the coronary artery. The difference between the first and last frames is used to calculate TIMI frames. 9 Many parameters have been developed for coronary artery disease risk assessment, one of which is the LGI. ILG was calculated as serum glucose [mg/dl] × leukocytes [106/L]/1000. Patients with high LGI have been shown to develop adverse cardiac events. 10 Diagnosis of CSY is difficult because it is a costly and invasive method; low-cost methods such as LGI can be predictive of CSY in those with angina complaints. The LGI became an accessible and clearly interpretable test with no major expenditures for patients and health systems due to its ease of calculation and routine assessment of involved variables on admission among MI patients.11–13 There is a limited number of studies investigating the relationship between CSY phenomenon and LGI levels. The aim of our study was to evaluate the relationship between LGI level and CSY in patients.
Patients and methods
Study design and population
A case–control, retrospective, and comparative study was conducted. Consecutively selected individuals over the age of 18 years who presented between June 2020 and July 2023 to the cardiology outpatient clinic with complaints of angina and exertional dyspnea and who had a positive treadmill exercise stress test or ischemia on myocardial perfusion scintigraphy were included in the study. The study was conducted in Ümraniye Training and Research Hospital, Istanbul, Turkey. We examined the clinical data and CAG results of these patients. Patients who had an emergency CAG operation were excluded from the research. The patients were divided into two groups: coronary slow flow and normal coronary arteries without occlusive CAD. Exclusion criteria were previous occlusive coronary artery disease (percutaneous intervention and coronary by-pass), heart failure, the presence of chronic renal failure, and chronic liver failure. The study protocol was approved by the Ethics Committee of Istanbul Ümraniye Training and Research Hospital (Approval Date: July 4, 2023; Approval Number: B.10.1.TKH.4.34.H.GP.0.01/249) in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines, and the reporting of this study was done in accordance with STROBE guidelines. 14
Definitions and laboratory measurements
Venous blood samples were taken within the first 24 hours of admission. White blood count (WBC) and fast plasma glucose (FPG) were calculated from the sample. ILG was calculated as serum glucose [mg/dl] × leukocytes [X103/µl]/1000.
Coronary angiography
The AXIOM Sensis (Siemens AG, Munich, Germany) digital angiographic system was utilized for coronary angiography. Images from coronary angiography were evaluated. CSY was determined using Beltram's criteria, which included (a) no obstructive epicardial CAD, (b) TFC > 27 frames, and (c) delayed distal vascular contrast opacification of epicardial coronary arteries. All angiograms were examined by one independent and experienced interventional cardiologist who was blind to the study, and the TFC was calculated for each patient. Gibson defined TFC as the number of cine frames required for the contrast material to reach the distal landmarks. The distal bifurcation of the LAD (known as the “mustache”), the distal bifurcation of the longest lateral left ventricular wall artery branch for the circumflex artery (Cx), and the first posterolateral artery branch of the right coronary artery (RCA) were among the distal markers. The right and left oblique cranial pictures for LAD, right oblique caudal images for Cx, and left oblique cranial images for RCA were used to calculate the TIMI-frame. Angiography was done at a rate of 30 frames per second. Given that the acquisition rate in our sample was 15 frames/second, we multiplied the TCF values by 2 to convert them to the 30 frames/second values. 15
Statistical analysis
SPSS 22.0 (Statistical Package for Social Sciences for Windows, version 22.0, IBM Corp, Armonk, NY, USA, 2013) was used for the statistical analyses. Student t test was used for parameters that fit the normal distribution, and Mann–Whitney U test was used for parameters that do not fit the normal distribution. Normal distribution data were expressed as mean standard deviation (SD). Categorical variables were compared with the Chi-square test and presented as percentages. Continuous variables were presented as mean ± standard deviation or median (interquartile range-IQR). To determine the normal features of the continuous variables, the Kolmogorov–Smirnov test was applied. The cut-off values of the LGI index that can be employed in the prediction of CSY were determined using a receiver operating characteristic (ROC) curve analysis. The independent predictors of CSY were identified using univariate and multivariate logistic regression analysis tests. Probability (P) values of .05 showed statistical significance. Pearson's correlation coefficient was employed in the correlation analysis between the CSFP independent indicators.
