Abstract
Objectives:
The present research was accomplished to characterize probiotics from broiler gastrointestinal tract (GIT) by profiling biochemical, antimicrobial, and antibiotic sensitivity properties. Eventually, probiotic potentiality was evaluated as a substitute for antibiotic supplements in broiler focusing growth performance, carcass characteristics, and serum lipid profile.
Methods:
Probiotic bacteria were characterized based on morphological, physiological, and several biochemical tests. Antibacterial activity against a broad spectrum of antibiotics and bacterial pathogens was detected. An in vivo trial was conducted on 40-day-old Ross 308 broiler strains during 21 days in an in vivo trial. The chicks were divided into total of five groups, a control group and four experimental groups (Antibiotic1, Antibiotic2, Probiotic1, and Probiotic2) in a completely randomized design. Probiotic was supplemented in broiler feed (2× 109 CFU/g feed) or by direct oral gavage (1× 109 CFU/chick). The variables of production performance like body weight (BW), average daily gain (ADG), feed intake (FI), and feed conversion ratio (FCR), carcass characteristics and serum lipid profile were measured.
Results:
10 probiotic bacteria were presumptively identified as Lactobacillus sp. based on the morphological, physiological, and strong resistance properties in several biochemical tests. The mixture of Lactobacillus had favorable effects on productive performance of broilers regarding BW, ADG, and FCR (p < .05) compared with chickens that had no additive or had antibiotic during overall period of in vivo trial. Additionally, noteworthy efficacy on carcass characteristics and serum lipid profile were found (p < .05) in Lactobacillus mixture fed chicken groups of in vivo trial.
Conclusion:
Mixed Lactobacillus sp. can be considered as a potential additive for broiler diet attributable to noteworthy efficacy on growth performance, carcass characteristics, and serum lipid profile. Accordingly, the research highlights the need for suitable alteration of antibiotics through probiotic characterization and proper inclusion in broiler diet.
Introduction
Antibiotics are extensively fed as a growth promoter at a sub-therapeutic level to prevent infections caused by pathogenic microbes to improve production of broiler chickens.1,2 Antibiotics that can improve the growth of chicken and kill pathogenic microorganisms in the chicken gut are known as antibiotic growth promoters (AGPs), 3 sub-therapeutic antibiotics (STAs), 4 or in-feed antibiotics (IFA). 5 But, due to contemporary biosecurity threats arising from the production and transmission of antibiotic-resistant genes in microbes, and drug residues in poultry meat, the continuous feeding of antibiotics as a growth promoter has been forbidden in the major developed countries.6,7
Therefore, the poultry industry needs proper alternative products that can improve chicken growth, intestine health, and immunity while confirming the safety of the meat. 8 Many probiotics have growth-promoting effects that can be equivalent to antibiotic like bacitracin methylene disalicylate, oxytetracycline, avilamycin, virginiamycin, and chlortetracycline.9,10 In recent years, substantial demand has been raised in supplementing probiotics in animal feeds as a substitute for antibiotics to enhance productivity.11–13
Probiotic supplementation for chicken can be provided by supplementing lactic acid bacteria (LAB) as they can retain viability during transit in GIT such as Lactobacillus strains. Most of the Lactobacillus strains are well-characterized, dominant, and nonpathogenic residents among LAB.14,15 Numerous species of the genera Lactobacillus are “generally recognized as safe” by the (Food and Drug Administration (FDA), USA and acknowledged as the “qualified presumption of safety” status by the European Food Safety Authority (ESFA).16,17 Moreover, their antimicrobial activity towards broad-spectrum pathogens 18 and potential antibiotic susceptibility pattern 8 have proved their colonization abilities and competitiveness in gut which makes Lactobacillus strains suitable for use in chicken diet.19,20
Several authors recently specified that a mixed Lactobacillus or specific probiotic strain favorably improved body weight (BW), average daily gain (ADG), feed conversion ratio (FCR), feed intake (FI), carcass characteristics, and reduced cholesterol level of chickens.19,21,22 Moreover, researchers suggest that probiotic supplementation during early age has more improved productive performances in broilers than advanced age. 23 A recent work suggests that probiotics supplementation has improved chicken intestinal morphology, strengthened immune system, and lessened liver enzymes. 24 Another research reported significantly better blood cholesterol levels, high-density lipoprotein, and low-density lipoprotein condition in chicken after feeding probiotic to chickens. 25
To fulfill the demand for acquiring functional products with better probiotic features, the current analysis of probiotic feeding aimed to analyze overall ability of probiotics to withstand harsh or competitive conditions of in vitro tests like biochemical or antimicrobial activities, to expand probiotic exploitation as a dietary supplement, and to assess their collective impact on productive performance and serum lipids of broiler chicks as a substitute for antibiotics.
