Abstract
Objective
The risk factors associated with hematoma formation following ultrasound (US)-guided vacuum-assisted excision (US-VAE) of breast fibroadenoma (FA), particularly those related to the US features of the lesions, remain unclear. This study aimed to investigate the US features influencing hematoma formation after US-VAE of FAs.
Materials and methods
This retrospective analysis included 137 patients with 145 FA nodules who underwent US-VAE at our hospital from January 2018 to August 2024, and subsequently underwent US follow-up to monitor hematoma formation and absorption. The US features of the lesions, including size, number of nodules removed, location, depth, distance from the nipple, peripheral tissue structure, and color Doppler flow features, were retrospectively analyzed to identify the potential risk factors associated with hematoma formation.
Results
Hematomas formed in 78 nodules (53.8%) 24–48 h after the US-VAE procedure and disappeared within 6 months in 77 cases (98.7%). In the multivariate logistic regression analysis, FA depth ≥ 1.5 cm or <0.7 cm (OR = 2.490, 95% CI: 1.056–5.872; p < 0.05), surrounding tissue mainly composed of fat (OR = 2.471, 95% CI: 1.043–5.857; p < 0.05), and rich blood supply (OR = 3.164, 95% CI: 1.444–6.934; p < 0.01) were independent risk factors for hematoma formation.
Conclusion
Most hematomas that formed after US-VAE for breast FA disappeared within 6 months. FA depth ≥ 1.5 cm or <0.7 cm, surrounding tissue mainly composed of fat, and rich blood supply indicated the possibility of hematoma formation after US-VAE.
Introduction
Fibroadenoma (FA) is the most common type of benign breast tumor in women, accounting for 40.7%–56.7% of all benign breast tumors1–3 and mostly occurring in young and middle-aged women. Asymptomatic FAs do not require treatment or follow-up, while symptomatic FA can be removed surgically or by vacuum-assisted mammotomy. 4
Ultrasound (US)-guided vacuum-assisted excision (US-VAE) is a recognized minimally invasive therapeutic alternative for the treatment of benign breast lesions.5–7 While US-VAE shows the same excellent curative effect as traditional open surgery,8,9 it is associated with smaller scars, reduced bleeding, shorter operation times, lower treatment costs, and greater patient satisfaction than traditional open surgery.9,10
Hematomas are the most common complication after US-VAE for benign breast nodules. 11 Although previous studies have reported the risk factors associated with hematoma formation following US-guided VAE interventions for benign breast lesions, including breast shape, number of nodules removed, duration of bandaging, and menstrual history,1,2 these factors do not include US features. Since VAE is guided by US imaging, the US characteristics of the lesions are a crucial factor worth consideration. However, the existing literature contains limited information regarding the correlation between US characteristics and the post-interventional occurrence of hematoma.
FAs show unique histopathological characteristics in comparison with other benign breast nodules and are composed of dense fibrous tissue and expanded acinar cells, which are wrapped by a fibrous capsule and form a clear boundary with the surrounding breast tissue. 12 Although the presence of the pseudocapsule can, in theory, facilitate US-VAE for FAs, some patients still develop hematomas after the procedure, and the incidence, outcomes, and risk factors of hematoma formation remain unclear.
Thus, the objective of this study was to retrospectively analyze US features to identify the risk factors for hematoma formation after US-VAE of breast FA.
Material and methods
Study design and patient selection
This study involved a retrospective investigation of consecutive female patients with breast FAs removed by US-VAE at the Department of Ultrasound of our hospital from January 2018 to August 2024. Our study adhered to the provisions of the 1975 Declaration of Helsinki, as revised in 2024. The study protocol was approved by the ethics committee, which waived the need to obtain informed patient consent considering the retrospective nature of the study. All patient data were de-identified. The reporting of this study conforms to STROBE guidelines. 13
The data were retrieved from the Picture Archiving and Communication System. The inclusion criteria were as follows: (1) The pathological assessment indicated FA; (2) complete follow-up records were available; and (3) US reports and retained images could be used to extract the observation indicators completely and accurately. The exclusion criteria were as follows: (1) non-standardized US images, resulting in unavailable information; (2) presence of nodules requiring a second US-VAE treatment due to residual lesions (multiple invasive procedures can increase the probability of hematoma, so we excluded these nodules); and (3) presence of coagulation dysfunction.
