Abstract
Objective
The aim of this study is to establish a population pharmacokinetic (PK) model for patients undergoing continuous veno-venous hemodiafiltration (CVVHDF) and optimize the dosing regimen of colistin sulfate.
Methods
A prospective observational study in a single center was conducted on patients who were administrated with colistin sulfate and CVVHDF for at least 48 h. Blood samples were obtained prior to dosing and four to six blood samples (primarily C0.5h, C1h, C2h, C4h, and C6h) after dosing. The blood concentration of colistin sulfate was determined by ultra-high performance liquid chromatography-tandem mass spectrometry assay. The NONMEM program was used to establish the population PK model and perform Monte Carlo simulations. The predictability and stability of the model were internally evaluated by the goodness of fit plots, visual prediction check, and bootstraps.
Results
A total of 86 plasma concentrations from 20 patients were used for population PK modeling. A two-compartment model with first-order linear elimination best described the population PK characteristics of colistin sulfate. Cystatin C (CysC) and body weight (WT) were identified as covariates for clearance (CL). Internal evaluation results showed that the final model had good stability and prediction performance. Monte Carlo simulations showed that only when the body WT was 50 kg with CysC ≥3.07 mg/l, and when the body WT was 65 kg with CysC = 5.11 mg/l, and minimum inhibitory concentration (MIC) = 0.25 mg/l, the target attainment probability (PTA) of the daily dose of 1.5 million U regimen was ≥90%. All treatment regimens fail to achieve the target PTA when MIC = 1 mg/l.
Conclusions
With the decrease of CysC levels and the increase of WT, the dose of colistin sulfate may need to be increased. It may be prudent for colistin sulfate to consider an initial dose doubling and subsequent maintenance dosing regimen of 200–225 million unit daily, administered in 2–3 divided doses, to attain PTA standard. This study was registered at the Chinese Clinical Trial Registry (www.chictr.org.cn) (trial registration number ChiCTR2300072191).
Keywords
Introduction
The inappropriate use of antimicrobial drugs has resulted in a significant threat to global public health through the emergence of bacterial resistance. 1 This has necessitated the increased utilization of older polymyxin drugs for the treatment of carbapenem-resistant organisms (CRO) infections, which are frequently encountered in intensive care units. Furthermore, approximately 50% of critically ill patients experience acute kidney injury, with 5% requiring continuous renal replacement therapy (CRRT) and facing a mortality risk exceeding 50%. 2 Infections in this patient population frequently signify an unfavorable prognosis and elevated mortality rates, presenting a significant challenge to the clinical management and treatment.
Colistin, a polymyxin antibiotic, has been increasingly used to treat infections caused by CRO, especially in cases where other treatment options are limited. Colistin is available in two forms: colistin sulfate and colistimethate sodium (CMS). 3 While colistin sulfate is approved for intravenous administration in China. It disrupts the outer membrane of Gram-negative bacteria, leading to quick bacterial death and potentially enhancing the effectiveness of other antibiotics. In contrast to CMS, which functions as an inactive prodrug, colistin sulfate is administered in its active form, resulting in a more straightforward pharmacokinetic (PK) profile. 4 According to the CHINET surveillance network report, colistin demonstrates a sensitivity rate exceeding 95% against prevalent drug-resistant bacteria found in hospitals, including Acinetobacter baumannii, Pseudomonas aeruginosa, and Klebsiella pneumoniae. 5 However, some regions, such as the U.S. and Europe, recommend some recent agents, such as beta-lactam-beta-lactamase inhibitors (BLBLIs). Although BLBLIs have shown promising efficacy in the treatment of CRO infections, it is worth noting that certain BLBLIs such as ceftazidime–avibactam are available in China; not all are accessible or affordable, for example, ceftolozane–tazobactam, imipenem–cilastatin, and meropenem–vaborbactam. Hence, colistin sulfate is frequently used as a first-line regimen for the treatment of CRO in China. However, PK and dosing data for colistin sulfate, particularly in critically ill patients undergoing continuous veno-venous hemodiafiltration (CVVHDF), remain limited. 6 In addition, since the antibacterial spectrum and activity of colistin sulfate, CMS, and polymyxin B sulfate are similar, experts have called for PK/pharmacodynamic (PK/PD) investigations of this antibiotic. 4 In light of the significance of personalized drug therapy for managing CRO infections in CRRT patients, there is an urgent need for PK data on colistin sulfate.
This study seeks to develop a population PK model for colistin sulfate in critically ill patients undergoing CVVHDF as part of CRRT and to assess the effectiveness of various dosing strategies using Monte Carlo simulation.