Materials and methods
The research was conducted at the Animal Cell Culture Laboratory of Biotechnology and Genetic Engineering Discipline, Khulna University by properly following necessary animal handling and management guidelines. The study was reported according to the relevant EQUATOR Network guidelines. 26 The experimental trial on broiler chicks was approved by the “Khulna University Animal Care and Ethics Committee” at September 12, 2018 (agreement no: KUACEC-369418). The study was reported according to the ARRIVE checklist.
Materials used in the study
For probiotic isolation, six healthy broiler chickens were collected from poultry farm of Rajshahi, Sylhet and Rangpur Divisions of Bangladesh. For Probiotics isolation and characterization, chemicals used were Man Rogosa and Sharp (MRS) medium, crystal violet, grams iodine solution, 95% ethanol, safranin, malachite green, 3% hydrogen peroxide, NaCl, bile salt, phenol, glucose, maltose, lactose, sucrose, fructose, galactose, raffinose, mannose, sorbitol, xylose, erythromycin (15 µg), tetracycline (30 µg), chloramphenicol (30 µg), gentamicin (10 µg), kanamycin (30 µg), penicillin G (10 µg), amoxicillin (10 µg), and nalidixic acid (30 µg), etc. For antimicrobial activity test, pathogens used were Vibrio cholera, Escherichia coli, Salmonella typhi, Staphylococcus aureus, and Listeria monocytogenes. For in vivo trial, distinct floor pens with a wood shaving floor, proper light, fan, and water provision were provided. As a basal feed for chicks, starter mash feed was provided. Chicks were adminstered with required vaccines in time. During in vivo trial, chemicals used were oxytetracycline, chlortetracycline, heparin, etc.
Isolation of bacteria
Bacteria were isolated from GIT contents (comprising caeca, crop, and small intestine) of six 15 and 30 days old broiler chickens collected from three divisions (Sylhet, Rajshahi, and Rangpur) of Bangladesh. Incubation was done by plating the bacteria onto Man, Rogosa, and Sharpe (MRS) medium 27 for 24–36 h at 37°C by ensuring pH = 6.5. Subculture was done for 7–8 times, and isolated pure cultures were assessed based on bacterial morphology, Gram staining, motility test, endospore test, coagulase test, and catalase test. Pure culture of bacteria was stored at −20°C in 20% (v/v) glycerol for long-term preservation.
Biochemical characterization
Screening of potential probiotic was done by performing several biochemical tests. Isolated pure cultures were evaluated for carbohydrates (glucose, maltose, lactose, sucrose, fructose, galactose, raffinose, mannose, sorbitol, and xylose) utilization test. 28 Then, isolates were assessed for their capability to resist NaCl, 29 bile salt, 30 phenol, 17 acidic pH, 30 and high temperature 31 to estimate the in vitro capability of probiotics to survive during transition in the intestine. In NaCl, bile and phenol tolerance test, MRS broth supplied with the inhibitory substances such as NaCl (2%, 4%, 6%, and 8%), bile salt (0.3% and 0.5%), and phenol (0.3% and 0.4%) were prepared for an individual test. Prepared chemicals were inoculated with 1% fresh overnight pure culture of probiotic. In high-temperature tolerance test, tolerance was evaluated by exposing probiotics to high temperature; 45°C, 50°C, and 55°C. All these above-described test was analyzed to all the inhibitory effects on probiotic by measuring optical density (OD) value. OD was determined at 620 nm every 4 h interval for 24 h by spectrophotometer for above described particular tests. The verification of all these biochemical tests was also performed on MRS agar for 24 h at 37°C by observing the presence or absence of bacterial growth.