Results
Both groups’ demographic characteristics, comorbidities, and laboratory test results are summarized in Table 1. Moreover, 268 consecutive individuals were included. Subjects with normal coronary flow (n = 132) and coronary slow flow (n = 136) were classified after confirmation of coronary angiography results. The mean age was 54 (48–64) years, and 138 (51.4%) of them were male. The age ranges were recorded as 57 (48–64) for the NCF group vs. 52 (46–58) for the CSY group, P = .010 (Table 1). There was no significant difference between the patient and control groups regarding comorbidities, diabetes mellitus, HT, and the mean BMI value. At the same time, there was no significant difference in Creatinine, BUN, LDL, glucose, triglyceride, ALT, and AST too. On the other hand, the mean HDL level was significantly higher in the NCF (45 mg/dl) group than in the CSY (42 mg/dl) group (0.043). The mean Hg 14.2 (g/dl), Hematocrit 40.7 (%), WBC 8.1 (×103/µl), and LGI 0,79 mg/dl.mm3 were significantly higher in the CSY group than in the NCF group (P < .001).
Demographic and clinical characteristics of the study population.
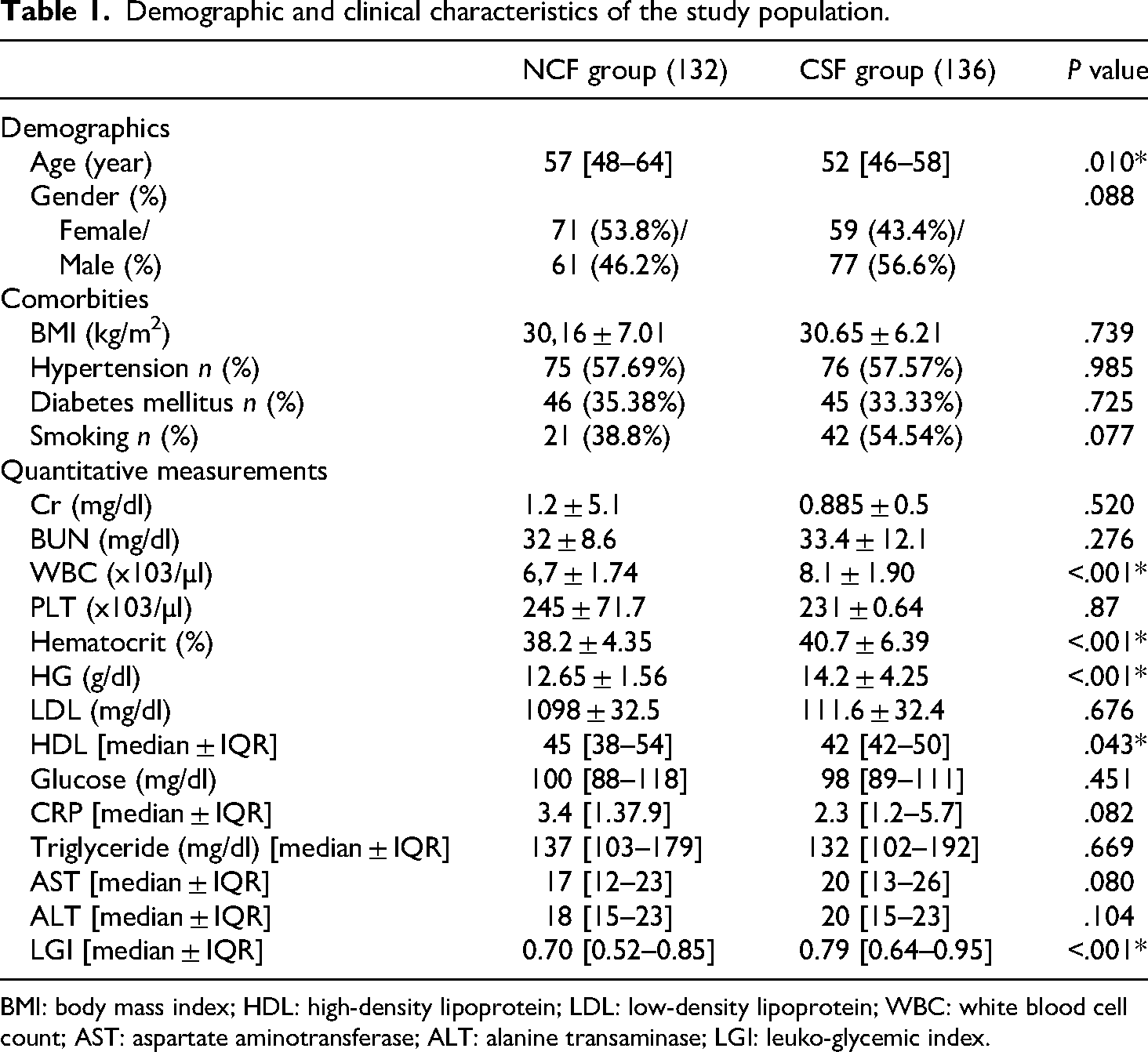
BMI: body mass index; HDL: high-density lipoprotein; LDL: low-density lipoprotein; WBC: white blood cell count; AST: aspartate aminotransferase; ALT: alanine transaminase; LGI: leuko-glycemic index.