Materials and methods
Patient setting
A prospective observational study was conducted at the First Affiliated Hospital of Guangxi Medical University between May 2023 and January 2024. The study was organized in accordance with the Helsinki Declaration of 1975 as revised in 2013. The study protocol was approved by the Medical Ethics Committee of the First Affiliated Hospital of Guangxi Medical University, Nanning, China (No. 2023-K131-01). Patients were consecutively enrolled as following inclusion criteria: (a) patients (≥18 years) with confirmed/suspected CRO infections; (b) receiving intravenous colistin sulfate (Shanghai New Asia Pharmaceuticals CO., LTD., Shanghai, China) ≥ 48 h; and (c) CRRT therapy ≥48 h. The exclusion criteria were as follows: (a) blood samples were not available; (b) died within 72 h after receiving colistin sulfate treatment; and (c) extracorporeal membrane oxygenation therapy. Written informed consent was obtained from all patients or patient representatives. All patient details have been de-identified. The reporting of this study conforms to STROBE guidelines. 7
Data collection
Demographic and clinical information was collected from electronic medical records, including acute physiology and chronic health evaluation (APACHE) II score, sequential organ failure assessment (SOFA) score, laboratory values, illness severity and treatment data, CRRT parameters, and residual diuresis. Creatinine clearance (CrCL) was calculated using the Cockcroft–Gault (CG) equation. 8
The main indicator for evaluating treatment outcomes is clinical treatment success, which is defined as improvement in clinical symptoms and laboratory indicators, including weaning from the ventilator, reduction or discontinuation of vasoactive drugs, body temperature ≤37.3°C, white blood cell count ≤10 × 109/L, procalcitonin <0.05 ng/ml or a decrease of >80%, and medical records indicating improvement in infection at the end of treatment. Otherwise, it is considered clinical treatment failure. The secondary indicator is microbiological clearance, which is only assessed in patients who have evidence of microbial infection and results of drug sensitivity tests. Microbiological clearance success is defined as a negative result when the same infected site specimen is cultured again during colistin sulfate treatment. Otherwise, it is considered microbiological clearance failure.
Continuous renal replacement therapy procedures
Continuous renal replacement therapy was performed with a Prismaflex system (Gambro; Baxter Medical Supplies Trading Co., LTD., Shanghai, China) using CVVHDF. Vascular access was through a double-lumen catheter inserted into the jugular or femoral vein. The hemofilter was a 1.5m2 polyacrylonitrile filter (AN69-ST150; Baxter Medical Supplies Trading Co., Ltd., Shanghai, China). A 100–200 ml/min blood flow rate (BFR), 500–1500 ml/h dialysate flow rate (DFR), 500–1500 ml/h replacement fluid rate (RFR), 50–300 ml/h ultrafiltration rate (UFR), 25–35 ml/kg/h CVVHDF dose intensity, 50–150 ml/h net removal rate, and 50–150 mmHg transmembrane pressure were set at the physicians’ discretion.
Drug administration and blood sampling
Colistin sulfate treatment was determined by the medical teams. One to 1.5 million units (MU) per day (1 MU = 44 mg), divided into 2 or 3 intravenous drops was recommended by the label sheet of colistin sulfate. All intravenous colistin sulfates were administered during CVVHDF treatment and infusion time 0.5 h or 2 h.
Patients underwent blood draws following CVVHDF and colistin sulfate treatment. All the blood sampling site was collected in dorsal artery of foot. During the maintenance phase, one blood sample (2 ml, Cmin) was obtained immediately prior to dosing, and four to six blood samples (primarily C0.5h, C1h, C2h, C4h, C6h) were obtained after dosing. Additional blood samples were collected as part of routine blood tests. Furthermore, AUC0−24h was calculated for individuals using the trapezoidal rule.
Quantification of colistin sulfate concentrations
All samples were processed by centrifugation for 5 min at 4000 rpm, with the supernatant collected and stored at −80°C until analysis. The plasma concentrations of colistin sulfate were measured using a validated method published previously. 9 Briefly, the quantification of colistin A and B was conducted utilizing ultra-high performance liquid chromatography-tandem mass spectrometry assay (DMT 9600BG, Hunan Demeter Instrument Co., Ltd., Changsha, China; ultra-high performance liquid chromatography-tandem mass spectrometry assay). Colistin sulfate isolated on K9 HILIC column (Hunan Demeter Instrument Co., Ltd., Changsha, China, 4.6 × 150 mm, 2.5 µm) at temperature of 40°C. The mobile phase consisted of eluent A: 0.1% formic acid aqueous solution and eluent B: 0.1% formic acid acetonitrile solution. The flow rate was 0.3 ml/min, and the total running time was 10 min. The relative standard deviation assays ranged from 0% to 10.8% for quality control samples, and their corresponding accuracy ranged from −10.4% to 10.1%. The limit of quantification was 0.027 mg/l for colistin A and 0.053 mg/l for colistin B. Accuracy, precision, extraction recovery, matrix effect, stability, carryover, dilution integrity, and incurred sample reanalysis all met the bioanalytical method validation guidance criteria. As colistin A and B have similar structures, molecular weight (WT), pharmacological activities, and PK characteristics, the plasma concentration of colistin was derived by summing the concentrations of colistin A and B. 10
Population PK modeling
The data modeling process was performed using NONMEM Version 7.4 (Icon Development Solutions, Ellicott City, MD, USA). R (version 4.2.1) software was applied to generate diagnostic plots. Based on previous population PK studies,10,11 one- and two-compartment models were first compared using the first-order conditional estimation. Interindividual variability was assessed using exponential function. Residual variability was described by additive, proportional, exponential, or mixed models. In addition, allometric scaling model was considered with the following formulation:
Model selection was based on diagnostic plots and parameters such as the objective function value (OFV), Akaike information criterion value, Bayesian information criterion value, and relative prediction error.