Antibiotic susceptibility
Antibiotic susceptibility pattern of isolated bacteria was screened to determine susceptibility or resistance profiles against eight antibiotics: erythromycin (15 µg), tetracycline (30 µg), chloramphenicol (30 µg), gentamicin (10 µg), kanamycin (30 µg), penicillin G (10 µg), amoxicillin (10 µg), and nalidixic acid (30 µg). Antibiotic susceptibility pattern was done by following disk diffusion method. 32 Briefly, antibiotic disc (6 mm) soaked with particular antibiotics were placed on MRS agar plates containing probiotic bacteria in it. Incubation was done at 37°C for 24 h. The diameters of inhibition zone were determined and the data (average of three test readings) were referred to as susceptible (S), intermediate (I), or resistant (R) based on the guidelines of the Clinical and Laboratory Standards Institute (CLSI). 33
Antimicrobial activity
Antimicrobial activity of 10 isolated bacteria was analyzed against five pathogens: Vibrio cholera, Escherichia coli, Salmonella typhi, Staphylococcus aureus, and Listeria monocytogenes. The experiment was done by modifying the “deferred cross streak” technique. 32 Briefly, on MRS agar plate, wells were prepared for inoculating pathogens. At first, streaking was performed with probiotic isolates (106 CFU/ml) on MRS agar plate and incubated at 37°C. One hundred micro litter of the particular tested pathogen was then inoculated on the wells at 107 CFU/ml for 24 h at 37°C. The diameter of the inhibition zone was measured around the wells (average of three test readings).
Mixed probiotic culture preparation and administration method
Administration of mixed probiotic culture was done in two ways; direct oral gavage and by mixing with broiler mash feed.
For oral gavage of probiotic culture, supplementation was prepared by following the modifications of Brisbin et al.,
34
Angelakis and Raoult,
35
and Higgins et al.
36
Briefly, each strain of probiotic was cultured in MRS broth separately at 37°C for 24 h. After incubation, CFU of probiotics were calculated. All cultures were centrifuged together at 6000×g for 6 min; the pellets were washed with phosphate-buffered saline (PBS) by centrifuging at 8000×g for 12 min. Then the supernatant was removed and the pelleted cells were diluted to a concentration of 1× 109 CFU/ml in PBS solution for administration by oral gavage (1 × 109 CFU/chick based on original CFU enumerated on MRS agar plate) using a tuberculin syringe with a stainless-steel animal feeding needle.
37
To prepare the probiotic culture mixed feed, modification of the method employed by Olnood et al.
38
and Jahromi et al.
39
was followed. Each culture was grown in MRS broth contained 5 g/L of yeast extract and 20 g/L of glucose, for overnight at 37°C. Pelleted cells were harvested by following the above described method of direct oral gavage supplementation. Then the pelleted cells containing probiotics were suspended in PBS and were immediately mixed for 10 min at a concentration of 2 × 109 CFU/g of mash basal feed based on actual CFU determined on MRS agar plate. The experimental basal diets (BDs) with isolated 10 probiotic strains were mixed daily after CFU counting to ensure viable probiotic cells in basal feed and supplemented as a one-phase mash feed to prevent inactivation of the probiotic isolates.
Chicks, dietary treatment and experimental design
Dietary treatment was administered to 40-day-old Ross 308 broiler strains (Gallus domesticus) during 21 days in an in vivo trial. The chicks were divided into total of five groups, a control group, and four experimental groups (Antibiotic1, Antibiotic2, Probiotic1, and Probiotic2) in a completely randomized design. Eight chicks in each dietary treatment group were divided in mixed sex. Sample size for the experimental groups were selected and calculated by following statistical guideline.
40
All animals were reared and transported under conditions of National Livestock Development Policy (NLDP), 2007 of Bangladesh.
41
Chicks were allotted in distinct floor pens with a wood shaving floor where the feed and water were provided as an ad labium consumption basis. During the 21 days of in vivo trial, different environmental influences (lightning, temperature, humidity, ventilation) were maintained on optimal levels and recordings were kept thrice daily after observation. The care and management of the chicks was done by following the Guide for the Care and Use of Laboratory Animals.
42
The BD was corn–soybean-based meal provided in a mash form prepared by following the formula of Shokryazdan et al.
43
BD can fulfill the nutrient demand of Ross 308 broiler chicks for starter periods (1–21 days) according to Nutrient Requirements of Poultry (NRC).
44
Experimental treatments for five groups were:
Control group: BD and drinking water (DW) provided without any additive. Antibiotic1 group: BD and DW were supplemented with antibiotic oxytetracycline at a dose of 500 mg/gallon in DW for the whole experimental period. Antibiotic2 group: BD and DW supplemented with antibiotic chlortetracycline at a dose of 500 mg/gallon were given continuously in DW. Probiotic1 group: BD, DW and probiotic mixed culture with an approximate concentration of 1× 109 CFU/chick were administrated daily by oral gavage. Probiotic2 group: BD supplemented with the mixed Lactobacillus culture at an approximate rate of 2× 109 CFU/g basal feed and DW were administrated daily.
