Abstract
Objective
The aim of this study was to assess the ability of plasma InterLeukin-6 (IL-6) monitoring to predict the failure to switch from volume-controlled ventilation to spontaneous ventilation (SV) in patients with COVID-19-related acute respiratory distress syndrome (ARDS).
Methods
We conducted an observational, single-center and prospective cohort study in the medico-surgical intensive care unit of Avignon Hospital Center. Participants were adult patients requiring invasive mechanical ventilation for COVID-19-related ARDS between August 2021 and August 2022, who were eligible for switching from volume-controlled ventilation to SV.
Results
Among the 35 patients included in the study, 13 (37%) successfully switched from controlled ventilation to SV, while 22 failed (63%). In the failure group, mean plasma IL-6 levels were higher than in the successful group from hour 0 (defined as the moment of the switch to SV mode) to 48 h. However, differences between groups became significant from 24 h (362.8 vs. 33.6 pg/mL, P = 0.002). Interestingly, between-group differences in plasma C-reactive protein (CRP) levels were only significant between groups from 48 h (129.3 vs. 52.2 mg/L, P = 0.017). Finally, IL-6 and CRP had a similar ability to predict the failure to switch to SV mode: area under the receiving operative curves 0.763 [95%CI: 0.633-0.893] and 0.753 [95%CI: 0.595-0.911], respectively (P = 0.87).
Conclusions
IL-6 and CRP are inflammatory biomarkers predictive of failure to switch to SV mode in COVID-19 ARDS patients. Our results showed that IL-6 can detect failure earlier than CRP. However, larger multicenter studies are needed to confirm our results, particularly in other ARDS models.
Introduction
During the four waves of the coronavirus disease 2019 (COVID-19) pandemic in France between February 2020 and September 2021, 460,000 patients were hospitalized for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection of varying severity. 1 Of them, 116,000 died mainly from acute respiratory distress syndrome (ARDS). The intensive care unit (ICU) surge in patients suffering from ARDS encouraged researchers to rapidly explore the pathophysiology of COVID-19, subsequently demonstrating that COVID-19-related ARDS was triggered by a cytokine storm.2–4
In the hope of finding novel therapeutic options to continue the advances made in ARDS management over the last decades, 5 research on COVID-19 patients focused on the cytokine storm.2–4 Among various cytokines, InterLeukin-6 (IL-6) received particular attention, as treatments such as tocilizumab were already available.2–4 Prior to the publication of the RECOVERY study, 6 which demonstrated the benefit of IL-6 antagonists on mortality, some studies showed that a higher plasma IL-6 concentration was associated with a poor prognosis in patients with severe COVID-19 such as ICU admission 7 or death.4,8 Interestingly, some authors9–11 reported that lower plasma IL-6 concentrations were also associated with successful weaning from mechanical ventilation.
In line with these findings, we hypothesized that IL-6 could be a useful biomarker for identifying COVID-19 patients who will fail to switch from volume-controlled to spontaneous ventilation (SV) mode. This study therefore aimed to assess the ability of elevated plasma IL-6 concentrations to predict the failure to switch from volume-controlled ventilation to SV mode in mechanically ventilated COVID-19 patients. The early identification of these patients who will go on to fail the weaning process could limit the risk of patient self-inflicted lung injury (P-SILI), which can worsen the pre-existing lung injury and lead to weaning failure. 12 However, to date, no study has investigated the correlation between IL-6 levels and successful switching to SV. Furthermore, if our results are positive and confirm IL-6 as a useful biomarker, they could serve as a model for broader investigations into other types of ARDS, even more so in this post COVID era, as there are many other diseases that cause ARDS.
Methods
Study design and setting
We conducted an observational, single-center and prospective cohort study in the medico-surgical ICU of Avignon General Hospital Center between August 2021 and August 2022. During each wave of COVID-19, our 20-bed ICU increased its capacity up to 30 beds. The same medical and paramedical teams managed the additional beds. This study was performed according to the COVIL-6 protocol (NCT05330845 https://clinicaltrials.gov/). The reporting of this study conforms to the STROBE guidelines. 13
Participants
During the study period, we screened all consecutive patients admitted to our ICU for COVID-19.14,15 Adult patients requiring invasive mechanical ventilation for COVID-19-related ARDS who were eligible for first weaning initiation from volume-controlled ventilation to SV mode were included.14,15 We excluded patients under the age of 18, those under legal guardianship, and pregnant women. We also excluded patients treated with tocilizumab, those requiring extra-corporeal membrane oxygenation (ECMO), those transferred to another hospital, and those who died before the first attempt to switch to SV.
