Abstract
Objectives
Group 1 pulmonary arterial hypertension (PAH) causes increased vascular resistance, right heart failure, and reduced survival. High-altitude PAH survival data are limited despite chronic hypoxia's potential impact on disease progression and risk stratification. The objective was to evaluate survival stratified by the European Society of Cardiology/European Respiratory Society (ESC/ERS) baseline risk score, REVEAL risk score 2.0, and etiologies in patients with PAH at high altitudes.
Methods
A retrospective cohort study was conducted to evaluate functional and hemodynamic variables and risk stratification using the ESC/ERS baseline risk score and REVEAL risk score 2.0. One-year and five-year survival rates were determined from admission to the pulmonary hypertension program until death or the end of follow-up.
Results
A total of 188 patients were included. The overall survival rate at one year was 95.8%, and at five years it was 86.8%. Precisely, 50% (95 out of 188) had congenital heart disease, 34% (63 out of 188) had idiopathic, and 13% (24 out of 188) had connective tissue disease. Risk stratification using REVEAL risk score 2.0 classified 58% (109 out of 188) of patients as low risk, 26% as intermediate risk, and 16% (30 out of 188) as high risk. Using the ESC/ERS baseline risk score, Stratification indicated 78% (146 out of 188) low risk, 21% (40 out of 188) intermediate risk, and 1% (2 out of 188) high risk. High risk according to ESC/ERS baseline risk score (p < 0.001) and REVEAL risk score 2.0 (p < 0.001) was associated with lower survival. Idiopathic etiology was associated with a lower survival rate (p = 0.011).
Conclusion
The survival of patients with PAH at a high-altitude expert center was 95.8% at one year and 86.8% at five years. Risk stratification using the REVEAL risk score 2.0 and the ESC/ERS baseline risk score was associated with higher mortality in patients classified as high risk.
Introduction
Pulmonary hypertension is characterized by a mean pulmonary artery pressure (mPAP) exceeding 20 mmHg and is classified into five groups.1,2 From a hemodynamic perspective, pulmonary arterial hypertension (PAH) is defined by a precapillary pattern, evidenced by a wedge pressure ≤15 mmHg and pulmonary vascular resistance (PVR) >3 wood units (WU), provided that other causes of precapillary pulmonary hypertension, such as chronic thromboembolic disease or pulmonary pathologies, are excluded. 2 Group 1 PAH is associated with high morbidity and mortality, with survival rates depending on the underlying etiology.1–3 Selective vasodilator therapies aimed at these patients have been shown to improve survival rates, as indicated by the REVEAL risk score 2.0, reporting rates of 85% at one year and 57% at five years, 1 significantly surpassing the survival rates observed in national health registries from 1980, which were 68% at one year and 36% at five years.2,3
In the 1980s, therapeutic options for PAH were limited to supportive care, including oxygen therapy and anticoagulation, resulting in poor survival rates.4–6 The introduction of prostacyclin analogs, followed by endothelin receptor antagonists and phosphodiesterase-5 inhibitors, significantly improved patient outcomes.4–6 These advancements have led to better disease management and increased survival, emphasizing the importance of ongoing research in PAH.4,6,7 Among patients diagnosed with congenital heart disease, the highest survival rates are observed at 88% at one year and 74% at five years.1,7 In contrast, those with PAH associated with connective tissue disease have the lowest survival rates, with 79% at 12 months and 43% at five years1,2,8; this difference may be explained by the fact that patients are diagnosed at an advanced stage of the disease.1,3,8 The establishment of specialized groups for managing complex pulmonary vascular diseases has been crucial for reducing mortality, thanks to early diagnosis, risk stratification, and the initiation of appropriate therapy.3,8
The survival of patients with PAH living at high altitudes, defined as ≥2500 m above sea level, is not well described, nor are the functional, clinical, and hemodynamic characteristics that determine mortality risk in these individuals fully understood.3,8,9 The aim of this study was to evaluate the survival of patients with PAH at high altitudes, analyzing clinical, functional, and hemodynamic characteristics, and comparing risk stratification using the European Society of Cardiology/European Respiratory Society (ESC/ERS) baseline risk score 4 and REVEAL risk score 2.0. 1
Methods
A retrospective cohort study was conducted to evaluate one- and five-year survival rates and to describe the functional, clinical, and hemodynamic characteristics used in the ESC/ERS baseline risk score and REVEAL risk score 2.0 at the time of enrollment in the pulmonary hypertension program.1,4 The primary objective of our study was to assess the survival of patients with PAH, stratified by age, sex, risk scores, and etiological subtypes.