The experimental trial on broiler chicks was approved by the “Khulna University Animal Care and Ethics Committee” at September 12, 2018 (agreement no: KUACEC-369418).
Growth performance
The variables of growth performance traits like BW, ADG, FI, and FCR evaluated were according to Costa et al.
45
BW: All chicks from different dietary treatment groups were individually weighed for the first time when they arrived at the trial site at Day 1. Further, chicks were weighted on a weekly basis for the whole experimental period (3 weeks). Thus, BW of chicks was calculated. ADG: ADG of the chicks in each group was calculated for every 7 days. The formula for calculating ADG is: subtracting initial BW from the final BW and dividing it by the total no. of days. FI: Feed consumption per group basis was logged daily by subtracting the quantity of feed spared from the amount provided for a specific day and FI was measured. FCR: FCR was determined per experimental unit as a ratio of daily FI (g) and ADG (g).
Carcass characteristics
To estimate the efficacy of probiotics on the carcass characteristics, chicks were weighed and euthanized individually by cervical dislocation after the termination of in vivo trial (Day 22). Then by removing the head, neck, and shanks from sacrificed chickens, the dressing yield was calculated. Breast meat, drumstick, wing, and abdominal fat from eviscerated chickens were excised, weighed, and recorded individually. Parameters of carcass yield were measured as a percentage of the weight of eviscerated carcass meat of chicks, regarding the live weight of particular chicks. 19
Serum lipid profile
At the end of in vivo trial, blood was collected from the wing vein of chicks by selecting randomly. Blood was collected in a clean vial containing heparin as an anticoagulant. All the collected blood samples were centrifuged at 1400 g for 10 min to separate serum. The serum was moved into vials, properly labeled, and stored for further studies at –20°C. Serum samples were evaluated for total cholesterol, high-density lipoprotein (HDL) level, and serum triglyceride (TG) level. Very low-density lipoprotein (VLDL) level was determined by using the Fried Wald formula: Serum VLDL level = (plasma triglycerides/5). The serum low-density lipoprotein (LDL) level was determined as the difference between total cholesterol and the sum of serum VLDL and HDL level; LDL (mg/dl) = total cholesterol – (HDL + VLDL).46,47 All the results (mean ± SEM) were expressed in mg/dl.
Statistical analysis
Data were compared between the groups by one-way analysis of variance (ANOVA) procedure. ANOVA was performed based on completely randomized design (CRD) followed by multiple comparisons among the means which was performed using Tukey’s honestly significant difference (HSD) range. All results were presented as means ± SEM. Software R (version 3.5.1) was used to perform F-test in one-way ANOVA by using STAT package. One-way ANOVA is performed when collected data includes one categorical independent variable and one quantitative dependent variable. The independent variable should have at least three different groups or categories. 40 Based on this, calculation and justification of the sample size were selected in this study. Dietary treatment effects on broiler performance were considered as significant at p < .05.
Results
Isolation of probiotic bacteria
In the present study, a total of 10 typical bacteria isolated from broiler chickens on MRS agar plates were initially selected on the basis of morphological and cultural properties. Ten isolated bacteria showed the specifications rod shapes, nonmotile, gram positive, and coagulase positive along with catalase and endospore negative (Table 1).
Physiological properties of 10 isolated probiotic bacteria from GIT of broiler chickens.

Note: ++ = excellent tolerance; + = moderate tolerance;
Biochemical characterization
The outcome of the biochemical characterization of isolated bacteria is given in Table 2.
Carbohydrate utilization test: In carbohydrate utilization test, 10 isolates were able to utilize glucose, maltose, lactose, sucrose, fructose, galactose, raffinose, and mannose. NaCl tolerance test: Lactobacillus isolates were able to tolerate 4%–6% NaCl concentration but could not survive in presence of 8% of NaCl (Figure 1). Bile tolerance test: In bile tolerance test, all isolated bacteria tolerated at 0.3% and 0.5% bile salts (Figure 2). pH tolerance test: Isolates also exhibited a higher growth pattern at low pH (pH = 2.4) (Figure 3). Phenol tolerance test: In addition, all isolates were able to withstand 0.3% and 0.4% phenol concentration (Figure 4). High temperature tolerance test: Isolates demonstrated excellent and moderate growth at 45°C and 50°C, respectively but poor growth at 55°C at OD620 nm (Figure 5).