Ultrasound examination and image analysis
Breast US examinations were performed using US equipment (Hitachi Arietta 70; Hitachi Medical, Japan) and linear-array transducers with variable frequencies (L64; 5–18 MHz) by a sonographer who had obtained a certificate of standardized training in residency, passed the training in breast US image quality control, and had 10–15 years of experience in breast US.
The size and number of nodules to be removed as well as their location, depth, distance from the nipple, peripheral tissue structure, and color Doppler flow (CDFI) features were recorded. When capturing CDFI features, the CDFI sampling frame included an area at least 5 mm around the nodules, and the scale was set between 3 and 5 cm/s. The Doppler gain was adjusted to its highest level, with no noise interference. The features were then classified on the basis of the following criteria. (1) Lesion size was categorized as ≥2 cm or <2 cm. This categorization was employed because the VAE equipment used in this study excised tissue strips with a fixed length of 2 cm. We believe that lesions larger than 2 cm may require additional tissue removal, which could potentially lead to an increased risk of bleeding. (2) The number of removed lesions was categorized as single or multiple. (3) Lesion location was categorized into medial or lateral regions of the breast. These regions were defined on the basis of clock positions, with the boundary being the line between 12 and 6 o’clock. (4) Lesion depth (the vertical distance from the center of the nodules to the skin surface) was used to categorize lesions as ≥1.5 cm or <0.7 cm or others. (5) The distance from the lesion to the nipple was categorized as >1 cm or ≤1 cm. Lesions located behind the areola complex were classified as “≤1 cm from the nipple.” (6) The CDFI analysis was based on the Adler grade, 14 and we classified the blood flow signal of the nodule into four levels. The CDFI characteristics of the lesion encompassed not only the lesion itself but also the normal breast tissue located within a 5-mm radius surrounding it. Adler 0, no obvious blood flow signal; Adler 1, flow observed in one or two pixels (usually less than 1 mm in diameter); Adler 2, visualization of 1–3 main blood vessels or several small vessels; Adler 3, visualization of four or more vessels. In this study, Adler 0 and 1 indicated nodules lacking blood supply, while Adler 2 and 3 indicated nodules with rich blood supply. (7) Peripheral tissue structure of the nodules was determined on the basis of the presence of adipose or gland tissue over more than half of the circumference of the nodules in maximum gray-scale US images. The evaluated categories are summarized in Table 1.
Ultrasound characteristics of the fibroadenomas.

CDFI: color Doppler flow; US: ultrasound.
Ultrasound-guided vacuum-assisted excision
Depending on the nodule location, the patients assumed a comfortable position, lying on the side or supine on the bed to fully expose the surface projection of the nodule. All procedures were performed at our institution by two breast interventional sonographers, of which one with more than 10 years of experience was in charge of removing lesions with a 7G VAE device (EnCor; SenoRX, Irvine, CA, USA), and the other with more than 3 years of experience was mainly tasked with providing US guidance using an L64 linear probe (Hitachi Medical, Japan). The insertion site for the needle was chosen to ensure the shortest distance to the nodules, while avoiding the areola as much as possible. The approach from the insertion site to the nodule was along the long axis of the nodule. Local anesthetic (10–30 mL of 1% lidocaine containing 1:100,000 epinephrine) was injected into the incision site and periphery of the nodule. For masses adjacent to the pectoralis major muscle or masses just beneath the skin, sufficient saline was administered between the structures and masses to artificially increase the distance for needle passage and to enhance safety. A 3-mm skin incision was made, through which the 7-gauge EnCor device was passed and positioned just beneath the nodule using real-time US guidance, and the nodule was excised layer-by-layer along the preset needle path to the lower edge of the nodule (Figure 1). The specimens were automatically collected into the sampling tank by vacuum assistance. The procedure was repeated until the nodule was no longer detectable on US. After confirming that the nodules were removed completely, negative pressure was used to suck out the fluid at the intervention site, and the site was bandaged with elastic bandages for 24–48 h.