The following variables served as covariates, including sex, WT, SOFA score, APACHE II score, total bilirubin, albumin, alanine aminotransferase, aspartate aminotransferase, blood urea nitrogen, uric acid, serum creatinine (SCR), Cystatin C (CysC), CrCL, BFR, DFR, RFR, and UFR residual diuresis. All covariates are tested on model parameters. They were considered using a stepwise regression to build the model. A decrease of OFV >3.84 (p < 0.05) for forward addition and an increase of OFV >6.63 (p = 0.001) for backward elimination were the inclusion criteria for covariates. The final model was internally validated for reliability, predictive ability, and stability using the goodness-of-fit (GOFs) method, the prediction-corrected visual predictive check (pcVPC) by simulating 1000 patients, the bootstrap method with 1000 samples, and all patients’ observed concentrations and individual predicted values versus time were also used to test the reliability of the final model.
Dosing regimen simulations
Based on the final population PK model, Monte Carlo simulations with 1000 subjects were performed for six fixed maintenance dosing regimens (0.5 MU q12h, 0.5 MU q8h, 0.75 MU q12h, 0.75 MU q8h, 1 MU q12h, 1 MU q8h) in conjunction with an initial loading dose equivalent to twice the maintenance dose across various minimum inhibitory concentration (MIC) levels ranging from 0.5 to 1 mg/l, accounting for multiple covariates.
According to the reference report, 12 the protein binding rate of colistin sulfate is about 50%, and fAUC/MIC ≥20 is set as the PK/PD target. A target attainment probability (PTA) of ≥90% is used as the selection for dose optimization.
Results
Patient information and treatment outcomes
The study included 20 patients treated with CVVHDF for acute kidney injury. The patients had high SOFA and APACHE II scores, indicating severe illness. Over 75% of patients used vasoactive drugs, mechanical ventilation, and had multisite infections, mainly in the lungs (90%). All isolated pathogens were CRO (meropenem MIC ≥8 mg/l), mainly including Klebsiella pneumoniae (35%), Acinetobacter baumannii (30%), and Pseudomonas aeruginosa (30%). Details are shown in Table 1.
Demographic and clinical data of patients (n = 20).

Mean ± standard deviation shown for continuous variables; proportion shown for categorical variables.
APACHE II: acute physiology and chronic health evaluation; ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALB: albumin; BFR: blood flow rate; BUN: blood urea nitrogen; CysC: Cystatin C; CrCL: creatinine clearance; CVVHDF: continuous veno-venous hemodiafiltration; DFR: dialysate flow rate; MIC: minimal inhibitory concentration; MU: million units; RFR: replacement fluid rate; SCR: serum creatinine; SOFA: sequential organ failure assessment; TBIL: total bilirubin; UC: uric acid; UFR: ultrafiltration rate; WT: weight.
All patients received intravenous infusion of colistin sulfate, and three patients with lung infections also received nebulized colistin sulfate. The Css, min of colistin sulfate was 0.30 ± 0.22 mg/l, and the AUCss, 0−24h was 12.51 ± 6.41 (mg·h/L). AUCss, 0−24h could not be acquired for four patients as there were too few blood sampling points. The concentration time profiles for the 16 patients are presented in Figure 1. Out of the 20 patients, 7 (35%) were considered to have successful clinical treatment; 13 patients had evidence of microbial infection and results of drug sensitivity tests, of these 13 patients, 3 (23%) were considered to have successful clinical treatment, and 5 (38%) were considered to have successful microbial clearance.

The concentration–time curves for 16 patients.
Population PK model
The PK characteristics of colistin sulfate may be well described by a two-compartment model with linear elimination. The allometric scaling model was not suitable for our data, so it was not considered finally. Upon examining interindividual variability, it was observed that incorporating variability into V1 and Q yielded values nearing zero, with shrinkage values surpassing 30%.