NaCl tolerance test. All bacterial isolates were able to tolerate 4%–6% NaCl concentration for total experimental period (A–C) but could not survive in the presence of 8% of NaCl (D). Here, results are demonstrated as average of the three readings.

Bile salt tolerance test. All bacterial isolates showed excellent growth in the presence of 0.3% and 0.5% bile salts (A–B). Here, results are demonstrated as average of the three readings.

pH tolerance test. All bacterial isolates exhibited a higher growth pattern at low pH (pH = 2.4) compared to control (pH = 7.4) (A–B). Here, results are demonstrated as average of the three readings.

Phenol tolerance test. All bacterial isolates were able to withstand 0.3% and 0.4% phenol (A–B). Here, results are demonstrated as average of the three readings.

High temperature tolerance test. All bacterial isolates demonstrated excellent and moderate growth at 45°C and 50°C, respectively (A–B). However, isolates showed poor growth at 55°C (C). Here, results are demonstrated as average of the three readings.
Biochemical properties of 10 isolated probiotic bacteria from GIT of broiler chicken.

Note: ++ = excellent tolerance; + =moderate tolerance;
Based on the positive results of all the tests carried out viz. morphological, physiological, and biochemical tests, 10 probiotic isolates with the best characteristics were presumptively identified as Lactobacillus sp.
Antibiotic susceptibility
Antibiotic susceptibility of 10 presumptively identified Lactobacillus isolates is summarized in Table 3. All 10 Lactobacillus isolates expressed susceptibility to erythromycin, chloramphenicol, penicillin G, amoxicillin, and nalidixic acid. Antibiotic resistance was detected for some isolates against gentamycin and kanamycin.
Antibiotic susceptibility test of 10 presumptively identified Lactobacillus sp. strains.

Note: Values are means ± SEM. R = resistant; I = intermediate; S = susceptible.
Antimicrobial activity
Table 4 demonstrates the antimicrobial property of probiotics as a zone of inhibition against five pathogens (Vibrio cholera, Escherichia coli, Salmonella typhi, Staphylococcus aureus, and Listeria monocytogenes). All 10 Lactobacillus isolates showed varying zones of inhibition from 0.00 to 27.38 mm depending upon the particular pathogen. Staphylococcus aureus was most sensitive with a 27.38 mm diameter of zone of inhibition. While Salmonella typhi had the least sensitivity with a zone of 0.00–19.67 mm.
Antimicrobial activity of 10 presumptively identified strains of Lactobacillus sp. against five bacterial pathogens.

Note: Values are means ± SEM.
Productive performances
All the results above mentioned in vitro tests specify the worth and potential of 10 isolated Lactobacillus isolates as a feed additive for supplementing in the chicken diet. The effect of Lactobacillus mixture supplementation on growth performance parameters of neonatal broilers is summarized in Table 5.
The effects of isolated Lactobacillus sp. supplementation on growth performances of broiler chickens.
