Abstract
Objective
The Vivosonic Integrity auditory brainstem response (ABR) system, featuring the Kalman-weighted averaging algorithm, was designed for awake testing in infants and young children. Despite reducing noise and state dependency, previous research has indicated that testing conditions still significantly affect ABR thresholds. Compared with the click stimulus most commonly used in clinical ABR testing, chirp stimuli compensate for the temporal dispersion occurring in the cochlea and overcome the traveling wave delay caused by the special anatomical structure of the cochlea. They enable more nerve fibers to discharge synchronously, thereby generating a stronger evoked waveform, which in turn enhances the anti-interference capability of ABR testing. The present study aimed to explore the application of chirp stimuli in Kalman-weighted averaging ABR and to optimize solutions for obtaining objective hearing results in nonsedated states.
Methods
Twenty-four adults (48 ears) with normal hearing aged 18–34 were enrolled for ABR testing using the Vivosonic Integrity system in three states: lying, sitting, and writing, which simulate the states of young children most commonly encountered during nonsedated ABR testing. Chirp/narrowband-chirp (NB-chirp) ABR thresholds and acquisition durations were recorded and compared with previous results of click ABR (cABR)/tone-burst ABR (tbABR) with Kalman-weighted averaging.
Results
(1) The thresholds and acquisition time of the chirp/NB-chirp ABR increased significantly generally in the order: lying < sitting < writing. (2) Chirp/NB-chirp ABR showed less variance across states, closer responses to the hearing threshold, and shorter testing durations than cABR/tbABR.
Conclusions
Chirp/NB-chirp ABR with the Kalman-weighted averaging algorithm demonstrated better noise resistance and shorter testing time than cABR/tbABR. These stimuli together with Kalman-weighted averaging provide advantages for threshold measurement in awake subjects, but it still requires respective normal and correction values for each testing state.
Introduction
Auditory brainstem response (ABR) is an essential tool for hearing assessment in infants and young children. In the clinic, sedatives are often used during tests to keep infants and young children in a sleeping state, minimize interference from brain and muscle activity, and obtain reliable test results. For subjects who cannot undergo conventional ABR testing due to various objective and subjective reasons, especially infants and young children who cannot cooperate with subjective hearing tests, finding a method to reduce the dependence of ABR on the testing state and environment is of great significance.
Due to the fact that ABR wave is time-locked to the stimulus, each repeated stimulus will elicit the same response, while noise is random and irregular. Traditional ABR improves the waveform by increasing the number of sweeps and performing signal averaging. At the same time, during the averaging process, an artifact rejection level at a certain voltage is usually set and sweeps with amplitudes greater than this level are excluded from the averaging process. Although this reduces the impact of noise on the ABR waveform, the exclusion of too many sweeps will require the collection of more data for adequate averaging, which may therefore prolong the test time. The Kalman-weighted averaging algorithm is executed using the commercialized Vivosonic Integrity System (Vivosonic Inc., Toronto, Ontario, Canada) during ABR testing. In this algorithm, no artifact rejection is performed, and each sweep recalculates the weighting according to the relationship between covariance in sweeps and residual noise. Scans with less noise are assigned much greater weights than those with higher noise amplitudes, thus enabling the acquisition of clearer waveforms in a shorter amount of time.1–5 As a result, the algorithm enhances the signal-to-noise ratio during testing and improves ABR recordings in challenging testing environments, such as with infants or individuals with high muscle activity. Along with in situ amplification technology and wireless acquisition technology, this system aims to enable ABR testing among awake infants and young children without the use of sedatives. 4 This approach is particularly beneficial for children who cannot consume sedative medications or adults who cannot remain calm during ABR testing. Additionally, nonsedative ABR mitigates medical risks, such as the potential for choking when sedatives are administered to infants, while also significantly reducing the overall testing time.