The study included patients with PAH treated at the Fundación Neumológica Colombiana in Bogotá, Colombia (located at an altitude of 2625 m above sea level) between 2017 and 2023. The study was conducted in accordance with the principles of the current Helsinki Declaration of 1975, as revised in 2024, as well as local, regional, and international regulations pertaining to clinical research, including Colombian Law on Biomedical Research. Ethical approval was obtained from the Medical Ethics Committee of the Fundación Neumológica Colombiana Ethics Committee (code: 289_27_octubre_2023). All personal data of patients have been deleted. This was a retrospective study, and informed consent was not required. The reporting of this study conforms to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 10
Eligibility criteria
The study included patients aged over 18 years with a diagnosis of PAH, treated in the pulmonary hypertension program, and diagnosed through right heart catheterization. Patients were included when residing permanently at altitudes above 2500 m above sea level during follow-up, regardless of the duration of the disease. Those with incomplete medical histories and the absence of essential functional, clinical, and hemodynamic data necessary for calculating the ESC/ERS baseline risk score and REVEAL risk score 2.0,1,4 as well as patients with mixed-etiology pulmonary hypertension, were excluded. This was a retrospective study in which patients were included consecutively.
Collected variables
The diagnosis of Group 1 pulmonary hypertension was defined by mPAP > 25 mmHg, PVR > 3 WU, and pulmonary artery wedge pressure (PAWP) ≤15 mmHg, according to the ESC/ERS guidelines. 3 Variables considered included age, sex, type of prostanoid, prostanoid dosage, and classification of Group 1 pulmonary hypertension by etiology. The risk was assessed based on the simplified version of the ESC/ERS baseline risk score and REVEAL risk score 2.0,1,4 including hemodynamic variables: pulmonary artery systolic pressure (PSAP), mPAP, PVR, PAWP, right atrial pressure (RAP), cardiac index (CI), and mixed venous oxygen saturation (SvO2). Additionally, the functional class was described according to the New York Heart Association (NYHA), along with B-type natriuretic peptide (BNP) and the Saint George's Respiratory Questionnaire (SGRQ). The 6-minute walk test (6MWT) was conducted on a flat, level, 30 m long course within the healthcare institution, free of obstacles and pedestrian movement.
Data collection and evaluation were conducted using a database obtained directly from electronic medical records and diagnostic test results. To identify the follow-up status of the patients, the General System of Health Security Resource Manager (ADRES) was consulted to report survival status. Precise eligibility criteria were established to mitigate selection bias. To reduce transcription bias, at least two team members reviewed the information, and in cases of inconsistency, a third team member reviewed the data and made the final decision. Furthermore, all variables were clearly defined, and the investigators responsible for data collection received prior training.
Statistical analysis
Qualitative variables were summarized as frequencies and percentages. Quantitative variables were presented as means and standard deviations if normally distributed or medians and interquartile ranges if not normally distributed. Overall survival was analyzed using the Kaplan–Meier estimation, 11 calculating survival rates at one year and five years. Survival was assessed from the date of enrollment in the pulmonary hypertension program until the event of interest (death) or the end of the follow-up period. Survival analysis was stratified by risk score (REVEAL risk score 2.0 and ESC/ERS baseline risk score),1,4 age (<65 years and >65 years), sex (male and female), and etiological subtype, including idiopathic, congenital heart disease, and miscellaneous causes (e.g. hereditary, connective tissue disease, human immunodeficiency virus (HIV) infection, portal hypertension, and pulmonary vasoreactivity test responders). Differences between strata were evaluated using the log-rank test—additionally, exploratory bivariate analysis compared age, sex, and risk scores between survivors and non-survivors. Qualitative variables were compared using the chi-square test, while quantitative variables were compared using Student's t-test or Mann–Whitney U test, depending on their distribution. A p-value < 0.05 was considered statistically significant.