13
The alterations in the OFV were not statistically significant. Therefore, interindividual variability was incorporated solely into CL and V2. The covariate model introduces CysC and WT as covariates for colistin sulfate CL, with interindividual variability and residual variability modeled using an exponential model. The scatter plots of interindividual variability versus covariates of base model and final model are shown in Figures S1 and S2, and the development process of the base model is shown in Table S1. The detailed modeling process of the covariate model is presented in Table 2. It should be stated that clearance decreases as the CysC level increases since this increase indicates reduced kidney function, and that clearance increases with increased body WT. The final population PK model is as follows:
The process of population pharmacokinetic modeling.

AIC: Akaike information criterion value; BIC: Bayesian information criterion; CysC: Cystatin C; UFR: ultrafiltration rate; IIV, interindividual variability; OFV: objective function value; TBIL: total bilirubin; WT: weight.
In this model, CL represents the drug clearance rate of colistin sulfate; V1 represents the volume of distribution in the central compartment; V2 represents the volume of distribution in the peripheral compartment; Q represents the intercompartmental clearance rate. The population typical values for CL, V1, V2, and Q were 3.69, 20.5, 33.2, and 25.3, respectively. CYSC and WT are included on CL in power form and exponential form, respectively. The values 2.31 and 65 were the medians for CYSC and WT, and in addition, −0.626 and 1.03 were their effects on CL.
Model evaluation
The GOFs diagnostic of the final model, as shown in Figure 2, indicates that the individual predicted values and the actual measured blood drug concentrations are evenly distributed around the line of identity in the plot, with the trend line close to the line of identity. This observation implies that the disparity between predicted and actual values is small, signifying the model's strong predictive capability. Furthermore, the conditional weighted residual scatter plots against time and population predicted values demonstrate that the majority of data points fall within the range of ±2, indicating the model's overall accuracy.

Goodness-of-fit (GOFs) plot for the final model. (a) Individual predictions versus concentrations; (b) Population predictions versus concentrations; (c) Conditional weighted residuals (CWRES) versus population predictions; and (d) CWRES versus time.
The results of pcVPC verification of the final model are shown in Figure 3, which represented that the model adequately described the overall trend and variability in the observed data. As shown in the figure, the majority of colistin sulfate concentration values were within the 90% prediction interval, indicating that the predictive performance of the final model and its use for subsequent dosing simulations were acceptable.

Prediction-corrected visual predictive (pcVPC) of the final model. Circles represent observations. Red solid line stands for the median of simulated concentrations, and red imaginary lines stand for the 90% prediction interval (PI) (5%, 95%) of the predictive colistin sulfate concentrations. Shaded areas represent the 95% confidence interval (CI) for each line.
Out of 1000 bootstrap datasets, the final model ran successfully 989 times, resulting in a robustness rate of 98.9%. The results are shown in Table 3. The median PK parameters from the bootstrap are close to the PK parameters summarized from the final model. The 95% confidence intervals include the parameter estimates from the final model, indicating good model stability, predictive accuracy, and robustness.
Pharmacokinetic parameters of the final model and bootstrap results.

CI: confidence interval; IIV: interindividual variability; RSE: relative standard error; RSV: residual variability.
The shrinkage values of interindividual variability and residual variability.
The plots of observed and individual predicted values versus time showed that almost all patients’ observations fall on the curve of the individual predicted, indicating a good predictive performance of the model (Figure 4).

Observed concentration (blue point) and individual predicted concentration (curve) versus time of 20 patients.
Monte Carlo simulation
Based on the final model results, CysC and WT were included as model covariates. Target attainment probability exhibited a positive correlation with both the colistin sulfate dosing and CysC levels, while demonstrating a negative correlation with WT and MIC.
In the 50 kg group, the 1 MU, q8h dosing regimen met the standard when MIC = 0.25 mg/l at all CysC levels, while the 0.5 MU, q12h regimen did not. Other dosing regimens met the standard at specific CysC levels.
In the 65 kg group, the 0.5 MU q8h, 0.75 MU q12h/q8h, and 1 MU q12h/q8h dosing regimens met the standard when CysC was ≥3.07 mg/l, 3.07/2.31 mg/l, and 3.07/1.4 mg/l, respectively, with MIC = 0.25 mg/l. Only the 1 MU q8h dosing regimen met the standard when CysC was 5.11 mg/l with MIC = 0.5 mg/l.
In the 80 kg group, the 0.75 MU q8h and 1 MU q12h/q8h dosing regimens met the standard when CysC was 3.07 mg/l, 5.11/2.31 mg/l, respectively, with MIC =0.25 mg/l. None of the regimens met the standard when MIC = 0.5/1 mg/l. The simulation results are shown in Figure 5. The recommended regimens for different CVVHDF patients when MIC = 0.25 mg/l are listed in Table 4.
