Abstract
The corrosion of metals, particularly rust on iron and its alloys, poses significant challenges across industries, with notable economic and environmental consequences. Traditional rust prevention methods, reliant on chemical inhibitors and coatings, often raise concerns regarding their environmental and health impacts. In response, advancements in corrosion science have emphasized the potential of o-phenylenediamine (OPD) derivatives and vaseline-based mixtures as innovative, eco-friendly solutions. This article reviews the efficacy of OPD derivatives in mitigating anodic and cathodic reactions, thereby preventing rust formation by creating robust protective layers on metal surfaces. These derivatives act as physical barriers, impeding corrosive agents and offering versatile applications, including integration into protective coatings for new and existing metal structures. The synergistic combination of OPD with other inhibitors enhances protection, supporting sustainable practices by reducing resource consumption and environmental impact. Vaseline-based layers, while providing satisfactory initial hydrophobicity and ease of application, face challenges such as achieving uniform coverage on polar metal surfaces and preventing pore formation. Integrating Fe(III)(BTA)3 into vaseline coatings represents a paradigm shift in sustainable corrosion prevention, with a 30% concentration identified as optimal for maximum protection. This development marks a pivotal advancement in materials science, offering robust corrosion protection aligned with sustainability principles. The use of OPD derivatives introduces a novel, environmentally benign approach, leveraging their ability to adsorb onto metal surfaces and create protective layers against moisture and oxygen. This innovation aligns with global initiatives to reduce industrial environmental footprints, promoting sustainable, effective corrosion prevention strategies.
Keywords
Introduction
The global impact of corrosion on infrastructure and industrial operations necessitates continuous innovation in rust prevention strategies. Corrosion not only undermines the structural integrity of metal components but also leads to substantial economic losses through repair and replacement costs. Moreover, the environmental implications of traditional rust inhibitors, often containing hazardous chemicals, underscore the urgency for sustainable alternatives. Amidst this backdrop, o-phenylenediamine (OPD) and its derivatives have garnered attention for their potential role in mitigating corrosion-related challenges. OPD, a versatile compound known for its antioxidative properties and metal-chelating abilities, offers a promising avenue towards eco-friendly rust prevention solutions. Unlike conventional inhibitors, which may introduce toxic residues into ecosystems and pose risks to human health, OPD derivatives present a safer alternative without compromising effectiveness. The efficacy of OPD in inhibiting rust formation lies in its ability to form stable complexes with metal ions on the surface of metals. This chelation process effectively creates a protective barrier that shields the metal from corrosive elements such as oxygen and moisture. 1 Moreover, OPD derivatives demonstrate a high affinity for metal surfaces, ensuring durable adhesion and long-term corrosion resistance. 2 These characteristics make OPD particularly suitable for applications across various industries, including automotive, maritime and infrastructure development. Recent studies have highlighted the superior performance of OPD-based inhibitors compared to traditional methods. For instance, research indicates that OPD derivatives not only delay the onset of rust but also significantly reduce the rate of corrosion propagation over time. 3 This dual functionality - preventing both the initiation and progression of rust - positions OPD as a versatile solution capable of extending the lifespan of metal assets whilst minimising maintenance costs. Practical applications of OPD-based coatings and treatments further validate their efficacy in real-world settings. Case studies across different environments demonstrate the adaptability and reliability of OPD derivatives in diverse climatic conditions and operational stresses. 4 The ability to integrate OPD into existing corrosion protection protocols underscores its seamless compatibility with industrial practices, facilitating smooth adoption and implementation. Looking forward, future research endeavours aim to optimise OPD formulations and application methodologies to enhance their performance and cost-effectiveness. Areas of focus include refining surface preparation techniques, exploring synergistic interactions with other corrosion inhibitors, and advancing nanotechnology-based approaches for targeted delivery of OPD derivatives. 5 Such advancements not only aim to bolster the efficacy of OPD-based inhibitors but also contribute to reducing their environmental footprint through sustainable manufacturing and application practices. The ongoing development and adoption of OPD derivatives represent a transformative step towards greener prevention strategies. By leveraging the inherent properties of OPD to inhibit corrosion effectively and sustainably, industries can mitigate operational risks, prolong asset lifespans and uphold environmental stewardship. Continued research and innovation will play a pivotal role in advancing OPD-based solutions and ushering in a new era of sustainable corrosion management practices globally. The rusting of tools and machine parts during storage and transportation causes particularly severe damage to our national economy, especially in the case of export shipments. Rust can degrade the functionality and appearance of metal components, resulting in significant financial losses. 6 New derivatives have developed a protective substance that, by forming a coating, shields metals from rusting. 7 It is well known that instruments, machine tools and general machine parts made of different metals often rust significantly during storage and transportation, thus reducing their value. 8 This rusting can become noticeable within days or weeks, representing a substantial loss, particularly for export shipments. Export shipments are often exposed to tropical temperatures and maritime air for extended periods, which exacerbates rusting. 9 Rusting during manufacturing is also problematic, as it necessitates additional machining that alters the dimensions of products and reduces their accuracy. 10 Rusting is generally caused by the moisture and oxygen content of the air, as well as gases from combustion products. 11 The moisture (adsorbed water vapour) deposited from the air onto the metal surface is corrosive, and this effect is exacerbated by dissolved gases. 12 Metals adsorb some of the active gases in the air, and when these gases settle on the metal surface, the water present dissolves them, creating highly concentrated solutions at specific points. 13 This leads to the formation of local corrosion elements and causes deep pitting corrosion. 14 The salt content of seawater, dispersed in the form of fine mist in maritime air, poses a particular danger during maritime transport due to the increased attacking effect of chloride ions. 15 Various methods used so far have proven inadequate for protecting against these agents, making corrosion during storage and transportation an unresolved problem. 16 Many metal objects, such as tools and machine lubrication surfaces, cannot be permanently protected by coatings of a constant nature due to their nature. 17 Temporary protection is provided by applying different durable materials. 18 Numerous such inhibitors have been isolated from natural sources. 19 However, these methods often fall short, requiring innovative solutions to address the challenge effectively. OPD derivatives 20 hold great promise as a versatile and eco-friendly solution for rust prevention. OPD's ability to inhibit corrosion, its compatibility with different materials and its environmental benefits make it an attractive option for industries seeking sustainable rust prevention strategies. OPD is an organic compound with the chemical formula C6H4(NH2)2, commonly used in the production of dyes, polymers and pharmaceuticals. Due to its chemical properties, OPD exhibits strong antioxidant and corrosion inhibition properties, making it suitable for rust prevention applications. The economic impact of rusting on export shipments is considerable. Rust can lead to rejected shipments, costly returns and damaged reputations. Protecting against rust not only preserves the integrity of the products but also ensures customer satisfaction and maintains international trade relationships. The development of new protective substances that form a durable coating on metal surfaces can mitigate these risks effectively. The formation of rust on metal surfaces is influenced by several factors, including humidity, temperature and the presence of corrosive agents. In tropical climates, where humidity levels are high, rust formation is accelerated. Similarly, exposure to maritime air, which contains high levels of salt, increases the rate of corrosion. Traditional protective measures, such as applying oil or grease, provide temporary protection but are not effective in the long term. Additionally, these substances can be messy and difficult to remove, complicating maintenance and repair processes. OPD derivatives offer a more sustainable and efficient solution for rust prevention. 