The statistical explorations were primarily exploratory, and no sample size calculation was performed. However, it was estimated that approximately 150 subjects would be included in the study. Data were transcribed into the Research Electronic Data Capture (REDCap) software.12,13 Subsequently, data analysis was performed using IBM SPSS Statistics version 25 (IBM Corp., Armonk, NY, USA) and STATA version 17 (StataCorp LLC, College Station, TX, USA).
Results
General characteristics of the study population at baseline
A total of 188 patients were included (Figure 1), with a mean age of 41 years (SD = 13.62), of which 85.6% (161 out of 188) were women. The overall survival rate at one year was 95.8% (163 out of 188), and at five years it was 86.8% (25 out of 188). Of the patients, 50% (95 out of 188) had congenital heart disease, 34% (63 out of 188) had idiopathic PAH, and 13% (24 out of 188) had connective tissue disease (Table 1). Hereditary PAH patients were identified through family anamnesis. However, specific information regarding other affected family members was not available. Additionally, the hereditary PAH patients included in our dataset were unrelated. Among patients with congenital heart disease, 32% (30 out of 95) had Eisenmenger syndrome, and 28% (27 out of 95) had closure of the congenital disability (Supplemental Table 1). Our cohort did not have any transplant patients. Pharmacological treatment at baseline was disclosed in Supplemental Table 2.
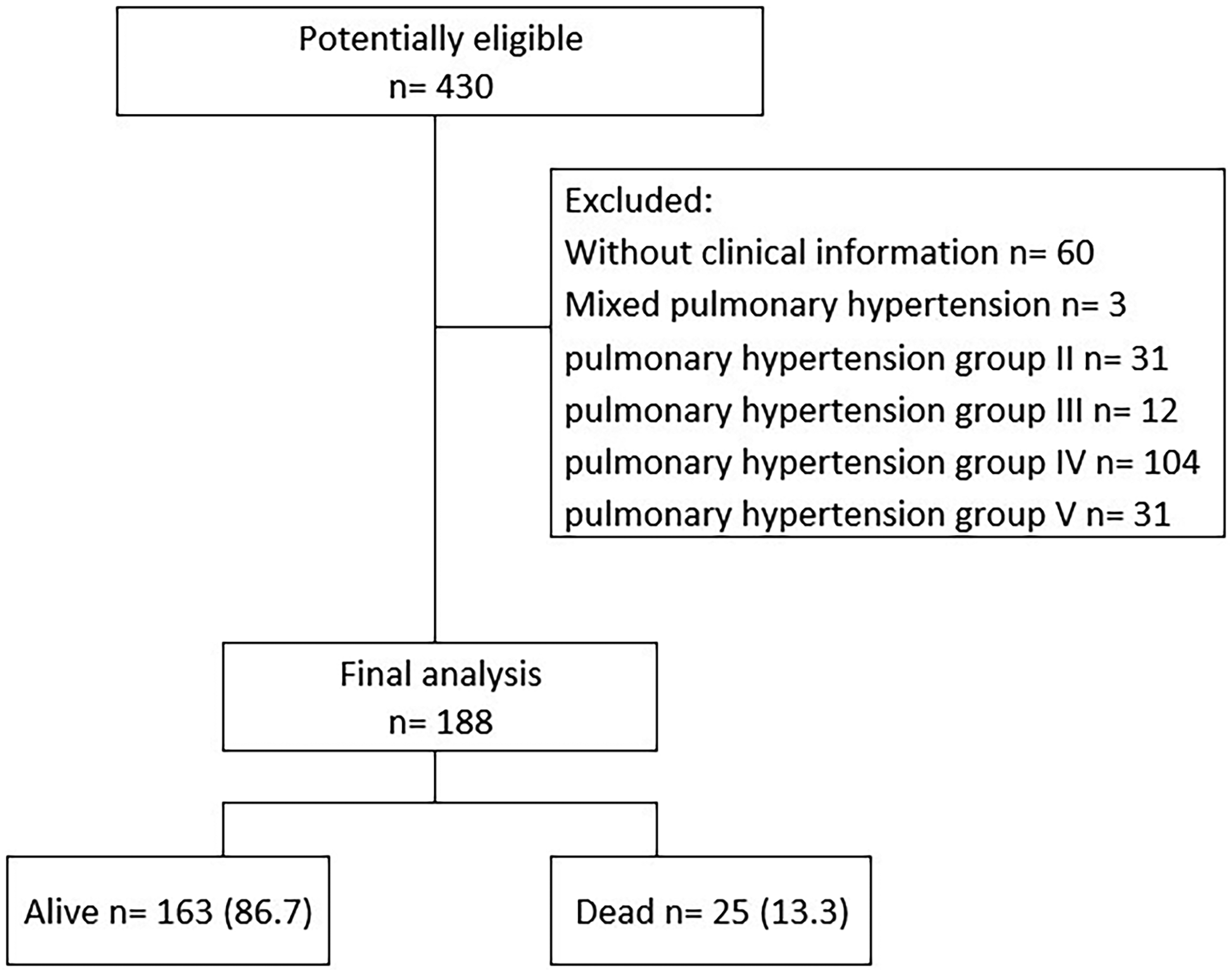
Study flowchart.
Baseline characteristics of the study patients.

m: mean; SD: standard deviation; n: numbers; %: percentage; kg: kilograms; cm: centimeters; m2: square meters; HIV, human immunodeficiency virus; PDE5i: phosphodiesterase 5 inhibitor; iEndothelin I: endothelin I inhibitor.
Clinical and functional characteristics at baseline
