Abstract
Objectives
Human induced pluripotent stem cells (hiPSCs) hold significant promise in regenerative medicine and drug discovery. However, single-cell dissociation, essential for genetic modification and clonal selection, often reduces hiPSC viability and colony formation. While various methods, including small molecules and feeder cells, have been developed to address this, their outcomes remain inconsistent. This study aims to develop more efficient methods to enhance hiPSC survival post-dissociation using a novel combination of well-characterized small-molecule inhibitors.
Methods
Human induced pluripotent stem cells were pretreated with Rho-associated protein kinase inhibitor (Y27632), SMC4 (PD0325901 + CHIR99021 + thiazovivin + SB431542), or SiM5 (PD0325901 + CHIR99021 + Thiazovivin + SB431542 + Pifithrin-α) for 1 h before subjected to single-cell dissociation by accutase. The dissociated single hiPSCs were then cultured in NutriStem or StemFlex medium supplemented with Y27632, SMC4, or SiM5. Cell viability, pluripotency marker expression, colony formation capacity, and karyotype were then compared between various treatments. The effect of SiM5 treatment on hiPSCs survival and colony formation capacity was also tested under hypoxic conditions and after fluorescence-activated cell sorting.
Results
The results show that SiM5 treatment significantly increases hiPSCs survival by approximately 2.5 and 25 times compared to those treated with SMC4 and Y27632, respectively. These results were consistently observed across different cell lines and culture media. Furthermore, SiM5 treatment also increased hiPSCs survival and proliferation after single-cell dissociation under hypoxic conditions. The withdrawal of SiM5 after treatment only temporarily hinders hiPSCs cell cycle progression, without impairing their subsequent expansion. Fluorescence-activated cell sorting analysis revealed that SiM5 does not affect the pluripotency of hiPSCs following treatment. Additionally, it was found that SiM5 has no effect on the colony-forming ability or chromosomal stability of hiPSCs.
Conclusion
SiM5 treatment significantly improves hiPSCs survival and colony formation after single-cell dissociation across various conditions. This approach could enhance the efficiency of genetic manipulation and single-cell cloning, advancing hiPSCs applications in research and clinical settings.
Keywords
Introduction
Human pluripotent stem cells (hPSCs), including human embryonic stem cells (hESCs) and human induced pluripotent stem cells (hiPSCs), have emerged as a powerful tool for investigating the mechanism underlying early human development and as a source for the derivation of various functional cell types for regenerative medicine.1–4 These hPSCs can also be used to study disease pathogenesis and determine the effectiveness and safety of various drugs. 5 Recent breakthroughs in gene editing, particularly the CRISPR-Cas9 system, have made precise alterations to the human genome much easier and faster than ever before.6,7 By combining hPSCs technology with CRISPR-Cas9, scientists can now correct various disease-causing mutations in specific patient-derived cells for research and clinical use.
Human pluripotent stem cells are highly sensitive to dissociation-induced apoptosis. This inherent characteristic necessitates their maintenance as cell clumps during passaging to prevent cell death and ensure their viability and functionality for subsequent studies. A significant advancement in overcoming this challenge was made with the discovery that inhibiting Rho-associated protein kinase (ROCK) can improve the survival of hPSCs during stressful procedures, such as clonal isolation, suspension culture, or cryopreservation.8–11 Subsequently, the combination of ROCK inhibitor (ROCKi; Thiazovivin) with MEK inhibitor (PD0325901), TGF-β inhibitor (SB431542), and GSK3 inhibitor (CHIR99021), termed the small molecule compound 4 (SMC4) cocktail, was shown to further improve hPSCs viability and maintain pluripotency post-dissociation. 12 Another factor that plays an important role in hPSCs survival is the tumor suppressor gene p53 which induces cell cycle arrest and triggers apoptosis through a mitochondrial pathway in response to various stresses under culture conditions.13–15 Inhibition of p53 activity, in combination with other small molecule inhibitors, could further improve the viability of hPSCs in culture. 15
Despite recent improvement in culture conditions by incorporating various small molecule inhibitors,11,12,16 extracellular matrixes,17,18 and feeder cells, 19 the survival of hPSCs after single-cell dissociation, which is essential for various applications, remains poor and inconsistent. Therefore, this study aims to develop more efficient methods to improve the hiPSCs survival after single-cell dissociation, using a novel combination of well-characterized small-molecule inhibitors. Our results suggest that the combination of five small molecule inhibitors, SiM5, significantly improves hiPSCs survival after single-cell dissociation under various culture conditions without affecting their pluripotency, colony formation capacity, and chromosomal integrity. We believe that our method could be used to increase the efficiency of large-scale production of genetically modified hPSCs for various future applications.