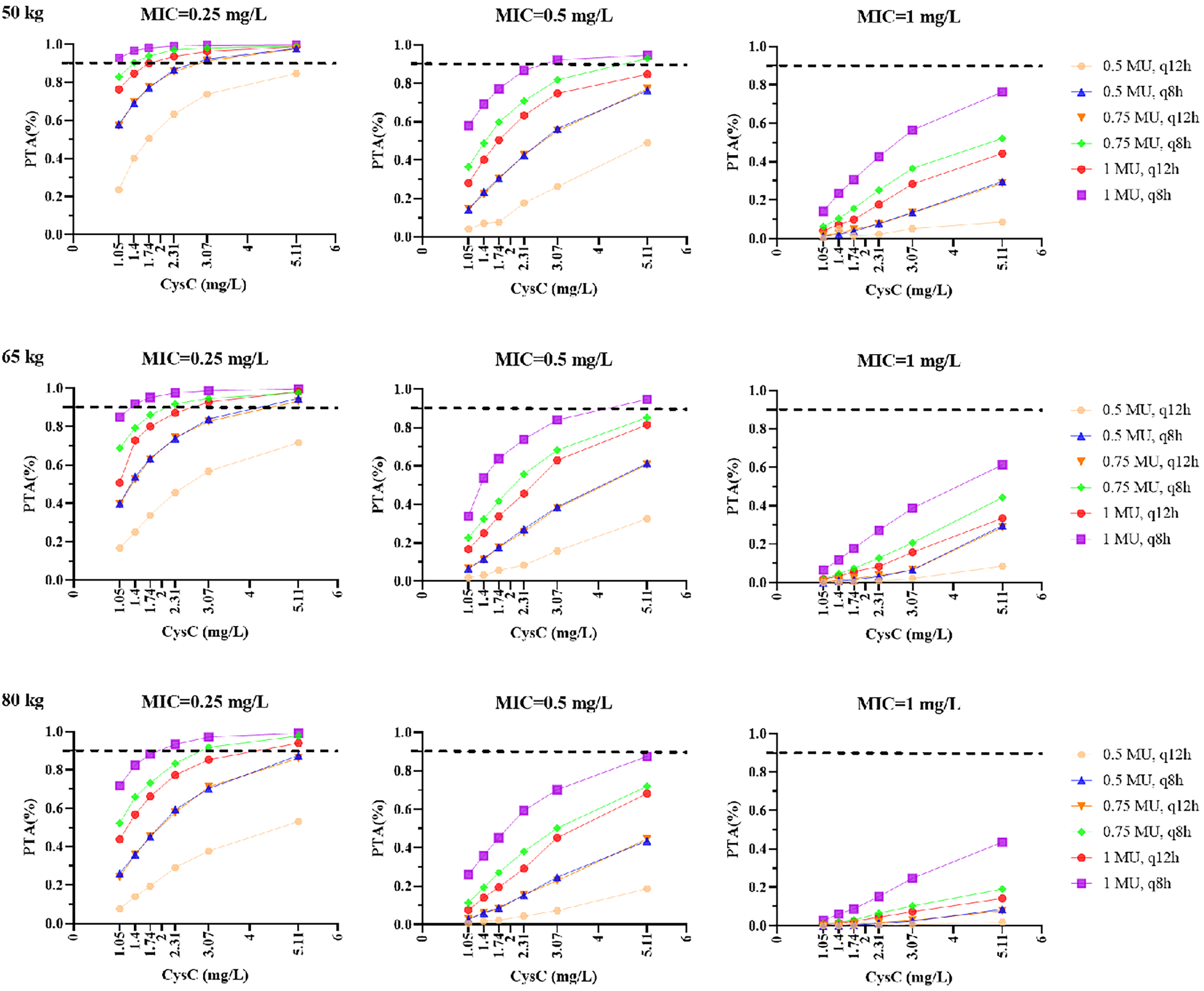
Simulated administration of colistin sulfate with target attainment probability (PTA) probability (fAUC/MIC ≥20) at six different dosing regimens note: the 5%, 25%, 50%, 75%, and 95% percentiles of CysC (range 1.05–5.11 mg/l) and the 5%, 50%, and 95% percentiles of WT (range 50–80 kg) from this dataset were grouped for simulation at different MIC values (0.25 mg/l, 0.5 mg/l, and 1 mg/l). MIC: minimal inhibitory concentration; CysC: Cystatin C; MU: million units; q12h: every 12 h; q8h: every 8 h; 1 MU = 44 mg.
The recommended dosage regimens for CVVHDF patients in different situations (MIC = 0.25 mg/l).

CVVHDF: continuous veno-venous hemodiafiltration; CysC: cystatin C; MIC: minimal inhibitory concentration; MU: million units; WT: weight; 1 MU = 44 mg.
Recommended only with therapeutic drug monitoring.