The mean SGRQ score was 41.1 (SD = 18.3) in patients who survived and 55.2 (SD = 21.4) in patients who died (p < 0.001) (Table 2). The mean distance walked by survivors was 489.1 m (SD = 101.2), compared to 434.4 m (SD = 120.1) in the deceased group (p = 0.016).
Functional variables at baseline.

m: mean; SD: standard deviation; SGRQ: Saint George's Respiratory Questionnaire; NYHA: New York Heart Association functional classification; FVC: forced vital capacity; FEV1: forced expiratory volume in the first second; CO % diffusion: carbon monoxide diffusion percentage; PaCO2: arterial carbon dioxide pressure; HCO3: serum bicarbonate; PaO2: arterial oxygen pressure; SaO2: arterial oxygen saturation; 6MWT: 6-minute walk test; HR: heart rate; SpO2: pulse oxygen saturation.
Hemodynamic characteristics at baseline
Left ventricular ejection fraction was 59.5% (SD = 6.87) in patients who survived versus 56.3% (SD = 9.15) in those who died (p = 0.041) (Table 3). The PSAP was 14.6 mmHg lower in survivors (75 vs. 89.6; p = 0.021). The mean RAP was 10.6 mmHg (SD = 5.23) in survivors compared to 14.6 mmHg (SD = 5.23) in deceased patients (p < 0.001). In survivors, the CI was 0.8 L/min/m² higher (4.6 vs. 3.8; p = 0.002) compared to those who died.
Right catheterization and echocardiographic variables at baseline.

m: mean; SD: standard deviation; LVEF %: left ventricular ejection fraction percentage; %: percentage; TAPSE: tricuspid annular plane systolic excursion; mm: millimeters; cm3: cubic centimeter; PSAP: pulmonary artery systolic pressure; mmHg: millimeters of mercury; PAPd: pulmonary artery diastolic pressure; mPAP: mean pulmonary artery pressure; mRAP: mean right atrial pressure; PAWP: pulmonary artery wedge pressure; RVP: right ventricular pressure; WU: wood units; CI: cardiac index; SvO2%: mixed venous oxygen saturation; BNP: B-type natriuretic peptide; ng/mL: nanograms per milliliter.