Materials and methods
Culture of human induced pluripotent stem cells
In this study, three independent hiPSC lines were used: MUSIi012-A (RRID:CVCL_YE49), MUSIi013-A (RRID:CVCL_A0ZV), and MUSIi024-A (RRID CVCL_E4E5).15,16 The study was approved by the Siriraj Institutional Review Board (COA no. Si 090/2020), Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand. Human induced pluripotent stem cells were cultured on Matrigel-coated plates in NutriStem (Biological Industries, USA) or StemFlex medium (ThermoFisher Scientific, USA) in a humidified incubator with 5% O2, 5% CO2 at 37 °C. Cells were passaged upon reaching 80% confluence using a nonenzymatic Versene solution (ThermoFisher Scientific, USA). To treat with small molecules, cells were cultured to reach 80% confluence and treated with ROCKi (Y27632, Sigma, USA), SMC4 [0.4 µM PD0325901 (Sigma, USA) + 1 µM CHIR99021 (TOCRIS, USA) + 5 µM Thiazovivin (Sellectchem, USA) + 2 µM SB431542 (Calbiochem, USA)] or SiM5 [0.4 µM PD0325901 + 1 µM CHIR99021 + 10 µM Thiazovivin + 4 µM SB431542 + 2 µM Pifithrin-α (TOCTIS, USA)] for 1 h before subjected to single-cell dissociation. For single-cell dissociation, hiPSCs were washed with phosphate buffer saline (PBS), treated with AccutaseTM (STEMCELL Technologies, Canada) at 37 °C for 5 min, and mechanically dissociated by pipetting several times. After dissociation, an equal volume of NutriStem medium was added, and the cells were centrifuged at 200 g for 10 min. The supernatant was then removed, the cell pellet was resuspended in NutriStem medium supplemented with various small molecules and cultured on a Matrigel-coated plate for an additional 24 h. Subsequently, the medium was replaced with small-molecule-free NutriStem medium and cells were cultured with daily media changes.
In vitro differentiation of hiPSCs
For embryoid body (EB) formation, hiPSC colonies were harvested by dispase (Stemcell Technology, Canada) and mechanically dissociated by pipetting. The small clumps of hiPSCs were then transferred to a nontreated 100 mm dish (ThermoFisher Scientific, USA) and cultured in DMEM (Invitrogen, USA) supplemented with 20% (v/v) serum replacement (Invitrogen, USA), 10 mM nonessential amino acids (Invitrogen, USA), 55 mM β-mercaptoethanol (Invitrogen, USA), 2 mM L-GlutaMAX (Invitrogen, USA), and 50 μg/mL penicillin/streptomycin (Millipore, USA) at 37 °C in an atmosphere containing 5% CO2 with media changing every 3 days to form EBs. On day 7, the EBs were transferred to gelatin-coated plates, allowed to attach and cultured for another 2 weeks. At the end of the culture, the expression of three primitive germ layer markers, NESTIN, SMA, and AFP, was evaluated by immunofluorescence. Images were acquired and analyzed with NIS-Elements software V.4.30 (Nikon Corporation, Japan) on a Nikon Eclipse Ti-U inverted fluorescent microscope using a Nikon digital sight DS-Fi2 camera (Nikon Corporation, Japan). The antibodies used in this experiment are listed in Supplementary Table 1.
Hoechst 33342 and Vybrant Ruby staining for cell sorting
Pluripotent cells were pretreated with small molecule cocktails for 1 h, washed with PBS to remove the small molecules, dissociated into single cells using TrypLE Select (ThermoFisher Scientific), and further processed with small molecule treatment. Cells were stained with 2.5 µg/mL Hoechst 33342 (Sigma-Aldrich) in NutriStem at 37 °C for 30 min. The cell pellet was harvested by centrifugation and resuspended with PBS + 15%KOSR. For Vybrant Ruby staining, cells were incubated with 5 µM Vybrant Ruby (ThermoFisher Scientific, USA) in phenol-red-free medium. The stained cells were filtered through a 70 µm cell strainer (BD Biosciences, USA) and sorted for 2n cells using a BD FACS AriaIII sorter. Sorted cells were seeded at a density of 20,000 cells/well in Matrigel-coated 6-well plates in the presence of specific small molecule cocktails for an additional 24 h under hypoxic conditions (5% O2, 5% CO2). Forty-eight hours after cell sorting, cells were fixed with 4% paraformaldehyde solution. Subsequently, they were stained with an OCT4 antibody (concentration detailed in Supplementary Table 1). Colony visualization, imaging, and counting were performed using a Nikon Eclipse Ti-U inverted fluorescent microscope (Nikon Corporation, Japan) with a Nikon digital sight DS-Fi2 camera. Image acquisition and analysis were performed with NIS-Elements image software V.4.30.