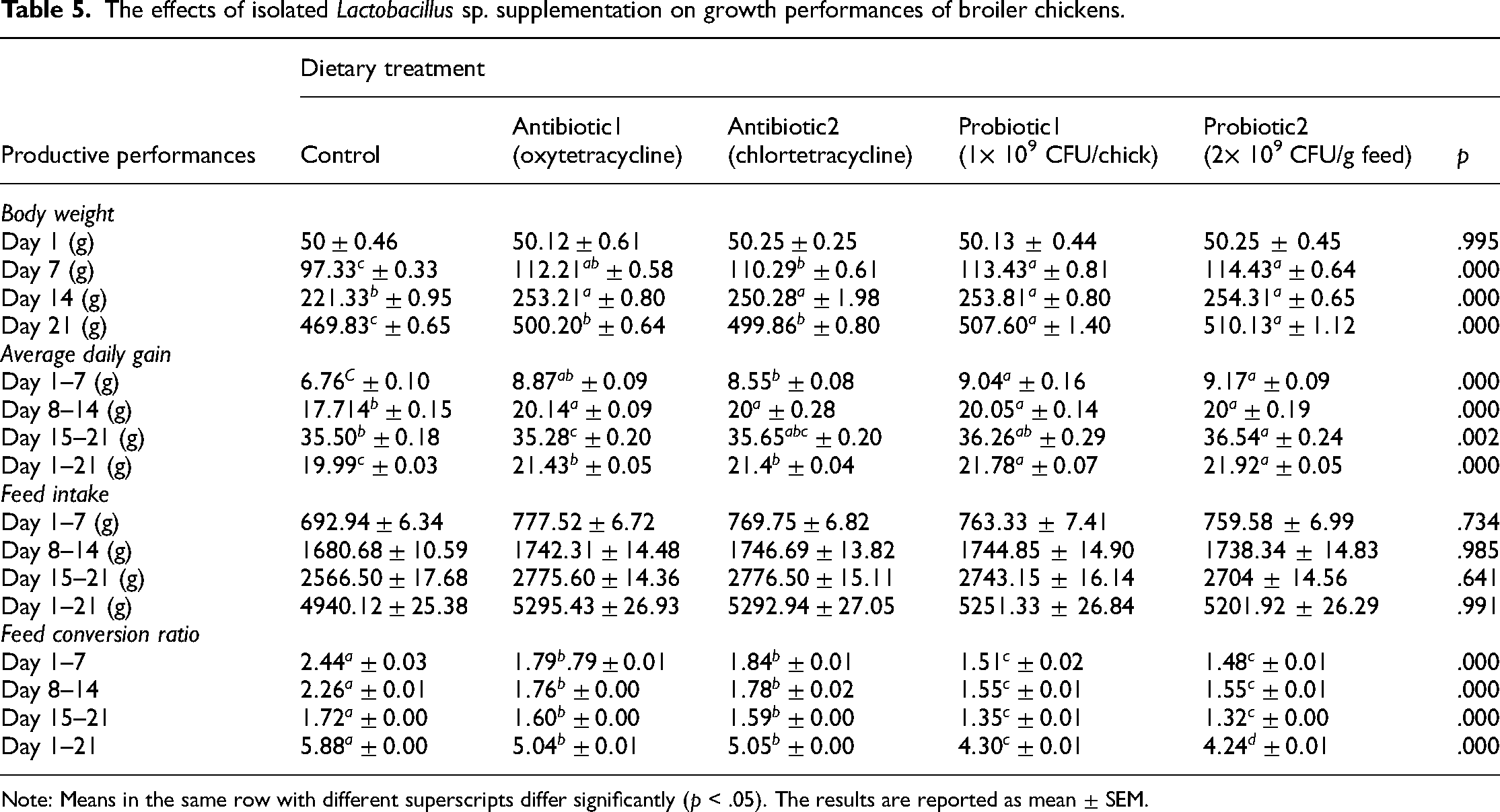
Note: Means in the same row with different superscripts differ significantly (p < .05). The results are reported as mean ± SEM.
BW
BW of broiler chicks was increased significantly (p < .05) in all experimental groups compared to control group on both Day 7 and 14 of in vivo trial. At the end of trial (Day 21), probiotic supplemented chicks had higher (p < .05) BW compared to control group and antibiotic groups. No significant difference was observed in regards to BW within two probiotic-fed groups at any period of the trial.
ADG
During Days 1–7 and 8–14, ADG of broiler chicks of all the experimental groups was significantly (p < .05) higher in comparison to the control group. During the whole period of the trial (Day 1–21), both probiotic groups had higher (p < .05) ADG in comparison to control and antibiotic groups. Probiotic fed groups showed no significant difference of ADG for overall experimental period (Day 1–21).
FI
FI of broiler chicks was observed for weekly basis (Day 1–7, 8–14, and 15–21) and for the overall period (Day 1–21) of the trial. However, no significant difference was found among all the experimental groups. Therefore, it can be concluded that Lactobacillus mixture feeding to neonatal broiler chicks couldn't make any difference on FI compared to either control or antibiotic groups.
Feed conversion ratio (FCR)
Between Days 1–7, 8–14, 15–21, and 1–21 of in vivo trial, Lactobacillus mixture supplemented groups had lower (p < .05) FCR compared to control and antibiotic groups. In addition, no significant difference was observed in FCR within probiotic fed groups for three consecutive weeks.
Carcass characteristics
The impact of probiotic mixture feeding during 21 days on carcass characteristics of broiler chicks is shown in Table 6.