ESC/ERS and REVEAL risk score 2.0 Risk Stratification at baseline
Risk stratification using REVEAL risk score 2.0 classified 58% (109 out of 188) of patients as low risk, 26% (26 out of 188) as intermediate risk, and 16% (30 out of 188) as high risk. Using the ESC/ERS baseline risk score, stratification indicated 78% (146 out of 188) low risk, 21% (40 out of 188) intermediate risk, and 1% (2 out of 188) high risk (Table 4).
REVEAL risk score 2.0 and ESC/ERS risk scores at baseline.

n: number; %: percentage; ESC/ERS: European Society of Cardiology/European Respiratory Society.
Survival
The overall survival was 95.8% (180 out of 188) at one year and 86.8% (163 out of 188) at five years (Figure 2). High risk according to ESC/ERS baseline risk score (p < 0.001) and REVEAL risk score 2.0 (p < 0.001) was associated with lower survival (Figures 3 and 4). Idiopathic etiology was associated with a lower survival rate (Figure 5; p = 0.011). Sex and age-stratified survival analyses were described in Supplemental Figures 1 and 2.

One-year and five-year survival.

Survival analysis by European Society of Cardiology/European Respiratory Society (ESC/ERS) baseline risk score.

Survival analysis by REVEAL risk score 2.0.

Survival analysis by etiology of pulmonary arterial hypertension group I.
Discussion
This study described the survival of patients with PAH at a high-altitude reference center, which was 95.8% at one year and 86.8% at five years. Clinical, hemodynamic, and functional variables associated with lower survival at enrollment in the pulmonary hypertension program included the NYHA, SGRQ score, PSAP, mean RAP, mPAP, CI, and 6MWT results. Risk stratification using the REVEAL risk score 2.0 and the ESC/ERS baseline risk score was associated with increased mortality at high-risk levels. The survival rates described in this study can be attributed to timely diagnosis, appropriate risk stratification, strict follow-up, and the initiation of suitable treatment provided by a multidisciplinary pulmonary hypertension team, as widely reported in the literature.3,7–9,14
Chronic exposure to high altitude induces changes due to increased PVR secondary to hypoxia-induced vasoconstriction and remodeling of the pulmonary arterioles.15–17 Therefore, individuals already diagnosed with pulmonary hypertension from any etiology may experience disease progression due to high-altitude exposure. It would be expected that survival would be lower in this patient group; however, in this study, survival was high and more significant compared to survival rates described in other pulmonary hypertension registries, such as those reported by Korsholm et al. 7 in Denmark, which were 86% at one year and 65% at five years. It was also higher than the survival described by Mazzei et al. 8 in Argentina, where a five-year survival rate of 52% was reported for males and 62% for females, with females showing lower survival rates, regardless of the subgroup of PAH.
The impact of altitude on cardiovascular mortality varies depending on elevation. As altitude increases, barometric pressure and oxygenation decrease.16,17 Observational studies have shown that living at moderate altitudes (1000–2000 m above sea level) is associated with lower mortality rates from coronary artery disease and stroke, possibly due to moderate hypoxia and its beneficial effects on cardiovascular adaptation.18–21 However, extreme altitudes (>3000 m above sea level) may increase overall mortality due to harsher environmental conditions.18,20,21 Additionally, altitude exposure has multiple physiological effects that can negatively impact individuals with chronic respiratory diseases. 22 Hypoxia-induced hyperventilation increases dyspnea and airway reactivity, affecting exercise tolerance and quality of life.22–24 Furthermore, severe hypoxemia at high altitudes may increase the need for chronic oxygen therapy. 24 Therefore, we believe that in clinical practice, the recommendation that patients migrate to lower altitudes depends on the severity of the disease, the oxygenation status, and the adaptive capacity of each individual.
