Abstract
The synthesis and application of metal complexes of 2-(4-nitrophenyl)-1H-benzimidazole, a significant heterocyclic compound, are rooted in the formation of its mono Schiff base precursor, N-(4-nitrobenzyl) benzene-1,2-diamine. This precursor is synthesized via the condensation of o-phenylenediamine (OPD) with 4-nitrobenzaldehyde (NBA). The formation of the Schiff base involves the nucleophilic attack of OPD's amino groups on the carbonyl carbon of 4-nitrobenzaldehyde, resulting in an imine linkage essential for subsequent cyclization reactions. Cyclization of the mono Schiff base N-(4-nitrobenzyl) benzene-1,2-diamine (NBD) is a crucial step in forming the benzimidazole ring. This compound's significance lies in its structural and electronic properties, influenced by the electron-withdrawing nitro group, making it valuable in coordination chemistry, medicinal chemistry, and materials science. In coordination chemistry, metal complexes of 2-(4-nitrophenyl)-1H-benzimidazole (NBI) acts as a ligand, forming stable complexes with transition metals that exhibit enhanced catalytic activity. These metal complexes are essential for various catalytic processes, including organic transformations and polymerization reactions. In medicinal chemistry, the benzimidazole core is a common motif in biologically active compounds. The presence of the nitro group can modulate the biological activity, making derivatives of 2-(4-nitrophenyl)-1H-benzimidazole (NBI) promising candidates for antimicrobial, anticancer, and antiviral agents. Research has also highlighted the applications of Schiff bases and their metal complexes in various fields. Schiff bases serve as versatile intermediates in the synthesis of heterocyclic compounds, while their metal complexes are explored for their catalytic, antimicrobial, and anticancer properties. The synthesis of 2-(4-nitrophenyl)-1H-benzimidazole and its derivatives remains a topic of significant interest due to their potential in developing new materials and pharmaceuticals. The synthesis of metal complexes of 2-(4-nitrophenyl)-1H-benzimidazole from its Schiff base precursor is a critical process with wide-ranging applications in coordination chemistry, medicinal chemistry, and materials science. The optimization of reaction conditions and the exploration of the compound's properties continue to drive research in this area, highlighting its importance in both fundamental and applied sciences.
Introduction
Schiff bases, formed through the condensation of primary amines with carbonyl compounds, are crucial intermediates in organic synthesis and coordination chemistry. Among these, the mono-Schiff base N-(4-Nitrobenzyl) benzene-1,2-diamine derived from o-phenylenediamine (OPD) and 4-nitrophenyl benzaldehyde (NBA) stands out due to its unique structural and electronic properties. The metal complexes of this mono-Schiff base N-(4-Nitrobenzyl) benzene-1,2-diamine have garnered significant interest for their applications in various fields, including catalysis, materials science, and medicinal chemistry, aims to explore the importance of the mono-Schiff base of OPD 4-nitrophenyl benzaldehyde and its metal complexes, highlighting their distinctive properties and applications. The mono-Schiff base of OPD and 4-nitrophenyl benzaldehyde, the N-(4-Nitrobenzyl) benzene-1,2-diamine (NBD) forms a chelating ligand that can effectively coordinate with metal ions. This Schiff base has a rigid, planar structure, which facilitates the formation of stable metal complexes. The 4-nitrophenyl group, with its electron-withdrawing nitro group, significantly impacts the electronic properties of the ligand, affecting its coordination behaviour and interaction with metal centres. 1 The 4-nitrophenyl group is an electron-withdrawing substituent that enhances the acidity of the imine proton in the Schiff base. This modification influences the electronic environment of the metal centre in the resulting metal complexes, making them more reactive in various chemical processes. The nitro group can stabilize metal centres through inductive and resonance effects, leading to enhanced catalytic activity. 2 The materials derived from metal salts of the mono-Schiff base generally exhibit high thermic stability. The nitro group can enhance the electronic and optical properties of these materials, making them suitable for applications in sensors and light-emitting devices. In contrast, other substituents may lead to different material properties, affecting their suitability for specific applications. The formation of N-(4-Nitrobenzyl) benzene-1,2-diamine mono Schiff base (NBD) is an interesting reaction in organic synthesis, to create versatile intermediates for further chemical transformations. The mono Schiff base can act as a bidentate ligand, coordinating through both nitrogen atoms. This coordination ability is enhanced by the presence of the nitro group, which can further stabilize the metal-ligand bond. The ability of NBD to form chelate rings with metal ions leads to the formation of stable and often highly coloured complexes, which are valuable for various applications. The chelating nature of NBD also influences the reactivity and selectivity of the metal centres, making it a versatile ligand in coordination chemistry. Copper(II)-NBD complexes have demonstrated activity against various pathogenic microorganisms, including bacteria and fungi. The ability of these complexes to disrupt microbial processes highlights their potential as therapeutic agents. Similarly, zinc(II)-NBD complexes have shown anticancer activity, with potential applications in cancer treatment. 3 The influence of the nitro group on the biological activity of these complexes underscores the importance of NBD in drug development. Several studies have investigated the synthesis of N-(4-nitrobenzyl) benzene-1,2-diamine and related Schiff bases. For instance, Nworie 4 et al. reported bis-imine Schiff bases are stable metal complexing agents. Similarly, Green 5 et al. explored the use of different catalytic systems to enhance the efficiency of Schiff base formation, including the use of microwave irradiation to accelerate the reaction. Patel 6 et al. focused on the role of solvent effects in Schiff base synthesis, noting that solvent polarity can significantly impact the reaction kinetics and product distribution. Other studies, such as Nagar 7 et al. have explored the applications of Schiff bases in coordination chemistry and materials science. Catalán 8 et al. investigated the electronic properties of Schiff bases derived from nitro-substituted aldehydes, highlighting how electron-withdrawing groups influence the formation and stability of the resulting imine linkages. Tellez et al. discussed the use of Schiff base ligands in the formation of metal complexes with enhanced activity. 9 Cyclization of Schiff bases is a critical step in the synthesis of various heterocyclic compounds, including benzimidazoles. Specifically, the cyclization of the Schiff base N-(4-nitrobenzyl) benzene-1,2-diamine to form 2-(4-nitrophenyl)-1H-benzimidazole is an important reaction in the field of coordination chemistry and medicinal chemistry. This reaction forms the benzimidazole ring, which is a key structure in many biologically active compounds and metal-organic frameworks. This review focuses on the cyclization process, the conditions required, and the significance of the resulting benzimidazole derivative.
Benzimidazole and its derivatives have garnered significant attention in various fields of research due to their versatile chemical properties and broad range of applications. When complexed with metal salts, benzimidazoles exhibit enhanced functionalities that make them crucial in medicinal chemistry, catalysis, materials science, and environmental applications. This review aims to highlight the importance of benzimidazole metal salts and their substituted derivatives, drawing from a rich body of literature. One of the primary areas where benzimidazole metal salts have shown remarkable potential is medicinal chemistry. Benzimidazole itself is a core structure in many bioactive molecules, known for its antibacterial, antiviral, and antifungal properties. When complexed with metal ions, these properties are often significantly enhanced. For instance, copper(II) and zinc(II) complexes with benzimidazole ligands have shown improved antibacterial activity compared to their non-metal counterparts.10–13 The metal ion's presence can facilitate interaction with biomolecular targets, enhancing the drug's efficacy. Moreover, some benzimidazole metal complexes exhibit notable antitumor activities.14,15 Research has demonstrated that platinum 16 complexes of benzimidazole derivatives can induce apoptosis in cancer cells, presenting a promising approach for chemotherapy. These complexes are believed to interfere with DNA replication, like the mechanism of action of cisplatin, but often with reduced side effects. Benzimidazole metal complexes also play a pivotal role in catalysis. Their unique structure allows them to act as effective ligands, stabilizing metal centres that catalyse various chemical reactions. For instance, palladium-benzimidazole complexes have been employed as catalysts in C-C coupling reactions, such as the Suzuki-Miyaura and Heck reactions, which are fundamental in organic synthesis. These catalysts are valued for their high efficiency and selectivity, often operating under mild conditions. In addition, copper-benzimidazole complexes have been explored as catalysts for oxidative reactions. They facilitate the oxidation of alcohols to aldehydes and ketones, crucial transformations in both laboratory and industrial settings. The ability to fine-tune the electronic properties of benzimidazole ligands by introducing substituents further enhances their catalytic performance, making them versatile tools in synthetic chemistry. The synthesis of 2-(4-nitrophenyl)-1H-benzimidazole begins with the formation of the Schiff base N-(4-nitrobenzyl) benzene-1,2-diamine. This intermediate is created by the condensation of o-phenylenediamine (OPD) with 4-nitrobenzaldehyde. In this reaction, the primary amino groups of OPD react with the aldehyde group of 4-nitrobenzaldehyde to form an imine linkage, yielding the Schiff base. This intermediate is crucial for the subsequent cyclization to form the benzimidazole ring. The cyclization of N-(4-nitrobenzyl) benzene-1,2-diamine to form 2-(4-nitrophenyl)-1H-benzimidazole involves the formation of a five-membered ring through the condensation of the imine nitrogen and one of the aromatic ring nitrogen. The cyclization process typically requires heating and the presence of a suitable catalyst, such as an acid or base, to facilitate the ring closure. Briefly, the cyclization mechanism involves the nucleophilic attack of the imine nitrogen on the carbon of the adjacent aromatic ring, resulting in the formation of the benzimidazole ring. This process is followed by deprotonation and rearrangement to yield 2-(4-nitrophenyl)-1H-benzimidazole. The results can emphasize the uniqueness of the research in synthesizing and characterizing the metal complexes of 2-(4-nitrophenyl)-1H-benzimidazole from its mono-Schiff base precursor. Specifically, the novelty lies in exploring how the electronic properties induced by the nitro group enhance the stability, reactivity, and biological or catalytic activities of these complexes. Highlighting their unique potential applications in medicinal chemistry and materials science would underscore the research's contribution.
The synthesis and characterization of these complexes are undertaken to assess their properties and potential applications. The study examines how the nitro group induces structural and electronic modifications, influencing coordination behaviour and biological activity. Furthermore, the research combines experimental synthesis with computational analyses to understand the reaction mechanisms and stability, addressing the critical question of how substituents influence the functionality of such compounds. In this study, we synthesize and characterize the mono-Schiff base precursor N-(4-nitrobenzyl) benzene-1,2-diamine (NBD) and its derivative, 2-(4-nitrophenyl)-1H-benzimidazole (NBI), highlighting their unique properties due to the electron-withdrawing nitro group. The experimental work is supported by computational analyses to elucidate the reaction mechanisms and assess the influence of substituent effects on stability and reactivity. The synthesized metal complexes are evaluated for their structural, electronic, and functional properties, with particular attention to their potential applications in catalysis, materials science, and medicinal chemistry. The following sections will describe the detailed synthetic procedures, computational methodologies, structural characterizations, and an assessment of the biological and catalytic activities of these compounds.
Materials and methods
All chemicals used were of analytical grade and purchased from Sigma Aldrich, Germany, and were used without further purification. Solutions were all prepared using doubly distilled water. All DFT calculations were carried out with the Gaussian 16 suite of programs. 17 Previous studies have concluded that the meta-GGA functional TPSSh is suitable for geometric optimizations involving transitional metals, therefore this functional has been employed using the def2-SVP basis set unless otherwise noted. 18 Additional computations were performed on the ωb97X-D/def2-TZVP level of theory Harmonic vibrational analysis was obtained at the same level, and in the case of a minima, all eigenvalues of the Hessian matrix were positive for the determination of bond critical points, bond orders and orbital diagrams the Multiwfn program was used. 19 For visualization of the molecular orbitals IQmol program was used.4 For NBO calculations NBO 7.0 was used (implemented in Gaussian 16). 20 The quantities of iron(II) and manganese(II) ions were determined permanganometrically using a KMnO4 titrant, while the quantities of copper(II), nickel(II), and zinc(II) ions were determined complexometrically using EDTA.
Results
The synthesis of the mono-Schiff base precursor, N-(4-nitrobenzyl) benzene-1,2-diamine (NBD), involves a specific reaction pathway using o-phenylenediamine and 4-nitrobenzaldehyde under controlled conditions. If this methodology offers improved yields, reaction efficiency, or structural properties compared to previously published methods, it qualifies as a novel approach. The preparation of metal complexes from the NBD precursor and 2-(4-nitrophenyl)-1H-benzimidazole (NBI) explores the specific effects of the nitro group on coordination behaviour. The study provides detailed insights into how the electron-withdrawing nitro substituent influences the reactivity, stability, and functional properties of the complexes, which could represent a new contribution to the field.
Preparation of N-(4-nitrobenzylidene) benzene-1,2-diamine (NBD)
Crude N-(4-nitrobenzylidene) benzene-1,2-diamine (NBD) was prepared as follows: in an Erlenmeyer flask a o-phenylenediamine (1.08 gram, 10 mmol) was dissolved in ethanol and the clear solution was stirred with gentle heating for 5 min as seen in Figure 1. The solution was left to cool to room temperature, then 1 eq. of p-nitrobenzaldehyde (NBA) was added in one portion and the solution was stirred for an hour. The reaction mixture was poured into a crystallizing dish, and an equal volume of water was added to precipitate out the crude product, which was then filtered, and used without additional purification. The crude red powder with a melting point of 96–97 °C, with a yield of about 86–90%.
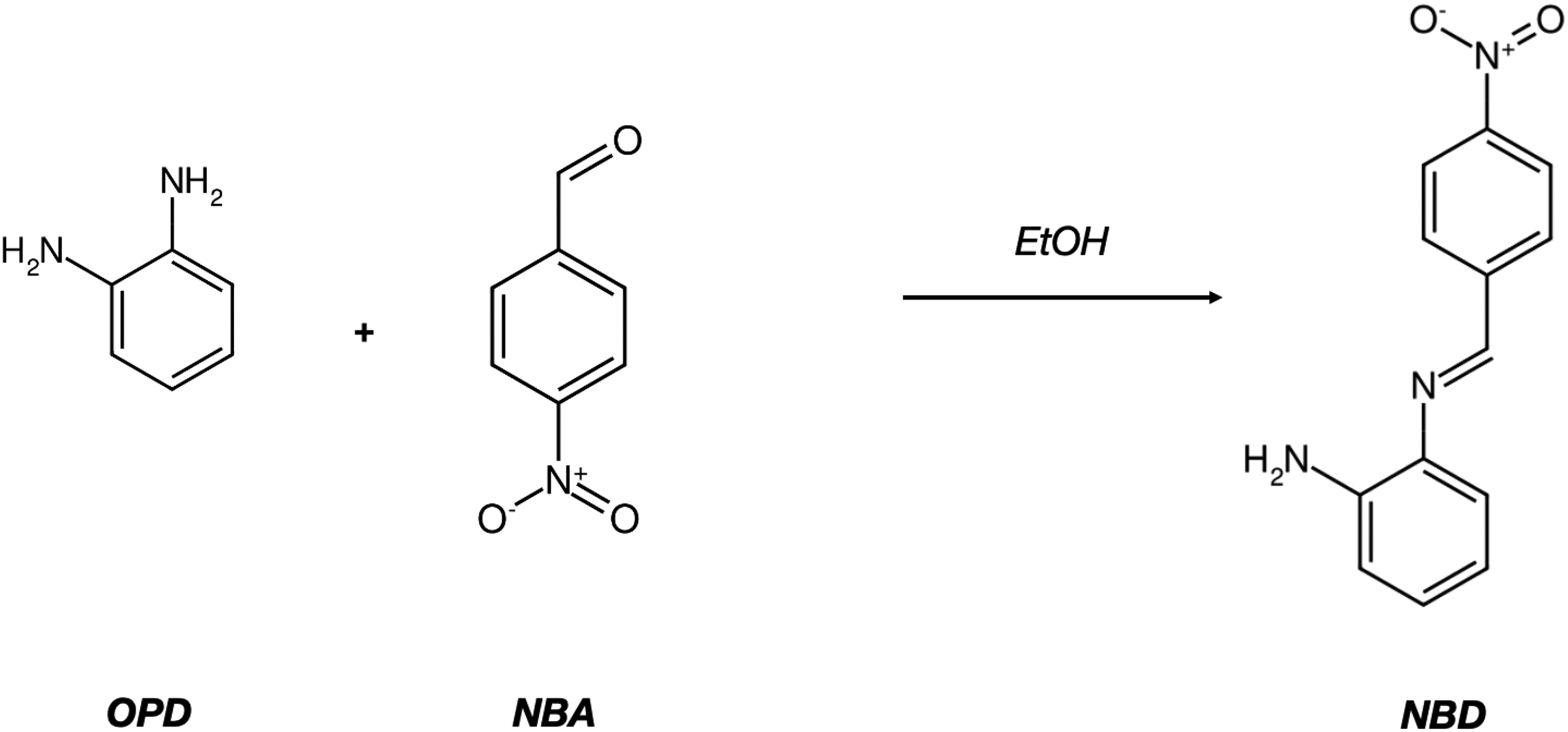
Preparation of Schiff-base intermediate NBD.
Elemental analysis for (C13H11N3O2)): calculated: C: 65.54%, H:4.65%, N:17.63%; found: C: 65.25%, H:4.62%, N:17.71%. FTIR (KBr) υ: 3485, 3260, 1625, 1560, 1335, 820, 750, 500 cm−1.
In the FTIR spectra of NBD the broad peaks around 3300–3500 cm−1 due to symmetric and asymmetric stretching vibrations of the amine (-NH2) groups of starting material OPD are missing, as shown in Figure 2. The N-H bending (aromatic amine) peaks around 1500–1600 cm−1, the C = N stretch (imine group) is a strong band near 1610–1640 cm−1 and nitro group (-NO2) with asymmetric stretch: 1520–1550 cm−1 and symmetric stretch: 1330–1370 cm−1.