21 These compounds form a protective layer on the metal surface, preventing moisture and oxygen from reaching the metal and initiating the rusting process. 22 Moreover, OPD derivatives are environmentally friendly, reducing the reliance on hazardous chemicals traditionally used in rust prevention. 23 This makes OPD an attractive option for industries seeking to adopt greener practices without compromising on performance. Research has shown that OPD derivatives can be effectively incorporated into coatings and paints, enhancing their protective properties. 24 These coatings provide a barrier against environmental factors that contribute to rust formation, such as moisture and corrosive gases. Furthermore, OPD-based coatings are durable 25 and resistant to abrasion, ensuring long-lasting protection for metal surfaces. 26 In addition to its protective properties, OPD is also cost-effective. The use of OPD derivatives in rust prevention can reduce maintenance costs and extend the lifespan of metal components. 23 This is particularly beneficial for industries with large-scale operations, where the cost of rust damage can be substantial. By investing in OPD-based solutions, companies can achieve significant savings in the long term. 27 One of the key advantages of OPD derivatives is their versatility. 28 Their effectiveness in different environments makes them suitable for a wide range of industries, including automotive, aerospace and marine. 29 Despite the promising potential of OPD derivatives, further research and development are essential to fully understand their capabilities and optimise their use in rust prevention. Studies are needed to explore the mechanisms by which OPD inhibits corrosion, as well as to identify the most effective formulations for different applications. Collaboration between industry and academia can drive innovation and accelerate the adoption of OPD-based solutions. The rusting of tools and machine parts during storage and transportation poses a significant challenge, especially for export shipments. Traditional methods of rust prevention are often inadequate and environmentally harmful. OPD derivatives offer a promising alternative, providing effective and eco-friendly protection against rust. Continued research and development in this field are essential to unlocking the full potential of OPD and advancing the transition towards greener corrosion control methods. By adopting OPD-based solutions, industries can enhance the durability and reliability of their products whilst minimising their environmental impact. This article significantly advances the field of corrosion science by focusing on OPD derivatives and Vaseline-based mixtures, offering a fresh perspective on eco-friendly corrosion inhibitors. Whilst traditional inhibitors like chromates are effective, they pose environmental and health risks. OPD derivatives, however, represent a shift towards greener alternatives, forming durable protective layers on metal surfaces to prevent corrosion. The article emphasises the versatility of OPD in various applications, including its integration into coatings and treatments that extend the life of metal assets whilst reducing environmental impact. Moreover, the exploration of Vaseline-based mixtures as rust inhibitors adds a practical dimension to the subject. Whilst not new, these mixtures are revisited in the context of sustainable corrosion protection. The article discusses their advantages, such as ease of removal and initial hydrophobic properties but also critically examines their limitations, particularly on polar metal surfaces where wetting is less effective. The susceptibility to pore formation after application is another challenge identified, which has implications for long-term durability. By addressing these practical challenges, the article provides valuable insights into how these materials can be optimised for broader industrial use. It bridges the gap between laboratory research and real-world applications, emphasising the need for continued innovation to refine these green corrosion inhibitors, making them more effective and widely applicable across various industries.
This research delves into the synthesis process of various OPD derivatives, exploring how different chemical modifications can enhance their properties. By characterising these derivatives through techniques such as spectroscopy and surface analysis, the study ensures a comprehensive understanding of their structural and chemical attributes, which are critical to their function as inhibitors. The study evaluates the efficacy of the synthesised OPD derivatives in real-world corrosion scenarios. It highlights how these compounds form protective layers on metal surfaces, effectively blocking corrosive agents like oxygen and moisture. The research also explores the ability of OPD derivatives to be incorporated into coatings and treatments, providing both immediate and long-term rust prevention. By comparing the performance of OPD derivatives to traditional rust inhibitors, the research demonstrates the advantages of these novel compounds, particularly in terms of environmental sustainability and application versatility. The study not only confirms their effectiveness but also positions OPD derivatives as viable alternatives to more harmful traditional inhibitors; provides the synthesis and characterisation of OPD derivatives but also thoroughly evaluating their practical application in corrosion prevention, making a compelling case for their use in industrial settings.
Materials and methods
All chemicals used were of analytical grade and purchased from Sigma Aldrich, Germany, and were used without further purification. Solutions were all prepared using doubly distilled water. Molecular models were calculated with AVOGADRO 1.2.0.
Benzotriazole 30 (BTA) was prepared in a high yield (82%) via diazotisation of OPD with sodium nitrite (NaNO2) in glacial acetic acid and water with minor modifications as described earlier as follows: 1.08 g (0.01 mol) OPD was dissolved in water, then glacial acetic acid was added 1.20 g (1.2 mL), cool down to 3 °C, then 0.7 g (0.01 mol) sodium nitrite (NaNO2) was added to the stirred reaction. The mixture was allowed to warm up to room temperature for 2 hours, cooled, filtered out the crystals, washed with ice water, dried to obtain the crude product and then recrystallized to obtain BTA with a melting point of 96–97 °C, with a yield of about 81% as shown in Figure 1.

Preparation of BTA.
Benzotriazole salts 31 were prepared from the BTA and equimolar metal nitrates in ethanol/water as shown in Figures 2 and 3.

Preparation of bivalent salts (Fe(II)BTA, Cu(II)BTA).

Preparation of three-valent salts (Fe(III)BTA, Al(III)BTA).
Briefly, 1.19 g (0.01 mol) BTA was dissolved in a 5 w% ethanol:water mixture, and then the appropriate metal-nitrate salts were added slowly. The mixture was stirred at room temperature for 2 hours, then the solvent was evaporated to obtain the crude product. Crude product was recrystallized, to obtain pure benzotriazole salts. The yields, melting points and ultraviolet (UV) maxima are indicated in Table 1.
Yields and chemical properties of BTA salts.

UV: ultraviolet; BTA: benzotriazole.
The cupric benzotriazole (Cu(II)BTA) complex was formed from the cupric salt copper(II) nitrate trihydrate (Cu(NO3)2·3H2O), resulting in a Cu(II)BTA complex formed with having a composition corresponding stoichiometrically with one metal atom to two molecules of BTA: a cupric derivative with the basic formula (C6H4N3)2Cu. X-ray diffraction (XRD) gives no pattern as shown in Figure 4; thus, the compound is either amorphous or microcrystalline, but suggested that bonding was achieved by the formation of a coordination complex between the cupric ion and the BTA molecule. BTA itself might not produce distinct peaks in XRD due to its non-crystalline or amorphous nature. The relevant peaks of the starting material Cu(NO3)2·3H2O (2θ = 10.8°, 14.6°, 18.3°, 22.0°, 25.1°) are missing from the spectra. The Cu(II) ion is probably bonded with a link formed by the replacement of the proton from the NH group and a coordinate bond with a lone pair of electrons from one of the nitrogen atoms. The proposed structure of Cu(II)BTA is shown in Figure 5. The XRD pattern of the sample Cu(II)BTA, which is light greenish-white in colour, is novel. Even with the assistance of the PDF5+ (Release 2024) international powder XRD database, neither starting materials nor other presumed crystalline products could be identified.

XRD spectra of Cu(II)BTA.

Proposed structures of Cu(II)BTA and Fe(II)BTA.
The ferrous benzotriazole (Fe(II)BTA) complex was formed starting from iron(II) nitrate dihydrate (Fe(NO3)2·2H2O). The Fe(II)BTA complex is formed having a composition corresponding stoichiometrically with one metal atom to two molecules of BTA: a ferrous derivative with the basic formula (C6H4N3)2Fe. XRD gives no specific pattern as shown in Figure 6. The relevant peaks of the starting material Fe(NO3)2 (2θ = 7.5°, 12.4°, 16.7°, 20.2°, 23.3°) are missing from the spectra. Suggested that the Fe(II) ion is bonded with a link formed by the replacement of the proton from the NH group and a coordinate bond with a lone pair of electrons from one of the nitrogen atoms, with the proposed structure is shown in Figure 5.