Elsayed et al. 6 used the Vivosonic Integrity system to determine click ABR (cABR) and tone-burst ABR (tbABR) thresholds in both awake and sleeping infants. They reported that there were no significant differences in thresholds between the two states. However, Cone and Norrix 7 reported threshold variations based on different test conditions. The mean threshold differences between the quiet and reading states were 18.5 dB for the conventional method and 11.0 dB for the experimental method (Kalman-weighted ABR). These threshold differences were statistically significant. In our initial research on Kalman-weighted ABR, we demonstrated that the physiological noise introduced by different testing conditions significantly influences the thresholds. 8 This aligns with the findings of Cone and Norrix. Given the challenges of maintaining nonsedated infants and young children in a stable state during actual testing, it is necessary to develop a more effective approach to mitigate the impact of the subject's state on the ABR threshold, thereby achieving greater stability.
Chirp stimulus has gained increasing prominence in clinical practice since its introduction into auditory electrophysiological response testing by Dr Shore and Nuttall. 9 Various studies have reported that chirp stimuli elicit larger wave V response amplitudes than click or tone-burst.10–14 This phenomenon is attributed primarily to the entire frequency range contributing to the chirp response, whereas in the case of clicks, lower frequencies may not effectively contribute to the response. Chirp ABR induces a more robust neural response and exhibits superior stability, facilitating more accurate determination of auditory thresholds.15,16 Additionally, the stability of the waveform gives examiners more confidence in the recorded results.17,18 Some studies have shown that it is precisely because of this characteristic that the response threshold of chirp/NB-chirp ABR is lower than that of cABR/tbABR.19,20
High frequencies stimulate the base of the cochlea, whereas low frequencies excite the apex of the cochlea. One consequence of this type of “cochleotopic” representation is that the high-frequency (basal) cochlear regions are stimulated first, with more apical regions stimulated after a bit of delay. 21 Thus, when a broadband sound signal is given, the hair cells in different parts of the cochlea and their corresponding nerve fibers cannot be activated simultaneously, and the compound neural response will be temporally smeared. Chirp stimulation was designed to compensate for the temporal dispersion that occurs in the cochlea, with its phase characteristic being “low-frequency sounds come out early, and high-frequency sounds come out late.” 22 This phase characteristic overcomes the traveling wave delay caused by the special anatomical structure of the cochlea and can cause more nerve fibers to be discharged synchronously. Therefore, the electrophysiological response evoked by the chirp is greater than that evoked by the click, and it is easier to judge the waveform. Elberling, Callø, and Don 23 reported that in individuals with normal hearing, the average ABR wave V amplitude evoked by chirp stimuli is generally greater than that evoked by click stimuli at various intensities. Maloff and Hood 24 reported similar results in adults with sensory hearing loss. These results suggest that using chirp stimulation may improve the resistance of interference in ABR testing due to more robust waveforms.
Although chirp and narrowband-chirp (NB-chirp) are promising stimuli that may promote the efficiency of auditory electrophysiological response testing, to our knowledge, no study has investigated the application of chirp/NB-chirp in Kalman-weighted ABR. This study aims to apply chirp and NB-chirp combined with Kalman-weighted averaging in ABR testing to explore whether their use can further reduce the impact of electrical interference generated by the brain and muscle on ABR thresholds in nonsedated infants and young children under different conditions.
Methods
This study was conducted at Peking Union Medical College Hospital, Beijing, China from April 2020 to June 2020. This is a prospective study.
Subjects
A total of 24 subjects (48 ears) with normal hearing participated in this study. The ages of the subjects ranged from 18 to 34, and there were 18 females and 6 males. The binaural air-conduction pure tone audiometry (PTA) thresholds were less than or equal to 20 dB HL across the 0.25–8 kHz frequency range. The tympanograms were type A, the acoustic reflexes were successfully elicited, and the cABR waveforms were reliably obtained in all participants while they were in a quiet supine position, with thresholds ≤25 dB HL. None of the subjects had a medical history of otological or neurological diseases. All the participants were able to cooperate smoothly during the tests. The research protocol received approval from the Ethics Committee of Peking Union Medical College Hospital, and informed consent was obtained from all participants before enrollment.
Test method
Pure tone audiometry
Pure tone audiometry was conducted in a standard soundproof booth with background noise less than 30 dB (A) SPL (GB/T 4854/ISO 389) using a Madsen Conera audiometer (GN Otometrics A/S, Taastrup, Denmark).