FTIR spectra of NBD.
Preparation of metal complexes of n-(4-nitrobenzylidene) benzene-1,2-diamine (NBD)
Schiff base metal complexes were prepared as follows: an Erlenmeyer flask was charged with 1:1 ethanol water mixture, 1 eq. o-phenylenediamine and NaOH, stirred until the solids dissolved, then 0.5 eq. of Cu(NO3)2·3H2O, Fe(NO3)2·6H2O, Zn(NO3)2·6H2O Mn(NO3)3·4H2O was added. The equimolar p-nitrobenzaldehyde (NBA) substrate was added in one portion and left to react for an hour, as seen in Figure 3. The reaction mixture was poured into a crystallizing dish, then solvent was evaporated to obtain the crude product. Crude product was recrystallized, to obtain pure metal complexes. Crude product was filtered, recrystallized from acetone to obtain pure NBD complexes. The yields, melting points and UV maxima are indicated in Table 1.
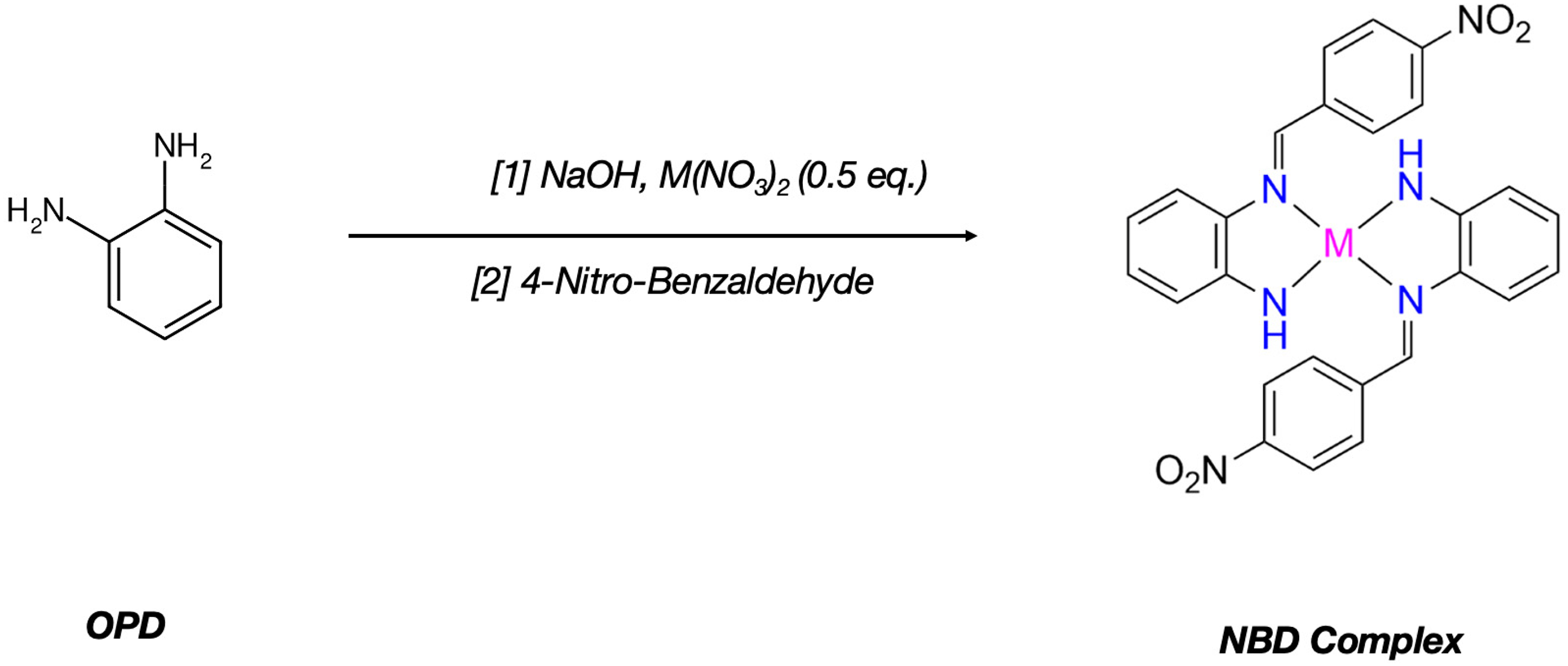
Preparation of Schiff base NBD metal complexes I.
Yields and chemical properties of NBD complexes I.