XRD spectra of Fe(II)BTA.
Ferric benzotriazole (Fe(III)BTA) was formed from iron(III) nitrate nonahydrate (Fe(NO3)3·9H2O), having a composition corresponding Fe(III) stoichiometrically with one metal atom to three molecules of BTA: a ferric derivative with the basic formula (C6H4N3)3Fe. The reddish-brown powder pattern of a single crystalline component is iron(III)-benzotriazole complex salt (2θ = 14.6°, 17.2°, 19.8°, 22.5°, 28.2°, 29.0°) as shown in Figure 7. The proposed structure is shown in Figure 7. The relevant peaks of the starting material Fe(NO3)3·9H2O (2θ = 10.8°, 15.2°, 19.1°, 21.8°, 24.6°) are missing from the spectra.
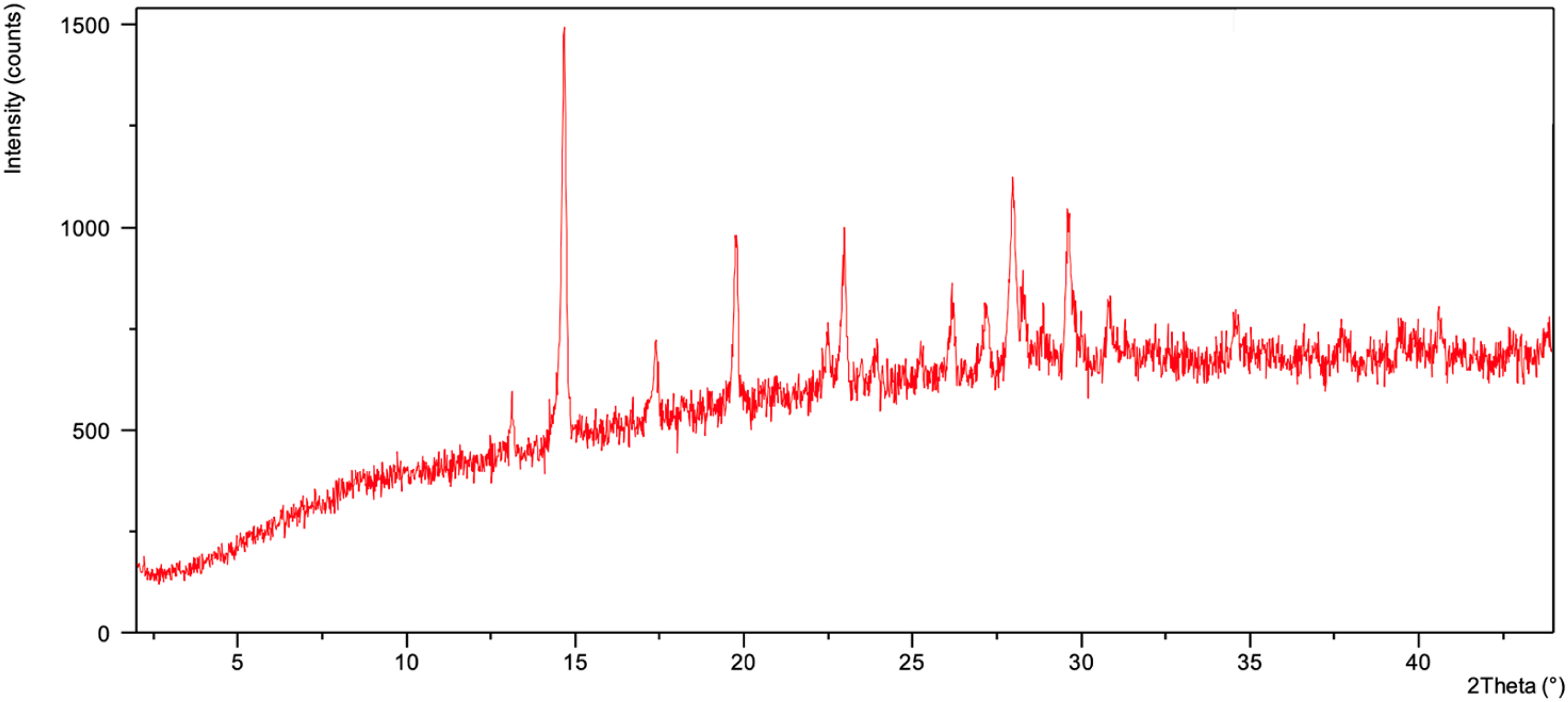
XRD spectra of Fe(III)BTA.
Aluminium benzotriazole (Al(III)BTA) was formed from aluminium(III) nitrate nonahydrate (Al(NO3)3·9H2O), having a composition corresponding Al(III) stoichiometrically with one metal atom to three molecules of BTA: an alumina derivative with the basic formula (C6H4N3)3Al. The proposed structure is shown in Figure 8. XRD gives no pattern as shown in Figure 9. The XRD pattern of the Al(III)BTA, which is strongly rose-pink in colour, is novel. The main peaks of starting material Al(NO3)3·9H2O (2θ = 8.8°, 11.6°, 14.1°, 16.8°, 19.7°) are missing from the spectra. Even with the assistance of the PDF5+ (Release 2024) international powder XRD database, neither starting materials nor other presumed crystalline products could be identified.

Proposed structures of Fe(III)BTA and Al(III)BTA.

XRD spectra of Al(III)BTA.
Benzimidazole 32 derivatives (1–5) have also been obtained by condensation of various benzaldehyde compounds with OPD as shown in Figure 10. Briefly, benzimidazole (1) was prepared as follows 33 : in a 100 mL round-bottomed flask 10.8 g (0.1 mole) of OPD was treated with 4.5 mL (7.00 g) of 90 w% formic acid (0.1 mole). The mixture is heated in a water bath at 100° for three hours. After cooling, 5 w% sodium hydroxide (NaOH) solution was added. The crude benzimidazole was filtrated, then washed with 50 mL of cold water, and was recrystallized from boiling water. The melting point was 170–171 °C, and the yield was 10.5 g, 82% of the theoretical. Briefly, benzimidazole derivatives (2–5) were prepared as follows: in a 100 mL round-bottomed flask 10.8 g (0.1 mole) of OPD was treated with (0.1 mole) of appropriate substituted benzaldehyde in 50 mL of 1:1 water:glycerol. The mixture is heated in a water bath at 100 °C until precipitates are observed. Solvent and solids were separated, crystals were washed with 25 mL of cold water and were recrystallized from ethanol to water. The yields, melting points and UV maxima are indicated in Table 2.

Preparation of benzimidazole derivatives (1–5).
Yields and chemical properties of benzimidazole salts.