Apoptosis assay
For the apoptosis assay, hiPSCs were treated with SiM5, SMC4 or Y27632 for 1 h, enzymatically dissociated into a single cell using AccutaseTM, and stained with 2.5 µg/mL Hoechst33342 (ThermoFisher Scientific, USA) for 30 min. The 2n diploid peak was then sorted using BD FACS AriaIII sorter (BD Biosciences, USA) and seeded at a density of 80,000 cells/well in a 24-well plate coated with Matrigel in NutriStem medium supplemented with SiM5, SMC4, or Y27632 for another 24 h. The cells were then dissociated into single cells with accutase and stained with annexin V-FITC and propidium iodide to determine cell viability, and apoptosis using the BD FACS CantoTM flow cytometer and analyzed with FlowJoTM software (BD Biosciences, USA).
Cell cycle analysis
Cells were pretreated with small molecule cocktails for 1 h, dissociated into single cells using TrypLE Select (ThermoFisher Scientific), and harvested by centrifugation. Cell cycle analysis was performed using the BD Cycletest™ Plus DNA reagent kit (BD Biosciences) according to the manufacturer's protocol. Flow cytometric analysis was carried out on 50,000 gated events using a BD FACS Canto™ flow cytometer (BD Biosciences). Cell cycle analysis was performed using FlowJoTM software.
Immunofluorescence staining
Cells were fixed with 4% (w/v) paraformaldehyde in PBS and their membranes were permeabilized with 0.1% (w/v) Triton X-100 in PBS. Cells were then incubated in blocking solution containing 3% (w/v) BSA in PBS at room temperature for 2 h to prevent nonspecific antibody reaction before incubation with primary antibodies against human OCT4, NANOG, SOX2, TRA-1-60, TRA-1-81, SSEA4, Nestin, smooth muscle actin and α-fetoprotein at 4 °C overnight. After incubation, cells were washed twice with PBS and incubated with appropriate secondary antibodies at room temperature for an additional 1 h. The nuclei were stained with Hoechst 33342 for 10 min, and the fluorescence image was visualized by fluorescence microscopy. Fluorescence images were acquired using a Nikon Eclipse Ti-U Inverted fluorescence microscope with a Nikon digital sight DS-Fi2 C-mount microscope camera, and subsequent image analysis was conducted using NIS-Elements software V.4.30. The antibodies used in this experiment are listed in Supplementary Table 1.
Single-cell colony formation
Cells were treated with SiM5, SMC4, or Y27632 for 1 h before being subjected to single-cell dissociation using AccutaseTM. Each single hiPSC was then seeded into an individual well of 96-well plates coated with Matrigel (Corning, USA) to form a single-cell colony. Cell morphology was observed with a Nikon Eclipse Ti-U inverted microscope. Images were captured using a Nikon Digital Sight DS-Fi2 C-mount Microscope Camera and visualized with NIS-Elements software V.4.30.
Karyotype analysis
Cells were expanded until their density reached 100% confluence and sent to Siriraj Central Cytogenetic Laboratory, Faculty of Medicine, Siriraj Hospital, Mahidol University for karyotyping.
Alkaline phosphatase staining
Cells were fixed with 4% (w/v) paraformaldehyde in PBS for 10 min and washed four times with PBS. Alkaline phosphatase staining was performed using the Vector® Red kit (Vector Laboratory, USA). Stained cells were observed using a Nikon Eclipse Ti-U inverted microscope equipped with a Nikon digital sight DS-Fi2 camera. Images were acquired using NIS-Elements image software V.4.30.
Statistical analysis
All data analyses were performed using GraphPad Prism 9.0 software. Kolmogorov–Smirnov normality test was used to determine the normality distribution of the groups. A Mann–Whitney U test was used for two-group comparisons. One-way ANOVA with Tukey's multiple comparison or Kruskal–Wallis test was used for multiple comparisons. The data represent mean ± SD from three or more independent experiments. Statistical significance is denoted at the p < 0.05.
Results
A combination of PFT-α and Y-27632 improves hiPSC survival after single-cell dissociation
We first treated hiPSCs with a combination of PFT-α, a small molecule that prevents cell apoptosis by inhibiting p53, and 10 µM Y-27632, a ROCKi that is widely used to increase hPSCs viability, to determine whether PFT-α can increase hiPSCs viability after single-cell dissociation compared to those treated with Y-27632 alone (Figure 1(a)). The results show that 2 µM PFT-α significantly increases hiPSCs viability on day 3 of culture compared to those treated with Y-27632 alone, as determined by alkaline phosphatase staining (Figure 1(b) and Supplementary Figure 1) and cell counting (Figure 1(c)). On the contrary, higher concentrations of PFT-α, 10 and 20 µM, did not increase hiPSCs viability after single-cell dissociation compared to those treated with Y-27632 alone (Figure 1(b) and (c)). Based on these results, 2 µM PFT-α which significantly increased the survival of hiPSCs after single-cell dissociation was chosen for further optimization of the culture condition.

