Abstract
Objective
The infection of the endograft after thoracic endovascular aortic repair (TEVAR) is a rare but potentially life-threatening complication. Unfortunately, clinical reports, large case-series or randomized studies are scarce or not available yet, and the optimal management of this complication is under debate. The aim of this review is collecting and analysing data on current practice and outcomes for the treatment of infected endografts after TEVAR.
Methods
A search on PubMed/MEDLINE and EMBASE for studies reporting endograft infections after TEVAR was conducted up to January 2023. Data on study design, patients demographic, infection (time from TEVAR to infection, vascular imaging, bacterial samples), clinical strategies, outcome and follow-up were extracted and analysed. The outcome after surgical and conservative treatment was compared.
Results
Thirty-six articles (5 case-series; 31 case reports) were deemed eligible for this review. A total of 57 patients (mean age: 62.5 ± 13.9 years) with endograft infections after TEVAR were included. Fever and pain were the two major clinical presentations. Mean time from TEVAR to diagnosis was 19.7 ± 36.4 months. The top four isolated microorganism were Staphylococcus aureus (25%), Candida albicans (15.6%), Streptococcus (12.5%) and Pseudomonas (7.8%). Patients were treated conservatively (36.8%) or surgically (63.2%). Surgical treatments were extra-anatomic aortic bypass (50%), in-situ aortic replacement (38.9%) and graft preservation (11.1%). Overall hospital mortality was 33.3% (30-day mortality: 26.4%). The mortality was 52.4% after conservative treatment and 22.2% after surgery (p = 0.0397). Among patients who underwent surgery, the mortality was 28.6% after in-situ aortic replacement and 22.2% after extra-anatomic aortic bypass (p = 0.5387). Major in-hospital causes of death after surgery were multiple organ failure and respiratory failure (6/8 patients).
Conclusion
Endograft infection after TEVAR is rare but it carries a high risk of mortality. In selected patients, the surgical treatment seems to be safer, with a lower mortality rate when compared to alternative conservative options.
Keywords
Introduction
The thoracic endovascular aortic repair (TEVAR) represents the treatment of choice for patients suffering from thoracic aorta diseases such as aneurysms, type-B dissections, intramural hematomas (IMH) or penetrating atherosclerotic ulcers, and it carries lower morbidity and mortality rate with shorter hospital stay when compared to standard open surgery. 1 The most common complications after TEVAR are endoleaks, stroke, stent graft-induced aortic new entries, endograft migration and aortic rupture. 2 The infection of the endograft after TEVAR is rare but it is associated with a high mortality rate after both conservative and surgical treatments. An endograft infection was at first reported in 1993 by Chalmers and colleagues who described an infected pseudoaneurysm at the site of an iliac stent. 3 Prosthetic vascular graft infection following open or endovascular aortic procedures occurs in 1–6% of patients and is associated with a high mortality rate, reaching the 75% in the worst scenario.4,5 A meta-analysis by Argyriou et al. reported an incidence of endograft infections after endovascular aneurysm repair (EVAR) of 0.6%, and a study by Li et al. showed that the incidence of infected endograft after EVAR was significantly higher compared to the number of infected TEVAR (87% vs 13%), but the TEVAR group had a significantly lower survival rate compared to the EVAR group (27% vs 58%; p < 0.001).6,7
The current evidence on the management of infected TEVAR is mainly based on case reports or small case-series and, therefore, the optimal treatment is still under debate and undertaken on a case-by-case basis without consensus guidelines. Little is known about infected TEVAR, including the real incidence, the clinical presentation, the optimal therapeutic options and the clinical outcomes after either medical treatments or high-risk surgical procedures. The aim of this systematic review is to investigate and compare data and outcomes of different therapeutic options for infected endografts after TEVAR.
Methods
Ethical considerations
This systematic review is exempt from the ethics approval as we have collected and summarized data taken from published studies in which informed consent was obtained by the study investigator.
Search strategy
This systematic review was performed and reported in line with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement. 8 A search of MEDLINE and EMBASE was carried out to find all cases of thoracic endograft infections after TEVAR published in the available literature with time point set to end of January 2023 using medical subject headings and text words supplemented by scanning the bibliographies of the reviewed articles. We used ‘management/treatment of infected TEVAR’ as the initial retrieval. The key search words included ‘TEVAR’, ‘thoracic endovascular aortic repair ‘, ‘endovascular aneurysm repair’, ‘endovascular stent graft’, ‘endograft’, ‘stent-graft’, ‘aortic stent-graft’ and ‘infection’. Two co-authors reviewed and selected relevant articles for inclusion. Differences were resolved in consensus discussions. All titles, abstracts and full papers were sequentially reviewed against predefined inclusion criteria prior to attempt data extraction. Additional hand-searching was undertaken.