Results
To further elaborate on the theoretical reasoning and experimental findings regarding the effectiveness of Me(II)(BTA)2 and Me(III)(BTA)3 compounds in corrosion inhibition, it is essential to delve into the specific requirements for an effective rust inhibitor, the mechanisms of inhibition and the practical applications and results obtained from experimental studies. The fundamental premise guiding the search for effective corrosion inhibitors revolves around the compound's ability to adhere strongly to polar metal surfaces whilst simultaneously repelling water molecules. This dual characteristic ensures that the inhibitor forms a robust protective layer on the metal, impeding the penetration of corrosive agents. Ideal inhibitors often feature polar groups that can interact strongly with metal atoms or ions through adsorption mechanisms. Additionally, the presence of hydrophobic, non-polar groups enhances the inhibitor's ability to repel water, thereby reinforcing its protective efficacy. Organic compounds containing long alkyl chains are particularly suitable candidates for corrosion inhibition due to their insolubility in water and their ability to exhibit dipole characteristics. These compounds can effectively adsorb onto metal surfaces, forming a stable protective film that acts as a barrier against corrosion-inducing factors. The compounds Me(II)(BTA)2 and Me(III)(BTA)3, where Me represents a metal ion such as iron (Fe), aluminium (Al) or copper (Cu). These compounds are known to form complex structures where BTA ligands coordinate with metal ions, resulting in the formation of polymeric films on metal surfaces. The mechanism of inhibition involves the formation of an inert and coherent polymer film, typically Me(II)(BTA)2 or Me(III)(BTA)3, which overlays and reinforces the existing oxide film naturally present on metal surfaces. The protective function of this polymer film is crucial as it enhances the durability and resistance of the metal against corrosive attacks. By closely adhering to the metal surface and conforming to its contours, the film effectively seals off potential corrosion sites and prevents the diffusion of moisture and oxygen, which are critical components in the corrosion process. Experimental studies were conducted to validate the corrosion-inhibiting properties of Me(II)(BTA)2 and Me(III)(BTA)3 compounds, particularly when blended with Vaseline. These experiments involved immersing polished metal sheets (steel, aluminium, copper) coated with various concentrations of Me(II)(BTA)2 and Me(III)(BTA)3 salts mixed with Vaseline into different corrosive environments, such as:
Atmospheres saturated with water vapour: This environment mimics high humidity conditions where moisture plays a significant role in accelerating corrosion processes. Salt spray atmosphere: This setup simulates coastal or marine environments where metal structures are exposed to saline aerosols, which can lead to accelerated corrosion. Immersion in 3.5% NaCl solution: A standard corrosive environment that represents moderate to severe conditions encountered in industrial settings. In these experiments, the metal sheets were carefully prepared and coated with the Vaseline mixture, ensuring uniform coverage and adhesion. After immersion, the mass loss of the metal sheets was monitored at regular intervals to assess the effectiveness of corrosion inhibition over time.
The immersion experiments were implemented as follows: pre-measured (with analytical precision), polished 2 × 5 cm surfaces, 1 mm thick metal sheets (steel, aluminium, copper) were immersed in different Fe(III)(BTA)3 containing molten Vaseline mixtures, then removed from the mixtures and rapidly cooled. A film formed on the sheets. The sheets were immersed in a 3.5 w% NaCl solution, and the mass of the metal sheets was measured at regular intervals. Figure 11 illustrates a characteristic example (steel sheet, DC01-A 1.0330, EN 10131), where the percentage decrease in mass is plotted on the ordinate (%) and the duration (days) of the experiment is plotted on the abscissa. The curves presented below contain the evaluation data of these experiments conducted with 3.5 w% NaCl solution (Figure 11).
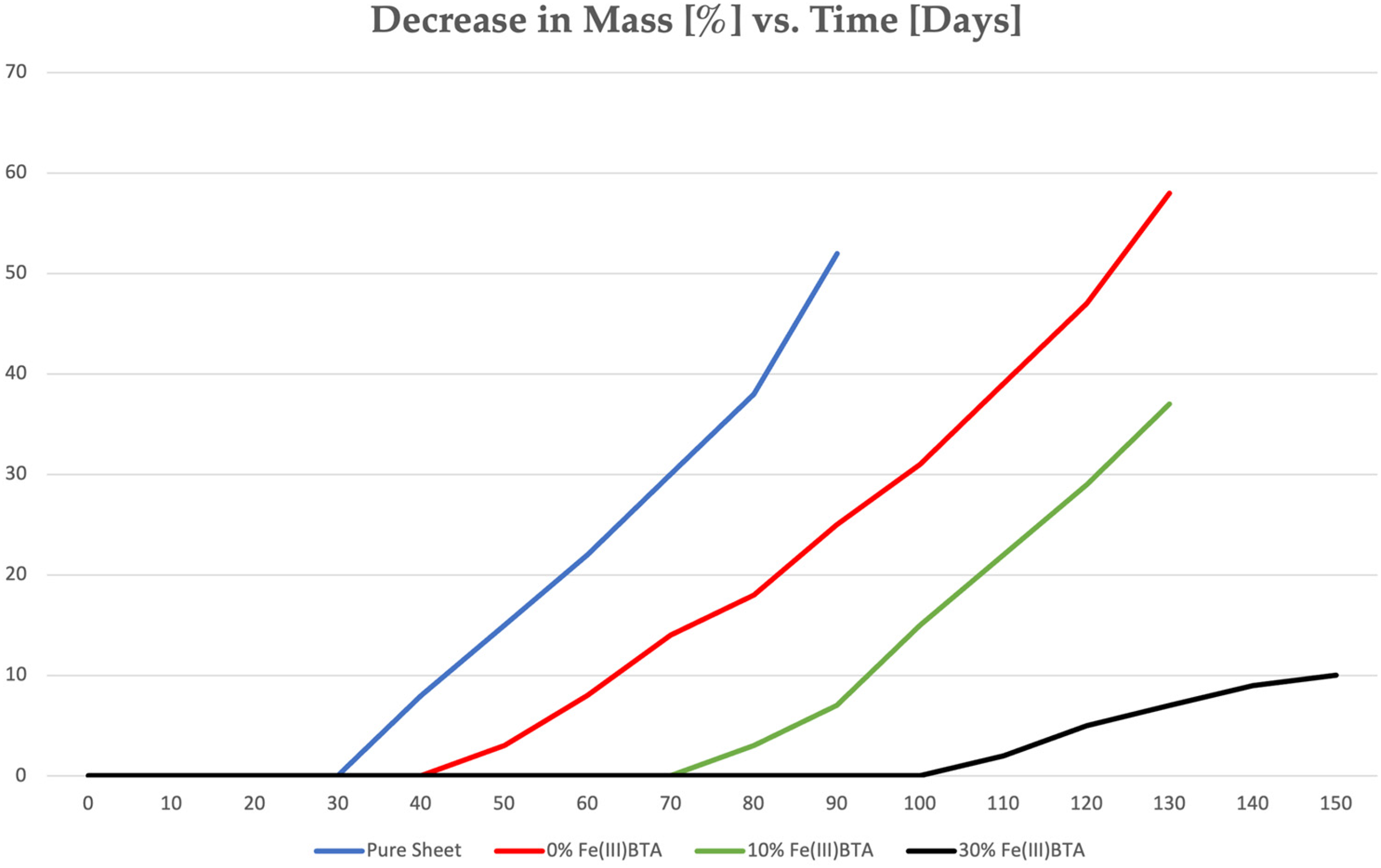
Decrease in mass in time of steel sheets.
Figure 11 illustrates a typical example from the experimental data, showing the percentage decrease in mass (%) on the vertical axis and the duration of the experiment (days) on the horizontal axis. The curves depicted in the figure represent the evaluation data obtained from experiments conducted using a 3.5 w% NaCl solution. These curves serve to visually demonstrate the protective effects of the Fe(III)(BTA)3 compounds when incorporated into Vaseline blends compared to untreated or conventionally protected metal surfaces.
Where, ‘Curve 0%’ represents the baseline corrosion rate observed without Fe(III)(BTA)3 inhibitor, ‘Curve 10%’ demonstrates the effect of a protective coating containing 10% Fe(III)(BTA)3 dissolved in Vaseline and ‘Curve 30%’ highlights the significant corrosion inhibition achieved with a coating containing 30% BTA in Vaseline, showing the most substantial reduction in mass loss over time. From practical experience and extrapolation of results, it is evident that the coating containing 30% Fe(III)(BTA)3 in Vaseline offers robust corrosion protection lasting several months even under harsh environmental conditions.