Acoustic impedance measurement
Acoustic impedance measurement (226 Hz) was executed in a quiet room with background noise less than 45 dB (A) SPL using a Madsen OTOflex 100 middle ear analyzer (GN Otometrics A/S, Taastrup, Denmark).
Establishment of the normalized hearing level for chirp stimuli
Since there is no standard acoustic reference zero for chirp and NB-chirp stimuli, this study enrolled normal-hearing students aged 18–25 years from Peking Union Medical College as participants to establish the normalized hearing level in our laboratory. The test was implemented under the same circumstances as the PTA, and the equipment was consistent with the ABR testing. The testing environment and equipment used were consistent with those in the formal experiment. The stimuli used in the test were chirp and NB-chirp, presented at a rate of 39.3/s. The subjects were instructed to raise their hand whenever they heard the sound. The average hearing thresholds of these subjects to chirp and NB-chirp stimuli were obtained, and these thresholds were used as the hearing zero level for chirp and NB-chirp stimuli.
Kalman-weighted averaging ABR recording
The test was performed in a quiet room with background noise less than 45 dB (A) SPL using a Vivosonic Integrity system (Vivosonic, Inc., Toronto, Ontario, Canada). The test system was calibrated annually before the study. The acoustic transducer was inserted earphone (ER-3A). The equipment was calibrated for the intensity of cABR, tbABR (0.5, 1, 2, and 4 kHz), on the basis of GB/T 4854.6/ISO 389-6. The intensities of the chirp and NB-chirp were calibrated on the basis of the abovementioned normalized hearing level.
In this experiment, the placement of ABR electrodes is as follows and has been added:
Active Electrode (Noninverting Electrode): Frontal high forehead (Fz) Reference Electrode (Inverting Electrode): Mastoid left/right (M1/M2) Ground Electrode: Frontal lower forehead (Fpz)
The thresholds of cABR, tbABR (0.5, 1, 2, and 4 kHz), chirp ABR, and NB-chirp ABR (0.5, 1, 2, and 4 kHz) were tested. Considering that the cooperation status of the subjects deteriorated with increasing test time, 12 subjects completed the cABR and tbABR tests (1, 4 kHz) as well as the chirp ABR and NB-chirp ABR tests (1, 4 kHz), whereas the other subjects completed the tbABR test (0.5, 2 kHz) and the NB-chirp ABR test (0.5, 2 kHz). The testing parameters for cABR and tbABR were consistent with those used in our previous study.
8
The chirp and NB-chirp stimuli were presented at a repetition rate of 39.3/s. A bandpass filter of 30–1500 Hz was used, and the data were recorded in a 20 ms time window for NB-chirp and 10 ms for chirp. Each subject was tested under three states as follows:
Lying: The subject was lying flat on a bed and was instructed to remain quiet and relaxed, with encouragement given to facilitate falling asleep. Sitting: The subject sat in a chair in a relaxed manner and watched silent videos or read quietly. Writing: The subject sat in a chair and wrote during the test.
The testing sequence of different stimuli and conditions for each participant was randomized. We took the following steps to acquire the thresholds: Starting at an initial sound intensity of 60 dB nHL, we decreased it with a step of 20 dB until a wave V could not be detected and then increased it with 10 dB at this level to close to the threshold. Then, 5 dB steps were used to find the threshold. The lowest stimulus intensity that could repeatedly detect wave V was the response threshold. We also measured the time required to obtain the threshold for each stimulus of each ear. We started timing when the initial sound intensity was applied and stopped timing when the lowest stimulus intensity at which Wave V could be repeatedly detected was reached, that is, when the response threshold was observed.
In this study, the ABR trial was terminated manually after a clear response was detected, in accordance with the American Academy of Audiology Practice Guidelines. 25 All tests were conducted by two audiologists, each of whom had more than 8 years of clinical experience. One audiologist was responsible for performing the tests and identifying wave V, whereas the other audiologist independently labeled wave V on blank waveforms without prior knowledge of the initial identification. The threshold duplication rate was 92.4%, with the threshold difference between the two assessors for all responses falling within ±5 dB.