Dressing yield percentage: Broiler chicks of all groups presented significantly (p < .05) higher dressing yield percentage compared with the control group. Breast yield percentage: There was a significant increase (p < .05) in the breast yield percentage of all groups when compared to control group along with the difference (p < .05) between antibiotic and probiotic groups was found. Drumstick yield percentages: Drumstick yield percentages of broilers of all groups were also found to be significantly improved (p < .05) in comparison to control group. Abdominal fat yield percentage: Abdominal fat yield percentages of broiler chicks decreased significantly (p < .05) in all the groups compared to control group.
The effects of Lactobacillus sp. supplementation on carcass characteristics of broiler chickens.

Note: Means in the same row with different superscripts differ significantly (p < .05). The results are reported as mean ± SEM.
However, no significant difference was observed within probiotic-fed groups in carcass characteristics of broiler chicks.
Serum lipid profile
The impact of dietary supplementations on serum total cholesterol, HDL, LDL, VLDL, and triglyceride levels in broiler chicks at the end of the trial is shown in Table 7.
Total cholesterol level: The serum total cholesterol level did not decrease in all the dietary treatment groups compared to control group. HDL level: Serum HDL level was significantly (p < .05) increased in probiotic groups compared to control and antibiotic groups. LDL level: There was no significant difference was found in serum LDL level among the treatment groups. VLDL level: A significant reduction (p < .05) of serum VLDL level were observed in probiotic groups compared to antibiotic groups. Triglycerides level: A significant reduction (p < .05) of serum triglycerides level were recorded in probiotic groups compared to antibiotic groups.
The effects of Lactobacillus sp. supplementation on serum lipid profile of broiler chickens.

Note: Means in the same row with different superscripts differ significantly (p < .05). The results are reported as mean ± SEM. HDL = high-density lipoprotein; LDL = low-density lipoprotein; VLDL = very low-density lipoprotein.
Discussion
As the GIT of broiler chicken is an abundant source for LAB, several Lactobacillus sp. screenings has been performed from different intestinal parts such as crop, large intestine, small intestine, and caecum. 48 In this current study, probiotic was isolated from caeca, crop, and small intestine. Advantageous impacts of probiotics are only achievable if they can survive in the harsh conditions of the GI tract like the high concentration of lysozyme, low pH, and bile salts. In the present study, all Lactobacillus isolates inhibited low concentration of NaCl (2% and 4%) and showed resistance to bile salt (0.3% and 0.5%) and phenol (0.3% and 0.4%) which is in accordance with related findings.49,50 Generally, the growth pattern and fermentation of Lactobacillus create an acidic condition that gives them the capacity to be resistant to acidic conditions. In the present research, all Lactobacillus isolates endured low pH (2.4) exposure for 24 h. These findings suggest a strong discriminative pH for the determination of low pH tolerant strains. 17 Moreover, Lactobacillus isolates showed survivability at high temperature (45°C and 50°C) which is important to survive in in vivo environment of broiler gut and also for in vitro conditions like growth and transport.
In the present study, all Lactobacillus isolates showed susceptibility against the tested antibiotics but drug resistance was detected for some isolates against both gentamycin and kanamycin. In a study of probiotic characterization, where antimicrobial activity of probiotic was measured and antibiotic resistant was also detected. 51
It was previously reported by several authors that, probiotic supplementation has a positive effect on growth performance, 11 carcass trait 52 and serum lipid profile 53 of broiler chickens. In the current study, mixed Lactobacillus supplementation has significantly (p < 0.05) improved BW of broiler chicks. In line with the present research, different strains of Lactobacillus such as L. subtilis, 54 L. salivarius, 18 L. acidophilus and L. casei, 52 and L. reuteri 55 supplementation have reported to increased BW of broilers significantly (p < .05) compared to the control group. In the current research, probiotic feeding has significantly (p < .05) improved BW compared to antibiotic group. A significant difference (p < .05) in BW was observed between probiotic and antibiotic fed groups of broilers22,56 which is in accordance with the findings of present research.