A 45-year-old female patient with a right breast fibroadenoma who underwent ultrasound-guided vacuum-assisted excision (US-VAE). (A) A 7G needle was placed under the nodule. (B) The cutter was withdrawn into the sheath, and the nodule was sucked into the cutter groove by vacuum negative pressure. (C) The nodule was partially removed. (D) The nodule was completely removed. US-VAE: ultrasound-guided vacuum-assisted excision.
Follow-up assessments
The US follow-up assessments were performed by one of five sonographers who had received a certificate of standardized training for resident doctors and had at least 5 years’ experience in breast US assessments. These five doctors were blinded to the US characteristics of the lesions and the previous review results. Hematoma was defined as fluid accumulation > 1 cm in the intervention area at 24–48 h after the procedure. US evaluations were performed 1 week, 1 month, 3 months, and 6 months after the intervention to observe the absorption of hematomas; follow-up assessments ended either when the hematoma disappeared or at the 6-month time point, whichever came first.
The hematoma images obtained by US examinations at different time points are shown in Figure 2.
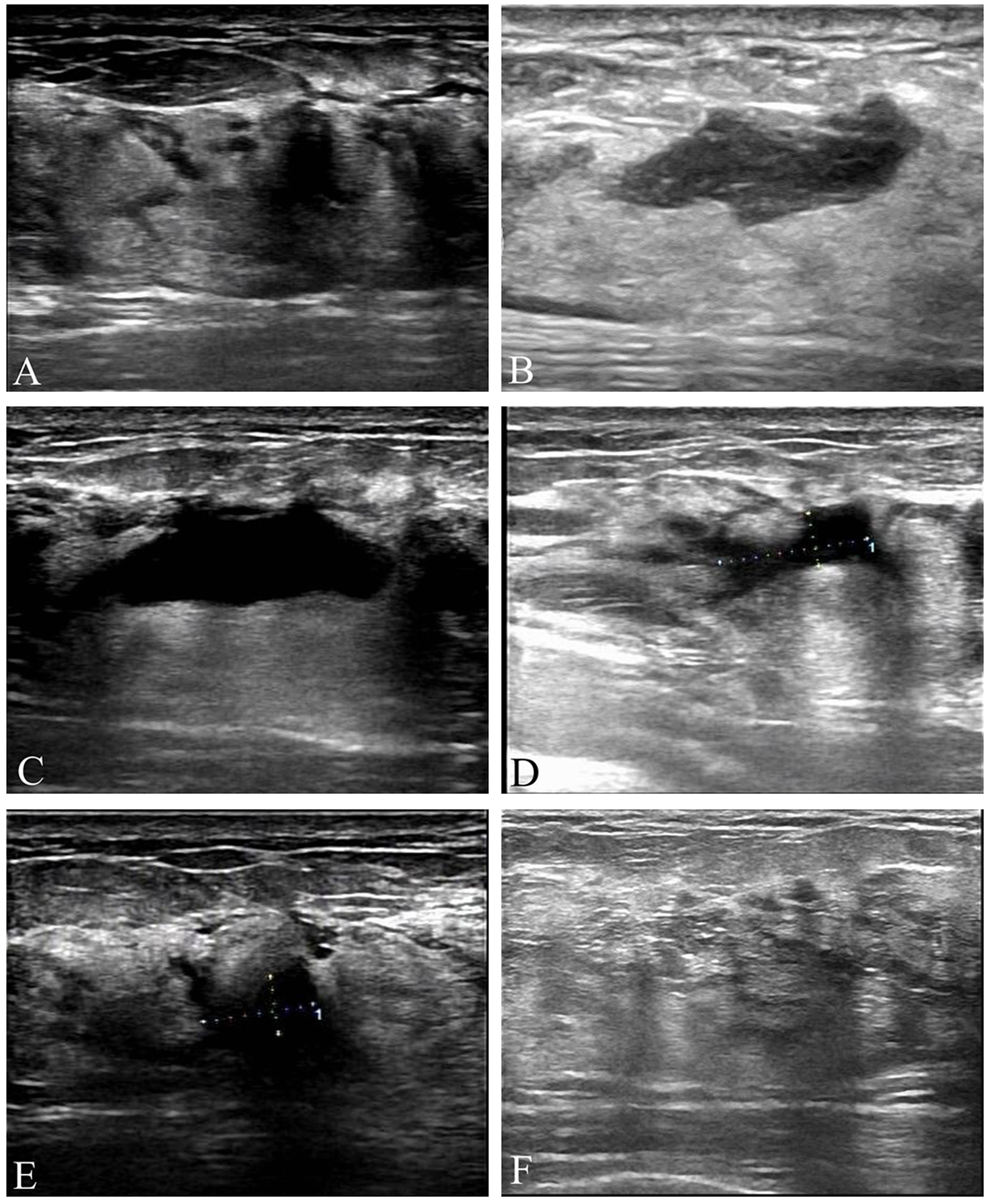
A 42-year-old female patient with a left breast fibroadenoma who underwent ultrasound-guided vacuum-assisted excision (US-VAE). Figures (A), (B), (C), (D), (E), and (F) show the ultrasound images obtained immediately after the intervention and at 24 h, 1 week, 1 month, 3 months, and 1 year after the intervention, respectively. At 3 months after the intervention, the maximum diameter of the hematoma was less than 1 cm, so subsequent ultrasound review was not required. Image (F) shows the breast ultrasound image obtained during the patient's annual health examination, which was performed exactly 1 year after the intervention. US-VAE: ultrasound-guided vacuum-assisted excision.
Statistical analysis
Data were analyzed using SPSS 17.0 (IBM, Armonk, NY, USA). Continuous data were presented as median (p25, p75) and were analyzed using the Mann–Whitney U test. Categorical variables were presented as frequencies and percentages and were analyzed using Pearson chi-square test. To assess the independent risk factors associated with hematoma, binary, forward multivariable logistic regression analysis was performed. All differences were considered statistically significant at p < 0.05.