Accordingly, CSY was positively correlated with hematocrit, Hg, WBC, and LGI values and negatively correlated with HDL values. In addition, there was a significant positive correlation between LGI values and CSY. The CSY group mean LAD TFC 40.5 ± 3.5, CX TFC 38.9 ± 2.3, RCA TFC 33.4 ± 3.3, and mean TFC 37.6 ± 2.3 was respectively.
The univariate logistic regression analysis revealed a significant relationship between CSY and HDL, WBC, Hematocrit, Hg, and LGI. Further analysis of these variables using the multivariate correlation analysis revealed that LGI values are independent predictors of CSF (Table 2). ROC curve analysis indicated that a cut-off value of ≥9.28 for the LGI predicted CSF with a sensitivity of 78% and a specificity of 78.1% [Area under the curve (AUC): 0.626 and 95% CI: 0.559–0.693] (Figure 1).

ROC analysis of the LGI index levels for predicting slow-flow coronary circulation. AUC: area under the curve; ROC: receiver operating characteristic; LGI: leuko-glycemic index.
Univariate and multivariate logistic regression analyses of the independent indicators of slow coronary flow.

CI: confidence interval, HDL: high-density lipoprotein, LDL: low-density lipoprotein, LGI: leuko-glycemic index.
Discussion
In this study, we demonstrated that the leuko-glycemic index (LGI) is significantly associated with the presence of the coronary slow flow (CSY) phenomenon. Furthermore, LGI was identified as an independent predictor of CSY. To the best of our knowledge, this is the first study to establish such an association, contributing novel insights into the pathophysiology of CSY.
The underlying mechanisms of CSY are complex and not yet fully understood, but proposed theories include endothelial dysfunction, microvascular abnormalities, increased vasomotor tone, and inflammatory processes.1,3,5,6 Endothelial dysfunction, in particular, has emerged as a pivotal factor in the genesis of CSY, with evidence of reduced flow-mediated dilation in affected patients.16–24
The role of inflammation in CSY pathogenesis is also well-supported. Increased leukocyte counts can exacerbate endothelial damage through the release of free radicals and proteolytic enzymes, which can impair vascular integrity.18,25,26 Leukocytes also promote capillary occlusion and hypercoagulability, potentially contributing to delayed coronary flow. 27 Similarly, hyperglycemia has been implicated in vascular dysfunction by promoting vasoconstriction, oxidative stress, and inflammatory mediator release.28–31 Together, these mechanisms suggest a critical interplay between metabolic and inflammatory factors in CSY.
The LGI combines fasting plasma glucose levels and leukocyte counts, serving as an accessible marker of systemic inflammation and metabolic dysregulation. Previous studies have shown that higher LGI levels are associated with adverse cardiovascular outcomes, including severe coronary artery disease (CAD) and acute myocardial infarction.23,24 In one meta-analysis conducted by Sadeghi et al. in 2023, the results demonstrated that the LGI could predict mortality and acute cardiac complications after MI. 10 Inspired by these findings, we hypothesized that LGI could also be a predictor of CSY. Indeed, our results showed significantly higher LGI levels in patients with CSY compared to those with normal coronary flow, and multivariate analysis confirmed LGI as an independent predictor of CSY. Our study further highlighted the diagnostic utility of LGI. Using ROC curve analysis, we determined that an LGI cut-off value of ≥9.28 (mg/dL.mm³) predicted CSY with 78% sensitivity and 78.1% specificity (AUC: 0.626; 95% CI: 0.559–0.693). While the moderate AUC value suggests limited discriminatory power, it is comparable to other noninvasive indices previously studied in CSY, such as the platelet-to-lymphocyte ratio (PLR) and neutrophil-to-lymphocyte ratio (NLR). 7 This finding underscores the potential role of LGI as a cost-effective and easily calculated marker for identifying patients at risk for CSY. In fact, if used together with indices such as PLR and NLR, CSY can be predicted more accurately. The pathophysiology of CSY remains an area of active investigation. While its exact mechanisms are still unclear, CSY is often considered a clinical syndrome linked to small vessel disease, microvascular dysfunction, and endothelial abnormalities.5–7 Endothelial dysfunction is particularly relevant, given its role in vascular homeostasis, platelet activation, and the progression of atherosclerosis.16–21 Reduced flow-mediated dilation in CSY patients further supports this theory. 22 The strong correlation between LGI and CSY observed in our study suggests that the inflammatory and metabolic changes reflected by LGI may play a central role in CSY pathogenesis.