Discussion
In comparison to previous clinical studies examining the efficacy of colistin sulfate,10,11,14,15 the treatment success rate in the current study is notably lower. This discrepancy may be attributed to the heightened severity of illness within the study population, as indicated by a mean APACHE II score of 26.64 ± 10.12, a high prevalence of lung infections and multiple site involvement (90% and 75%, respectively), and the reduced exposure to colistin sulfate among patients undergoing CRRT so that it is difficult to achieve the PK/PD efficacy. International guidelines 16 recommend that the PK/PD index of polymyxin drugs be AUC/MIC and that for the sensitive breakpoint of Klebsiella pneumoniae, Acinetobacter baumannii, and Pseudomonas aeruginosa is MIC ≤ 2 mg/l; the AUC of colistin is recommended to reach 50 mg·h/L, and the AUC of polymyxin B is recommended to reach 50–100 mg·h/L. It can be seen that the AUC of this study was 12.51 ± 6.41 (mg·h/L), far below the above recommended range. Although the PK/PD target value of colistin sulfate has not been established, considering that it is very close to the antibacterial activity of polymyxin B and CMS in vitro, the PKPD index and target value of the three drugs may be mutually referable. 17 Therefore, the inappropriate administration of colistin sulfate doses of anti-infective medications in patients undergoing CVVHDF may impede the assessment of both efficacy and safety, potentially resulting in treatment ineffectiveness. The frequent blood sampling intervals employed in this study provide a comprehensive depiction of the PK profile, allowing for a detailed characterization of PK variability across various clinical scenarios. The population typical value CL obtained from the final model is 3.69 L/h, which is higher than the previous research results of 0.994 L/h and 1.5 L/h,10,11 which may be related to whether the study population is treated with CRRT.
The clearance of drugs in CRRT is primarily influenced by factors including molecular WT, protein binding rate, apparent distribution volume, and drug clearance pathway. Colistin sulfate, 12 a water-soluble antimicrobial drug, has a molecular WT of approximately 1100 Da and a protein binding rate of around 50%. A lower apparent distribution volume indicates that the drug predominantly remains within the bloodstream. According to drug instructions, colistin sulfate is primarily eliminated through renal excretion, suggesting that CRRT may impact the clearance rate of this drug. Colistin sulfate and polymyxin B sulfate are both active forms of drugs. After entering the body, they directly exert their effects. Infectious Diseases Society of China's consensus suggests that the PK characteristics of the two drugs are similar. 4 The findings of a population PK study on polymyxin B sulfate in critically ill patients undergoing continuous veno-venous hemofiltration (CVVH) mode 18 indicate that the population typical value of CL in the CVVH group (4.52 L/h) is significantly higher than in the non-CVVH group (1.95 L/h). Additionally, AUC of the drug is significantly lower in the CVVH group compared to the non-CVVH group (27.94 ± 10.92 vs. 77.89 ± 35.66 mg·h/L, p < 0.001), suggesting that CVVH leads to a significant increase in the clearance rate of polymyxin B sulfate. Another population PK study of CMS suggests that patients on CVVHDF require higher doses compared to patients with normal renal function due to suboptimal colistin concentrations. 19 Similarly, the CVVHDF CL from the above study was determined to be 2.98 L/h for colistin, which is higher than the previous research results10,11 for non-CRRT patients and closer to the CL of 3.69 L/h found in our study. These observations may provide insight into the higher CL values observed and the higher dosing recommended in the population PK model utilized in this study. It is worth noting that the study on the effect of CRRT on the blood concentration of colistin sulfate by Peng Danyang et al. 20 suggests that CRRT has a small impact on drug elimination. There is no statistical significance in the difference in steady-state trough concentration between the CRRT group and the non-CRRT group (0.58 ± 0.30 vs. 0.64 ± 0.25 mg/l, p = 0.328), but the steady-state AUC was not compared.
Previous research on the population PK model of colistin sulfate has indicated that renal function plays a crucial role in drug clearance,10,11 with CrCL estimated by CrCL as a key covariate. However, CrCL calculated by the CG equation has limitations when used in critically ill individuals, where significant fluctuations in renal function are common. It may underestimate the CrCL, leading to insufficient drug dose. 21 Although, our study also found no relationship between CrCL and clearance. Besides, a method of a 24-h or shorter urine collection to evaluate drug clearance is considered the reference standard for assessing renal function in PK studies. 22 This study reveals that serum CysC levels and body WT also significantly influence the clearance of colistin sulfate. CysC, a small molecular protein produced consistently by nucleated cells throughout the body, is present in all tissues and can be filtered by the glomeruli, rather than being secreted into renal tubules or reabsorbed into the bloodstream. In comparison to SCR, CysC exhibits reduced reliance on factors such as age, gender, race, and muscle mass, rendering it a superior alternative indicator for assessing glomerular filtration rate. 23 Due to its larger molecular WT of 13250 Da, CysC may not be effectively cleared through traditional hemodialysis or diffusion-based dialysis methods. 24 In contrast, creatinine, with its smaller molecular WT of 113 Da, can be effectively eliminated through dialysis. The CVVHDF treatment mode of CRRT in this study utilizes both convection and diffusion mechanisms, resulting in effective clearance rates for small and medium molecular- WT drugs. This may explain the observed impact of CysC and blood creatinine, key serum markers for assessing kidney function, on CL in the CRRT population studied, with the former showing significance while the latter did not.