Statistical methods
SPSS 23 was used for statistical analysis. For data that meet the assumption of normality, repeated-measures analyses of variance were conducted to assess differences in thresholds and testing time of chirp/NB-chirp ABR across the three testing states, followed by post hoc tests with Bonferroni correction to explore significant differences between pairwise states. For data that do not meet the assumption of normality, Friedman test was conducted and followed by Wilcoxon signed-rank test to explore significant differences between pairwise states. Paired-sample t tests (data meet the assumption of normality) or Wilcoxon tests (data do not meet the assumption of normality) were employed to analyze the differences in thresholds and testing time between chirp/NB-chirp ABR and cABR/tbABR.
Results
The normalized hearing level of chirp/NB-chirp
The normalized hearing level of the chirp was 28 dB SPL, whereas the normalized hearing level was 26 dB SPL, 23.5 dB SPL, 28.5 dB SPL, and 27.5 dB SPL at 0.5–4 kHz for NB-chirp. All the thresholds of chirp/NB-chirp ABR were calibrated on this basis.
Comparison of response thresholds and time required for chirp/NB-chirp ABR among the three testing states
The ABR thresholds and their differences from the PTA thresholds are shown in Table 1. Figure 1(a) compares the ABR response thresholds evoked by chirp stimuli in the lying, sitting, and writing states. The response thresholds in the writing state are larger than that in the sitting state, which is larger than that in the lying state. All the frequencies showed statistically significant differences between each pair of states. Furthermore, as shown in Figure 1(b), the duration required to achieve reliable threshold results in the writing state is greater than that in the sitting state, which is greater than that in the lying state.

(a) The means and standard errors of the thresholds of chirp/NB-chirp ABR in the three testing states. (b) The mean and standard error of the time required for chirp/NB-chirp ABR in the three testing states.
Response thresholds of chirp/NB-chirp-evoked Kalman-weighted ABR testing under three testing states.

Correction values were determined by subtracting PTA thresholds from chirp/NB-chirp ABR thresholds.
Comparison of response thresholds between different stimuli under three testing states
Figure 2 shows the response thresholds of ABR evoked by each stimulus under the three testing states. Under the lying state, the thresholds of chirp/NB-chirp ABR were lower than those of cABR/tbABR at all frequencies except 1 kHz. There was a statistically significant difference at 0.5 kHz (p = 0.005). Under the sitting state, the thresholds of chirp/NB-chirp ABR were lower than those of cABR/tbABR at all frequencies, and significant differences were observed at 0.5 kHz, 2 kHz and 4 kHz (p = 0.001, 0.023, and 0.033). Under the writing state, the thresholds of chirp/NB-chirp ABR were significantly lower than those of cABR/tbABR except at 0.5 kHz (p = 0.004, 0.03, 0.000, and 0.000 for chirp/click, 1 kHz, 2 kHz, and 4 kHz, respectively).

Comparison of chirp/NB-chirp ABR thresholds with cABR/tbABR under three states.
Figure 3(a) shows the proportion distribution of threshold differences between the writing and lying states for different stimuli. Generally, a higher proportion of threshold differences >10 dB was observed for cABR/tbABR, except at 0.5 kHz. As shown in Figure 3(b), the threshold differences between chirp/NB-chirp ABR in the two states were also generally smaller than those for cABR/tbABR except at 0.5 kHz, and there were significant differences at 0.5 kHz, 2 kHz, and 4 kHz (p = 0.004, 0.044, 0.001). This finding indicates that the chirp/NB-chirp ABR waveform is more stable and has stronger anti-interference ability.