In the present research, Lactobacillus mixture feeding also significantly (p < .05) increased ADG of broiler chicks compared to control or antibiotic groups. Some recent researches reported similar outcome which has corresponds with the present study.39,52 On the other hand, FI of chicks were increased in probiotic fed groups. FI of broiler chicks was increased compared to the control group may be due to the reason that broilers are genetically known for their urge of high FI. 39 In the present study, Lactobacillus mixture was also able to significantly (p < .05) decrease FCR in all groups compared to the control group for the overall period of trial (Days 1–21) and also in every 3 weeks. In line with this result, Lactobacillus mixture supplementation significantly (p < .05) decreased FCR in broiler chicks compared to the control group 55 or antibiotic groups. 8
Improvements in growth performance and carcass characteristics with probiotic supplementation can be caused by enhancement in ingestion, ability to digest the feed, enrichment of beneficial intestinal microflora, and renovation of intestinal health. In the present research, carcass characteristics regarding all selected parameters were found to significantly increase (p < .05) compared to control group after feeding Lactobacillus mixture to broiler chicks. A recent study has reported to significantly improve carcass characteristics in broiler fed with Lactobacillus which is in accordance with the current study. 19 Abdominal fat yield percentages of broiler chicks were significantly decreased (p < .05) in all the groups compared to the control group in the current study. In accordance with the current result, dietary Lactobacillus acidophilus was reported to lower carcass fat content in broiler chicken. 57 Overall serum lipid profile of broiler chicks was accounted to be improved due to Lactobacillus feeding by several authors43,58 which is in line with the findings of present study.
Consistent with the above findings, analyzed probiotic mixture supplemented in present research has the capability to replace antibiotics as supplemented Lactobacillus mixture either improved the productive performance of chicks similarly to the effectiveness of antibiotics or significantly (p < .05) better than them which is in accordance with the findings of other authors.18,22,59
It is very hard to directly compare different researches comprising various probiotic species and administration levels. Because, the variations in the efficiency of probiotic supplementation will furthermore depend on numerous other factors like diversities in the strains, sources, viability, and concentrations of used probiotics, processes of administration, and conditions of chickens.43,48,59 Another determining factor for probiotic efficacy could be the broiler age. At initial age, colonization is significant to host due to residing in the gut permanently as the major microflora, and accordingly, influencing the further colonization of other microbes during grower and finisher age. 60
The limitations of the study includes isolated 10 probiotics were presumptively identified as Lactobacillus sp., species-level identification of isolated probiotic bacteria through molecular characterization is still required. In addition to that, in vivo trial consisting of higher number of broilers are still requisite to confirm the beneficial role of Lactobacillus analyzed in the present research work.
Conclusion
The mixture of Lactobacillus had favorable effects on productive performance of broilers regarding BW, ADG, and FCR (p < .05) compared with chickens that had no additive or had antibiotic. Additionally, noteworthy efficacy on carcass characteristics and serum lipid profile were found (p < .05) in Lactobacillus mixture fed chicken groups. Hence, probiotics to the diet of broiler chicks significantly improved productive performance, carcass characteristics, and serum lipid profile compared to the control group or antibiotic groups suggests that they can be used as a substitute for antibiotics. Accordingly, this research highlights the potential of probiotic through characterization, and inclusion in broiler diet by evaluating productive performances.
Supplemental Material
sj-pdf-1-sci-10.1177_00368504241276259 - Supplemental material for Effects of probiotics on productive performances and serum lipid profile of broiler as substitute of antibiotics
Supplemental material, sj-pdf-1-sci-10.1177_00368504241276259 for Effects of probiotics on productive performances and serum lipid profile of broiler as substitute of antibiotics by Umama Khan, SM Mahbubur Rahman, Salauddin Khan, Swarna Roy and Khondoker Moazzem Hossain in Science Progress
Footnotes
Author agreement statement
All authors have seen and approved the final version of the manuscript being submitted. The article is the authors’ original work, hasn’t received prior publication and isn't under consideration for publication elsewhere.
Authors’ contributions
Khondoker Moazzem Hossain served as lead for conceptualization, experiment design, methodology, resources, supervision, project administration, and manuscript review; Umama Khan served as lead for original manuscript writing, conceptualization, experiment design, methodology, data collection, data curation, analysis, visualization, and manuscript review; S.M. Mahbubur Rahman served as lead for resources, supervision, and manuscript review; Md. Salauddin Khan served as lead for data curation, analysis tools, software, and manuscript review; Swarna Roy served as lead for data collection and manuscript review.
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy/ethical restrictions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
The experimental trial was approved by “Khulna University Animal Care and Ethics Committee” at September 12, 2018 (agreement no: KUACEC-369418). The care and management was also done following Guide for the Care and Use of Laboratory Animals, 8th edition, 2011.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