Consistent with other clinical studies, it is suggested that CVVHDF or hemofiltration has a lesser impact on CysC levels compared to SCR. Baas et al. 25 studied the serum CysC concentration of 18 oliguric acute kidney injury patients receiving CVVH treatment over three consecutive sampling periods. The clearance rate of CysC was 17 ml/min, equivalent to a clearance rate of 2 mg/h, which is less than 30% of the production rate of CysC. Consequently, it is posited that CVVH treatment may have minimal impact on serum CysC concentration, thereby suggesting its potential utility as a marker for residual renal function during CVVH therapy. Kim et al. 26 found that serum CysC at the time of stopping CRRT is an independent predictor of successful withdrawal from CRRT in critically ill patients with acute kidney injury, and believe that serum CysC levels may be used to monitor residual renal function. Balik et al. 27 studied the role of CVVHDF in critically ill patients and concluded that CysC was not significantly cleared. The findings of these studies indirectly imply that the elimination of colistin sulfate could be influenced by the remaining renal function as estimated by CysC, and that CysC may exhibit superior predictive capabilities in the population PK model compared to SCR.28,29
Modifying drug dosage according to body WT is a medication approach that demonstrates favorable adherence. While international guidelines advocate for adapting the dosing regimen of polymyxin B sulfate in accordance with body WT, 16 there remains discordance in the findings of subsequent modeling studies. 30 However, in the present investigation, the incorporation of body WT as a covariate for the clearance of colistin sulfate resulted in a significant reduction in the population typical value CL and IIVCL, a practice not observed in prior research.10,11 In addition, the dosing regimen of CMS is not adjusted according to body WT. 16 So, the PK of colistin sulfate may be analogous to those of polymyxin B, indirectly implying that WT could influence its PK profile. Further investigation is warranted to elucidate the correlation between colistin sulfate exposure and body WT. Additionally, the study indicated that CVVHDF parameters did not exert a significant influence, possibly attributed to the high flow rate of CVVHDF, which could lead to a platform effect on the elimination of colistin sulfate.31,32
The results of the Monte Carlo simulation show that the exposure of colistin sulfate is significantly positively correlated with CysC and significantly negatively correlated with body WT. Considering that there are many formulas for calculating the glomerular filtration rate based on CysC and the influencing factors are different,33–35 it is more reliable to use CysC directly to simulate the dosing regimen without related conversion. Currently, the maximum recommended daily dose of colistin sulfate in the instructions is 1.5 MU. This dosing regimen only has a PTA of ≥90% in patients in the 50 kg group with MIC = 0.25 mg/l and CysC ≥3.07 mg/l. However, when MIC ≥0.5 mg/l, the PTA of the six simulated dosing regimens basically does not meet the standard under different simulated body WTs and CysC levels. Different from previous studies, it is suggested that the regimen of a 1.0–1.5 MU daily dose recommended by the current label of colistin sulfate reach PTA ≥90% only for MIC values ≤0.5 mg/l or with renal impairments in critically ill patients.10,11 Importantly, the recommended dosing regimen of label was insufficient for patients with normal renal functions (CrCL ≥80 ml/min) or those infected by pathogens with MIC ≥1.0 mg/l. This discrepancy may be attributed to the influence of CRRT on drug clearance in the population. Specifically, CRRT may enhance drug clearance due to its impact on the apparent distribution volume, which is further exacerbated by the heightened systemic inflammatory response observed in critically ill CRRT patients. 2 In addition, although no statistical analysis was conducted, it was noted that the plasma concentration range of colistin sulfate in non-CRRT patients, 11 which spanned from 0.28 to 6.20 mg/l, was significantly higher than the concentration range of 0.09–2.32 mg/l observed in this study, thereby supporting the aforementioned statement. Furthermore, the low-dose unit conversion of colistin sulfate (1 MU = 44 mg) also partially explained the possible low blood concentration (mg/l) of individuals. 4