The effect of Y-27632, PFT-α and SiM5 on human induced pluripotent stem cells (hiPSCs) survival after single-cell dissociation. (a) Schematic representation of the experimental protocol to investigate the effects of Y-24632, Y-27632 + PFT-α, and SiM5 on the viability of hiPSCs after single-cell dissociation. (b) Representative images of alkaline phosphatase staining in cultured hiPSCs treated with 10 µM Y-27632 alone, 10 µM Y-27632 supplemented with various concentrations of PFT-α, or SiM5 on days 1 and 3 post single-cell dissociation. Magnification: 40×. (c) Graphs show the number of viable hiPSCs treated with Y-27632 alone, Y-27632 supplemented with various concentrations of PFT-α, or SiM5 on days 1–3 after single-cell dissociation. Data are mean ± SD of 6 biological replicates. Statistical significance was determined using ANOVA with Kruskal–Wallis test *p < 0.05, **p < 0.01, ***p < 0.001, ***p < 0.0001, ****p < 0.0001. Scale bars: 200 µm.
A combination of PFT-α with a specific combination of four other small molecule inhibitors further improves the survival of hiPSCs after single-cell dissociation
Although 2 µM PFT-α significantly increases the survival of hiPSCs treated with Y-27632, the increase is quite modest, only 25% higher than those treated with Y-27632 alone (Figure 1(b)). To further improve hiPSCs survival, we supplemented culture medium with 4 additional small molecule inhibitors (PD0325901; 0.4 µM, CHIR99021; 1 µM, Thiazovivin; 10 µM, SB431542: 4 µM) together with 2 µM PFT-α, namely SiM5 (small molecule inhibitors of transcription factor mixed 5). Our findings demonstrated that SiM5 significantly reduced cell death and increased cell viability of hiPSCs on both day 2 and day 3 after single-cell dissociation compared to those treated with Y-27632 + 2 µM PFT-α (Figure 1(b) and (c)).
To determine the long-term effect of SiM5 on hiPSCs cell cycle progression, we performed a cell cycle analysis for four consecutive days after the single-cell dissociation of hiPSCs (Figure 2(a)). On day 0 (immediately after single-cell dissociation) and day 1 (24 h after single-cell dissociation, when cells were still treated with small molecules), SiM5 did not significantly alter the progression of the hiPSCs cell cycle compared to Y-27632 alone (Figure 2(b) and (c)).

The effect of Y-27632, PFT-α and SiM5 on human induced pluripotent stem cells (hiPSCs) cell cycle progression after single-cell dissociation. (a) Schematic illustration of the experimental design to investigate the effects of Y-24632 and SiM5 on cell cycle phase distribution in small molecule-treated cells. (b) Cell cycle distribution of hiPSCs treated with Y-27632 or SiM5 post-dissociation. Flow cytometry analysis was performed on days 0, 2, 3, and 4. (c) Effect of Y-27632 and SiM5 on hiPSC cell cycle distribution (G1, S, G2/M) over 4 days. Data represent the mean ± SD of three biological replicates. Statistical significance was determined using Mann–Whitney U test.
On day 2 (48 h after single-cell dissociation and 24 h after removal of small molecules), there was an increase in G1-phase cells and a decrease in S-phase cells in SiM5-treated hiPSCs compared to those treated with Y-27632 alone (Figure 2(b) and (c)). However, this alteration in cell cycle progression was transient and was no longer observed on day 3. On day 4 (96 h after single-cell dissociation and 72 h after removal of small molecules), there was a decrease in G1-phase cells and an increase in S-phase cells in SiM5-treated hiPSCs compared to those treated with Y-27632 alone (Figure 2(b) and (c)). These findings suggest that the removal of SiM5 after treatment only transiently inhibits hiPSCs cell cycle progression without compromising their subsequent expansion.
We then investigated the effect of SiM5 on hiPSCs survival after single-cell dissociation and fluorescence-activated cell sorting (FACS) sorting compared to that of the commercially available SMC4 supplement. The results revealed that SiM5 increases hiPSCs viability approximately 2.5 times compared to those treated with SMC4 and approximately 25 times compared to those treated with Y-27632 (Figure 3(a)). Consistent with this, SiM5 also reduced the percentages of late apoptotic hiPSCs by approximately 1.7 times (25% vs. 43%) compared to those treated with SMC4 and approximately 3.5 times (25% vs. 87%) compared to those treated with Y-27632 (Figure 3(b) and Supplementary Figure 2). These findings suggest that SiM5 increases hiPSCs after single-cell dissociation by reducing apoptotic cell death.