Data collection and variables
All patient data were anonymized so that patients could not be identified in any way. We performed as-treated analysis and did not exclude patients with missing data. We included all patients for analysis. In Tables 1 and 2, we specify the number of patients with missing data for each variable.
Clinical features of the study population and difference between patients who successfully switched from volume-controlled ventilation to spontaneous ventilation mode and those who failed.

*P = p-value: Student t test, Mann-Whitney or Wilcoxon tests for Mean comparisons; χ² tests or Fisher exact tests for the analysis of contingency tables. In bold: significant differences.
95%CI: 95% confidence interval; BMI: body mass index; bpm: beats per minute; cm: centimeter; cmH2O; centimeter of water; FiO2: fraction of inspirated oxygen; HX: data at time (hour) X; kg: kilogram; mmHg: millimeter of mercury; mL: milliliter; NA: not applicable; NMBA: neuromuscular blocking agent; PEEP: positive end expiratory pressure; P/F ratio: partial pressure of arterial oxygen/fraction of inspirated oxygen; RASS: Richmond Agitation Sedation Scale; RR: respiratory rate; SAPS II: Simplified Acute Physiology Score II; SBP: systolic blood pressure; SV: spontaneous ventilation; Vt: tidal volume.
Biological features of the study population and difference between patients who successfully switched from volume-controlled ventilation to spontaneous ventilation mode and those who failed.
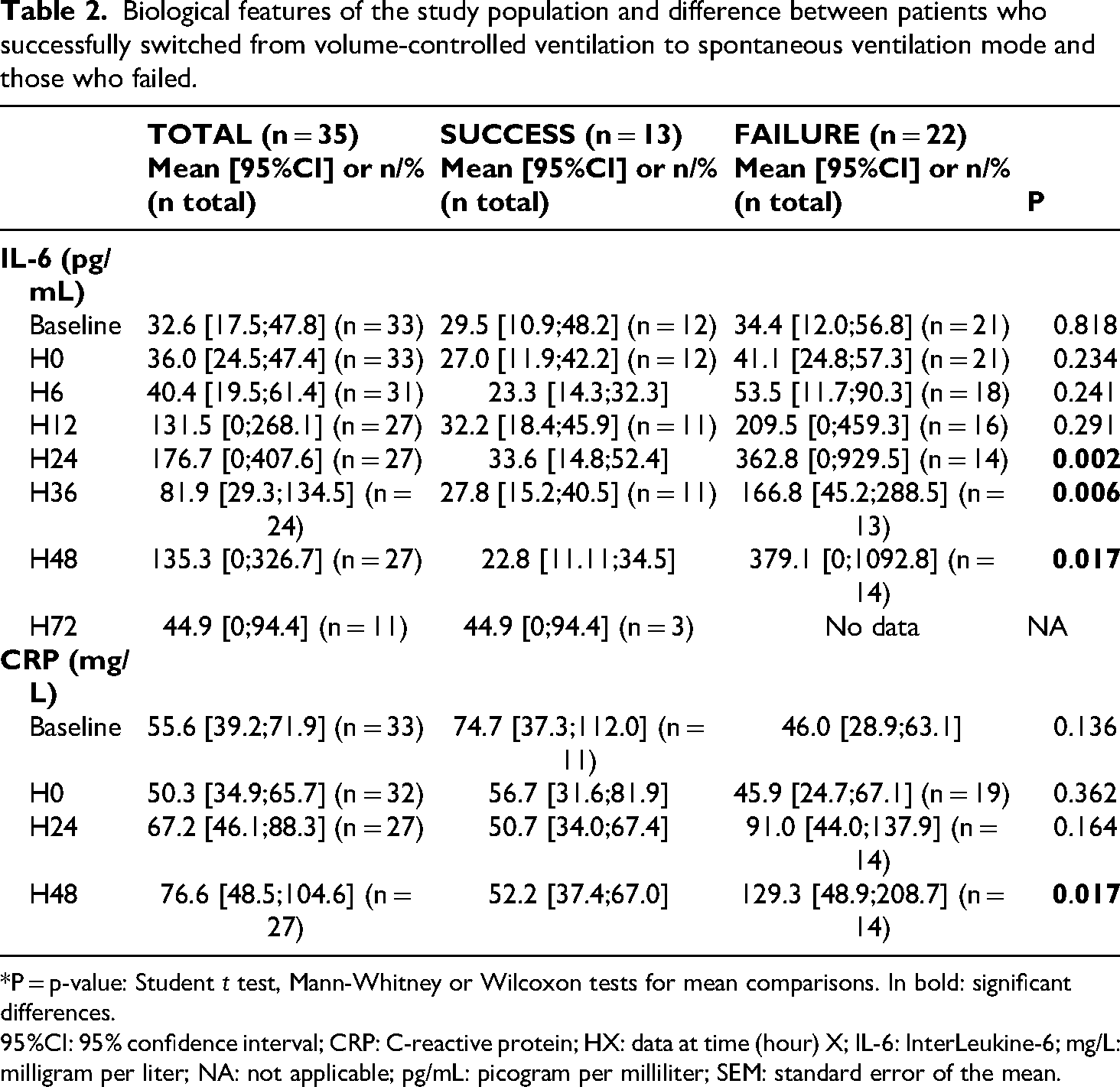
*P = p-value: Student t test, Mann-Whitney or Wilcoxon tests for mean comparisons. In bold: significant differences.
95%CI: 95% confidence interval; CRP: C-reactive protein; HX: data at time (hour) X; IL-6: InterLeukine-6; mg/L: milligram per liter; NA: not applicable; pg/mL: picogram per milliliter; SEM: standard error of the mean.
We collected general demographic parameters: sex, age, comorbidities, body mass index (BMI), and Simplified Acute Physiology Score (SAPS II).16,17 We also collected parameters specific to COVID-19 ARDS patients in the ICU: tidal volume (Vt), positive end expiratory pressure (PEEP), and respiratory rate (RR) corresponding to the ventilator settings; invasive mechanical