The Vaseline-Fe(III)BTA mixture can be easily removed from metal surfaces using hydrocarbon solvents or by heating. However, this removal is often unnecessary as the coating is thin and possesses lubricating properties. This dual functionality means it can remain on lubricated surfaces without interfering with their primary function. Moreover, the mixture is highly efficient in its application, requiring significantly less quantity per unit area compared to pure Vaseline. For instance, whilst 40 g/m² of pure Vaseline is needed, only 20 g/m² of the Vaseline-Fe(III)(BTA)3 mixture is sufficient to provide an adequate protective coating. Fe(III)(BTA)3 exhibits remarkable adhesion to metal surfaces due to its high content of active substances. This strong adhesion forms a dense and protective layer on the metal. It is crucial to note that the effectiveness of this adhesive layer is not determined by its thickness but by its adhesion strength. The robust adhesion of Fe(III)(BTA)3 ensures a long-lasting protective barrier against corrosive elements. The Vaseline-Fe(III)(BTA)3 mixture, which contains both apolar and polar groups, shows significantly higher adsorption affinity on metal surfaces compared to Vaseline. Vaseline mainly consist of polar components, whereas the mixture includes non-polar hydrocarbon chains as well. When applied, the polar groups in the mixture orient towards the metal surface, whilst the non-polar groups repel water, creating an effective barrier. This dual nature facilitates various application methods, including spraying, dipping and immersing, making the mixture versatile and easy to apply across different surfaces.
The experimental results consistently show that metal sheets treated with Fe(III)(BTA)3 in Vaseline exhibit significantly reduced rates of corrosion compared to untreated surfaces or those treated with Vaseline alone. This reduction in corrosion rate is indicative of the inhibitor's ability to form a durable protective barrier that effectively mitigates the corrosive effects of chloride ions 34 and atmospheric moisture. Furthermore, the synergistic effect observed between the metal ion (Fe3+) and BTA ligands underscores the importance of formulation design in optimising corrosion inhibition performance. Variations in concentration and composition of the inhibitor blend were studied to determine the optimal conditions for maximum corrosion protection across different metal types and environmental conditions. The theoretical framework supporting the selection and efficacy of Me(III)(BTA)3 compounds as corrosion inhibitors is firmly grounded in their dual functionality of strong metal adsorption and water repellence. Experimental validations using standardised corrosion testing methods have corroborated these theoretical insights, demonstrating significant improvements in corrosion resistance when these compounds are applied in practical settings. Future research directions should focus on refining the formulation of Me(II)(BTA)2 and Me(III)(BTA)3 inhibitors to enhance their performance under varying environmental conditions and across different metal substrates. Additionally, exploring novel application techniques and integrating these inhibitors into industrial coating processes could further optimise their effectiveness and broaden their applicability in diverse corrosion prevention strategies. By continuing to innovate and refine corrosion inhibition technologies such as Me(II)(BTA)2 and Me(III)(BTA)3 compounds, industries can advance towards more sustainable practices that mitigate the economic and environmental impacts of corrosion on infrastructure and metal assets globally.
In recent studies, significant advancements have been made in the development of corrosion inhibitors utilising Fe(III)(BTA)3 dissolved in Vaseline as the protective coating. This compound has demonstrated remarkable efficacy in mitigating corrosion on various metal surfaces, surpassing traditional protective coatings in terms of durability and effectiveness. This section delves into the theoretical underpinnings, experimental findings and practical implications of using Fe(III)(BTA)3 in Vaseline-based coatings for corrosion prevention. The effectiveness of Fe(III)(BTA)3 in corrosion inhibition lies in its ability to adhere strongly to metal surfaces whilst forming a dense and cohesive protective layer. Unlike conventional coatings that rely solely on physical barriers, Fe(III)(BTA)3 molecules contain both polar and non-polar groups. The polar groups facilitate strong adsorption onto the metal surface, enhancing the coating's adherence and stability. Meanwhile, the non-polar hydrocarbon chains contribute to a water-repellent effect, crucial for preventing moisture ingress and subsequent corrosion.
Experimental investigations have substantiated the theoretical framework by demonstrating the superior corrosion protection afforded by Vaseline-based coatings containing varying concentrations of Fe(III)(BTA)3. Figure 12 illustrates experimental results conducted on copper sheets (Cu-ETP (CW004A), EN 13601) exposed to a 3.5% NaCl solution over time. The curves plotted depict the percentage decrease in mass (%) against the duration (days) of exposure for coatings with different Fe(III)(BTA)3 concentrations: 0%, 10% and 30%, see Figure 12.
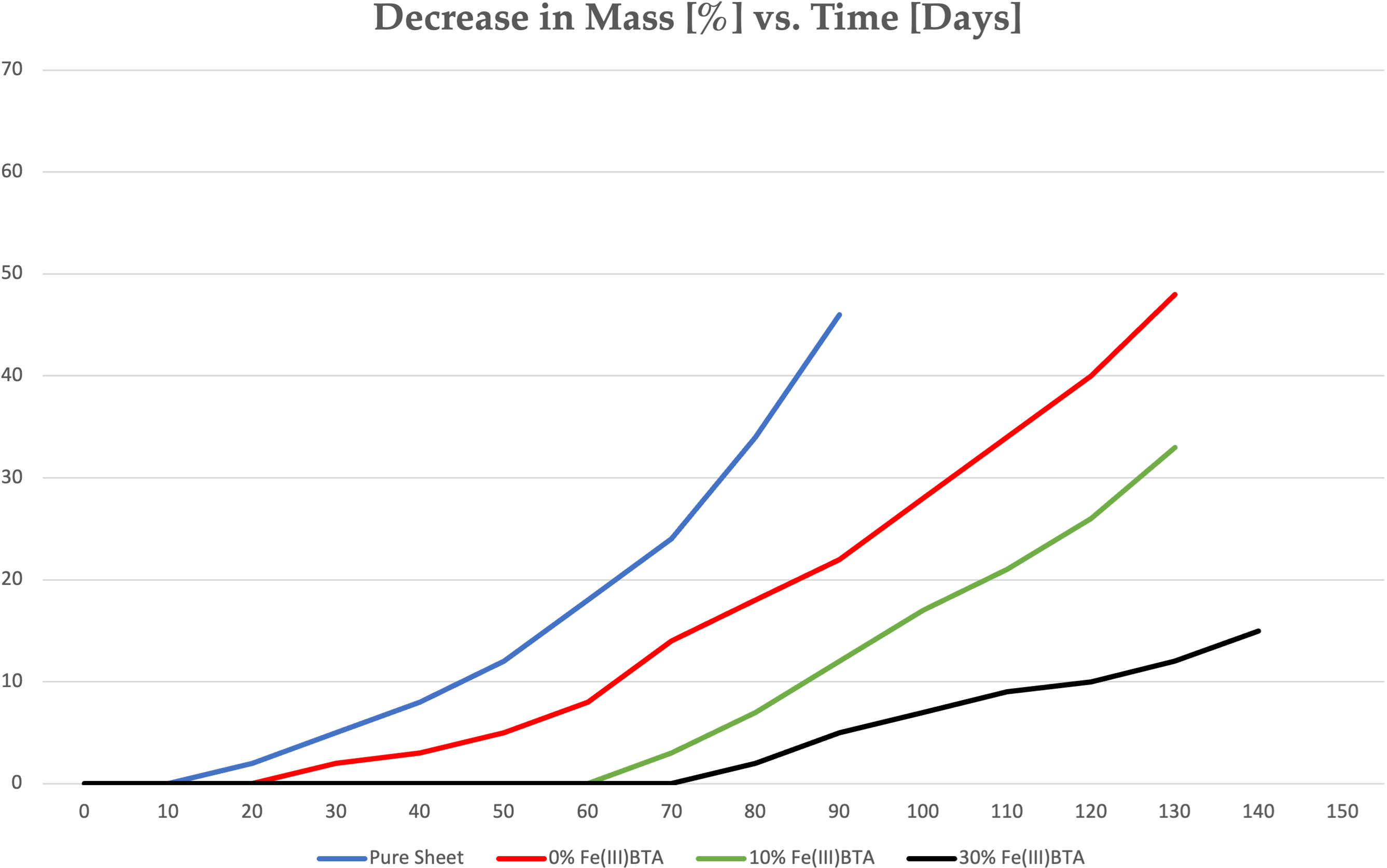
Decrease in mass in time of copper sheets.