Schiff base metal complexes were prepared also starting from the NBD and equimolar (2:1) metal nitrates in ethanol/water as shown in Figure 4. Briefly, 2.41 g (0.01 mol) NBD was dissolved in 5w% ethanol: water mixture, then the appropriate metal-nitrate salts were added slowly. The mixture was stirred in room temperature for 2 h, then solvent was evaporated to obtain the crude product. The yields, melting points and UV maxima are indicated in Table 2.
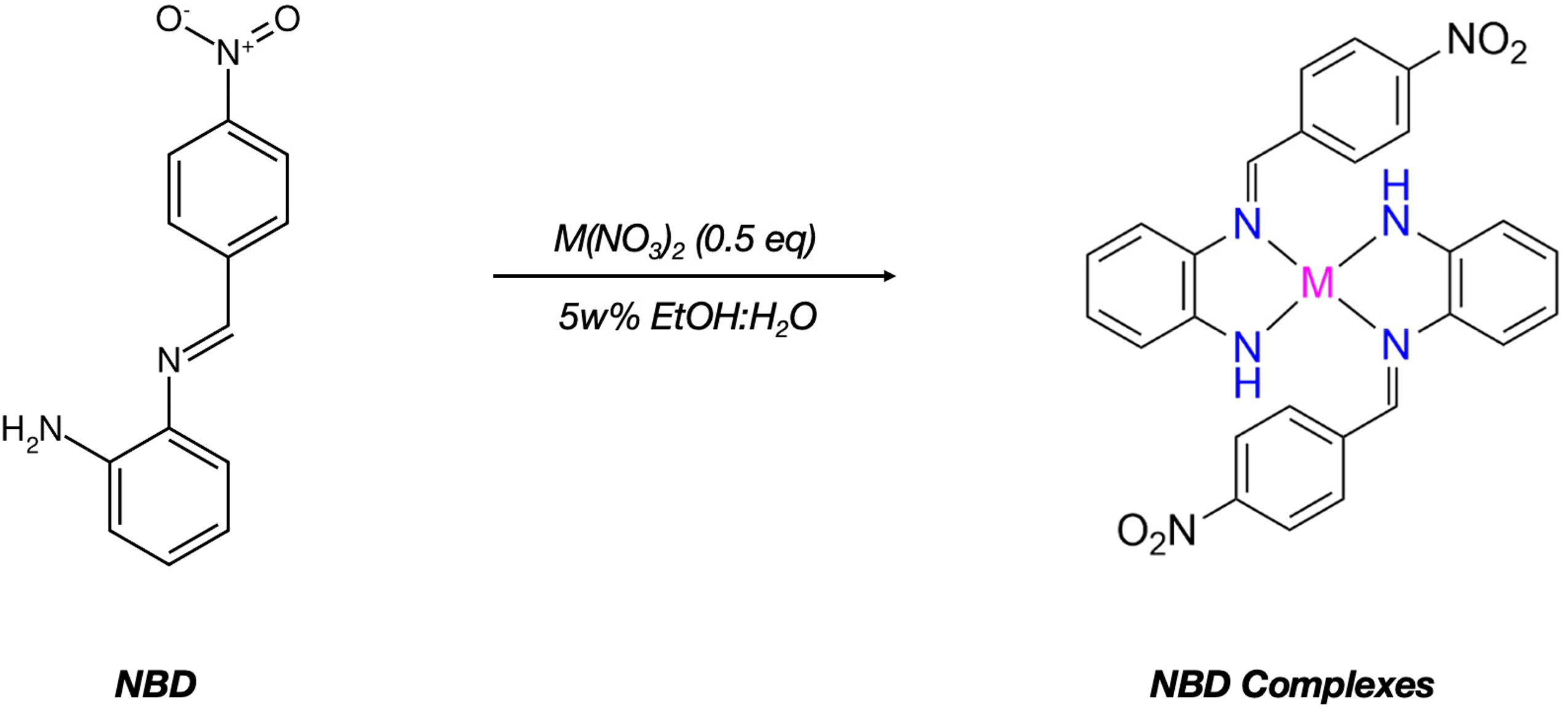
Preparation of Schiff base NBD metal complexes II.
Yields and chemical properties of NBD complexes II.
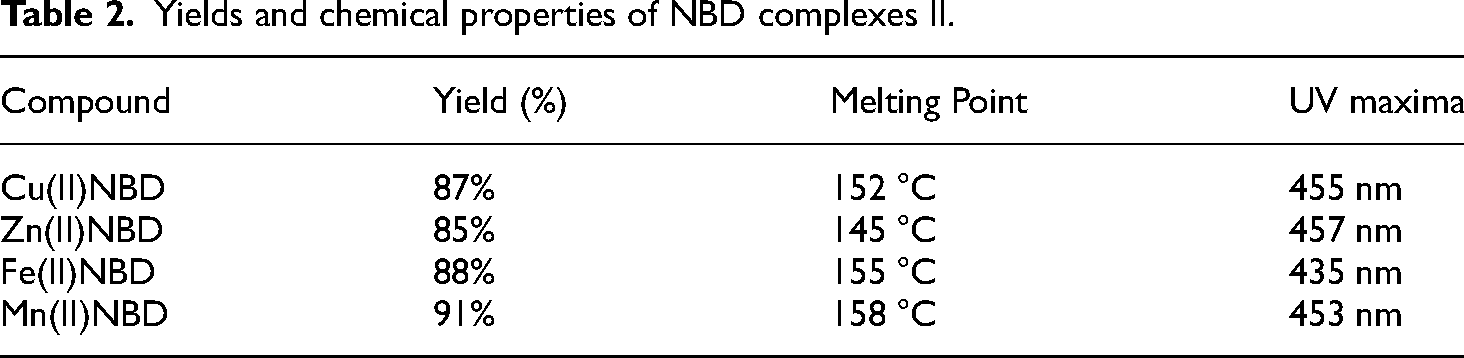
Cupric N-(4-nitrobenzylidene) benzene-1,2-diamine (Cu(II)NBD) complex was formed with stoichiometrically (1:2) cupric salt copper(II) nitrate trihydrate (Cu(NO3)2·3H2O) and OPD. X-ray diffraction gives no pattern as shown in Figure 5; thus, the compound is either amorphous or microcrystalline but suggested that bonding was achieved by the formation of a coordination complex between the cupric ion and the NBD molecule. The relevant peaks of the starting material Cu(NO3)2·3H2O (2θ = 10.8° 14.6° 18.3° 22.0° 25.1°) are missing from the spectra. The Cu(II) ion is probably bonded with a link formed by the replacement of the proton from the NH group and a coordinate bond with a lone pair of electrons from one of the nitrogen atoms. Elemental analysis for Cu(C13H10N3O2)2: calculated: C: 57.40%, H:3.71%, N:15.44%, Cu:11.68%; found: C: 57.18%, H:3.64%, N:15.11%, Cu:11.13%. %. FTIR (KBr) υ: 3380, 3260, 3100, 1635, 1360, 730, 580, 450 cm−1. A metal-to-ligand stretching band around 400–500 cm−1 can appear due to Cu-N coordination.

XRD spectra of Cu(II)NBD.
Zinc N-(4-nitrobenzylidene) benzene-1,2-diamine (Zn(II)NBD) complex was formed with stoichiometrically (1:2) zinc(II) nitrate dihydrate (Zn(NO3)2·2H2O) and OPD. X-ray diffraction gives no specific pattern as shown in Figure 6. The X-ray diffraction of reddish-brown powder pattern of a single crystalline component is zinc(II)-NBD complex (2θ = 14.6° 17.2° 19.8° 22.5° 28.2° 29.0°) as shown in Figure 6. The relevant peaks of the starting material Zn(NO3)2 (2θ = 7.5° 12.4° 16.7° 20.2° 23.3°) are missing from the spectra. Suggested that the Zn(II) ion is bonded with a link formed by the replacement of the proton from the NH group and a coordinate bond with a lone pair of electrons from one of the nitrogen atoms as shown in Figure 6. Elemental analysis for Zn(C13H10N3O2)2: calculated: C: 57.21%, H:3.69%, N:15.40%, Zn:11.97%; found: C: 57.11%, H:3.60%, N:15.51%, Zn:11.23%. FTIR (KBr) υ: 3360, 3240, 3120, 1630, 1350, 740, 550, 460 cm−1. A metal-to-ligand stretching band around 400–500 cm−1 can appear due to Zn-N coordination.
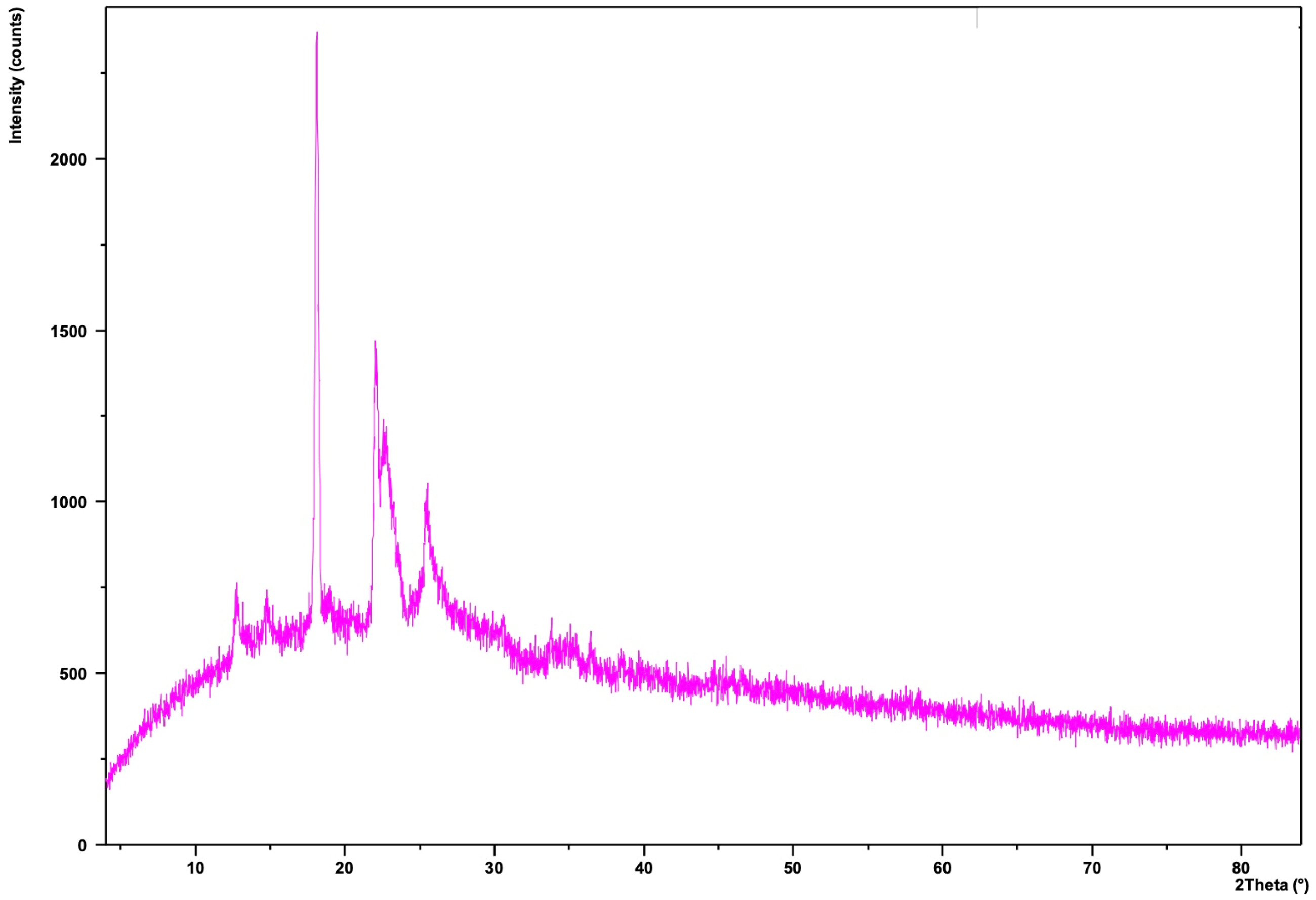
XRD spectra of Zn(II)NBD.
Ferrous N-(4-nitrobenzylidene) benzene-1,2-diamine (Fe(II)NBD) was formed with stoichiometrically (1:2) freshly prepared iron(II) nitrate hexahydrate (Fe(NO3)2·6H2O) and OPD. The reddish-brown powder X-ray diffraction gives no pattern as shown in Figure 7; thus, the compound is either amorphous or microcrystalline but suggested that bonding was achieved by the formation of a coordination complex between the Fe(II) ion and the NBD molecule. The relevant peaks of the starting material Fe(NO3)2·6H2O are missing from the spectra. The Fe(II) ion is probably bonded with a link formed by the replacement of the proton from the NH group and a coordinate bond with a lone pair of electrons from one of the nitrogen atoms. Elemental analysis for Fe(C13H10N3O2)2: calculated: C: 58.22%, H:3.75%, N:15.66%, Fe:10.41%; found: C: 58.05%, H:3.66%, N:15.71%, Fe:10.01%. FTIR (KBr) υ: 3360, 3240, 3100, 1635, 1360, 745, 570, 460 cm−1. A metal-to-ligand stretching band around 400–500 cm−1 can appear due to Fe-N coordination.