Inclusion criteria
The predefined inclusion criteria were the presence of a report of one or more patients affected by thoracic endograft infection after TEVAR, with details on treatment management and outcome. Only papers where we could extract data about the number of cases, the evidence and the details of the endografts infection, the treatment modalities and the outcomes were included. Language was limited to articles written in English for practical purposes.
Exclusion criteria
Predefined exclusion criteria in this research protocol determined whether studies were eligible for full-text analysis or not. Papers only focusing on clinical diagnosis, EVAR or peripheral artery disease, and without any description of patients with infected thoracic endografts were immediately excluded. Articles reporting on endovascular repairs of a primary aortic infection, such as mycotic aneurysms, infectious aortitis, primary aortoesophageal fistulae (AEF) or aortobronchial fistulae were also excluded. When data or essential information was unavailable, the study was excluded from the analysis. In order to avoid duplicated reporting, when institutions published several studies from the same registry we chose the latest article with more patients. Correspondence, expert opinions and review papers were not included as well.
Data extraction
We collected data on study type (case-series or case report), age, gender, number of patients, clinical presentation, time lapse from TEVAR implantation to infection, vascular imaging, bacteriological results, surgical procedures or medical treatments, postoperative course of intravenous antibiotic, hospital mortality, cause of death and follow-up time.
Statistical analysis
Data collected were organized on an Apple Numbers spreadsheet (version 6.6.2). Continuous variables were expressed as mean values ± standard deviation and categorical variables as numbers and percentages. The overall comparison of different therapeutic strategies was performed using the Fisher's exact test. Statistical analysis was performed using SPSS version 18.0 (IBM Corp., Chicago, IL, USA).
Results
The literature search identified a total of 496 studies and 10 additional articles were identified by manually retrieving the list of references (Figure 1). After the screening, 36 studies focusing on infected endografts after TEVAR were included in the final analysis (5 case-series and 31 case reports, spanning a period of time ranging from 2005 to 2023) (Table 1).9–44