Our results indicate a five-year survival rate of 86.8%, exceeding the 84.6% reported in COMPERA for young patients without comorbidities and significantly higher than that observed in groups with greater comorbidity burdens. 25 This difference may be explained by the lower mean age of our population and a lower burden of systemic comorbidities in our patients. The predominance of congenital heart disease in our cohort, in contrast to the higher prevalence of metabolic and cardiovascular diseases in COMPERA, could contribute to the observed difference in survival. 25 In terms of prognostic factors, our population showed a significant relationship between low survival and high-risk scores in REVEAL risk score 2.0 and ESC/ERS baseline risk score, such as COMPERA, where the phenotype with the worst prognosis (older patients with multiple comorbidities and low DLCO) exhibited the poorest treatment response and lowest survival. 25 These findings underscore the importance of considering regional and phenotypic differences when interpreting survival data. Altitude-related physiological adaptations and the distinct comorbidity profiles of different populations may influence the prognosis of PAH.22–24,25
Hemodynamic characteristics were assessed using right heart catheterization and echocardiography. In this study, PSAP was higher in the group with the lowest five-year survival. Likewise, a lower CI and higher mean RAP were observed in patients with reduced survival, reflecting greater disease severity and their inclusion in established mortality risk scores.25–29 However, due to the study design and the limited number of deaths, identifying independent risk factors through multivariate analysis was beyond the study's scope. Instead, survival was stratified based on key clinical variables in patients with PAH, providing a more detailed evaluation of how different clinical and demographic factors influence survival trends.30–36 Additionally, stratification helps mitigate the risk of overfitting in small-event multivariate models while still yielding clinically relevant insights into patient subgroups.
As for clinical variables, World Health Organization functional class and pulmonary function tests can serve as prognostic and survival predictors at the time of diagnosis and during clinical follow-up.37–39 In this study, no significant differences were found regarding spirometry values, including FEV1 and forced vital capacity (FVC), nor in DLCO values; however, the mean DLCO at enrollment in the program was lower. In the 6MWT, patients who walked shorter distances exhibited lower survival, possibly associated with a more significant increase in pulmonary artery pressures, a finding corroborated by other studies where the distance covered in the 6MWT is related to mortality outcomes, indicating that this parameter could be considered as a therapeutic target in rehabilitation programs.40,41
The potential impact of altitude on the 6MWT and functional status is an important consideration, as hypoxia at high altitudes could theoretically lead to lower exercise capacity and worse risk scores.42–44 However, despite these physiological challenges, a high proportion of our patients were classified as low risk according to REVEAL risk score 2.0 and ESC/ERS baseline risk score. This may be attributed to long-term adaptation to chronic hypoxia, optimal treatment strategies, and comprehensive multidisciplinary management. Additionally, while the 6MWT is a key component of risk assessment, other hemodynamic and clinical variables, such as CI and RAP, also play a crucial role in determining overall risk.42–44 These factors may explain why, despite potential altitude-related limitations in functional capacity, a significant proportion of patients remained in the low-risk category.
Ultimately, additional research is necessary to validate risk scores at high altitudes due to the need for concordance between current stratification scales (REVEAL risk score 2.0 and ESC/ERS baseline risk score) regarding the number of patients classified as low, intermediate, or high risk.44–46 Nevertheless, the observed survival was consistent, showing higher survival rates in patients classified as low risk and lower survival in those classified as high risk, regardless of the scoring system used.45–47
Previous studies have shown that in patients with PAH temporarily exposed to high altitude, exercise tolerance decreases due to reduced arterial oxygenation and increased ventilation and ventilatory inefficiency.48,49 While our study did not evaluate changes in exercise capacity at different altitudes, the findings suggest that patients with PAH may adapt to long-term high-altitude hypoxia, provided they receive timely diagnosis, appropriate treatment, and strict follow-up.48,49 Although PAH at high altitudes may present additional challenges due to environmental hypoxia, our results highlight that a multidisciplinary approach and individualized management can contribute to survival rates comparable to those reported at other altitudes. Future comparative studies across different altitudes could help better understand the impact of hypoxia on PAH progression and optimize therapeutic strategies for these patients.