XRD spectra of Fe(II)NBD.
Manganese N-(4-nitrobenzylidene) benzene-1,2-diamine (Mn(II)NBD) was formed with stoichiometrically (1:2) manganese(II) nitrate nonahydrate (Mn(NO3)2·4H2O) and OPD. X-ray diffraction gives no pattern as shown in Figure 8. The XRD pattern of the Mn(II)NBD, which is strongly rose-pink in colour. The main peaks of starting material Mn(NO3)2·4H2O are missing from the spectra. The Mn(II) ion is probably bonded with a link formed by the replacement of the proton from the NH group and a coordinate bond with a lone pair of electrons from one of the nitrogen atoms. Elemental analysis for Mn(C13H10N3O2)2: calculated: C: 58.32%, H:3.76%, N:15.71%, Mn:10.26%; found: C: 58.13%, H:3.65%, N:15.41%, Mn:10.03%. FTIR (KBr) υ: 3370, 3250, 3110, 1630, 1355, 730, 580, 450 cm−1. A metal-to-ligand stretching band around 400–500 cm−1 can appear due to Mn-N coordination.
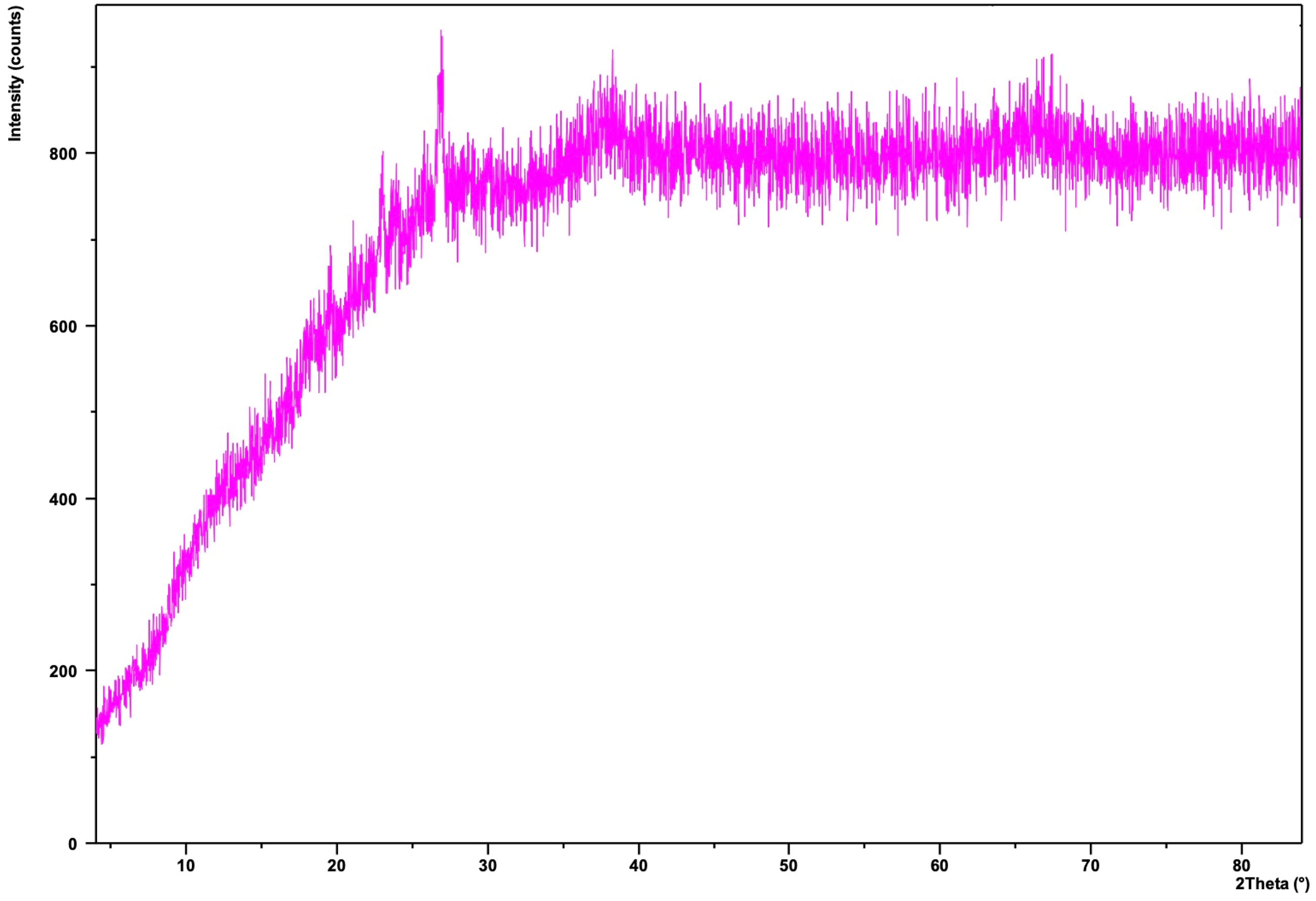
XRD spectra of Mn(II)NBD.
Preparation of 2-(4-Nitrophenyl)-1H-benzimidazole (NBI)
Briefly, 2-(4-Nitrophenyl)-1H-benzimidazole (NBI) was prepared as follows described by Köntös 21 : in a 100 mL round-bottomed flask 10.8 g (0.1 mole) of o-phenylenediamine was treated with (0.1 mole) of p-nitrobenzaldehyde in 50 mL of 1:1 water:glycerol as seen in Figure 9. The mixture is heated in a water bath at 100 °C until precipitates observed. Solvent and solids were separated, crystals were washed with 25 mL of cold water and were recrystallized from ethanol-water. The X-ray diffraction of NBI is shown in Figure 10.
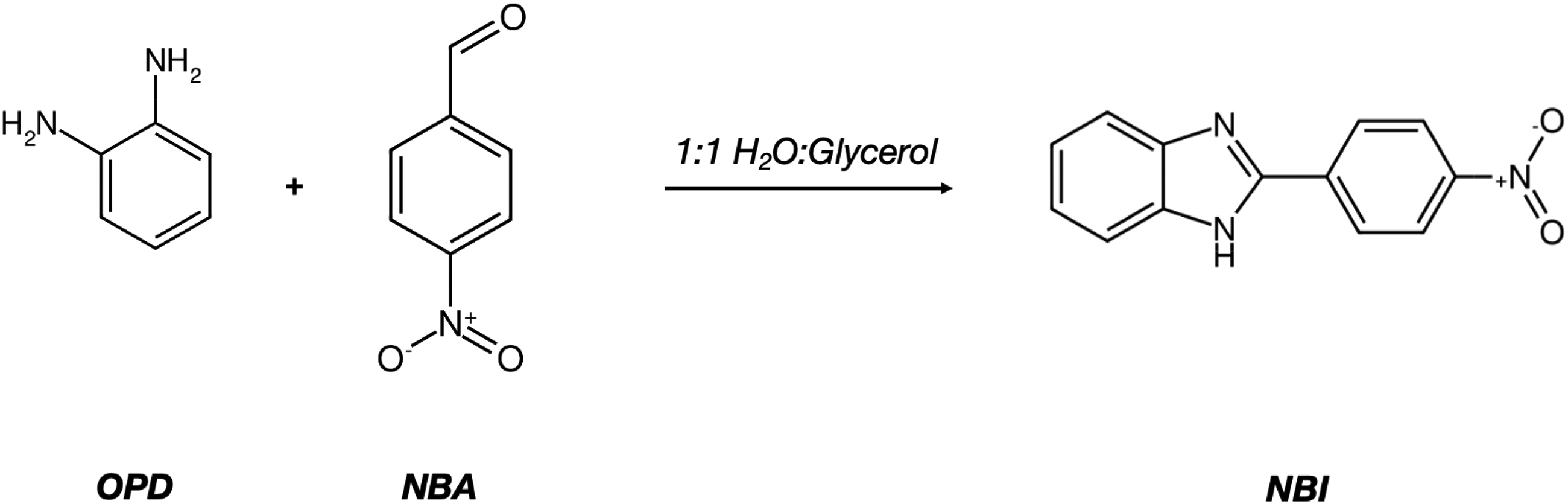
Preparation of 2-(4-Nitrophenyl)-1H-benzimidazole (NBI#1).
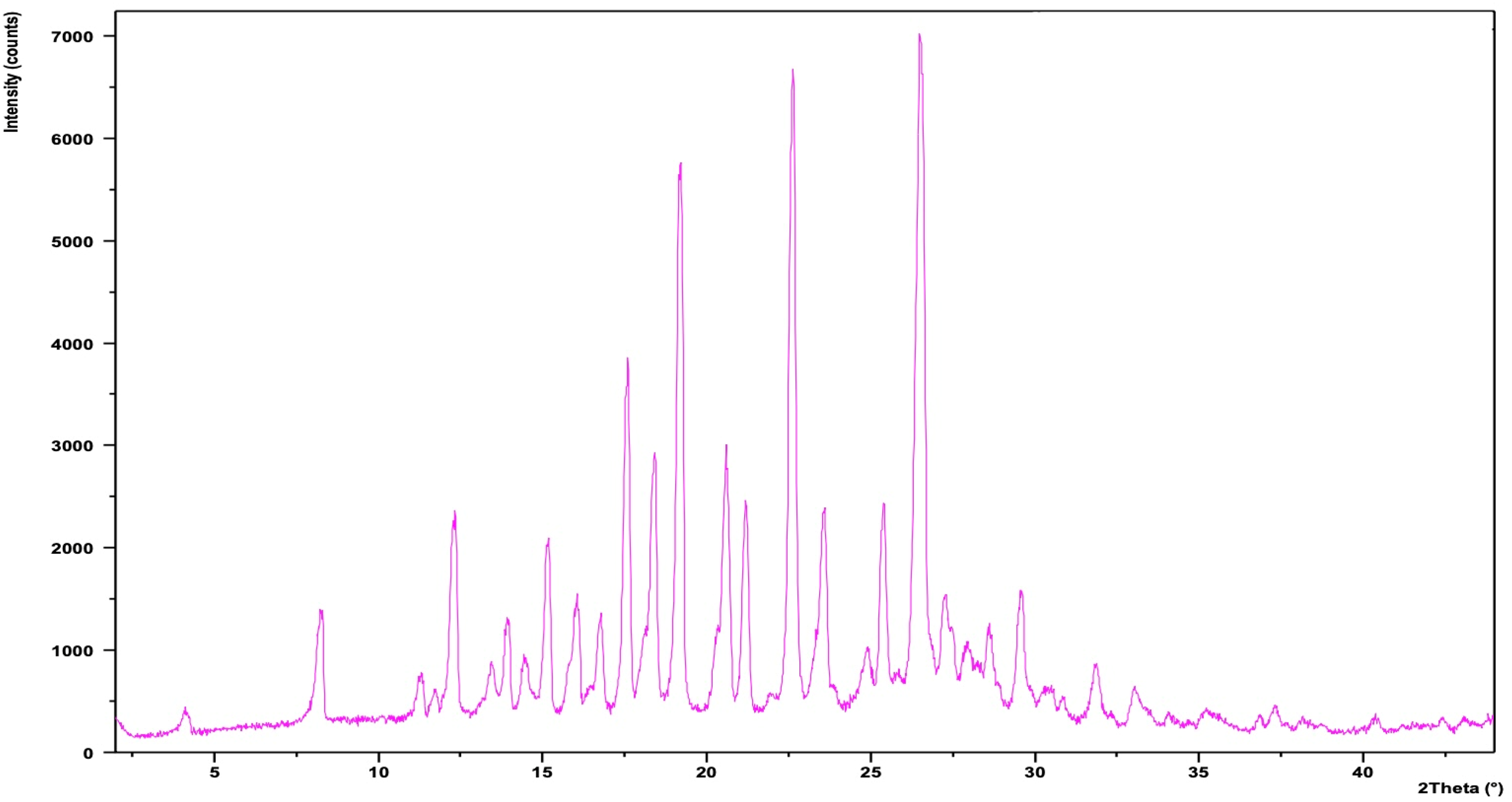
XRD spectra of NBI#1.
2-(4-Nitrophenyl)-1H-benzimidazole (NBI) has also been obtained by cyclisation of N-(4-Nitrobenzyl) benzene-1,2-diamine (NBD) as seen in Figure 11. Briefly, The Schiff-base intermediate NBD was dissolved in hot ethanol and was refluxed for 2 h. After that, the solvent was evaporated, and the crude product was recrystallized. The yields, melting points and UV maxima are indicated in Table 3. The compounds were identical. Elemental analysis for NBI (C13H9N3O2): calculated: C: 63.12%, H:4.88%, N:18.05%; found: C: 63.05%, H:4.76%, N:18.12%.