ventilation duration and ventilation mode; delay between discontinuation of neuromuscular blocking agents (NMBAs) and initiation of SV mode; number of ventilation weaning attempts; number of prone positioning sessions; SARS-CoV-2 variant of concern and SARS-CoV-2 antibody level in plasma. The occurrence of superinfections [day 1-2, day 2-3], fluid overload [day 1-2, day 2-3], and lung injury worsening [day 1-2, day 3-4] was noted. Medical and demographic data were collected as part of current practice and not gathered specifically for the study.
We also collected the biomedical features of the study population according to the protocol set up in the department to respond to our hypotheses. Thus, IL-6 and C-reactive protein (CRP) were collected specifically for this study in order to address our main hypothesis about the predictive ability of these biomarkers. We performed IL-6 and CRP blood tests according to a standardized workup for the study. Thus, for plasmatic assays, we defined H0 as the moment of the switch from volume-controlled ventilation to SV mode. We measured the plasma IL-6 level at H0, then after 6 h (H6), 12 h (H12), 24 h (H24), 36 h (H36), 48 h (H48), and 72 h (H72). We also measured CRP at H0, H24, and H48. The protocol included concomitant dosage of procalcitonin, but it was dropped because of substantial missing data at the initial stage of inclusions.
Finally, the other variables mentioned above were examined to better refine our strong criteria for IL-6 and CRP biomarkers and to ensure that the groups were comparable.
Study definition and endpoints
We used the clinical pulmonary infection score to define ventilator-associated pneumonia (VAP). 18 We defined bloodstream infection according to the published guidelines. 19 We defined sepsis and septic shock according to the sepsis-3 criteria. 20 Fluid overload was defined according to the 2016 European Society of Cardiology guidelines. 21 The worsening of lung injury was assessed by comparing chest X-rays or chest computed tomography scans and classified as “worsening yes or no” by two different blind reviewers (ED and GP) to minimize the risk of bias. In the case of discrepancy between reviewers, a third reviewer (NP) was called to achieve consensus.