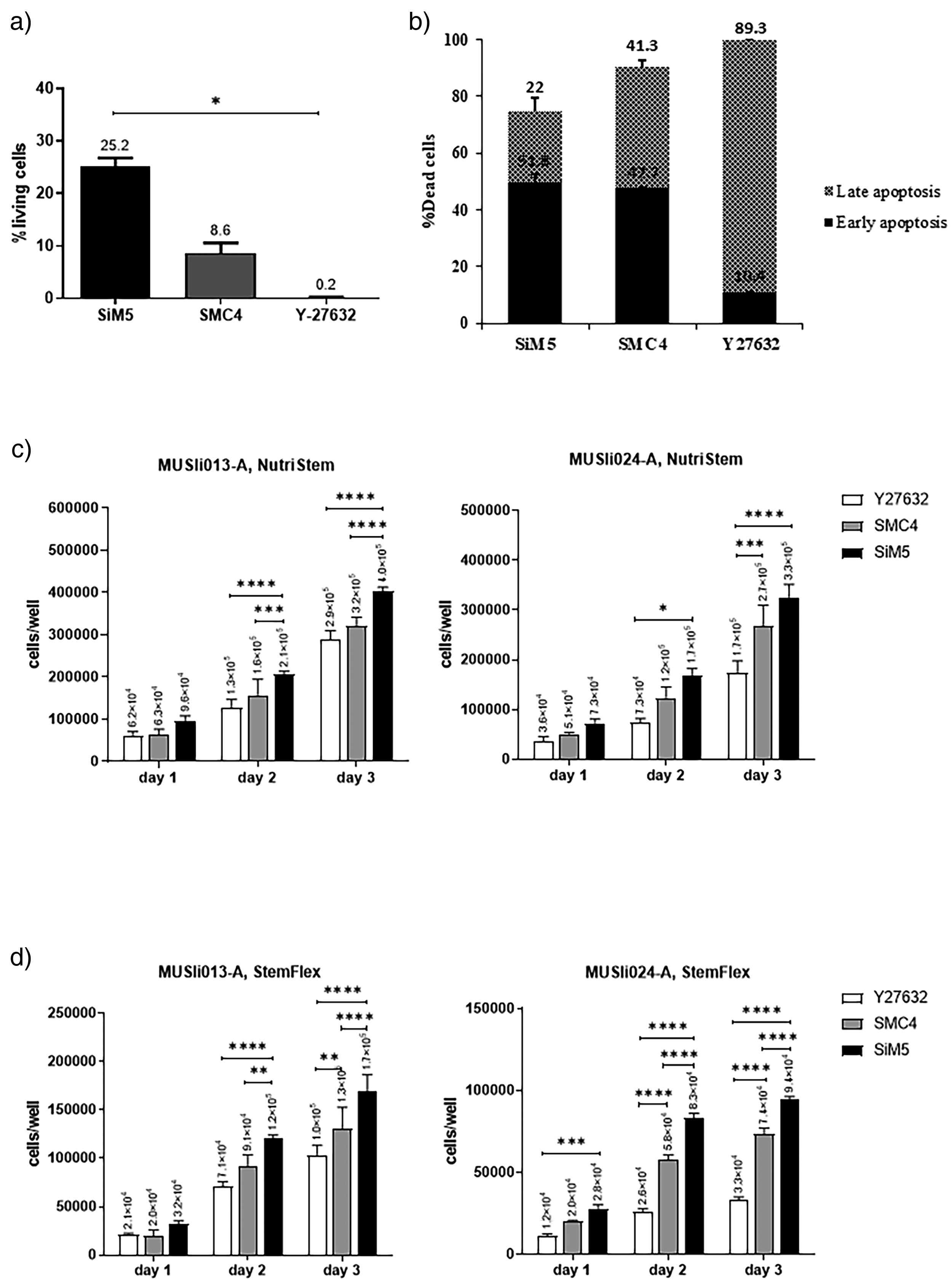
Effects of Y-27632, SMC4, and SiM5 on human induced pluripotent stem cells (hiPSCs) viability and apoptosis post-dissociation. (a) Percentage of viable hiPSCs treated with Y-27632, SMC4, or SiM5 following single-cell dissociation. Data are mean ± SD of three biological replicates. Statistical significance was determined using ANOVA with Kruskal–Wallis test *p < 0.05. (b) Rates of early and late apoptotic hiPSCs after treatment. (c and d) Comparative analysis of two hiPSC lines (MUSIi013-A and MUSIi024-A) exposed to Y-27632, SMC4, or SiM5, cultured in NutriStem or StemFlex media. Data represent the mean ± SD of six biological replicates and statistical significance was determined by one-way ANOVA with Tukey's multiple comparison test, *p < 0.05, **p < 0.01, ***p < 0.001, ***p < 0.0001, ****p < 0.0001.