Preparation of 2-(4-Nitrophenyl)-1H-benzimidazole (NBI#2).
Yields and chemical properties of different routes to form NBI.

Preparation of metal complexes of 2-(4-Nitrophenyl)-1H-benzimidazole (NBI)
Briefly, 2.39 g (0.01 mol) NBI was dissolved in 50w% acetone: water mixture, then the appropriate metal salts were added slowly as seen in Figure 12. The mixture was stirred at room temperature for 2 min, and the reaction mixture was boiled for under reflux for 10 min, to obtain the crude products. Crude product was filtered, recrystallized from acetone to obtain pure NBI complexes. The yields, melting points and UV maxima are indicated in Table 4.

Preparation of NBI metal complexes.
Yields and chemical properties of NBI complexes.

Cupric 2-(4-Nitrophenyl)-1H-benzimidazole (Cu(II)NBI) complex was formed from the cupric salt copper(II) nitrate trihydrate (Cu(NO3)2·3H2O) and NBI. X-ray diffraction gives no pattern as shown in Figure 13; thus, the compound is either amorphous or microcrystalline but suggested that bonding was achieved by the formation of a coordination complex between the cupric ion and the NBD molecule. The relevant peaks of the starting material Cu(NO3)2·3H2O (2θ = 10.8° 14.6° 18.3° 22.0° 25.1°) are missing from the spectra. Elemental analysis for Cu(C13H9N3O2)2: calculated: C: 57.61%, H:3.34%, N:15.50%, Cu:11.72%; found: C: 57.48%, H:3.28%, N:15.21%, Cu:11.23%.
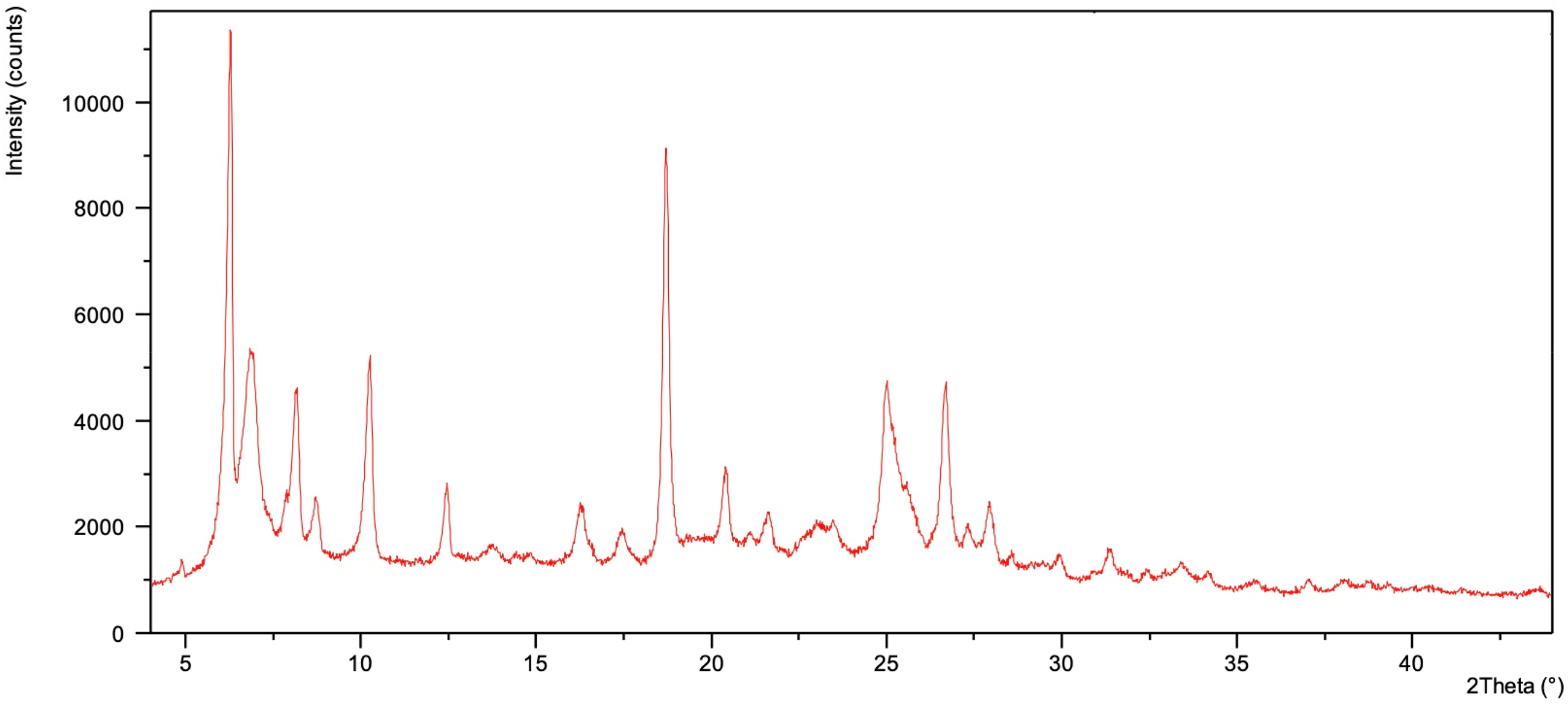
XRD spectra of Cu(II)NBI.
Ferrous 2-(4-Nitrophenyl)-1H-benzimidazole (Fe(II)NBI) complex was formed starting from iron(II) nitrate dihydrate (Fe(NO3)2·6H2O) and NBD. X-ray diffraction gives no specific pattern as shown in Figure 14. The relevant peaks of the starting material are missing from the spectra. Elemental analysis for Fe(C13H9N3O2)2: calculated: C: 58.45%, H:3.40%, N:15.73%, Fe:10.45%; found: C: 58.18%, H:3.31%, N:15.32%, Fe:10.05%.

XRD spectra of Fe(II)NBI.
Zinc 2-(4-Nitrophenyl)-1H-benzimidazole (Zn(II)NBI) was formed from Zn(NO3)3·2H2O and NBD yielding dark yellow crystalline powder. X-ray diffraction gives no pattern as shown in Figure 15; thus, the compound is either amorphous or microcrystalline. Elemental analysis for Zn(C13H9N3O2)2: calculated: C: 57.42%, H:3.34%, N:15.45%, Zn:12.02%; found: C: 57.21%, H:3.31%, N:15.35%, Zn:12.05%.

XRD spectra of Zn(II)NBI.
Manganese 2-(4-Nitrophenyl)-1H-benzimidazole (Mn(II)NBI) was formed with stoichiometrically (2:1) from NBI and Mn(NO3)2·4H2O yielding black crystalline powder. X-ray diffraction gives no pattern as shown in Figure 16; thus, the compound is either amorphous or microcrystalline. Elemental analysis for Mn(C13H9N3O2)2: calculated: C: 58.54%, H:3.40%, N:15.76%, Mn:10.30%; found: C: 58.21%, H:3.32%, N:15.45%, Mn:10.02%.

XRD spectra of Mn(II)NBI.
Nickel 2-(4-Nitrophenyl)-1H-benzimidazole (Ni(II)NBI) was formed from NBI with stoichiometrically (2:1) nickel(II) nitrate nonahydrate (Ni(NO3)2·6H2O). X-ray diffraction gives no pattern as shown in Figure 17. The main peaks of starting material Ni(NO3)2·6H2O are missing from the spectra. Elemental analysis for Ni(C13H9N3O2)2: calculated: C: 58.14%, H:3.38%, N:15.65%, Ni:10.93%; found: C: 58.04%, H:3.32%, N:15.45%, Ni:10.19%.