Where, ‘Curve 0%’ represents the baseline corrosion rate observed with untreated Vaseline-coated copper sheets, ‘Curve 10%’ demonstrates the effect of a protective coating containing 10% Fe(III)(BTA)3 dissolved in Vaseline and ‘Curve 30%’ highlights the significant corrosion inhibition achieved with a coating containing 30% Fe(III)(BTA)3 in Vaseline, showing the most substantial reduction in mass loss over time. From practical experience and extrapolation of results, it is evident that the coating containing 30% Fe(III)(BTA)3 in Vaseline offers robust corrosion protection lasting several months even under harsh environmental conditions. This extended protection period is crucial for industries where metal assets are exposed to corrosive agents on a continuous basis. The combination of polar and non-polar groups in the Fe(III)(BTA)3 molecule enhances its adsorption affinity on metal surfaces compared to coatings composed solely of Vaseline. The polar groups interact favourably with metal atoms, whilst the non-polar chains contribute to the hydrophobic barrier essential for repelling water and preventing corrosion initiation. Vaseline-based coatings containing Fe(III)(BTA)3 can be applied using various methods such as spraying, dipping, or immersion due to their versatile formulation. Moreover, these coatings can be easily removed from surfaces using hydrocarbon solvents or heating, if necessary, although their thin and protective nature often renders removal unnecessary. This characteristic minimises waste and facilitates maintenance procedures, particularly in industrial settings where efficiency is paramount. In practical tests, as illustrated in Figure 12, a copper sheet (Cu-ETP (CW004A), EN 13601) was used to evaluate the protective efficiency of various coatings. The mass percentage decrease was plotted against the duration of the experiment in days. The tests involved exposure to a 3.5% NaCl solution, simulating a corrosive environment.
The integration of 2-(4-nitrophenyl)-1H-benzimidazole derivative Fe(III)(BTA)3 into Vaseline-based coatings marks a significant step forward in corrosion prevention technology. This compound has demonstrated exceptional efficacy in inhibiting corrosion on metal surfaces by leveraging its unique properties of strong metal adsorption and water repellence. The practical applications of Fe(III)(BTA)3 in Vaseline coatings have been substantiated through rigorous experimental validations, highlighting its potential as a sustainable and effective alternative to traditional corrosion inhibitors. The effectiveness of Fe(III)(BTA)3 lies in its ability to form a dense and cohesive protective layer on metal surfaces. This layer adheres strongly due to the polar groups in Fe(III)(BTA)3 that interact favourably with metal atoms, whilst the non-polar hydrocarbon chains contribute to the formation of a hydrophobic barrier. This dual-action mechanism not only prevents moisture ingress, a key factor in corrosion initiation but also enhances the durability of the protective coating under various environmental conditions.
Experimental studies, such as those illustrated in Figure 12 for copper sheets exposed to a 3.5% NaCl solution, clearly demonstrate the superior corrosion resistance of Vaseline-based coatings containing Fe(III)(BTA)3. The curves depicting mass loss over time indicate that higher concentrations of Fe(III)(BTA)3 lead to greater protection, with the 30% Fe(III)(BTA)3 concentration showing the most significant reduction in corrosion compared to pure Vaseline coatings. Moreover, the practical applicability of Fe(III)(BTA)3 in Vaseline coatings is underscored by its versatility in application methods. Whether applied through spraying, dipping, or immersion, these coatings can be uniformly distributed on metal surfaces, ensuring comprehensive protection. The ease of application, coupled with the ability to use Vaseline as a solvent, enhances the feasibility of integrating Fe(III)(BTA)3 into existing industrial processes without major modifications. The integration of 2-(4-nitrophenyl)-1H-benzimidazole (5) into Vaseline-based coatings represents not only a technological advancement but also a paradigm shift towards sustainable corrosion prevention solutions. The protective efficacy of the coating increases proportionally with the concentration of Fe(III)(BTA)3, with a 30% concentration identified as optimal for achieving maximum corrosion inhibition. Due to its Fe(III)(BTA)3 content, the Vaseline-based coating forms a dense layer that adheres strongly to the metal surface. This adhesion strength is crucial for maintaining the integrity of the protective film, as it ensures long-term corrosion resistance even in challenging environments. These tests confirmed that Vaseline is the most effective solvent for the active substance. Additionally, the protective effect of the coating increased proportionally with the concentration of the active substance. An optimal concentration of 30% 2Fe(III)(BTA)3 was determined, offering the best balance between protective efficacy and material efficiency. The enhanced properties of the Vaseline-based mixtures present a significant improvement. Its superior adhesion, higher adsorption affinity, ease of removal and efficient application make it a more practical and effective solution for rust prevention. The reduced quantity required for effective coverage not only cuts down on material costs but also minimises environmental impact. By requiring less material, the Vaseline-based mixtures reduce waste and the need for frequent reapplications. This efficiency translates into economic savings and a more sustainable approach to corrosion prevention. Additionally, exploring environmentally friendly solvents and additives could align with the increasing emphasis on green chemistry and sustainable practices 35 in industrial applications. 36 The versatility of the Vaseline-based mixtures is suitable for a wide range of industries, from automotive to maritime. Its ability to provide long-lasting protection in various environmental conditions ensures that it can be tailored to specific industrial needs, thereby enhancing the durability and lifespan of metal components. The Vaseline-based mixtures represent a significant advancement in rust prevention technology. Its strong adhesion, high adsorption affinity and efficient application make it an ideal solution for protecting metal surfaces against corrosion. The practical benefits, combined with economic and environmental advantages, highlight its potential as a leading rust-inhibiting material for various industrial applications.