SiM5 improves survival of three different hiPSC lines after single-cell dissociation and expansion in two different media
To confirm the promoting effect of SiM5 on the survival of hiPSCs after single-cell dissociation, two additional hiPSC lines, MUSIi013-A and MUSIi024-A, were treated with SiM5 and subjected to the survival assay. Consistent with the findings on MUSIi012-A, SiM5 significantly reduced apoptotic cell death and increased cell the viability of both hiPSCs on day 2 and day 3 after single-cell dissociation compared to those treated with Y-27632 or commercially available SMC4 (Figure 3(c)).
To further investigate whether the promoting effect of SiM5 on hiPSCs survival depends on the culture medium, both hiPSC lines were cultured in either Nutristem, which is the standard medium used to culture all hiPSC lines in our previous experiments, or StemFlex, another commercial hiPSCs culture medium. MUSIi013-A and MUSIi024-A were expanded in StemFlex for two passages prior to the experiment to adapt the cells to the medium and minimize stress-induced effects. The results show that SiM5 significantly increases cell numbers compared to those treated with Y-27632 and SMC4, starting from day 2 of culture in both Nutristem and StemFlex medium (Figure 3(c) and (d)). It should be noted that MUSIi013-A exhibited greater tolerance to single-cell dissociation compared to MUSIi024-A in both Nutristem and StemFlex medium. These results suggest that there might be variation in an endogenous response to dissociation stress between hiPSC lines.
SiM5 promotes the survival and colony formation capacity of hiPSCs without compromising their pluripotency and chromosomal integrity after repeated single-cell expansion
Next, we determined whether SiM5 treatment affects hiPSCs colony formation capacity after single-cell dissociation under hypoxic condition compared to those treated with SMC4 or Y-27632. Colony formation capacity was determined by seeding a single hiPSCs in an individual well of Matrigel-coated plates and evaluating its ability to form a colony on culture day 8 (Figure 4(a)). Consistent with its effect on hiPSCs survival, SiM5 treatment increases hiPSCs colony formation capacity after single-cell dissociation under hypoxic conditions, compared to those treated with SMC4 (69% ± 4.8 vs. 56% ± 1.68) and Y-27632 (69% ± 4.8 vs. 20% ± 4.29) (Figure 4(b)).

The impact of SiM5 on pluripotency and chromosomal integrity of human induced pluripotent stem cells (hiPSCs) post-single-cell expansion. The effects of SiM5 on pluripotency and chromosomal integrity of hiPSCs after single-cell expansion. (a) The morphological progression of hiPSC colonies derived from single SiM5-treated hiPSCs over 8 days of culture. Magnification: 200×. (b) Colony formation capacity of hiPSCs treated with Y-27632, SiM5, and SMC4 after single-cell dissociation under hypoxic (5% O2, 5% CO2) and normoxic (21% O2, 5% CO2) conditions. (c and d) The expression levels of various pluripotency marker proteins in SiM5-treated hiPSCs post-single-cell expansion, as assessed by flow cytometry and immunofluorescence staining, respectively. Magnification: 200×. (e) Representative immunofluorescent staining of differentiated SiM5-treated hiPSCs expressing differentiation markers of the three primary germ layers. Magnification: 200×. (f) The karyotype analysis of single cell expanded SiM5-treated hiPSCs. Data are mean ± SD of four biological replicates. Statistical significance was determined using ANOVA with Kruskal–Wallis test. Scale bars: 50 µm.
To ensure that SiM5 promotes the survival and colony formation capacity of hiPSCs without compromising their pluripotency, we determined the expression of pluripotent markers and the multilineage differentiation capacity of SiM5-treated hiPSCs after four passages of single-cell expansion. Immunofluorescent and flow cytometric assays show that more than 95% of SiM5-treated hiPSCs maintain the expression of pluripotency marker proteins, OCT4, NANOG, SOX2, SSEA-4, TRA1-60, and TRA1-81 (Figure 4(c) and (d)). The SiM5-treated hiPSCs are also capable of differentiate into derivatives of the three primary germ layers (Figure 4(e)) and maintain normal diploid karyotypes even after repeated single-cell expansion (Figure 4(f)). These results suggest that SiM5 promotes the survival and colony formation capacity of hiPSCs without negatively affecting their pluripotency and chromosomal integrity.