The SV mode was defined as one of the following: pressure support ventilation (PSV), adaptive support ventilation (ASV), or airway pressure release ventilation (APRV).22,23 For the last two modes, the interviewer ensured that the patient had SV.
The primary endpoint was failure to switch to SV mode within 96 h of initiating ventilator weaning. Failure was defined by the occurrence of at least one of the following criteria: RR > 35/min, need to increase the fraction of inspired oxygen (FiO2) > 50% for a pulse oximetry (SpO2) goal > 90%, Richmond agitation sedation scale > 0, spontaneous Vt > 9 mL/kg, heart rate > 120/min, systolic blood pressure > 160 mmHg or < 90 mmHg. In the event of failure, the patient returned to volume-controlled mode. The clinician then decided to make a new attempt to switch to SV mode. In certain situations of prolonged ventilatory weaning, a decision to perform a tracheotomy could be taken at a medical meeting.
Secondary endpoints were failure to wean from mechanical ventilation, death in the ICU, and length of ICU and hospital stays.
Statistical analysis
Quantitative data were described as mean and 95% Confidence Interval (CI) or Standard Error of the Mean (SEM), and qualitative data as number and percentage. The study population was divided according to their success or failure to switch from volume-controlled ventilation to SV mode. Lilliefors’ test was used to assess normality. Continuous variables were compared using unpaired Student t-tests, Mann-Whitney, or Wilcoxon tests, while categorical variables were compared using χ 2 tests or Fisher exact tests, as appropriate.
Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) for the prediction of failure to switch from volume-controlled ventilation to SV mode were calculated, and the receiver operating characteristic (ROC) curves were plotted for IL-6 and CRP. Optimal cut-off values were estimated by maximizing the Youden index. The areas under the ROC curves (AUC) for the prediction of failure to switch from volume-controlled ventilation to SV mode were calculated for IL-6 and CRP along with their 95% confidence interval (95% CI). As our aim was to evaluate the predictive capacity of inflammatory markers, we used for the ROC curves: in failure group, closest values to the time of failure (from 0 to 48 h); in success group: value at H48.
All tests were two-sided, and P-values < 0.05 were considered statistically significant. Analyses were performed using XLStats software version 2019.1.1 (Addinsoft, Paris, France).
Ethics
The study protocol entitled “Role of InterLeukin-6 monitoring during weaning from volume-controlled ventilation in patients with COVID-19 acute respiratory distress syndrome” was approved by a central ethics committee (Comité de Protection des Personnes d’Ile de France I, Paris, France; number CPPIDF1-2021-ND67) on August 2, 2021, in accordance with French and European law and the Helsinki Declaration of 1975 as revised in 2024. Upon study inclusion, we obtained written informed consent from patients or their legally authorized representatives (if they did not have the decisional capacity to provide written informed consent themselves). If consent was not obtained, data were excluded from the analysis.
Results
Participants
During the study period, 160 COVID-19 patients were admitted to our ICU, including 77 requiring invasive mechanical ventilation. Ten of them weren’t included because they were transferred to another ICU or because they had a negative Covid test. Thirty-two patients were excluded due to one of the following reasons : died before attempting a ventilatory weaning test, needed an ECMO, under legal guardianship, receiving tocilizumab treatment, other clinical reasons. We finally included 35 of them in the final analysis (26 men and 9 women) (see the flowchart in Figure 1).

Study flowchart. COVID: coronavirus disease; ECMO: extra-corporeal membrane oxygenation; ICU: intensive care unit.
Descriptive data
Among the 35 included patients, mean age, BMI, and SAPS II were respectively 61.6 years [95%CI: 58.1;65.21], 29.6 kg/m2 [95%CI: 27.6;31.6], and 34.1 [95%CI: 29.3;39.0]. The main comorbidities were hypertension (n = 14) and diabetes mellitus (n = 11). Overall, 13 patients were successfully switched from controlled ventilation to SV mode (success group), while 22 failed to change ventilation mode (failure group). Mean failure time was 34 h 49 min ± 5 h 00 min. Failure occurred within the first 48 h in 16 patients (72.7%), up to 72 h in 20 patients (90.9%), and up to 96 h in 22 patients (100%). Table 1 summarizes the clinical features of the study population and the differences between patients who successfully switched from controlled ventilation to SV mode and those who failed.