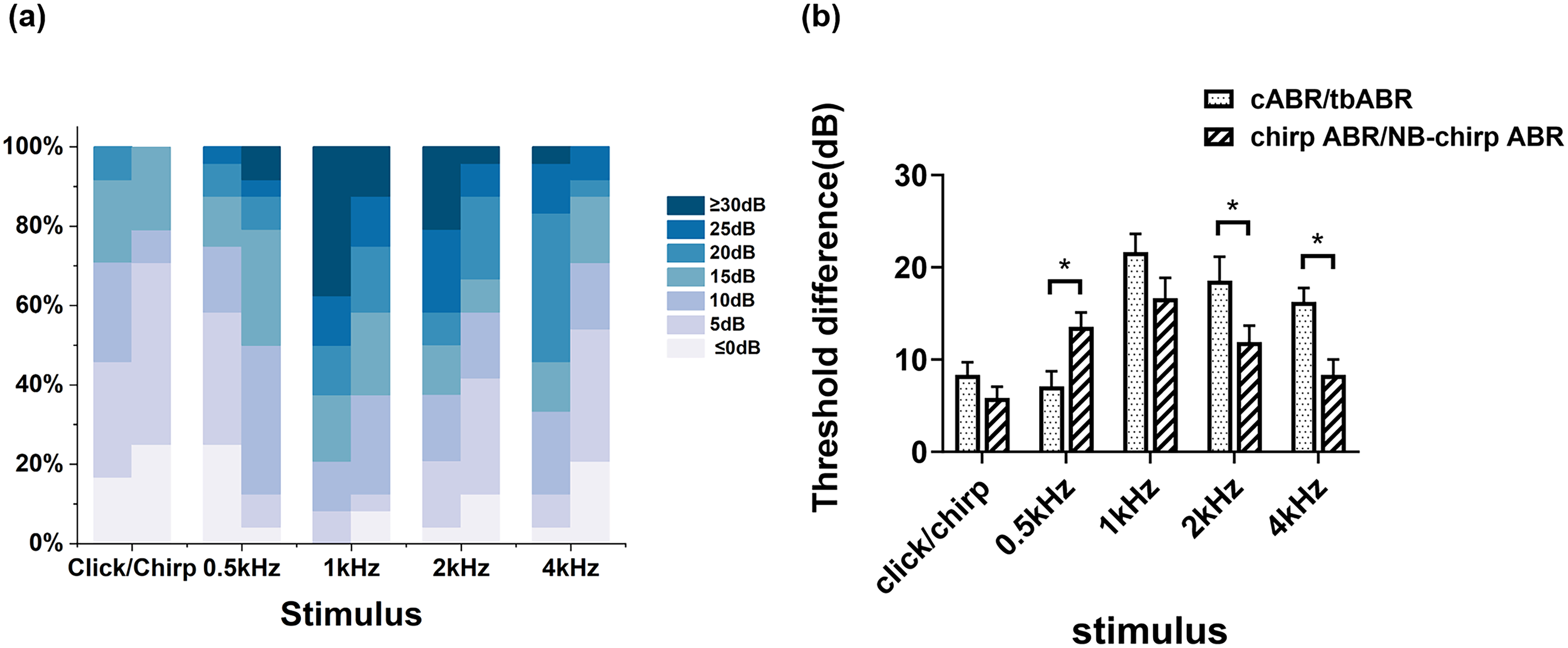
(a) The proportion distribution of threshold differences between the writing and lying states for different stimuli. (b) Threshold differences between the two states for different stimuli. The left column represents cABR/tbABR, whereas the right column represents chirp/NB-chirp ABR.
Comparison of testing time between different stimuli under three testing states
The testing time of the different stimuli under the three testing states is shown in Figure 4. In general, the test time of chirp/NB-chirp ABR under the three testing states is shorter than that of cABR/tbABR. There are significant differences at 2 kHz under the lying state (p = 0.000), at 1 kHz and 2 kHz under the sitting state (p = 0.035, 0.004), and at 4 kHz under the writing state (p = 0.03).

Comparison of the time required to obtain chirp ABR thresholds with cABR/tbABR under three states.