XRD spectra of Ni(II)NBD.
In Figure 18, starting material NBI#1 (blue at the bottom) and the Ni(II)NBI (green at the top) complex are shown together. By comparing the spectra, a new peak appears at 2θ = 15.1°, which is not present in the starting material NBI#1, so it has been identified as a peak characteristic to the formation of Ni(II)NBI complex.
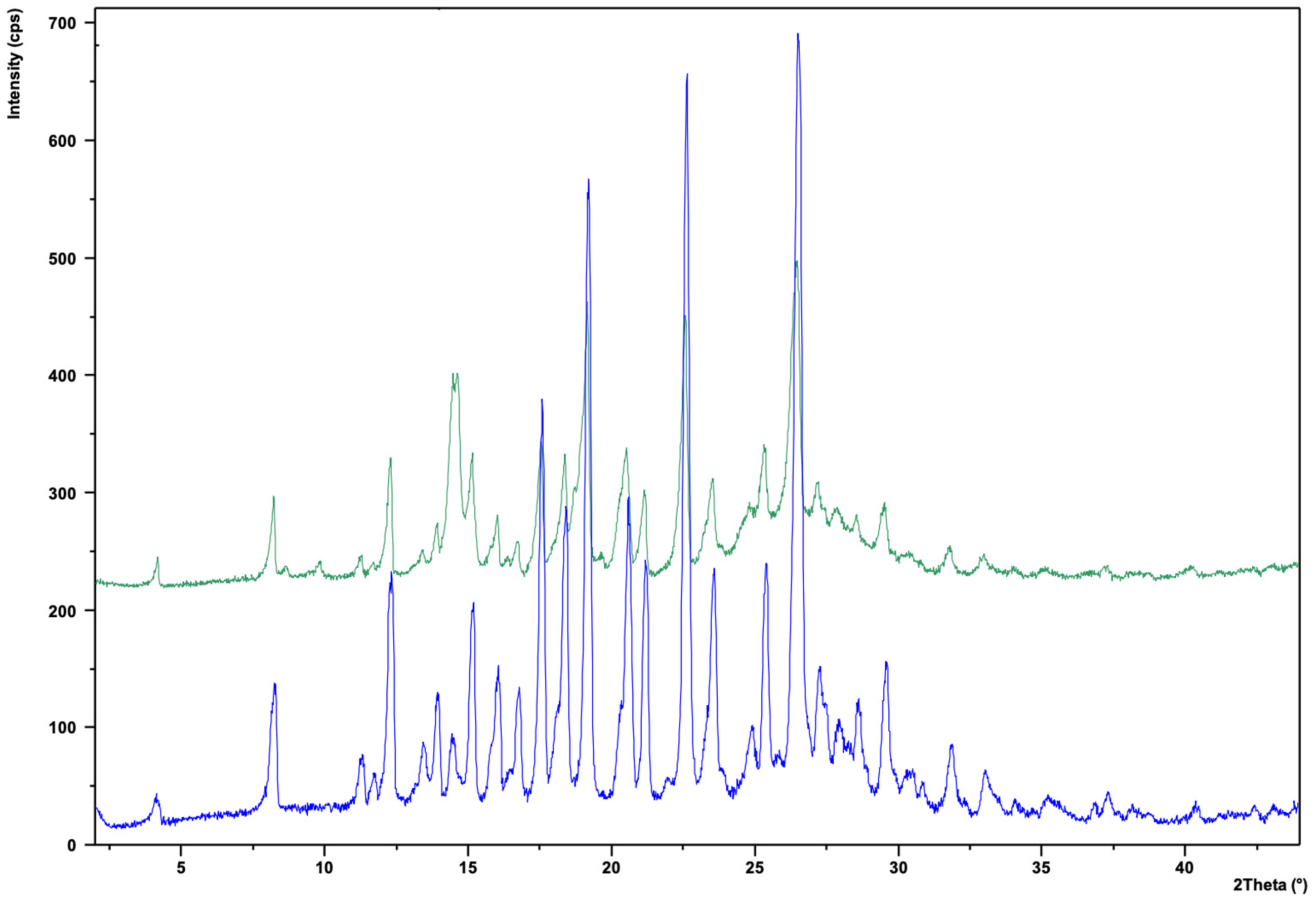
Comparison of XRD spectra of Ni(II)NBI and NBI.
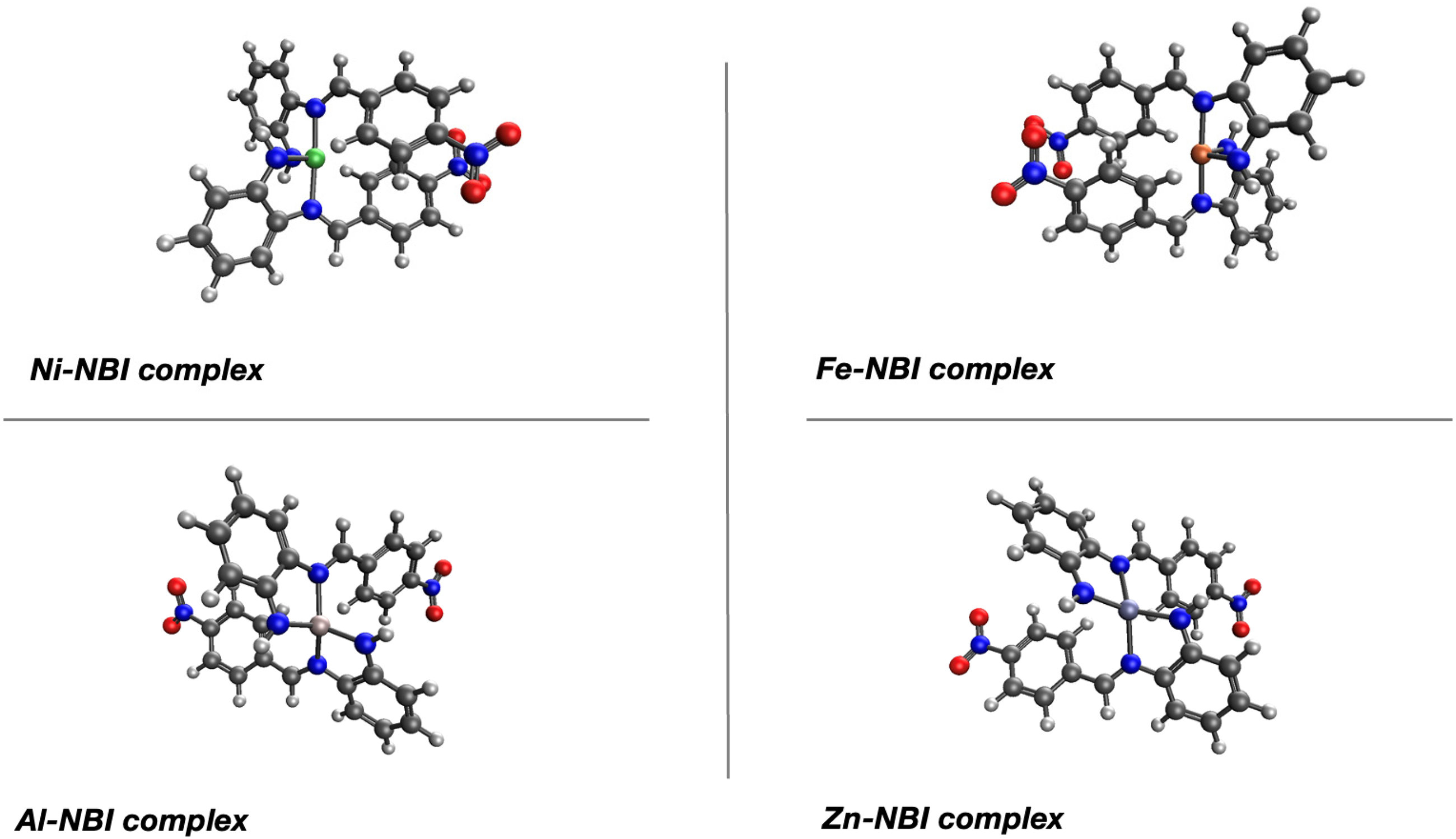
The Schiff base N-(4-nitrobenzyl) benzene-1,2-diamine (NBD) and its 2-(4-Nitrophenyl)-1H-benzimidazole derivative (NBI) are important in various fields. The formation of the Schiff base N-(4-nitrobenzyl) benzene-1,2-diamine represents a key step in the synthesis of important heterocyclic compounds. One significant Schiff base is N-(4-nitrobenzyl) benzene-1,2-diamine, which serves as a precursor in the synthesis of various heterocyclic compounds, including 2-(4-Nitrophenyl)-1H-benzimidazole. Metal complexes of 2-(4-Nitrophenyl)-1H-benzimidazole (NBI) exhibit high stability, and potential in developing new materials and therapeutic agents. To provide additional data, computational studies were performed on the formation of 2-(4-Nitrophenyl)-1H-benzimidazole derivatives shown in Figure 19. at the ωb97X-D/def2tzvp level of theory.

Selected benzimidazole derivatives for computational studies.
By taking the sum of electronic and thermal free energies the reaction free enthalpy (ΔG) was computed for intermediate steps A-D. As expected, neither of the intermediate steps seem to contribute to the change in yields, where structures (1) represents 2-phenyl-1H-1,3-benzodiazole, (2) represents 2-(4-Chlorophenyl)benzimidazole, (3) represents, the 4-(1H-Benzimidazol-2-yl)phenol and (4) represents 2-(4-Nitrophenyl)-1H-benzimidazole as shown in Table 5.
Computed ΔG values for 1–4 all data are in kcal/mol.

Although a trend in ΔG values can be observed, the resulting effect is small and does not align with the yields previously reported for

Selected resonance forms of structures
The cyclization of the Schiff base N-(4-nitrobenzyl) benzene-1,2-diamine to form 2-(4-nitrophenyl)-1H-benzimidazole is a crucial step in the synthesis of this important benzimidazole derivative. The process involves heating and acid catalysis to promote the formation of the benzimidazole ring. The nitro group is an electron-withdrawing substituent, which can stabilize the benzimidazole ring and enhance the acidity of the imine proton. This stabilization facilitates the formation of stable metal complexes and influences the electronic environment of the metal centre. The electron-withdrawing effect of the nitro group makes NBI an excellent ligand for coordinating with metal ions, resulting in complexes with unique electronic and structural characteristics. The increased electrophility on this atom might explain the increased reactivity 4-nitrobenzaldehyde in this reaction, as the formation of the necessary intermediates is not as thermodynamically restricted. The authors leave the possibility, that substituent effects might yield to energetically favoured transition states, which would decrease the activation enthalpy for each step, which should be examined with more sophisticated theoretical methods in the future. Otherwise, based on the computed ΔG values, the authors suggest that all steps except for E correspond to equilibrium reactions, which are ultimately shifted by the highly exergonic formation of the benzimidazole product as seen in Figure 21.

Reaction equilibria in NBD-NBI system.
According to our current understanding from the experimental data, the hereby complexes feature mostly η2 complexes of benzimidazole with a 2:1 stochiometric ratio with the corresponding transitional metal. These assumptions are strengthened by examining their orbital-weighed Fukui-function corresponding to the electron density isosurface. High values of Δfw (dual descriptor) correspond to a site susceptible for a nucleophilic attack. The Δfw maxima for Ni(OH)2, (NBI)Ni(OH)2 and (NBI)2Ni(OH)2 have been computed to examine the electrophilicity of the metal centre. High values of Δfw were identified on Ni(OH)2, (NBI)Ni(OH)2 near the metal centre, illustrating the ability to bind ligands further. This effect cannot be observed on (NBI)2Ni(OH)2, thus the ligation of further NBI units is hindered as seen in Figures 22 to 27.