Age, sex, BMI, comorbidities, SAPS II, use of prone positioning, use of NMBAs, and controlled-ventilation mode duration were not significantly different between the success and failure groups (P > 0.05). Moreover, ventilator settings were similar between groups, except for PEEP on day 3 (mean=11.2 vs. 9.4 cmH2O in the failure and success groups, respectively, P = 0.042) and FiO2, which was higher in the failure group (mean=44.2 vs. 35.8%, P = 0.004, 51.4 vs. 39%, P = 0.0002, and 49.8 vs. 37.3%, P = 0.005, on days 1, 2, and 3, respectively). Most importantly, the PaO2/FiO2 (P/F) ratio was significantly higher on days 1 and 3 in the success group than in the failure group (mean=213.7 vs. 177, P = 0.046, and 216.6 vs. 169.4, P = 0.032) (Figure 2).

Kinetics of inflammatory biomarkers and P/F ratio. CRP: C-reactive protein; F: failure; H0: the moment of transition from volume-controlled to SV mode; HX: number of hours after the moment of transition from volume-controlled to SV mode; IL-6: InterLeukin-6; P: p-value; P/F ratio: PaO2/FiO2; S: success; mg/L: milligram per liter; pg/mL: picogram per milliliter.
It should be noted that superinfections (13 VAP and one bloodstream infection) concerned 14 patients (63.6%) in the failure group versus three patients (23.1%, three VAP) in the success group (P = 0.035). On the contrary, the number of patients with fluid overload was balanced between the two groups (P > 0.99) (Table 1).
Outcome data
Of the 35 patients analyzed in the present study, 13 (37%) successfully switched from volume-controlled ventilation to SV mode, while 22 (63%) failed to change mode. All 13 patients (100%) from the success group as well as 15 patients (68%) from the failure group were eventually weaned from ventilation and discharged alive. Nine out of 15 patients in the failure group required a temporary tracheostomy (60%) compared with only one patient in the success group (8%). Finally, seven patients (20%) died in the ICU and two developed pulmonary fibrosis at ICU discharge; all of them were in the failure group.
Regarding the ICU length of stay, it was significantly longer in the failure group than in the success group (mean=45.6 vs. 24.8 days, P = 0.022). Hospital stay duration was not significant between groups (mean=57.9 vs. 39.8 days in the failure and success groups, respectively, P = 0.099).
Main results: value of inflammatory biomarkers during switch to Sv mode
In patients who failed to switch to SV mode, mean plasma IL-6 levels were consistently higher than in the success group from H0 to H48. These differences in plasma IL-6 levels became significant between groups from H24 onwards (mean=362.8 vs. 33.6 pg/mL, P = 0.002). Interestingly, in the failure group, CRP also increased, although differences in plasma CRP levels were only significant between groups at H48 (mean=129.3 vs. 52.2 mg/L, P = 0.017).
The kinetics of inflammatory biomarkers associated with the success or failure of switching to SV mode are presented in Table 2 and Figure 2.
IL-6 and CRP had a similar ability to predict failure in switching to SV mode: AUC 0.763 [95%CI: 0.633-0.893] and 0.753 [95%CI: 0.595-0.911], respectively (P = 0.87) (Table 3 and Figure 3). However, at a cut-off value of 75 mg/L, CRP exhibited better sensitivity at 76.5% (vs. 61.1%), whereas IL-6 at a cut-off value of 80 pg/mL had better specificity at 82.9% (vs. 73.8%). Both biomarkers had low PPV and good NPV (Table 3).

The ability of IL-6 and CRP to predict a failure in transition to SV mode. AUC: areas under the ROC curves; CI: 95% Confidence Interval; CRP: C-reactive protein; IL-6: InterLeukin-6; P: p-value.
Receiver operating characteristic (ROC) curve of InterLeukin-6 (IL-6) and C-reactive protein (CRP) for transition failure from controlled ventilation to spontaneous ventilation mode.