Flow diagram illustrating the identification, the selection and the exclusion of articles used in this review.
Case reports and case-series reporting infected endografts after TEVAR.
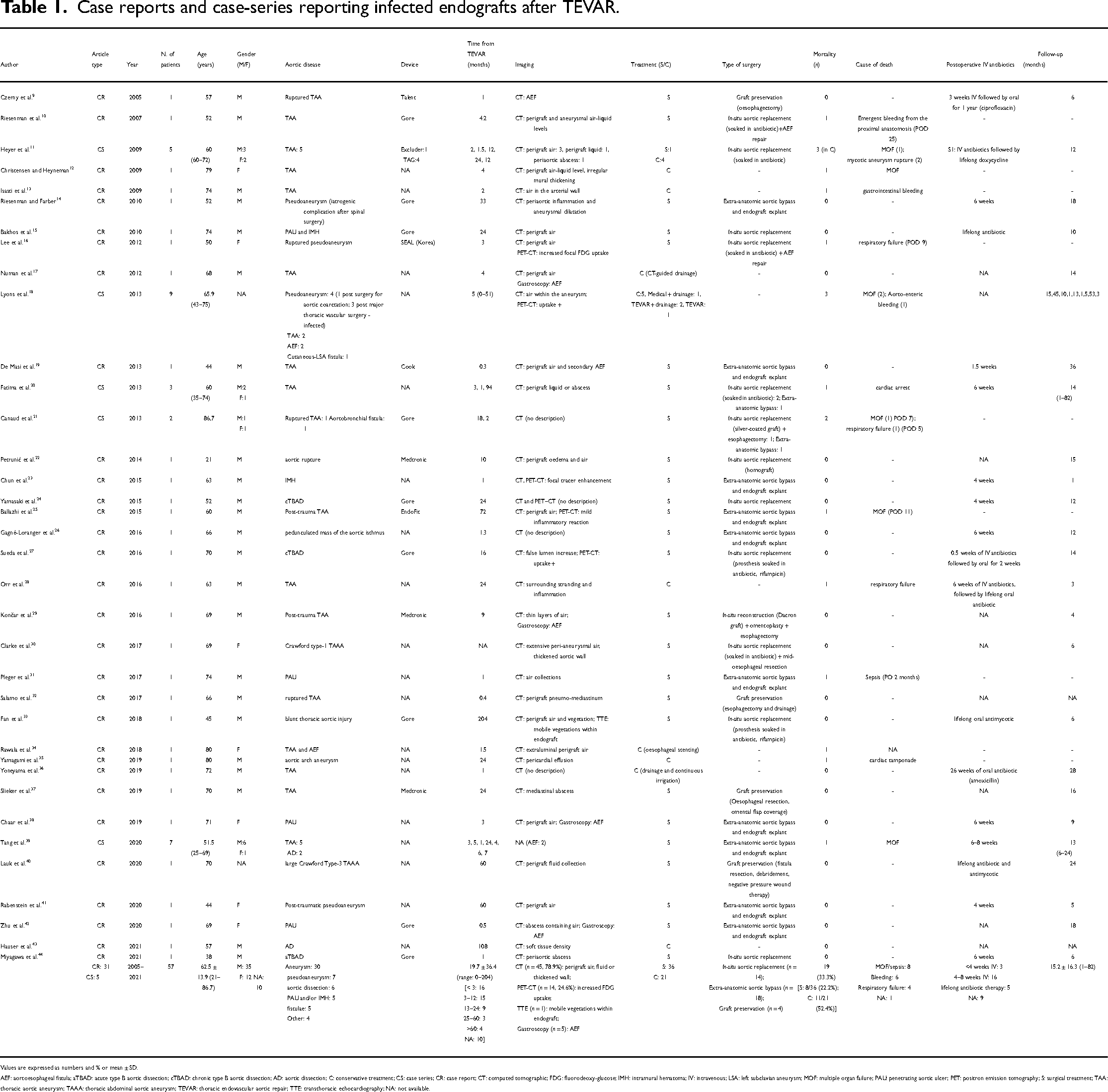
Values are expressed as numbers and % or mean ± SD.
AEF: aortoesophageal fistula; aTBAD: acute type B aortic dissection; cTBAD: chronic type B aortic dissection; AD: aortic dissection; C: conservative treatment; CS: case series; CR: case report; CT: computed tomographic; FDG: fluorodeoxy-glucose; IMH: intramural hematoma; IV: intravenous; LSA: left subclavian aneurysm; MOF: multiple organ failure; PAU: penetrating aortic ulcer; PET: positron emission tomography; S: surgical treatment; TAA: thoracic aortic aneurysm; TAAA: thoracic abdominal aortic aneurysm; TEVAR: thoracic endovascular aortic repair; TTE: transthoracic echocardiography; NA: not available.
Demographic data
A total of 57 patients were identified, with an average age of 62.5 ± 13.9 years (ranging from 21 to 86.7 years). In this group, 35 patients were male, 12 were female and the gender of 10 patients was not reported. Indications for primarily TEVAR included aneurysms (n = 30), pseudoaneurysms (n = 7), aortic dissections (n = 6), penetrating ulcers and/or IMH (n = 5) and fistulae (n = 5) (Table 1). Fever (n = 28, 77.8%) and pain (n = 17, 47.2%) were the two major documented clinical presentations. Other symptoms included haemoptysis, haematemesis or melena, fatigue, loss of weight, night sweats, nausea and dyspnoea (Table 2). The overall hospital mortality after conservative or surgical treatment was 33.3% (n = 19), with a 30-day mortality of 26.4% (n = 15). Multiple organ failure (MOF) and sepsis (n = 8) were the major cause of death. The other two causes of death were bleeding (n = 6) and respiratory failure (n = 4). Among patients who survived (n = 38), 16 (42.1%) received postoperative intravenous antibiotics for 4–8 weeks based on the results of bacterial cultures and sensitivity, 3 (7.9%) for less than 4 weeks and 5 (13.2%) patients underwent a lifelong antibiotic treatment. In nine cases, the use of intravenous antibiotic treatment was not documented. Mean follow-up time was 15.2 ± 16.3 months (range: 1–82 months) for 41 patients, and, during this time, patients did not undergo further surgical procedures (Table 1).
Symptoms and isolated microorganisms.

Values are expressed as numbers and %.
NA: not available.