Results
Characteristics of the patients and nodules
A total of 137 female patients (median age, 37 years; range, 20–67 years) with 145 nodules were included in this study. The mean maximum nodule diameter was 1.3 cm (range, 0.3–3.0 cm). The characteristics of the patients and nodules are presented in Table 2.
Characteristics of the patients and nodules.

FA: fibroadenoma.
The p-value represents the comparison between the groups with and without hematoma.
Hematomas
Among the 145 nodules, 78 (53.8%) showed hematoma formation at 24–48 h after the procedure. The hematomas ranged from 1.1 to 4.5 cm in size, including 44 cases (56.4%) with hematomas 1.1–2.0 cm in size; 31 cases (39.7%), 2.1–3.0 cm; two cases (2.6%), 3.1–4.0 cm; and one case (1.3%), > 4 cm (Table 3).
Hematomas formed 24 to 48 h after US-VAE.
US-VAE: ultrasound-guided vacuum-assisted excision.
The hematomas were not observable at 1 week after the procedure in 17 cases (21.8%), 1 month after the procedure in 45 cases (57.7%), 3 months after the procedure in 72 cases (92.3%), and 6 months after the procedure in 77 cases (98.7%) (Figure 3).

Among the 145 nodules included in the study; hematomas were found at 24–48 h after the procedure in 78 nodules. The number of hematomas decreased gradually over time, and at 6 months after ultrasound-guided vacuum-assisted excision (US-VAE), only one hematoma was not fully absorbed. However, the ultrasound assessment performed 12 months after US-VAE showed that the hematoma had been completely absorbed in this case. US-VAE: ultrasound-guided vacuum-assisted excision.
Risk factors for hematoma formation
Univariable analyses showed no significant associations between hematoma formation and nodule size (p = 0.277), number of removed nodules (p = 0.053), nodule location (p = 0.585), and distance to the nipple (p = 0.157) (Table 4). However, hematoma formation was associated with lesion depth (p = 0.016), peripheral tissue structure (p = 0.008), and CDFI (p = 0.001). The multivariable logistic regression analysis showed that nodule depth ≥ 1.5 cm or <0.7 cm (p = 0.037; odds ratio [OR] = 2.490; 95% confidence interval [CI]: 1.056–5.872), surrounding tissue mainly composed of fat (p = 0.040; OR: 2.471; 95% CI: 1.043–5.857), and the presence of a rich blood supply (p = 0.004; OR: 3.164; 95% CI: 1.444–6.932) was independently associated with hematoma occurrence after US-VAE (Table 5).