Discussion
The present study applied chirp and NB-chirp stimuli in Kalman-weighted averaging ABR to investigate the thresholds and testing time under different states in a group of normal hearing subjects. Three different testing states were used to simulate the testing state of nonsedated children. Writing was used to mimic the state of children playing with toys, while sitting and watching silent movies or reading books simulated watching animated cartoons, and lying supine represented children resting quietly. The findings indicated that the response thresholds of chirp/NB-chirp ABR varied significantly across the different states in the following order: lying < sitting < writing. Specifically, the thresholds were lowest during the supine position, intermediate during the sitting position, and highest during the writing task. The duration for acquiring thresholds was shortest during the supine position, intermediate during the seated position, and longest during the writing task. Additionally, we compared the results of chirp/NB-chirp ABR with the cABR/tbABR results obtained using the Kalman-weighted averaging algorithm under the three corresponding conditions in our previous studies. The results showed that chirp/NB-chirp ABR generally presented lower thresholds than cABR/tbABR. Simultaneously, we calculated the difference in thresholds between the writing and supine states for each subject, which represents the range of threshold variation across different testing states. The results indicated that, compared with cABR/tbABR, chirp/NB-chirp ABR exhibited a smaller range of threshold variation across different testing states. Moreover, it takes a shorter time to obtain the threshold for chirp/NB-chirp ABR than it takes to obtain the threshold for cABR/tbABR. These results indicate that applying chirp/NB-chirp stimuli for Kalman-weighted ABR testing may further enhance its anti-interference capability.
In our previous study, we conducted Kalman-weighted averaging ABR using click and tone-burst stimuli under three testing conditions in adults to mimic the conditions most often encountered in infants and young children during nonsedated ABR testing. Our results indicate that although Kalman-weighted averaging can reduce the influence of interference on the ABR results across different testing states, the physiological noise introduced by different testing states significantly impacts the thresholds. Therefore, in this study, we applied chirp/NB-chirp stimuli to Kalman-weighted ABR testing in hopes of further enhancing its resistance to interference and reducing the impact of physiological noise introduced by different states of the subjects on the ABR threshold.
This study compared the thresholds of chirp/NB-chirp ABR with those of cABR/tbABR. The results revealed that, under otherwise identical conditions, the threshold of chirp/NB-chirp ABR is lower than that of cABR/tbABR. Moreover, this trend is more pronounced in states with higher physiological noise, such as sitting and writing. Moreover, the chirp/NB-chirp ABR threshold was closer to the behavioral threshold than the cABR/tbABR threshold. Maloff and Hood 24 compared the ABRs elicited by click and chirp using the traditional summating and averaging method in normal-hearing adults and adults with sensory hearing loss and reported that the chirp ABR threshold was closer to the behavioral threshold than the cABR threshold was. Shi et al. 20 reported that among young people with normal hearing, the response threshold of chirp ABR was lower than that of cABR, and the average difference was 8.59 dB. Zheng et al. 19 compared the characteristics of NB-chirp ABR and tbABR in premature infants and found that the threshold of NB-chirp ABR was significantly lower than that of tbABR. Our results further indicate that when Kalman-weighted averaging is used, the chirp/NB-chirp signals still effectively reduce the difference between response and behavioral hearing thresholds. Wilson et al. 26 reported that when both the Kalman-weighted averaging algorithm and NB-chirp were used in 40-Hz sinusoidal auditory steady-state response (s-ASSR), a smaller correction factor than tbABR was used when estimating the low-frequency behavioral thresholds. This finding is consistent with our findings to some extent.
To investigate whether the adoption of chirp/NB-chirp stimuli further enhances the resistance of the Kalman-weighted ABR to interference, we compared the difference in thresholds between the writing and supine states for each subject, which represents the range of threshold variation across different testing states. The range of threshold variation across different testing states of chirp/NB-chirp ABR for each stimulus is generally lower than those of cABR or tbABR. The proportions of the threshold variation range that were 10 dB or less were 79.1%, 50%, 37.5%, 58.4%, and 70.8% for chirp ABR and 0.5–4 kHz NB-chirp ABR, respectively. In comparison, the proportions of the threshold variation range that were 10 dB or less were 70.9%, 75%, 20.8%, 37.6%, and 33.3% for cABR and 0.5–4 kHz tbABR, respectively. These findings indicate that chirp/NB-chirp ABR has better anti-interference capabilities, likely due to a more prominent and stable wave V. Without sedation, infants and young children often cannot remain quiet and still for a long time during ABR tests, and they are inevitably affected by physiological noise from different states of the subjects. Our results suggest that the use of chirp/NB-chirp does help reduce the impact of physiological noise on the ABR threshold. However, comparing thresholds among different states still reveals significant differences across various states for chirp/NB-chirp ABR. Therefore, even when chirp/NB-chirp stimuli combined with Kalman-weighted averaging are used, the influence of different subject states on ABR thresholds cannot be completely eliminated.