Diagnostic modalities
The time from TEVAR to diagnosis ranged from 0 to 204 months (mean 19.7 ± 36.4 months). Sixteen patients got infected during the first 3 months, 15 patients between 3 and 12 months, 9 patients between 13 and 24 months, 3 patients between 25 and 60 months and 4 patients after 60 months (NA = 10).
Imaging modalities included computed tomography (CT) in 78.9% patients (n = 45), followed by positron emission tomography (PET) scan in 24.6% (n = 14). The main findings on CT included presence of air or fluid collections around the stentgraft, or a thickened aortic wall. In one case, the transthoracic echocardiography was used to identify two mobile vegetations in the thoracic aortic endograft. Gastroscopy was used to diagnose five AEF.
In five cases, the details of blood cultures were not reported. And there was no bacteria growth in four cases. A total of 64 bacteria were isolated from blood, endografts, aortic wall, fluid collection or tissue. Staphylococcus aureus was isolated in 16 cases (25%), Candida albicans in 10 (15.6%), Streptococcus in 8 (12.5%) and Pseudomonas in 5 (7.8%) (Table 2). In 22 patients, the infection was polymicrobic.
Treatment modalities
Treatment modalities were reported in all cases. In 21 cases (36.8%), a conservative medical treatment was undertaken using a long-lasting antibiotic therapy with or without continuous percutaneous drainage and irrigation of the infected site, placed percutaneously under echographic or CT guidance (five cases). Thirty-six patients (63.2%) underwent a surgical procedure: 14 (38.9%) underwent in-situ aortic replacement, 18 (50%) were treated with extra-anatomic aortic bypass and 4 (11.1%) received a graft preservation treatment (oesophagectomy with fistula resection and additional surgical drainage). In-situ aortic replacement was performed using a cryopreserved allograft (homograft) in one case, prosthetic grafts soaked in antibiotic in nine cases and non-soaked prosthetic grafts in four cases (Table 1). There were no available data concerning whether the prosthetic grafts were rifampicin-soaked or silver-coated.
The mortality was 52.4% (n = 11/21) after conservative treatment and 22.2% (n = 8/36) after surgical treatment (p = 0.0397) (Table 3). Among surgically treated patients, the mortality was 28.6% (n = 4/14) after in-situ aortic replacement, 22.2% (n = 4/18) after extra-anatomic aortic bypass and 0% (n = 0/4) after graft preservation (p = 0.5387). The majority of surgically treated patients who died, died of MOF (3/8) and respiratory failure (3/8) during hospitalization (Table 4).
Alternative treatments for infected TEVAR.
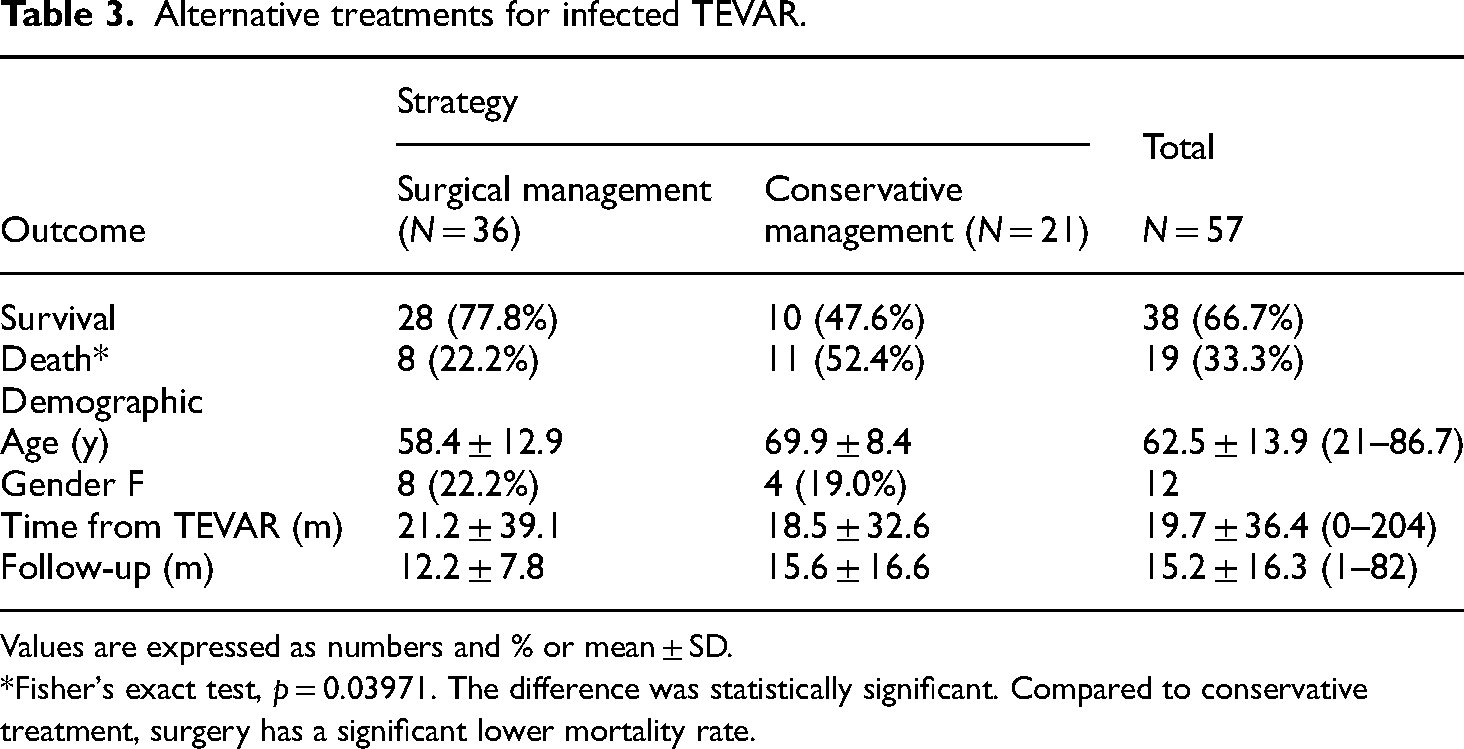
Values are expressed as numbers and % or mean ± SD.
*Fisher's exact test, p = 0.03971. The difference was statistically significant. Compared to conservative treatment, surgery has a significant lower mortality rate.
Alternative surgical treatments for infected TEVAR.

Values are expressed as numbers and %.
*Fisher's exact test, p = 0.5387. The difference was not statistically significant between the mortality rates of the three different surgical treatments.
Discussion