To strengthen the methodology and ensure more robust and reliable conclusions, there are several specific improvements and additional controls:
Surface preparation significantly impacts the effectiveness of corrosion inhibitors. Variations in surface cleanliness, roughness and pre-treatment can influence how well the inhibitor adheres to the metal surface. During future experiments, authors should standardise and rigorously document the surface preparation process. Techniques such as sandblasting, chemical etching, or mechanical polishing should be consistently applied and tested for their impact on inhibitor performance. Moreover, examining how different surface finishes affect the protective layer's formation and durability would provide valuable insights. Laboratory tests often do not fully replicate real-world environmental conditions, which can include fluctuations in temperature, humidity and exposure to various pollutants. During future experiments, the authors should include long-term environmental exposure tests, such as salt spray tests, cyclic corrosion testing, or outdoor exposure trials in different climates. These tests would simulate real-world conditions more accurately and provide a better understanding of the long-term effectiveness and durability of the inhibitors. Whilst the study highlights the advantages of OPD derivatives and Vaseline-based mixtures, it does not adequately compare these new inhibitors against well-established, commercially available corrosion inhibitors under identical conditions. During future experiments, authors should include a series of comparative studies where OPD derivatives and Vaseline-based mixtures are tested alongside traditional inhibitors (e.g. chromates, phosphates) under the same experimental conditions. This comparison would help quantify the performance improvements and better position the new inhibitors within the existing market of corrosion protection solutions. The effectiveness of corrosion inhibitors can be significantly affected by the thickness and uniformity of the applied film. Inconsistent application can lead to weak points susceptible to corrosion. During future experiments, authors should implement precise measurement techniques, such as ellipsometry or scanning electron microscopy, to quantify the thickness and uniformity of the inhibitor film across different samples. Ensuring consistent application would lead to more reliable results and help identify optimal application methods. Basic electrochemical tests like dynamic polarisation or electrochemical impedance spectroscopy might not provide a complete understanding of the inhibitor's performance. More advanced electrochemical techniques, such as localised electrochemical impedance spectroscopy or scanning vibrating electrode technique are useful, these methods would allow for the assessment of localised corrosion processes and give deeper insights into the inhibitor's protective mechanism. Corrosion studies often focus on a single type of metal substrate, typically steel. However, industrial applications often involve alloys or mixed-metal systems, which can exhibit different corrosion behaviours. During future experiments, authors should expand their study to include various metal substrates, such as aluminium alloys, copper and stainless steel, to assess the generalisability of OPD derivatives and Vaseline-based mixtures. Additionally, testing the inhibitors on mixed-metal systems would provide valuable information on their effectiveness in complex industrial scenarios. Various studies promote the eco-friendliness of the new inhibitors but lack thorough evaluation of their environmental impact and potential toxicity. A more detailed analysis of the environmental impact and toxicity of the OPD derivatives and Vaseline-based mixtures could include biodegradability tests, leaching studies and assessments of the inhibitors’ impact on aquatic life. Such evaluations would strengthen the claim of environmental benefits and ensure compliance with regulatory standards. To fully understand the protective capability of the inhibitors, it is crucial to have a control group where the metal surfaces receive no treatment. This control would provide a baseline for evaluating the effectiveness of the corrosion inhibitors and help quantify the extent of protection offered by the treatments.
By incorporating these improvements and additional controls, the future studies methodology would be more rigorous, leading to more comprehensive and reliable conclusions about the effectiveness and applicability of OPD derivatives and Vaseline-based mixtures in corrosion prevention.
Conclusions
Recent advancements in corrosion science have underscored the efficacy of OPD derivatives as potent inhibitors capable of mitigating both anodic and cathodic reactions fundamental to the corrosion process. By forming a robust protective layer on metal surfaces, OPD effectively impedes the onset of rust and corrosion. This protective mechanism operates through the adsorption of OPD derivatives onto metal surfaces, creating a physical barrier that restricts the ingress of corrosive agents such as oxygen and moisture. Moreover, OPD exhibits versatility in its application, commonly integrated into various protective coatings and treatments aimed at providing enduring corrosion resistance for metal substrates. These coatings serve as durable shields, safeguarding metals from environmental stressors and preventing the initiation and propagation of rust formation. The versatility of OPD-based treatments extends beyond newly manufactured products to include the restoration and preservation of existing metal structures, where they penetrate and adhere to surfaces, fortifying them with long-lasting protective layers. In industrial settings, the synergistic combination of OPD with other corrosion inhibitors has proven particularly advantageous. The collaborative action between OPD and complementary inhibitors enhances overall corrosion protection, thereby extending the service life of critical metal assets. This integrated approach not only enhances efficacy but also supports sustainable practices by minimising resource consumption and reducing environmental impact, aligning with global initiatives for eco-friendly corrosion mitigation strategies. The adoption of OPD-based coatings and treatments signifies a significant paradigm shift towards greener alternatives in rust prevention. Unlike conventional chemical-intensive methods, OPD derivatives offer a more environmentally benign solution. Incorporating electrochemical techniques and natural corrosion inhibitors alongside OPD further enhances its effectiveness whilst concurrently reducing the ecological footprint associated with corrosion prevention efforts. Recent research has highlighted the exceptional effectiveness of Vaseline-based mixtures as superior rust inhibitors compared to previously utilised materials. This innovative combination demonstrates an order of magnitude greater rust-inhibiting capability, emphasising its immediate recommendation for widespread production and application across various industries. However, to optimise its efficacy across diverse industrial contexts, further investigations are warranted. These should focus on refining application methodologies and surface preparation techniques tailored to specific operational environments and metal substrates.
Continued research and innovation are imperative for advancing greener rust prevention strategies and fostering a sustainable future. Ongoing efforts to refine corrosion inhibitors, develop enhanced application methodologies and embrace eco-friendly coatings are pivotal. Such initiatives not only contribute to mitigating the environmental impact of industrial activities but also ensure the long-term durability and integrity of valuable metal assets worldwide. In conclusion, the evolution towards OPD derivatives and Vaseline-based mixtures as advanced corrosion inhibitors represents a pivotal advancement in materials science and engineering. By integrating these innovations into practical applications, industries can achieve robust corrosion protection whilst adhering to sustainability principles, thereby contributing to a more resilient and eco-conscious industrial landscape. Recent research has identified specific criteria that rust-inhibiting substances must fulfil to effectively protect metal surfaces from corrosion. These criteria include the formation of a durable compact layer, adequate wetting or adsorption capacity, porelessness, hydrophobicity, ease of application and considerations regarding removal from surfaces.
The integration of OPD derivatives and Vaseline-based mixtures into corrosion prevention strategies represents a significant expansion of the subject area compared to existing published materials on rust prevention and corrosion science. Traditionally, research in this field has focused on well-established methods involving chemical inhibitors and coatings, often with an emphasis on the effectiveness of various compounds like chromates, phosphates and nitrites. These methods, whilst effective, have been scrutinised for their environmental impact and potential health risks, particularly in the case of chromate-based inhibitors, which are known to be carcinogenic. The current focus on OPD derivatives and Vaseline-based mixtures, however, introduces a new dimension to corrosion science by prioritising eco-friendly, sustainable solutions that do not compromise on performance. The primary objective of a rust inhibitor is to create a robust and tightly adherent layer on metal surfaces to shield them from environmental factors that promote corrosion. Vaseline-based layers have been found to generally achieve satisfactory compactness, providing a protective barrier against moisture and oxygen. The ability of a rust inhibitor to wet the metal surface effectively and facilitate the adsorption of its active components is crucial for ensuring uniform coverage and long-term protection. Vaseline-based materials, predominantly composed of apolar and slightly polar hydrocarbons, exhibit limitations in wetting polar metal surfaces efficiently. This limitation can affect the uniformity and durability of the protective layer, particularly in environments with fluctuating humidity and temperature conditions. A poreless coating is essential to prevent the penetration of corrosive agents such as water and salts, which can accelerate rust formation. Research indicates that Vaseline-based layers can establish relatively poreless coatings, especially when applied with solvents that promote the formation of a homogeneous film. However, it is noted that after solvent evaporation, there is a risk of pore formation, potentially compromising the inhibitor's effectiveness over extended periods. Hydrophobic properties are critical for rust inhibitors as they enable the repellence of water and moisture from the metal surface. Vaseline-based materials typically exhibit satisfactory hydrophobic behaviour initially. However, due to the inherent polarity of metal surfaces, water molecules may adhere more readily compared to nonpolar protective materials previously used, which can displace the inhibitor and undermine its protective efficacy over time. Practical considerations regarding the ease of application play a significant role in the adoption of rust inhibitors across various industrial and domestic applications. Vaseline-based layers are generally considered straightforward to apply, although they may not be ideal for spraying applications due to their viscosity and consistency. In contrast, ceresin-like materials often present challenges during application, requiring meticulous attention to achieve uniform coverage. The ability to remove or clean the inhibitor from surfaces without causing damage is crucial for maintenance and reapplication purposes. Vaseline-based materials are typically removable from protected surfaces using hydrocarbon solvents, facilitating periodic maintenance and reapplication. Conversely, the removal of ceresin-like coatings, especially from lubricated surfaces, demands meticulous cleaning procedures to prevent operational issues and ensure surface integrity.