Univariable analyses of hematoma after US-VAE.
CDFI: color Doppler flow; US-VAE: ultrasound-guided vacuum-assisted excision.
Multivariate analysis of hematomas after US-VAE.

CDFI: color Doppler flow; CI: confidence interval; OR: odds ratio; SE: standard error; US-VAE: ultrasound-guided vacuum-assisted excision.
Discussion
The findings of this study revealed that the incidence rate of hematoma following US-VAE treatment for breast FA was 53.8%. Independent risk factors for hematoma formation included FA depth ≥ 1.5 cm or <0.7 cm, predominant surrounding adipose tissue, and abundant blood supply. The distinguishing feature of this study is its focus on breast FA specifically, rather than benign breast lesions in general. The influence of factors affecting postoperative hematoma formation and outcomes in breast FA has been insufficiently addressed in prior studies. Furthermore, unlike previous studies that emphasized factors subject to artificial interventions, such as the menstrual period, compression time, and the use of hemostatic drugs, we placed greater emphasis on the US characteristics of the lesions themselves and the breast tissue during the selection of influencing factors. These intrinsic lesion characteristics cannot be artificially altered. By analyzing the US characteristics of each lesion, we identified risk factors associated with hematoma formation.
At the 1-week follow-up, only 21.8% of the hematomas had disappeared, and the hematomas remained unchanged in most cases. In the follow-up assessments, 57.7% of the hematomas had disappeared within 1 month, 92.3% within 3 months, and 98.7% within 6 months. These results were similar to the findings of most previous studies. According to Huo et al., 2 98.4% of hematomas completely disappear within 6 months. Ko ES et al. 15 reported that approximately 81.5% of hematomas disappear within 1 month, with the remaining hematomas ranging in size from 7 mm to 12 mm, while 93.8% of hematomas disappear within 6 months, with the remaining hematomas being <10 mm in size. In this study, only one patient did not show disappearance of the hematoma at the 6-month follow-up; however, the maximum diameter of the hematoma in this patient was only 1.2 cm.
In this study, most hematomas were small, and only three cases showed hematomas with maximum diameters > 3 cm. Surgical treatment is usually required for symptomatic, painful, or infected hematomas because puncture drainage is often unsuccessful due to the presence of clots or dense contents in such cases.16,17 However, none of the patients is this study experienced notable discomfort, so they did not require treatment or painkillers for the hematomas, and only US follow-up was performed.
The application of elastic bandages is the most effective approach to prevent hematoma formation. 2 Any factor that reduces the effectiveness of elastic bandages can trigger hematoma. However, due to the various factors contributing to biofilm resistance against mechanical force, such as viscoelasticity, 18 tissue strain tends to decrease with increasing depth during compression. 19 Consequently, lesions located too deep may exhibit suboptimal response to compression, thereby increasing the likelihood of hematoma formation. In addition, deeper FAs require a longer puncture path and involve a higher vascular injury rate, increasing the likelihood of hematoma.
Breast blood supply is mainly distributed in the subcutaneous tissue, which is relatively shallow, and the venous system can even be observed through the skin, 20 potentially explaining the higher incidence of hematoma when lesions are shallow. Furthermore, for FA close to the skin, a large amount of isolation fluid is injected around the FA, especially between the skin and the nodule, to prevent skin damage. However, when the nodule is removed, a part of this isolation fluid may penetrate into the residual cavity, showing hematoma-like manifestations during US review.
The incidence rate of hematomas also differed depending on the type of tissue surrounding the FA. The incidence rate of hematoma was 70.5% when the tissue surrounding the FA was dominated by fat, while it was significantly lower (46.5%) when the tissue surrounding the FA was dominated by glands. The higher incidence of hematoma when fat tissue predominates around the lesion can be attributed to two possible reasons: (1) Fat tissue, in comparison with gland tissue, has a lower density and hardness, making it more prone to deformation21,22 and resulting in suboptimal filling of the residual cavity; (2) fat tissue predominantly surrounds the fibroglandular tissue like a “pocket.” 23 As mentioned earlier, this adipose-rich region contains a higher concentration of blood vessels, increasing the risk of vessel damage and subsequent hematoma formation.