SiM5 improves the survival and colony formation capacity of hiPSCs after fluorescent-activated cell sorting
Fluorescence-activated cell sorting is critical for selecting specific hiPSCs populations for various applications but is well known to negatively affect hiPSCs viability and colony formation capacity. Therefore, we determined whether SiM5 treatment could increase hiPSCs viability after FACS compared to those treated with SMC4. The hiPSCs were pretreated with SiM5 or SMC4 for 1 h prior to FACS, and the hiPSCs were continuously treated with SiM5 or SMC4 for an additional 24 h. Compared to SMC4, hiPSCs treated with SiM5 showed increased cell survival post-seeding as observed under a microscope (Figure 5(a)), exhibited higher alkaline phosphatase activity (Figure 5(b)), and have a higher number of OCT-4-expressing colonies (Figure 5(c)). SiM5 increases hiPSCs survival after FACS by approximately 2.5 times compared to those treated with SMC4 (Figure 5(d)). To further optimize the FACS procedure, we also determined the effect of SiM5 on the survival of hiPSCs labeled with two fluorescent dyes commonly used to stain nuclear DNA, Hoechst 33342 and Vybrant® DyeCycleTM Ruby, after FACS. The results show that Hoechst 33342-labeled hiPSCs generate a higher number of OCT-4-expressing cell colonies after FACS compared to those labeled with Vybrant® DyeCycleTM Ruby (Figure 6(a) and (b)). These results suggest that Hoechst 33342 may be more appropriate for labeling hiPSCs for FACS than Vybrant® DyeCycleTM Ruby due to its lower toxicity.

Effect of SiM5 and SMC4 on human induced pluripotent stem cells (hiPSCs) viability following single-cell dissociation and fluorescence-activated cell sorting (FACS). (a) Morphology of hiPSC colonies treated with SiM5 or SMC4 on days 1 and 2 post-FACS. Magnification: 40× (b) Alkaline phosphatase (AP) staining of treated hiPSC colonies on day 2. (c) Representative fluorescent images of OCT4-positive colonies derived from SiM5- and SMC4-treated hiPSCs at day 1 post-FACS. Magnification: 40× (d) quantifies the number of OCT4-positive colonies at day 2 post-FACS. Data represent the mean ± SD of three biological replicates. Statistical significance was determined using Mann–Whitney U test. Scale bars: 200 µm.

The effect of Hoechst33342 and Ruby vital dyes on human induced pluripotent stem cell (hiPSC) viability following fluorescence-activated cell sorting (FACS). (a) Representative fluorescent images depict OCT4-positive colonies derived from hiPSCs stained with Hoechst33342 or Ruby on day 2 post-FACS. Magnification: 40× and 100× (b) Quantitative analysis of OCT4-positive colonies from Hoechst33342- and Ruby-stained hiPSCs on day 2 post-FACS. Data represent the mean ± SD of three biological replicates. Statistical significance was determined using Mann–Whitney U test. Scale bars: 200 µm.
Discussion
Although single-cell expansion of hPSCs is essential for many of their applications, it is known that hPSCs require survival signals from both cell-to-cell and cell-to-matrix interactions, which makes it difficult for them to survive as single cells.17,18 Despite recent progress in improving the survival and colony formation capacity of hPSCs after single-cell dissociation using various defined matrices and soluble factors, especially a ROCKi, such as Y-27632, the results remain generally poor with modest efficiency gains of only 5–20%.11,12,19–22
Our study aimed to improve the viability of hiPSCs after single-cell dissociation by incorporating specific small molecules that target key signaling pathways involved in cell survival into culture medium. p53 activation is known to hinder the derivation of hiPSCs by triggering cell cycle arrest and apoptosis after DNA damage.23–26 Our findings demonstrate that a combination of the p53 inhibitor PFT-α with ROCKi Y-27632 modestly increases hiPSCs viability after single-cell dissociation compared to those treated with Y-27632 alone. To further improve hiPSCs survival, we modified a commercially available SMC4, consisting of PD0325901 (MEK/ERK inhibitor), CHIR99021 (GSK3 inhibitor), thiazovivin (ROCKi), and SB431542 (TGF-β inhibitor), by adjusting the concentration of thiazovivin and adding PFT-α. We demonstrate that our optimized SMC4, named SiM5 (Supplementary Table 2), improved hiPSCs survival 2.5 times compared to cells treated with SMC4, and 25 times compared to those treated with Y-27632. The enhancing effect of SiM5 on hiPSCs viability can be attributed to the synergistic effect of its five components of small molecules, which collectively reduce hiPSCs apoptosis to a greater level than SMC4 and Y-27632. While the p53 inhibitor PFT-α, one of the components of SiM5, is known to induce cell cycle arrest, we did not observe this effect throughout the treatment period of SiM5. This result suggests that other components of SiM5 may counteract the PFT-α effect and prevent cell cycle arrest in hiPSCs. Although the removal of SiM5 after treatment slightly inhibits the hiPSCs cell cycle progression, the effect is transient and did not compromise their subsequent expansion.