In our study, a possible explanation for the high proportion of patients classified as low risk is the heightened clinical suspicion of pulmonary hypertension in our specialized medical center, which may lead to earlier diagnosis. Additionally, physiological adaptation to chronic hypoxia at high altitudes could alter hemodynamic parameters, potentially affecting the prognostic accuracy of current risk stratification models.16–24 As a result, a greater proportion of patients may meet low-risk criteria despite ongoing disease progression, which may differ from findings in previously published studies and populations living at sea level.20,24,25 Future research should focus on validating risk scores in our population to ensure precise prognostication and optimal clinical decision-making.
Limitations
Among the study's limitations is the exclusive inclusion of patients from a single hospital, which may restrict the external validity of the findings. However, given that the characteristics of our population are very similar to those of other patient series and that a standard technique was used, the impact of this limitation is likely minimal. The study's observational nature carries the risk of confounding by uncontrolled factors. Strengths of our study include the rigor in data collection from medical records and the verification of the diagnosis of PAH. Additionally, the study was conducted in a multidisciplinary center experienced in pulmonary hypertension, ensuring timely diagnosis and appropriate follow-up during the study period.
A high proportion of our cohort had PAH associated with congenital heart disease, a subgroup known for its relatively better survival when reaching adulthood.50,51 This could have contributed to the overall high survival rates observed. However, our study included patients with different PAH subtypes, and survival was also influenced by factors such as early diagnosis, appropriate risk stratification, and comprehensive multidisciplinary management. While the presence of PAH associated with congenital heart disease may have played a role in the favorable outcomes, our findings highlight the importance of specialized care in improving survival at high altitudes.
It is worth noting that the research was conducted in a high-complexity institution, which introduced a selection bias by not uniformly including patients across all stages of the disease. Therefore, prospective studies are required to evaluate survival rates in more homogeneous subgroups. To mitigate the risk of biases, measures such as training of data collection personnel, manuscript preparation according to the STROBE checklist, 52 use of standard techniques for procedures, and adjustment for potential confounding variables were implemented.
Conclusion
The survival of patients with PAH at a high-altitude expert center was 95.8% at one year and 86.8% at five years. Risk stratification using the REVEAL risk score 2.0 and the ESC/ERS baseline risk score was associated with higher mortality in patients classified as high risk. Clinical, hemodynamic, and functional variables at enrollment in the pulmonary hypertension program were linked to lower survival. However, further studies are needed to better evaluate the relationship between these risk factors and mortality.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251336066 - Supplemental material for Survival rates at one and five years for patients with group 1 pulmonary arterial hypertension at high altitude: A retrospective cohort study
Supplemental material, sj-docx-1-sci-10.1177_00368504251336066 for Survival rates at one and five years for patients with group 1 pulmonary arterial hypertension at high altitude: A retrospective cohort study by Rafael Conde-Camacho, Eduardo Tuta-Quintero, Alirio Bastidas-Goyes, Luis F Giraldo-Cadavid, Oscar Barón and Angelica Mora in Science Progress
Footnotes
Acknowledgments
The authors are most thankful for the Universidad de La Sabana and Fundación Neumológica Colombiana.
Ethical considerations
The study was conducted in accordance with the principles of the current Helsinki Declaration of 1975, as revised in 2024, as well as local, regional, and international regulations pertaining to clinical research, including Colombian Law on Biomedical Research. Ethical approval was obtained from the Medical Ethics Committee of the Fundación Neumológica Colombiana Ethics Committee (code: 289_27_octubre_2023).
Consent to participate
This is a retrospective database analysis study and the consent to participate is waived by the institutional review board.
Author contributions/CRediT
RCC, ETQ, ABG, LFGC, OBC, and AM contributed to the conception and design. They also supervised the whole process, data collection, analysis, and interpretation of the patient data. ETQ and LFGC wrote major parts of the manuscript, and OBC revised it. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Universidad de La Sabana (Grant: MEDEsp-107-2023) and Fundación Neumológica Colombiana (Grant: 202310-28931).
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