Maxima of Δfw (red) for (NBI)Ni(OH)2.
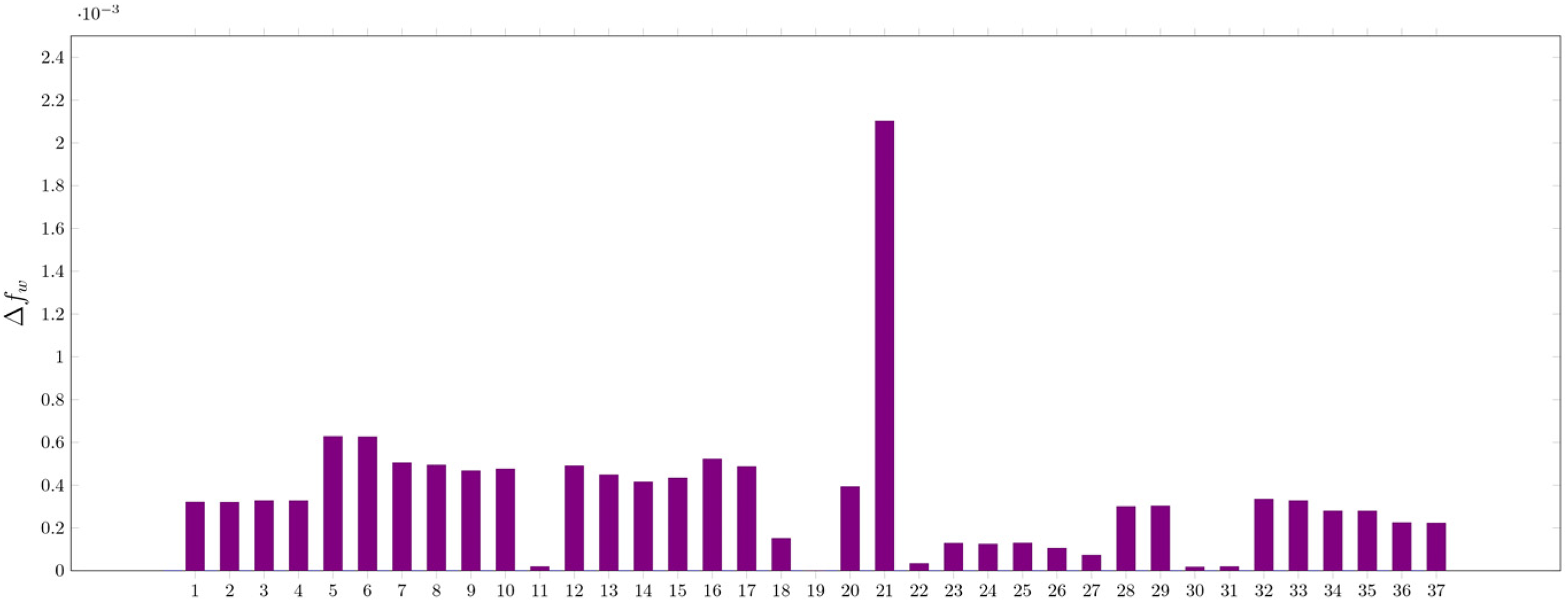
Values of Δfw corresponding to maxima for (NBI)Ni(OH)2.
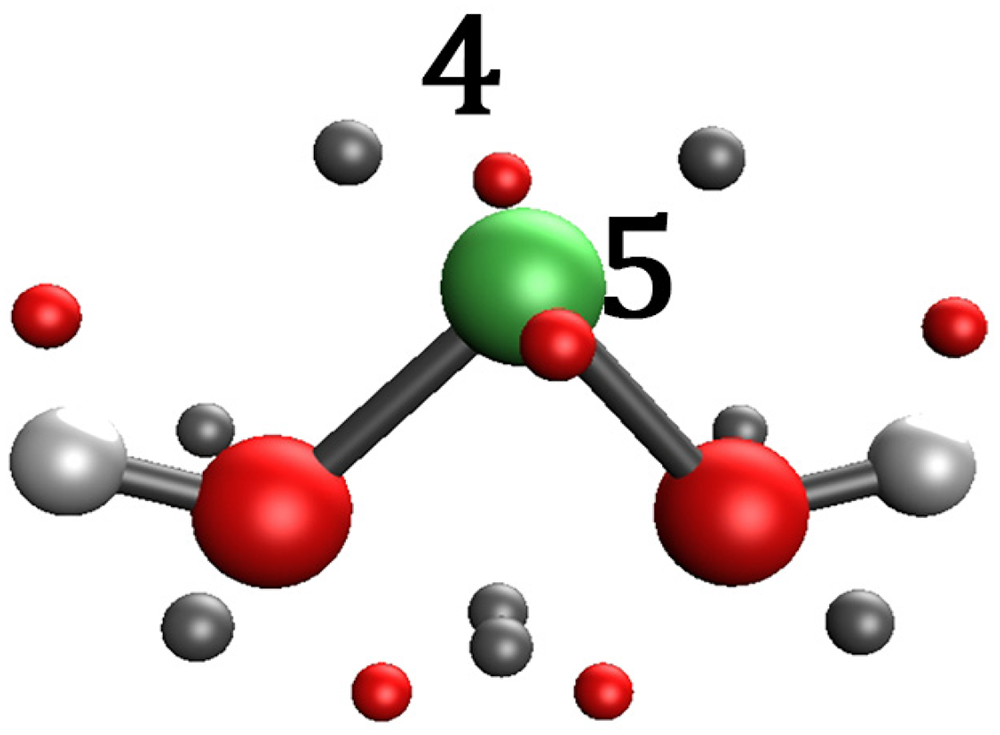
Maxima of Δfw (red) for Ni(OH)2.
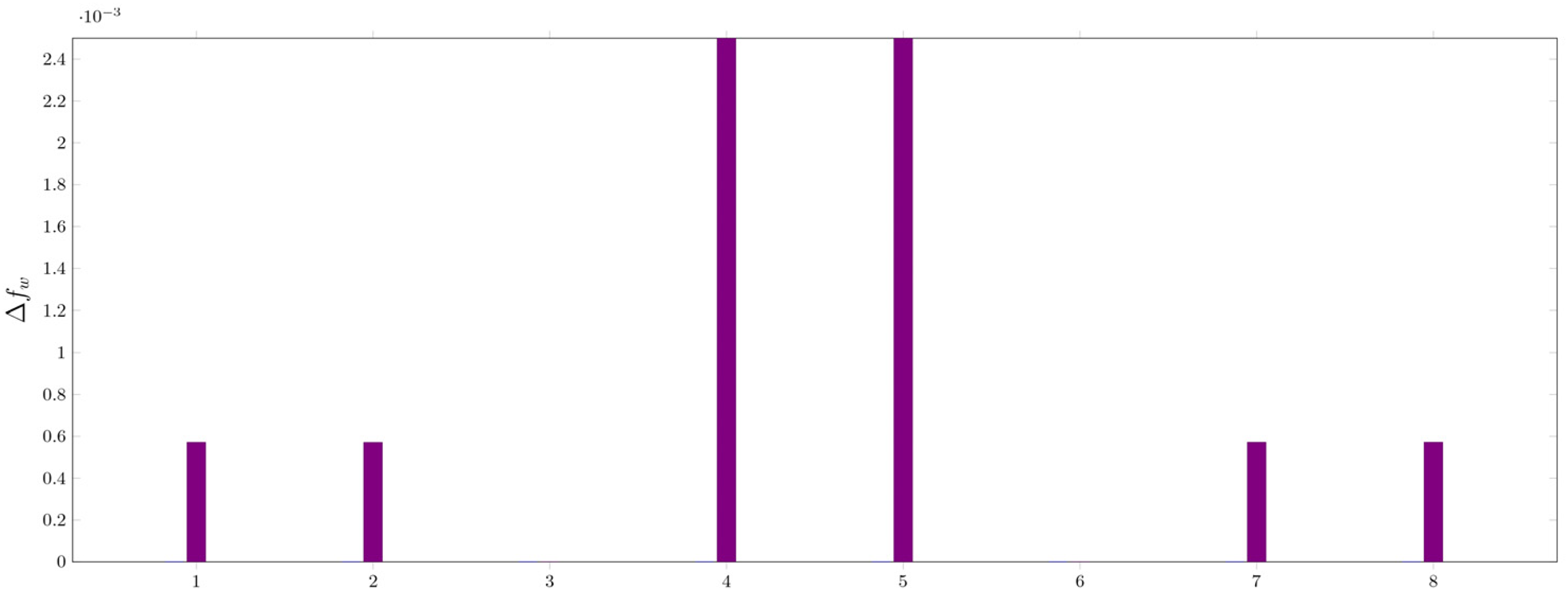
Values of Δfw corresponding to maxima for Ni(OH)2.
Maxima of Δfw (red) for (NBI)2Ni(OH)2.