Endograft infection after TEVAR is rare, and the incidence is largely undefined at this time. As a rare complication, randomized trials with relevant caseload do not exist and there are few case reports and short case-series available in literature. Therefore, the knowledge on TEVAR infections is limited, and, so far, the treatment is still undertaken on a case-by-case basis with no consensus guidelines. The 2022 ACC/AHA Guidelines for the Diagnosis and Management of Aortic Disease recommend that, in patients with an infected prosthetic aortic graft who are hemodynamically stable or unstable, it is reasonable to perform open surgery (2a and B-NR or C-LD). 1 Following our review of the literature, the surgical management of infected TEVAR shows a lower mortality rate compared to conservative treatment (22.2% vs 52.4%; p = 0.03971), and this finding is in line with previous published data where the survival rate of surgical and conservative group was 58% and 33%, respectively (p = 0.002). 7 However, one important clinical fact is that patients with infected endografts are usually severely compromised by long-lasting sepsis and malnutrition, and therefore in many cases, they are considered unsuitable for surgery. In this contest, the conservative treatment remains the only available strategy, which will inevitably lead to a higher mortality rate.
Another important result from our review is that, as the two major surgical strategies, both in-situ aorta replacement and extra-anatomic aortic bypass with complete removal of the infected endograft have similar outcomes (p = 0.5387). Extra-anatomic bypass needs ascending-to-descending aortic bypass graft or ascending-to-abdominal aorta/bi-iliac/bi-femoral bypass grafts, thus avoiding the infected area and leading to a reasonably low rate of postoperative re-infection of the grafts.45,46 The prosthetic grafts used during in-situ aortic replacement include rifampicin or silver-impregnated prosthetic grafts, non-soaked prosthetic grafts and cryopreserved allografts (homografts). In our review, prosthetic grafts soaked in antibiotic were used in nine cases (62.3%), non-soaked grafts in four, and only one case was treated with a homograft. All four deaths in the in-situ aortic replacement group occurred in prosthetic grafts soaked in antibiotic. From a recent retrospective, multi-centric study of secondary aorto-enteric fistulae from 2002 to 2014, none of these graft options is clearly superior to the others and, as such, in a stable patient without extensive infection with resistant microorganisms, the use of any of these alternative graft options is acceptable. 47
Graft preservation with only oesophagectomy or fistula resection and drainage was used in a small number of surgically managed patients (n = 4, 11.1%), and it was associated with promising results (may be due to the low number of cases). In a recent report by Kouijzer et al., the mortality rate was 8%, with a curative rate of 67%, and no re-infection occurred after a median follow-up of 24 months in 24 patients with TEVAR graft infection treated without graft removal. 48 Moreover, an intensive antibiotic treatment without graft removal may be a non-inferior option for patients at high surgical risk.