Since infants and young children cannot remain quiet for long periods, testing time is also a critical issue. In the present study, the scanning was manually stopped once a stable and discernible wave V was obtained. Consistent with our previous Kalman-weighted study in which click and tone-burst were used, the testing time increased in the following order: lying < sitting < writing. Additionally, there was increased physiological noise for both chirp and NB-chirp stimuli. Moreover, compared with cABR/tbABR, the time required to acquire the chirp/NB-chirp ABR threshold is shorter under otherwise identical conditions. Cebulla et al. 27 conducted chirp and cABR tests on newborns who had undergone hearing screenings. He reported that chirp ABR had a greater amplitude near the threshold, making it easier to detect responses and expectedly resulting in a shorter measurement time. Stuart and Cobb 28 reported that using chirp stimuli to evoke ABR in newborns can significantly improve response amplitude and reduce testing time. Chirp/NB-chirp has also been applied in ASSR to help reduce the testing time. For example, Zhang et al. 29 reported that the average detection time of chirp ASSR binaural is 424 s, whereas the average detection time of tbABR is 1266 s. The use of chirp for ASSR can reduce the testing time by approximately 66%. In summary, using chirp/NB-chirp in ABR and other electrophysiological tests can help save testing time and obtain more audiological clues in limited duration, especially in infants and young children.
This study has several limitations. First, the primary goal of the Vivosonic Integrity system is to conduct ABR testing on infants and young children without sedation. Although studies in adults under various testing states, which mimic different states most often encountered in infants and young children, offer valuable information. It is still necessary to establish normal and correction values for ABR using chirp/NB-chirp together with Kalman-weighted averaging in infants and young children in future studies. Additionally, different testing states, which can vary in terms of noise levels, may correspond to varied correction values. Since the state of infants and young children often changes during the testing process, the classification of testing states is particularly important. Subsequent research should further explore the noise levels associated with different states to provide more objective criteria for state classification during testing. Lastly, it is important to mention that we did not perform a formal sample size calculation or power analysis in our study. This may affect the generalizability and statistical power of our results. Future studies should consider conducting a thorough sample size calculation to ensure adequate power and more reliable findings.
Conclusion
The present study demonstrated that chirp/NB-chirp ABR using the Kalman-weighted averaging algorithm demonstrated better resistance to interference, a smaller difference between response and hearing thresholds, and shorter testing time under various nonsedated conditions than did Kalman-weighted cABR/tbABR. However, the physiological noise generated by the brain and muscles under different testing states still affects the response thresholds and testing durations. Thus, it is still necessary to establish normal and correction values separately for each testing state of the subjects.
Footnotes
Acknowledgements
This work was supported by the National Natural Science Foundation of China [Grant Nos. 82171156 and 32371051] and National High Level Hospital Clinical Research Funding [Grant Nos. 2022-PUMCH-A-002 and 2022-PUMCH-B-095].
Ethical considerations
The study was approved by the Ethics Review Committee of Beijing Union Medical College Hospital (Protocol ZS-2861, approved 23 March 2021). All participants provided written informed consent prior to enrollment.
Author contributions/CRediT
Conceptualization, Yingying Shang; methodology, Jiayan Yang, Suju Wang, and Yingying Shang; validation, Wen Sun; formal analysis, Yingying Shang and Daofeng Ni; investigation, Jiayan Yang, Suju Wang, and Xinheng Ru; resources, Zhiyuan Zhang and Hong Jiang; data curation, Jiayan Yang and Suju Wang; writing – original draft preparation, Jiayan Yang; writing – review & editing, Jiayan Yang and Yingying Shang; supervision, Zhiqiang Gao; project administration, Yingying Shang and Zhiqiang Gao; funding acquisition, Yingying Shang. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 32371051 and 82171156) and National High Level Hospital Clinical Research Funding (grant number 2022-PUMCH-A-002 and 2022-PUMCH-B-095).
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The raw data supporting the conclusions of this article will be made available by the authors on request.