Hence, considering the significant variability in CysC levels influenced by alterations in clinical status and CRRT modality, it may be prudent for the administration of colistin sulfate to consider an initial dose doubling and subsequent maintenance dosing regimen of 0.75 MU, q8h or 1 MU, q12h, with a 0.5-h infusion time to attain PTA standard, rather than the labeled dose of 1–1.5 MU. Three retrospective cohort studies were conducted to evaluate the safety of colistin sulfate and polymyxin B, encompassing a total of 909 patients. These studies36–38 administered colistin sulfate at a maintain dosage range of 1–1.5 MU per day, while polymyxin B was maintained within a therapeutic dosage range of 0.02–0.03 MU/kg per day, with the initial dose either loaded or unloaded. The findings consistently demonstrated a significantly lower incidence of acute kidney injury or drug-related nephrotoxicity associated with colistin sulfate compared to polymyxin B, with p < 0.05 (18.8% vs. 51.5%; 7.0% vs. 21.1%; 5.9% vs. 20.5%). Given the close similarity in molecular structures and PK properties between polymyxin B and colistin, it is plausible to infer that the nephrotoxicity safety threshold for colistin sulfate is comparable to that of polymyxin B (Cmin 3.13 mg/l). 11 In the present study, the Cmin of all patients was observed to be below 3.13 mg/l. Furthermore, colistin sulfate did not exhibit adverse effects on skin pigmentation, which may be attributed to the absence of phenylalanine in its molecular structure. Nevertheless, the safety profile of administering colistin sulfate at high dosages, specifically 3 MU per day, remains unsubstantiated. While our research indicates that a dosage of 3 MU may enhance the probability of PTA ≥90%, its use is not advisable without the implementation of therapeutic drug monitoring. 39 Simultaneously, it is highly advised, in accordance with the guidelines for infection diagnosis and treatment, that infections caused by multidrug-resistant bacteria be treated with a combination of one or even two drugs demonstrating activity or synergistic effects in vitro.40,41
However, this study is subject to certain limitations. Firstly, the sample size of patients and blood samples obtained is relatively small, necessitating further research and external validation to assess the study findings. Secondly, the study did not monitor blood drug concentrations outside of CRRT states, thus lacking analysis of the effect of CRRT on the clearance rate of colistin sulfate. Thirdly, the PK/PD target and protein binding rate were derived from preclinical studies, and PTA endpoint of the dosing regimen requires additional clinical validation. Lastly, the study excluded patients who died, which could potentially bias the assessment of efficacy.
Conclusion
This study represents the initial investigation into the population PK of colistin sulfate in patients undergoing CRRT, revealing that CysC levels and body WT exert a significant influence on the PK of colistin sulfate. Patients undergoing CVVHDF may require higher doses of the drug to achieve the desired PK/PD target attainment. It is advisable to employ therapeutic drug monitoring to individualize dosing regimens and consider combination therapy for optimal treatment outcomes. Further clinical research is imperative to enhance our understanding of the PK/PD profile of colistin sulfate.
Supplemental Material
sj-docx-2-sci-10.1177_00368504251325334 - Supplemental material for Population pharmacokinetics of colistin sulfate in patients on continuous veno-venous hemodiafiltration
Supplemental material, sj-docx-2-sci-10.1177_00368504251325334 for Population pharmacokinetics of colistin sulfate in patients on continuous veno-venous hemodiafiltration by Tianmin Huang, Yilin Luo, Yun Wu, Lulu Niu, Yang Xiao, Tingqing Wu, Xin Chen, Yongjun Liu, Jiejiu Lu, Donglan Zhu and Taotao Liu in Science Progress
Supplemental Material
sj-doc-3-sci-10.1177_00368504251325334 - Supplemental material for Population pharmacokinetics of colistin sulfate in patients on continuous veno-venous hemodiafiltration
Supplemental material, sj-doc-3-sci-10.1177_00368504251325334 for Population pharmacokinetics of colistin sulfate in patients on continuous veno-venous hemodiafiltration by Tianmin Huang, Yilin Luo, Yun Wu, Lulu Niu, Yang Xiao, Tingqing Wu, Xin Chen, Yongjun Liu, Jiejiu Lu, Donglan Zhu and Taotao Liu in Science Progress
Footnotes
Authors statement
Tianmin Huang: Data curation, Project administration, Supervision, Writing—review & editing. Yilin Luo: Data curation. Yun Wu: Formal analysis. Lulu Niu: Software. Yang Xiao: Methodology. Tingqing Wu: Methodology. Xin Chen: Investigation. Yongjun Liu: Investigation. Jiejiu Lu: Software. Donglan Zhu: Data curation. Taotao Liu: Resources, Supervision, Writing—review & editing.
Acknowledgments
The authors thank the ICU team (physicians and nurses) that selected the patients and collected the samples, making this work possible.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author upon reasonable request
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The study protocol was approved by the Medical Ethics Committee of the First Affiliated Hospital of Guangxi Medical University in May 2023, Nanning, China (No. 2023-K131-01).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Chinese Research Hospital Association Project: Research Project on Monitoring and Evaluation of the Use of Key Clinical Drugs (No. Y2023FH-YWPJ03-207) and Natural Science Foundation of Guangxi, China (No. 2021JJA140122).
Informed consent
Written informed consent was obtained from all patients or patient representatives. All patient details have been de-identified.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