The treatment of infected TEVAR is challenging and needs multidisciplinary collaboration. When a TEVAR infection is diagnosed, comprehensive treatment is demanding, which includes several medical therapies based on targeted intravenous antimicrobial therapy and nutrition support treatment associated with various surgical options. In our research, the results of bacterial cultures showed a wide variety of 21 types of bacterial species, and polymicrobial infection occurred in nearly half of the patients, with Staphylococcus aureus as the most reported pathogens (25%). The source of the infection is rarely identified. A perioperative contamination, a graft enteric erosion, a fistula to the adjacent oesophagus or airway, or a haematogenous spread from remote infections are common risks of endograft infection. 1 Susceptible, adequate and long-term antimicrobial therapy is needed throughout the entire treatment. A 6-week course of intravenous antimicrobial therapy on infected surgical or stented grafts has been recommended in multiple reports and scientific statements.45,47,49 In our review, half of survival patients received postoperative intravenous antibiotics for 4–8 weeks, and 13.2% of patients underwent lifelong antibiotic therapies.
Diagnosis of infected TEVAR has certain difficulties and should be highly vigilant, timely and accurate. From our investigation, fever and pain were the two major clinical presentations with or without symptoms of haemoptysis, haematemesis or melena, fatigue and loss of weight. In addition to that, infectious factors, such as white blood cell count, erythrocyte sedimentation rate and C-reactive protein level were elevated in some case reports. However, these signs and symptoms are non-specific, and the initial workup should include complete blood tests, blood cultures and imaging examination.
Computed tomography and PET-CT scans were the most used imaging examinations in this review. Findings of graft infection on CT included perigraft air, fluid collection around the stentgraft, inflammatory reactions, thickened aortic wall, pseudoaneurysms or haemorrhage. A published study has shown that CT scan has a sensitivity of 94% and specificity of 85–100% in case of graft infection. 50 With the use of PET-CT scan, sensitivity and specificity for low-grade infection may be increased from 77% to 93% and from 70% to 89%, respectively.51,52 In those patients with haematemesis or melena, the gastroscopy may be useful for the diagnosis of AEF or other causes. In this review, the use of the magnetic resonance was not reported.
There are several significant limitations to consider when interpreting the results described in this study. Being a retrospective systematic review on management of infected stentgrafts after TEVAR, the data has inherent deficiencies. The risk of publication bias appears inevitable due to the small sample size (small case-series and case reports), and there is a wide variety of different pathologies, techniques, strategies and outcomes described. This may also represent other limiting factors related to the presence of several reporting centres, such as different patient selection criteria, varying pre-, intra- and postoperative patient management strategies or different surgical experiences. Given these limitations, a general conclusion based on a solid statistical analysis with adequate sample size is not possible at present.
Conclusion
Thoracic endograft infection after TEVAR is a rare complication associated with a high risk of mortality, regardless the type of adopted treatment. The available literature on infected TEVAR still consists of small case-series and case reports, showing a wide variety of treatment options and outcomes. However, following our findings, the surgical treatment seems to have a lower mortality rate when compared to the conservative treatment, in selected patients.
Supplemental Material
sj-pdf-1-sci-10.1177_00368504241296312 - Supplemental material for Treatment of infected endografts after thoracic endovascular aortic repair: a systematic review
Supplemental material, sj-pdf-1-sci-10.1177_00368504241296312 for Treatment of infected endografts after thoracic endovascular aortic repair: a systematic review by Changtian Wang, Ludwig K. von Segesser, Denis Berdajs, Alberto Pozzoli and Enrico Ferrari in Science Progress
Footnotes
Authors contributions
CW: data collection, data analysis, writing the article. LKS: data analysis, study conceptualization, critical revision of the article, supervision. DB: data analysis, critical revision of the article. AP: data analysis, critical revision of the article, supervision. EF: data analysis, writing the article, study conceptualization, supervision.
Data availability statement
Data are taken from the available literature giving the nature of this review article. Data are available upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