Furthermore, SiM5 also promotes the colony formation capacity of hiPSCs after FACS and repeated single-cell expansion without affecting their pluripotency and chromosomal integrity. This greatly improved survival and colony formation capacity of hiPSCs after single-cell dissociation is probably due to the synergistic effects of inhibiting p53 signaling together with modulation of other signaling pathways, including MAP kinase, Rho kinase, TGF-β, and Wnt signaling that have been shown to promote self-renewal, suppress differentiation, and prevent anoikis of hiPSCs.20,21,27
The synergistic effects between each SiM5 component are complex and involve multiple signaling pathways. PD0325901 and CHIR99021 inhibit MEK and GSK3β, which play an important role in regulating pluripotency and cell fate decisions. 12 Inhibition of these pathways could promote the survival and self-renewal of hPSCs. Thiazovivin, a potent inhibitor of Rho-associated kinase, could decrease apoptosis and promote the survival of hPSCs by reducing cell contractility, facilitating cell attachment to the extracellular matrix, and promoting cell–cell adhesion by stabilizing the expression of E-cadherin, which is crucial for the survival of hPSCs after dissociation. 28 SB431542 may promote pluripotency and prevent hPSCs apoptosis by inhibiting the TGF-β type I receptor ALK5, as well as the Activin/Nodal receptors ALK4 and ALK7, which are critical for the TGF-β/Activin/Nodal signaling pathway, 29 while Pifithrin-α, a potent inhibitor of p53, can protect cells from stress-induced cell death and promote cell survival. 23
The synergistic effects of these five small molecules likely contribute to the improved ability of SiM5 to promote hiPSCs survival after single-cell dissociation. However, additional studies are required to fully elucidate the precise mechanisms of action of these small molecules and to optimize their utility for diverse stem cell applications. Furthermore, it is crucial to note that our investigation of the effects of SiM5 is focused on short-term treatment (less than 48 h), and the long-term effects of SiM5 on the hiPSC properties should be addressed in a future study.
Conclusions
In this study, we demonstrate that SiM5 treatment greatly improves the survival and colony formation capacity of hiPSCs after single-cell dissociation under various culture conditions without affecting their pluripotency and chromosomal integrity. We believe that our optimized SiM5 agent could be used to improve the efficiency of genetic manipulation and single-cell cloning of hiPSCs for various clinical and research applications.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251330956 - Supplemental material for The novel combination of small-molecule inhibitors increases the survival and colony formation capacity of human induced pluripotent stem cells after single-cell dissociation
Supplemental material, sj-docx-1-sci-10.1177_00368504251330956 for The novel combination of small-molecule inhibitors increases the survival and colony formation capacity of human induced pluripotent stem cells after single-cell dissociation by Chuti Laowtammathron, Pimjai Chingsuwanrote, Pimonwan Srisook, Phatchanat Klaihmon, Patcharee Meaunpim, Chanchao Lorthongpanich, Pakpoom Kheolamai and Surapol Issaragrisil in Science Progress
Footnotes
Acknowledgements
The authors thank Ms. Sirinart Buasumrit for laboratory management and administration. The authors also thank all the SiSCR members for their technical assistance.
Ethical considerations
This study was approved on January 28, 2020, by the Siriraj Institutional Review Board (COA no. Si 090/2020), Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand. The protocols used in this study complied with the principles established in the Declaration of Helsinki, the Belmont Report, the CIOMS Guidelines, and the ICH-GCP.
Informed consent
Author contributions/CRediT
CL conceived and designed the study, performed the experiments, interpreted the results, wrote the manuscript, and acquired funding. PC, PS, and PK performed experiments and conducted statistical analysis. PM conducted flow cytometry for cell cycle analysis. CLo interpreted the results, edited the manuscript, and PKh analyzed the results, and edited the manuscript. SI supervised the project, edited the manuscript, and acquired funding. All authors read and approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Faculty of Medicine Siriraj Hospital, Mahidol University, the Siriraj Foundation for Stem Cell Research (grant number: D003276). CL was also supported by a grant from the National Research Council of Thailand (NRCT; grant number: NRCT5-RSA63015-13).
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data following publication will only be shared upon receiving a written request and approval from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