Whilst Vaseline-based inhibitors demonstrate advantages such as compactness, ease of removal and initial hydrophobicity, challenges remain regarding their application on polar metal surfaces and the potential for pore formation. These findings underscore the ongoing need for research and development to refine rust-inhibiting technologies, ensuring robust protection against corrosion whilst addressing practical challenges in application and maintenance. Rust prevention is crucial for preserving the functionality and longevity of metal tools and machine parts, especially during storage and transportation. Evaluating the properties of rust-inhibiting materials based on their ability to meet specific criteria is essential to determine their effectiveness. Vaseline-based layers are known to form sufficiently compact coatings. The dense nature of Vaseline-based substances provides a protective barrier that prevents moisture and oxygen from reaching the metal surface, thereby inhibiting rust formation. However, these layers can sometimes become less effective when exposed to high temperatures, which may cause them to melt and lose their protective integrity. Vaseline-based materials consist largely of apolar and slightly polar hydrocarbons, which means they wet polar metal surfaces relatively poorly. The poor wetting capability results in less effective adhesion to the metal surface, reducing the overall protective performance. This limitation is particularly significant when the metal surface has high surface energy, leading to inadequate coverage and potential exposure to corrosive agents. Materials with better wetting and adsorption properties are needed to enhance the protective efficacy. For instance, certain organic inhibitors and synthetic polymers exhibit superior wetting characteristics, enabling them to form more uniform and adherent coatings on metal surfaces.
Vaseline-based layers establish relatively poreless coatings, especially when applied with solvents to the surface to be protected. The solvent helps spread the Vaseline evenly, filling micro voids and creating a seamless barrier. However, as the solvent evaporates, the layer may form pores that diminish its effectiveness. This pore formation can be mitigated by using multiple coats or by employing materials that inherently form nonporous films. In terms of hydrophobicity, Vaseline-based materials behave satisfactorily on their own. They repel water to a certain extent, which helps in preventing moisture from reaching the metal surface. However, because the metal surface itself is polar, water tends to be more adsorbed on it than on the previously used non-polar protective materials. This interaction allows water to penetrate the coating, displacing the protective layer and exposing the metal to rusting. Enhancing the hydrophobic properties of rust inhibitors can significantly improve their performance. Incorporating fluorinated compounds or silane-based treatments can increase the water-repellent ability of the coating, providing better protection against moisture-induced corrosion.
Vaseline-based mixtures can be easily applied using brushes or rollers but are unsuitable for spraying due to their high viscosity. This limitation affects their usability in large-scale industrial applications where efficient and uniform coating methods are required. Despite this, Vaseline-based layers remain a practical choice for small-scale or manual applications. Vaseline-based materials can be easily removed from the protected surface using hydrocarbon solvents. The ability to remove the coating without damaging the underlying metal is advantageous for maintenance and reapplication purposes. However, for lubricated surfaces, the coating must be removed entirely to ensure proper lubrication and avoid contamination. This requirement necessitates the use of rust inhibitors that can be dissolved or washed away without leaving residues. The environmental impact of rust-inhibiting materials is a significant consideration. Traditional rust inhibitors often contain hazardous chemicals that can harm the environment and human health. For instance, certain chromate-based inhibitors, whilst effective, pose serious environmental risks and are subject to strict regulations. Therefore, the development of eco-friendly rust inhibitors that do not compromise performance is essential. Recent research has focused on the development of novel rust inhibitors that meet the criteria outlined above whilst minimising environmental impact. One promising avenue is the use of organic inhibitors derived from natural sources. These inhibitors often exhibit excellent adsorption properties and form compact, poreless coatings. Examining real-world applications of rust-inhibiting materials can provide valuable insights into their practical effectiveness. For instance, in the automotive industry, rust prevention is crucial for maintaining the structural integrity and appearance of vehicles. Modern rust inhibitors used in automotive applications often combine multiple protective mechanisms, including hydrophobic coatings, sacrificial anodes and corrosion-resistant alloys. These comprehensive strategies ensure long-lasting protection even in harsh environmental conditions. Evaluating the properties of rust-inhibiting materials based on criteria such as compactness, wetting capacity, porelessness, hydrophobicity, ease of application and removability is essential for determining their effectiveness. Vaseline-based mixtures, whilst offering certain advantages, have limitations that must be addressed through innovation and the development of new materials. The exploration of organic inhibitors, advances in nanotechnology and environmentally friendly formulations represent promising directions for future research and application. By continuing to improve rust prevention technologies, we can protect valuable metal assets, reduce economic losses and minimise environmental impact. Looking ahead, future research should prioritise several key areas to further enhance the efficacy and applicability of Vaseline-based coatings. Continued research should focus on refining the formulation of BTA in Vaseline to achieve maximum stability and performance. This includes exploring additives or modifying the composition to improve adherence and durability on different metal substrates. Exploring synergies with other green technologies, such as renewable energy sources or eco-friendly surface treatments, could further enhance the sustainability profile of Vaseline-based coatings. Collaborative research efforts could lead to integrated solutions that promote holistic environmental stewardship in corrosion prevention practices. The shift towards eco-friendly corrosion inhibitors aligns with global efforts to reduce carbon footprints and enhance resource efficiency in the manufacturing and infrastructure sectors.
Moreover, the article advances the subject area by discussing the synergistic combination of OPD with other corrosion inhibitors. This approach is relatively underexplored in existing literature, where the focus has largely been on individual inhibitors and their standalone efficacy. The collaborative action between OPD and complementary inhibitors, as highlighted in the article, enhances overall corrosion protection and extends the service life of metal assets. This integrated strategy not only improves the performance of corrosion inhibitors but also contributes to resource efficiency and environmental sustainability, offering a more holistic approach to corrosion prevention.
In addition, the incorporation of Vaseline-based mixtures into the discussion expands the subject area by addressing practical challenges associated with corrosion protection. Vaseline-based inhibitors, whilst not entirely new, have been revisited in this context to explore their potential as eco-friendly alternatives to traditional coatings. The research presented in the article sheds light on both the advantages and limitations of these mixtures, particularly in terms of their application on polar metal surfaces and their susceptibility to pore formation. This nuanced exploration of Vaseline-based coatings fills a gap in the literature by providing a more detailed analysis of their performance under varying environmental conditions, offering insights into how these materials can be optimised for broader industrial use. Furthermore, the article's emphasis on real-world applications and the practical effectiveness of these materials in diverse industrial contexts offers a fresh perspective that is often lacking in more theoretical studies. By discussing the integration of OPD derivatives and Vaseline-based mixtures into existing rust prevention strategies, the article bridges the gap between laboratory research and practical implementation, highlighting the feasibility and benefits of these eco-friendly solutions in real-world scenarios.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