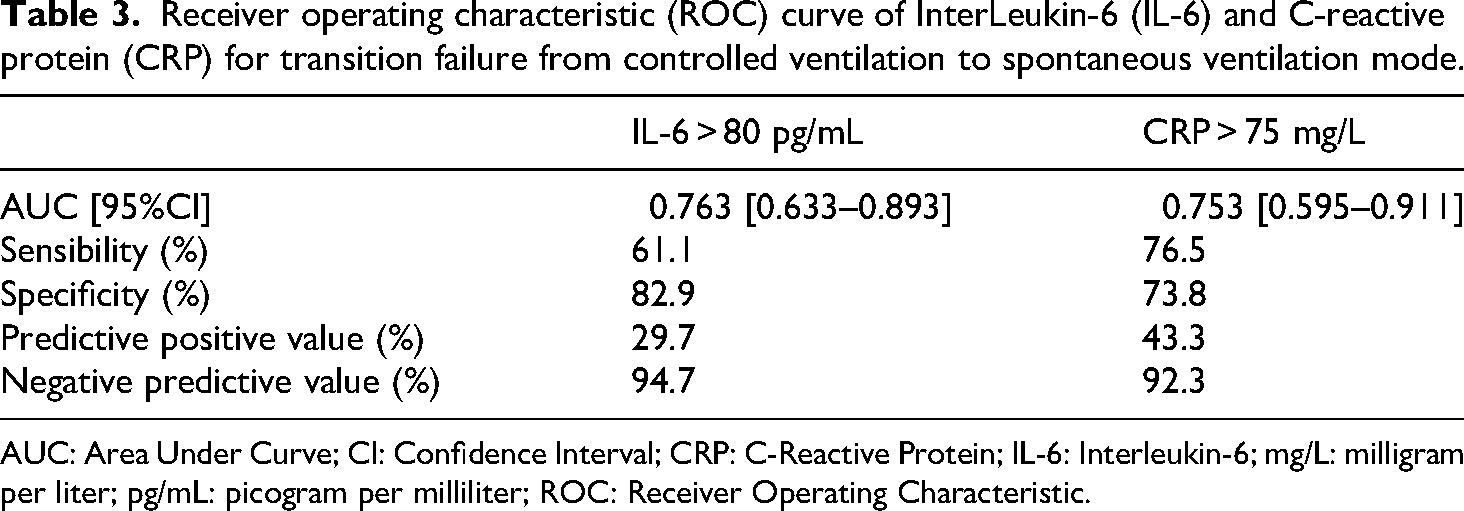
AUC: Area Under Curve; CI: Confidence Interval; CRP: C-Reactive Protein; IL-6: Interleukin-6; mg/L: milligram per liter; pg/mL: picogram per milliliter; ROC: Receiver Operating Characteristic.
Regarding the ROC curve (Figure 3), in the failure group, we analyzed the IL-6 and CRP data closest to the time of failure. In the success group, we analyzed the values taken at H48, since they corresponded to the mean failure time in the other group, which was 34 h 49 min ± 5 h 00 min. The event of interest was indeed failure or success. Our prediction therefore had to be made at the time of the event. Consequently, we choose to analyze the ROC curve at the time of the event's occurrence.
Other analyses
In the failure group, elevated IL-6 levels were measured comparatively in two subgroups with VAP and without VAP. We used the last value before failure. The mean did not significantly differ between the groups: 325.8 in the group without VAP and 403.3 in the VAP group (P = 0.395). Thus, elevated IL-6 was associated with failure to switch to SV mode, regardless of whether VAP occurred (Figures 4 and 5).

InterLeukin-6 (IL-6) values (pg/mL) by subgroups. HX: data at time X in hour; pg/mL: pictogram per millimeter; VAP: ventilator-associated pneumonia.

Last InterLeukine-6 (IL-6) value (pg/mL) before failure. One-tailed t test p-value = 0.395. Mean without VAP group = 325.8; mean with VAP group = 403.3. HX: data at time X in hour; pg/mL: pictogram per millimeter; VAP: ventilator-associated pneumonia.
Discussion
In our cohort of 35 COVID-19 ARDS, we observed that elevated IL-6 and CRP levels were associated with ventilatory weaning failures. Both biomarkers had a similar ability to predict the failure of switching to SV mode. However, we showed that elevated IL-6 levels differentiated patients in the failed group earlier than CRP. Moreover, due to ROC curve analysis, our results were able to determine cut-off values for CRP and IL-6, respectively 75 mg/L and 80 pg/mL. Furthermore, we demonstrated that most patients who failed subsequently developed VAP.