The findings reported by Yao et al. 24 revealed no significant association between the blood supply characteristics of the lesion and hematoma occurrence; however, these results may be plausibly attributed to the vascular network surrounding the lesion. Lesions with higher internal blood flow may possess a greater number of internal vascular branches, but this does not necessarily imply an increased likelihood of peripheral vessel blood leakage into the residual cavity following mass removal in comparison with masses showing poor blood supply. Unlike previous investigations, we analyzed the blood flow inside the lesion and within a 5-mm region of breast tissue surrounding the lesion. For curative resection of the lesion, normal breast tissue surrounding the lesions is also inevitably excised or damaged. Blood vessels are the source of bleeding. While vascular injury can directly result in leakage of blood, it can also induce inflammation and facilitate the formation of fragile blood vessels. 25 These “leaky” newly formed vessels may show micro-hemorrhages, resulting in fluid exudation into the residual cavity. 26 Our study demonstrated that a high Adler blood flow grade (2 and 3) was an independent risk factor for hematoma formation and when abundant blood supply (Adler grade 2 or 3) appeared around and inside the lesion, the probability of hematoma increased 3.16-fold (p < 0.05).
Although some studies have reported that simultaneous resection of multiple nodules is a risk factor for postprocedural hematoma formation,1,2,6,16,27 the results of this study showed that the number of nodules was not a risk factor for postprocedural hematoma. This difference between the findings of previous studies and the present study may be attributable to differences in factors such as the interventional doctors’ personal operating practices, diagnosis and treatment levels, and the distance between and relative positions of different nodules.
This study had some limitations. First, the retrospective nature of the analysis may have resulted in selection bias. Second, some factors could not be evaluated. For example, the maximum diameter of the hematoma was used instead of the hematoma volume. Some hematomas with a large maximum diameter and two other small vertical diameters are actually small and are absorbed within a short time, whereas some hematomas with a smaller maximum diameter and a larger volume may be absorbed slowly. However, since data for the hematoma volume were not available in this study, the influence of hematoma volume on the hematoma-absorption rate could not be analyzed. In addition, while the analysis of lesion blood flow in this study included breast tissue 0.5 cm around the nodule, the 0.5-cm cutoff was based on the experience of a single center, and was not verified by other centers. Furthermore, relying solely on data from a single center may have limited the generalizability of the findings.
Conclusions
Hematomas occurred in 53.8% of the patients with breast FA after US-VAE, and approximately 98.7% of these hematomas were absorbed within 6 months after the procedure. FA depth ≥ 1.5 cm or <0.7 cm, surrounding tissue mainly composed of fat, and rich blood supply of the lesion were independent risk factors for hematoma formation after US-VAE. Similar analyses of influencing factors based on US characteristics are rare in previous studies. US is a commonly used image-guidance method in VAE, and a thorough understanding of the US characteristics of images is of great significance. Although our study yielded some important results, additional data from larger-scale, prospective, multi-center studies are required for further discussion and analysis, which is the direction of our future research.
Footnotes
Author contributions
YD did substantial contributions to the conception or design of the work; acquisition, analysis, or interpretation of data for the work; drafting the work; final approval of the version to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. YH did acquisition of data for the work; reviewing it critically for important intellectual content; final approval of the version to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. XZ did substantial contributions to the conception or design of the work; analysis of data for the work; reviewing it critically for important intellectual content; final approval of the version to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. LQ did acquisition of data for the work; reviewing it critically for important intellectual content; final approval of the version to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. YY did acquisition of data for the work; reviewing it critically for important intellectual content; final approval of the version to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. XQS did substantial contributions to the conception or design of the work; interpretation of data for the work; reviewing it critically for important intellectual content; final approval of the version to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Acknowledgments
The authors thank all team members and colleagues in the Medical Center of Ultrasound for their cooperation.
Ethical considerations
This retrospective study was approved by the Bioethics Committee of Beijing Friendship Hospital affiliated to Capital Medical University, and the requirement for informed consent was waived by the committee. Address of the Ethics Committee: Room 95, Yongan Road, Xicheng District, Beijing, China. Approval number: 2022-P2-394-01. Date of approval: 28 November 2022.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