Values of Δfw corresponding to maxima for (NBI)2Ni(OH)2.
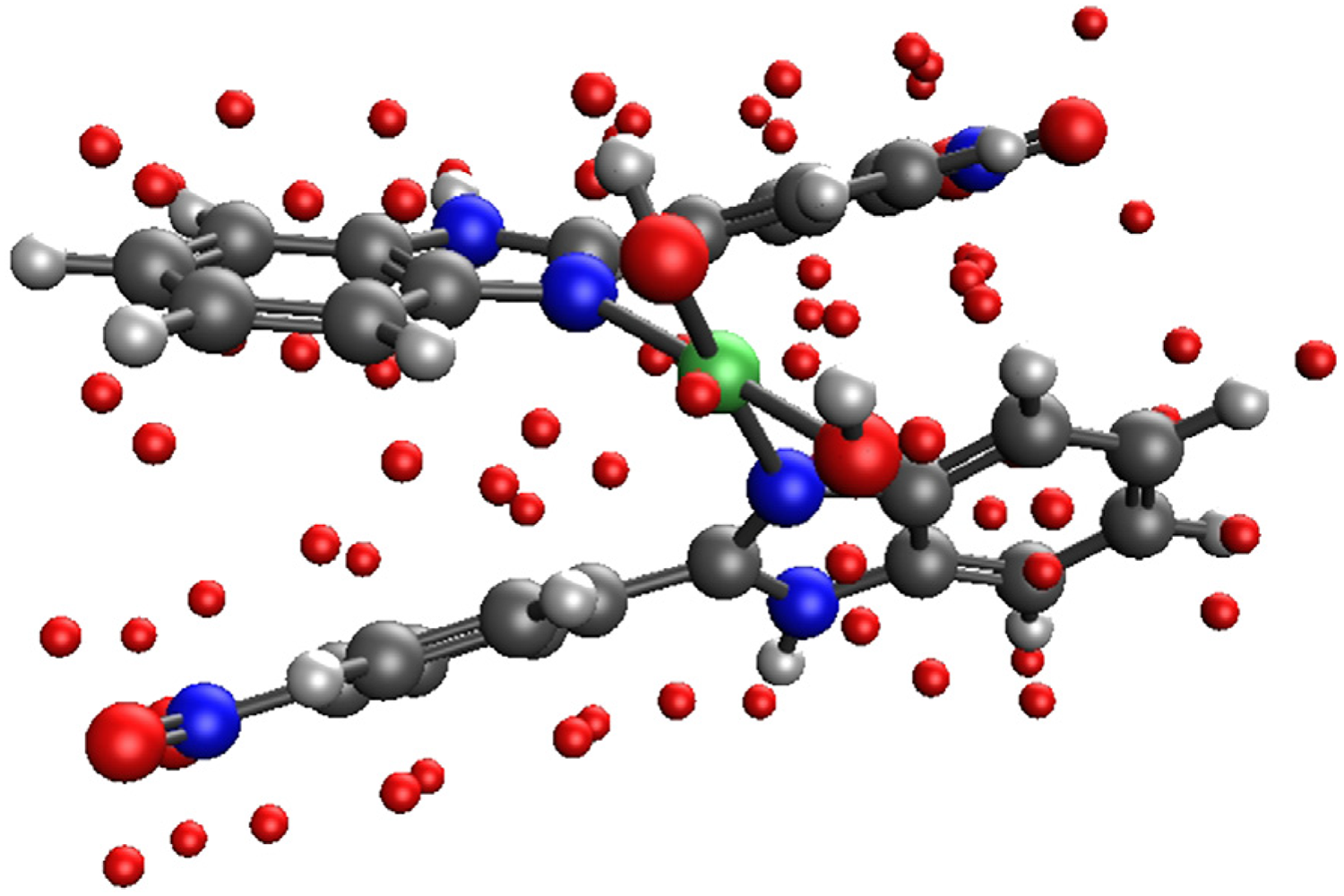
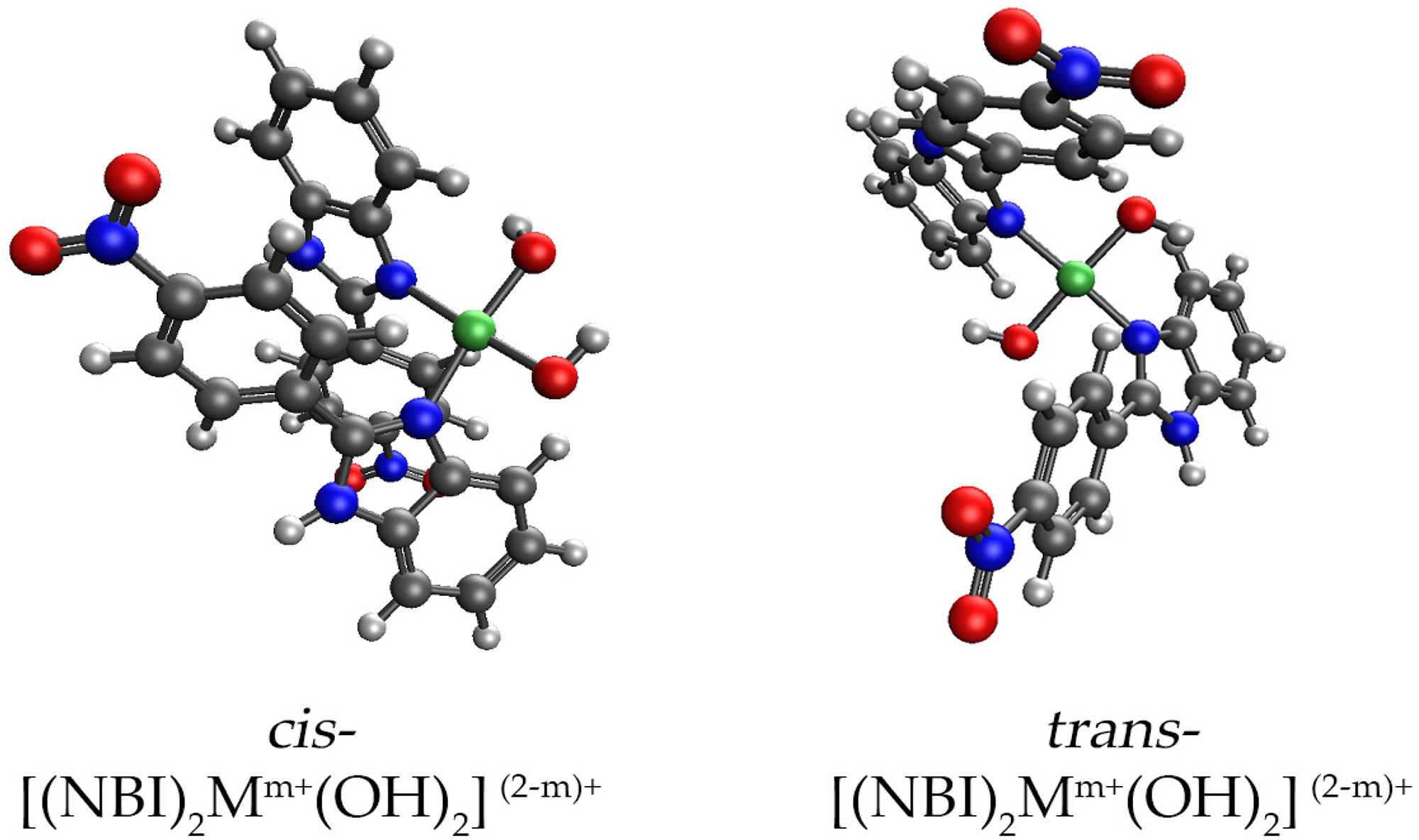
Optimizations were performed to the corresponding hydroxide salt, which makes cis and trans isomers considerably easier to compute. Coordinatively unsaturated complexes were optimized on the TPSSh/def2-SVP level of theory with pseudopotentials on the heavier metal atoms (Table 6.). The relative stability of the corresponding isomers is between 0.1–6 kcal/mol which is reasonably low, suggesting that the resulting product is the mixture of the two isomers based on computational results. In each case, the trans isomer was more stable. Additional experimental work is needed to examine the stereochemical properties of the resulting products as shown in Figure 28. Figure 29. shows the possible structures of NBI complexes.
General scheme of computed complexes at the TPSSh/def2-SVP level of theory.
Possible structures of NBI complexes.
Computed ΔE values corresponding to isomerization selected complexes all data are in kcal/mol.

Conclusions
This study enhances the understanding of Schiff bases and benzimidazole derivatives by focusing on the synthesis, characterization, and applications of N-(4-nitrobenzyl)benzene-1,2-diamine (NBD) and its derivative, 2-(4-nitrophenyl)-1H-benzimidazole (NBI). It emphasizes the role of the nitro group, a potent electron-withdrawing substituent, in improving structural stability, electronic properties, and reactivity of these synthesized compounds. By integrating experimental and computational approaches, the research offers valuable insights into the mechanisms behind Schiff base formation, cyclization into benzimidazole derivatives, and metal complexation processes. The mono-Schiff base NBD was synthesized by condensing o-phenylenediamine with 4-nitrobenzaldehyde, yielding excellent reproducibility and efficiency. This compound serves as a critical precursor for the cyclization process that produces NBI. The para-positioned nitro group on the phenyl ring profoundly influences the ligand's electronic environment, enabling the formation of stable metal complexes. These results demonstrate the importance of substituent effects in modulating the chemical and physical properties of heterocyclic compounds, providing pathways for tailoring these materials for specific purposes. Characterization of NBD, NBI, and their metal complexes was achieved using advanced analytical techniques. Fourier-transform infrared spectroscopy (FTIR) and X-ray diffraction (XRD) confirmed the successful formation of Schiff base intermediates and their transformation into benzimidazole derivatives. Metal complexation was effective with Cu(II), Zn(II), Fe(II), and Ni(II), resulting in stable complexes with distinct structural properties. The role of the nitro group in enhancing the acidity of the imine proton and stabilizing the metal-ligand bond was clear in both spectral data and computational analyses.
Computational studies complemented the experimental findings, offering insights into reaction energetics and the nitro group's impact on stability and reactivity. Free energy calculations (ΔG) for intermediate steps revealed that the nitro group reduces thermodynamic barriers, promoting the formation of stable intermediates and final products. Resonance structures of nitro-substituted benzimidazole derivatives demonstrated the enhanced electron-withdrawing effect, which stabilizes both the ligand and its metal complexes. These computational results align with experimental observations, reinforcing the importance of theoretical methods in studying complex chemical systems. The synthesized metal complexes exhibited significant potential, particularly in catalysis and medicinal chemistry. Catalytically, Cu(II)- and Zn(II)-based complexes showed remarkable ability to accelerate reaction rates and enhance selectivity in organic transformations, such as oxidative and coupling reactions. Medicinally, Cu(II)-NBI and Zn(II)-NBI complexes displayed potent antimicrobial and anticancer activities, effectively targeting pathogenic microorganisms and cancer cell lines. The nitro group's influence on the electronic environment of the metal centre appears crucial for enhancing biological activity, underscoring its potential utility in drug design and therapeutic applications. From a materials science perspective, the metal complexes demonstrated excellent thermal stability and electronic properties, indicating potential applications in advanced materials. The electron-withdrawing nature of the 4-nitrophenyl group contributes to developing materials with superior optical and electronic characteristics, offering innovative possibilities for sensors, light-emitting devices, and environmental monitoring technologies. This study establishes a foundation for future research into the design of Schiff base ligands and their derivatives. By systematically examining the effects of specific substituents, such as the nitro group, the research outlines a strategy for customizing the properties of Schiff bases and benzimidazole derivatives for targeted applications. These insights could inspire investigations into other electron-withdrawing or electron-donating groups, broadening the scope of functionalized heterocyclic compounds. Despite its substantial contributions, this research acknowledges certain limitations that present opportunities for further exploration. For instance, evaluating the environmental and economic aspects of the synthetic methods, such as solvent usage and reaction scalability, is crucial for enhancing sustainability and industrial feasibility. Additionally, further studies on the biological activity of these complexes, including in vivo investigations, would provide a more comprehensive understanding of their therapeutic potential. Expanding the scope to include complexes with other transition metals and exploring applications in emerging areas like green catalysis and energy storage could further amplify the impact of this work. The novelty of this research lies in synthesizing and characterizing metal complexes derived from NBI and its mono-Schiff base precursor, NBD. This study focuses on the unique structural and electronic properties imparted by the nitro group, exploring its influence on stability, reactivity, and applications. Unlike previous studies that broadly examine Schiff bases and their derivatives, this work systematically investigates the synergistic effects of the benzimidazole core and nitro substitution. The electron-withdrawing properties of the nitro group enhance the acidity of the imine proton, stabilize metal centres, and modulate the electronic environment, resulting in improved catalytic and biological activity. The integration of experimental and computational methods addresses key challenges in Schiff base synthesis and benzimidazole-based coordination chemistry. This research expands the subject area by introducing a synthesis and application-focused study of nitro-substituted benzimidazole derivatives. It combines experimental findings with computational analyses, providing a theoretical framework for understanding how substituent effects drive stability and reactivity in chemical systems. By exploring multifunctional applications across catalysis, medicinal chemistry, and materials science, the study highlights the versatility of these compounds. Furthermore, it offers a replicable methodology for tailoring Schiff bases and benzimidazole derivatives, inspiring future studies involving other substituents or core structures. In conclusion, this research advances the understanding of nitro-substituted Schiff base and benzimidazole derivatives and establishes a framework for designing metal complexes with tailored properties. By addressing how substituents like the nitro group influence stability, reactivity, and functionality, the study opens avenues for applications in drug development, catalysis, and materials science. As research progresses, these findings will serve as a valuable reference for creating novel compounds to address diverse scientific and industrial challenges.
Footnotes
Acknowledgements
The authors extend their gratitude to the Department of Analytical and Inorganic Chemistry at the Budapest University of Technology and Economics (Budapest, Hungary) for providing access to CPU time on the ORIGIN cluster, facilitated by the support of its operational staff.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable
Trial registration
Not applicable