Finally, we confirmed that patients who succeeded in their first attempt to switch from volume-controlled ventilation to different types of weaning ventilatory modes (PSV, ASV, or APRV) had better outcomes than those who failed (Figure 2). This is consistent with recent data from two larger retrospective studies.24,25 In the first, Perez et al. found that failure to switch from volume-controlled ventilation to PSV was associated with worse outcomes, with COVID-19 diagnosis and hypoxemia being independent risk factors. 24 The second observational study published in 2024 analyzed the factors predicting successful weaning to PSV in mechanically ventilated COVID-19 patients and their impact on patient outcomes. 25 This study emphasized that the P/F ratio was an independent factor associated with a failed switch from volume-controlled ventilation to PSV, which was also associated with worse outcomes. In comparison with these existing data, our study has the advantage of addressing the relevance of biomarker assays during the switch from volume-controlled ventilation to SV mode in COVID-19 ARDS patients.
An imbalanced host response to SARS-CoV-2 infection was reported in early 2020 3 as the main cause of COVID-19 ARDS. It is now widely accepted that SARS-CoV-2 induces hyperinflammation that can destroy lung tissue. 26 Importantly, elevated IL-6 drives this maladaptive immune response, preceding lymphopenia, elevated CRP, procalcitonin, lactate, and D-Dimer.26–35 Increased IL-6 levels are associated with worse disease severity and poorer outcome in COVID-19 patients.26,36–38 Some authors reported that IL-6 levels could be useful to help to determine the COVID-19 patients later requiring invasive mechanical ventilation. 39 Others 40 already demonstrated the value of monitoring IL-6 kinetics in ARDS. However, to the best of our knowledge, this is the first study to confirm that IL-6 and CRP could be reliable biomarkers during weaning from volume-controlled ventilation to SV mode in COVID-19 ARDS patients.
The search for a biomarker able to predict at an early stage the failure of switching from volume-controlled ventilation to SV could help to improve the outcomes of COVID-19 patients, mainly by limiting the occurrence of P-SILI. 12 Indeed, switching to SV mode is a delicate and crucial moment in the weaning procedure from mechanical ventilation. At this stage, the risk of worsening COVID-19-related lung injuries is extremely high and could have a negative impact on outcomes.41,42 Early detection of patients at risk of failure would help to limit this phenomenon by switching back to protective ventilation at an early stage.41,42 Such a biomarker could also ultimately save time in the weaning procedure, which would be a significant advantage in the context of a pandemic by decreasing workload in the ICU.
Our work has shown that IL-6 and CRP have an especially good NPV for detecting the failure to switch to SV mode. This suggests that monitoring CRP and IL-6 levels could help clinicians by giving them an additional argument for pursuing a trial of switching to SV mode. In addition, our results suggest that increased IL-6 levels can detect failure earlier than CRP. However, it is important to bear in mind that in critically ill patients, elevated IL-6 (and CRP) levels can be driven by COVID-19 itself and other conditions such as VAP, sepsis, 43 or P-SILI, 12 which are several causes of failure to switch to SV mode or to wean from mechanical ventilation. Nevertheless, we found that elevated IL-6 levels were associated with failure to switch to SV mode, regardless of whether VAP occurred (Figures 4 and 5).
In the failure group, the increase in IL-6 appeared to be delayed in patients without VAP. It may therefore be hypothesized that this difference is related to a progressive mechanism of biotrauma aggravated by P-SILI.