In comparison to other inflammatory markers, such as the neutrophil-to-lymphocyte ratio, LGI might be superior to others. Vascular problems and prothrombotic situations are caused by the production of procoagulant and proinflammatory chemicals in response to hyperglycemia.20,23 Being a crucial inflammatory indicator, the leukocytes, the blood glucose level is significant since it is a part of standard tests, in contrast to other indicators that are not easily available for real-world use. 32 Our findings also align with previous studies investigating inflammatory and hematological markers in cardiovascular disease. For instance, elevated neutrophil counts have been shown to correlate with endothelial dysfunction and adverse cardiac events, including acute myocardial infarction.25,26 Hyperglycemia, another component of LGI, is also associated with endothelial injury and poor cardiovascular outcomes.28–31 Thus, LGI provides a composite measure that integrates these interrelated processes, making it a potentially valuable tool for predicting CSY. In conclusion, this study highlights LGI as a simple, cost-effective, and independent predictor of CSY. Given the challenges of diagnosing CSY using invasive methods, LGI could serve as a valuable non-invasive tool for risk stratification, especially in patients presenting with angina and ischemic symptoms. By incorporating LGI into routine clinical practice, clinicians may improve early detection and optimize management strategies for CSY, potentially reducing unnecessary angiographic procedures. However, larger-scale studies are needed to confirm these findings and refine the clinical utility of LGI in CSY.
Study limitations
Despite its strengths, our study has several limitations. First, it was a single-center, retrospective analysis, which may limit the generalizability of our findings. The relatively small sample size may also have reduced the statistical power of our results. Additionally, the moderate AUC value suggests that while LGI is a useful predictor, its performance could be improved by combining it with other biomarkers or diagnostic tools. Performing power analysis could have made an additional contribution to our study. Future research should focus on validating these findings in larger, multicenter, prospective studies and exploring potential synergistic effects of LGI with other indices.
Future directions
To validate the findings of this study, future research should focus on large-scale, prospective studies conducted across multiple centers. This would enhance the generalizability of the results and help mitigate biases associated with retrospective, single-center studies.
Additional research should investigate other readily available and inexpensive inflammatory markers such as NLR, PLR, and systemic immune-inflammation index (SII) and compare their predictive value with LGI for CSY to see if improved predictive accuracy can be achieved when used together.
Longitudinal studies could explore how elevated LGI levels and other markers correlate with the progression of CSY to more severe cardiovascular conditions, including myocardial infarction or heart failure.
More studies are needed to elucidate the underlying mechanisms of CSY, particularly the role of microvascular dysfunction, endothelial dysfunction, and inflammatory pathways, which are suggested contributors.
Future research could evaluate the predictive accuracy of LGI in combination with other non-invasive diagnostic methods, such as echocardiographic flow-mediated dilation (FMD) or advanced imaging techniques, to create a comprehensive, low-cost diagnostic framework for CSY.
Investigating how therapeutic interventions targeting inflammation or hyperglycemia (e.g., anti-inflammatory drugs, glucose-lowering agents) affect LGI levels and CSY outcomes would provide valuable insights into potential treatment strategies.
Further research could focus on specific populations (e.g., patients with diabetes, metabolic syndrome, or chronic inflammatory diseases) to determine whether LGI has differential predictive value in these subgroups for CSY.
Conclusion
Inflammation plays an important role in the formation and progression of atherosclerosis. In this process, it is important to detect subclinical cardiac changes before early cardiac structural changes develop. In this study, we show for the first time that elevated LGI levels can independently predict CSF formation in the subclinical process. In this study, high LGI levels could independently predict CSY formation in the subclinical process. LGI is a non-invasive, easy, inexpensive, and repeatable examination.
Footnotes
Ethical considerations
This study was approved by the Istanbul Ümraniye Training and Research Hospital ethics committee (Approval Date: July 4, 2023; Approval Number: B.10.1.TKH.4.34.H.GP.0.01/249).
Author contribution
Conception and design of the research and Statistical analysis: MBO, EE, MBT. Acquisition of data: MBO, SN. Analysis and interpretation of the data: MBO, EE, KT, MBT. Writing of the manuscript and Critical revision of the manuscript for important intellectual content: MBO, KT, MBT. All the authors have approved the final version of the manuscript to be published.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Patients’ consent
The retroactive nature of the study eliminated the requirement that each participant sign a written informed consent form.