Thus, in a pragmatic approach, our results suggest that the lack of elevated IL-6 provides valuable information that can be used at the bedside to continue the SV trial. 44 On the contrary, if IL-6 is elevated, the cause of failure to switch to SV mode should be sought and treated, including VAP, sepsis, fluid overload, or P-SILI.45–48 At this stage, physicians should ensure that the patient is on protective ventilation to limit the worsening of lung injuries.45–48
Our study has several limitations. First, it is a single center prospective cohort, which could limit the generalizability of its results. Second, the small cohort size may have limited the detection of statistical differences between groups for clinically relevant variables such as IL-6 levels at H6 and H12. It also limited our ability to carry out multivariate analysis, including the occurrence or non-occurrence of VAP or the type of SV mode (which was at the physician's discretion), which may have impacted the outcomes and influenced the kinetics of IL-6. Indeed, considering that no data were available for IL-6 values in COVID-19 patients during ventilator weaning, the magnitude of the effect was unknown, and it was not possible to calculate the sample size to test our hypothesis. To be conservative, we included all eligible patients admitted to the intensive care unit during the inclusion period. Third, numerous descriptive data, which may have influenced the main outcome such as plateau pressure and airway occlusion pressure, could not be collected in the ICU, as the department was overstretched during the pandemic period. Fourth, the high screening failure rate (16%, 12 patients), as encountered in several studies during the COVID-19 pandemic due to ICU surge, may have induced a selection bias. In this context, there may have been fewer inclusions, particularly on weekends or at night. There is therefore a risk that the patients most affected by the ICU surge may not have been included. Our study may therefore have underestimated the rate of ventilatory weaning failures in patients with COVID ARDS. Last, we published this study after the end of the COVID-19 pandemic.
Nevertheless, we believe that it may serve as a model for other future research hypotheses in ARDS, in this post-pandemic COVID era. Indeed, to date, no study has investigated the correlation between IL-6 levels and successful switching to SV. That's why we believe that it would be relevant to study it to guide the correct conduct of ventilatory weaning in these patients with ARDS as a rule.
Conclusion
IL-6 and CRP may be useful biomarkers to predict failure of switching to SV mode in COVID-19 ARDS patients. Elevated IL-6 levels can discriminate failure earlier than CRP (H24 vs. H48). Given the small size and the monocentric nature of the sample, further studies are needed to confirm our results and to determine the relevance of a systematic IL-6 measurement during the weaning process.
Footnotes
Acknowledgments
The authors thank the patients for their participation in the clinical studies. We also thank all the doctors at the ICU of Avignon Hospital for their dedication and significant contributions during data collection. We especially want to thank the clinical research nurses and paramedical staff for their investment and hard work in the overstretched department during the pandemic period.
Ethical considerations
We obtained written informed consent from the participants for the publication of this study as well as any accompanying data and images. Where the patients provided consent themselves, we confirm that they had the capacity to do so and that any mental or physical disabilities were taken into consideration in the process of informing and obtaining written informed consent. In the case of participants who died, we confirm that the consent provided still allows for publication.
We confirm that all content presented in this study along with its associated data and images have been de-identified and anonymized to the best possible extent.
The original signed and dated consent form is held by the treating institution or appropriate governing national body and will be retained according to the policies and procedures of the institution or governing body.
The participants were given the opportunity to ask questions regarding the publication of the study. Their questions were fully answered, and they consented to publish all associated data and images. The participants were informed that their consent and participation in the publication of this study were entirely voluntary and that they had the right to withdraw their consent at any time.
We confirm that our article complies with the appropriate national law on consent and privacy.
Consent to participate
The study was approved by a central ethics committee (Comité de Protection des Personnes d’Ile de France I, Paris, France; number CPPIDF1-2021-ND67) on August 2, 2021, in accordance with French and European law and the Helsinki Declaration of 1975 as revised in 2024. We obtained written informed consent from patients or their legally authorized representatives (if they did not have the decisional capacity to provide written informed consent themselves) upon study inclusion. If consent was not obtained, data were excluded from the analysis.
Author contributions/CRediT
Every author meets the ICMJE guidelines.
Delaunay, Estelle: conceived and designed the study, collected the data, contributed to data/analysis tools, performed the analysis, wrote the paper, and submitted the article for publication.
Poussard, Nicolas: conceived and designed the study, collected the data, and contributed to data/analysis tools.
Gourjon, Géraud: contributed to data/analysis tools, performed the analysis, and wrote the paper (statistics).
Frouin, Antoine: collected the data and revised the paper.
Ducq, Pierre: collected the data and revised the paper.
Di Vico, Lynda: collected the data and revised the paper.
Moschietto, Sébastien: conceived and designed the study, collected the data, contributed to data/analysis tools, and revised the paper.
Larcher, Romaric: conceived and designed the study, contributed to data/analysis tools, performed the analysis, and wrote the paper.
Pradel, Gaël: contributed to data/analysis tools, performed the analysis, wrote the paper, and submitted the article for publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available from the corresponding author [ED] upon reasonable request.
