Abstract
Objectives
Injection laryngoplasty (IL) has been widely used as an initial treatment option for unilateral vocal fold paralysis (UVFP). An additional (second) IL is considered a salvage treatment for unsatisfactory outcomes of initial IL resulting from inadequate injection or early resorption of the injection material. This study aims to evaluate the efficacy of additional IL, distinguishing between “salvage” (within 4 months) and “repeated” injections (beyond 4 months), and to analyze prognostic factors for successful outcomes.
Methods
This retrospective study involved patients who received IL at Asan Medical Center from January 2014 to December 2020. Voice parameters were collected after each procedure, and those who conducted the statistical analysis were blinded to the study subjects. Among the 65 patients who underwent additional IL, 51 patients were enrolled in this study. Postinjection grade, roughness, breathiness, asthenia, strain (GRBAS) scales were used to determine satisfactory treatment outcomes. Success of the additional IL was defined as a postinjection grade of dysphonia score of 0 or 1, with a reduction in grade compared with the preinjection grade.
Results
The mean age of the patients was 61.6 years. Out of a total of 51 patients, 37 were men participating in the study. The odds ratio represents the likelihood of success in the second IL. Improved voice outcome after the additional IL was maintained in 23 (45%) patients. Compared with the failure group, the success group had a longer injection time interval between the initial and additional injection (9.1 vs. 7.4 months, respectively, p = 0.010). The success group had a higher proportion of patients with injection intervals >6 months (73.9% vs. 42.9%, p = 0.026). Logistic regression analysis revealed an injection interval >6 months had an odds ratio of 0.265 (confidence interval: 0.080–0.874, p = 0.029).
Conclusions
Additional injections would benefit the patients whose voice outcomes are maintained for a longer period (>6 months) after the first injection.
Introduction
Unilateral vocal fold paralysis (UVFP) is usually caused by thyroid surgery, non-thyroid surgery, trauma, neurologic disease, malignancy, intubation, infection, and other idiopathic factors. 1 UVFP can lead to complications, such as dysphonia, dysphagia, and risk of aspiration during oral intake. Considering the negative effect on patients’ quality of life and additional complications owing to glottal insufficiencies, such as aspiration pneumonia and asphyxia, several treatment modalities have been used to improve glottal insufficiency. 2 Injection laryngoplasty (IL) is widely used as a first-line treatment for UVFP to medialize the paralyzed vocal fold.1,3 The benefits of IL are its cost-effectiveness and similar voice outcomes to open framework surgery.4–6
Patient-reported outcomes showed improved voice and swallowing function after an IL.7–9 However, reports of dissatisfaction can be attributed to inadequate injection or early resorption of the injection material. Moreover, predicting the degree of improvement in voice quality after IL is challenging. 10 Adjuvant voice therapy, laryngeal framework surgery, reinnervation surgery, and repeated IL can resolve unsatisfactory voice outcomes after the initial IL. 11 An additional IL is considered as a salvage treatment due to its convenience and efficacy. Several reports exist of patients with multiple ILs whose voice outcomes following each procedure have been evaluated and compared.6,12 Although favorable outcomes of recurrent IL have been observed after the failure of the initial IL, a large number of patients remain dissatisfied or unresponsive to IL. It is difficult to guarantee the successful outcome of additional IL and to identify the optimal patients who would benefit from them. This study aimed to evaluate the therapeutic efficacy of additional IL by distinguishing between “salvage injections” (within 4 months of the initial injection) and “repeated injections” (beyond 4 months). Additionally, it attempts to analyze the prognostic factors for successful additional IL to better understand who will benefit from these interventions.
Material and methods
Patient selection
We reviewed 300 patients who underwent IL at Asan Medical Center, Seoul, Republic of Korea, between January 2014 and December 2020. Among them, 25 were excluded due to the lack of voice lab results or long injection intervals between the first and second IL (> 35 months). We also excluded 224 patients who received only one IL, leaving 51 patients who received additional IL enrolled in this study (Figure 1). This study was approved by the institutional review board of Asan Medical Center (approval no.: S2022-1735-0001) and performed in accordance with the tenets of the Declaration of Helsinki. The requirement for patients’ informed consent was waived considering the low risk associated with the retrospective review of the data.

Patients who received injection laryngoplasty more than once at Asan Medical Center and their subsequent clinical courses.
Injection laryngoplasty procedure
Injection laryngoplasty was performed in the operating room under local anesthesia. The injection materials were hyaluronic acid (HA) and artificial collagen. Before injection, 4% xylocaine spray (AstraZeneca, Schaumburg, IL) and nasal packing gauze sprayed with 1% lidocaine and 0.1% epinephrine were used for anesthetizing the pharynx, larynx, and nasal cavity. These injection materials were administered through the cricothyroid membrane with a disposable 26G needle under the guidance of a transnasal flexible video laryngoscope. Depending on the defect site, the material was laterally infiltrated to the involved vocal process and vocal ligament at the midportion of the true vocal fold. The injection was considered adequate when the minimum glottal gap on phonation was observed via videolaryngoscopy. We chose Neuramis® (Medytox Inc., Cheongju, Korea) composed of 1.0 mL hyaluronic acid (HA) mixed with 0.3% lidocaine for the first IL considering its relative short duration of action and a viscoelastic profile that most closely resembles that of the human vocal fold. For the second IL, we used Artesense® (European Medical Contract Manufacturing B.V., Nijmegen, The Netherlands) composed of 80% denatured bovine collagen, and 20% polymethyl methacrylate (PMMA) microspheres for the VC filler, because of its relative long-lasting effect and low adverse effect profile.13,14 In most procedures, 0.8–1.0 mL of Neuramis and 0.7–1.0 mL of ArteSense were injected to achieve a slight over-correction of the UVFP. We ensured that no patient had any remaining gap due to insufficient injection material by examining the vocal cords during /e/ phonation using a flexible fiberoptic nasopharyngoscope. After the 1st vocal fold injection, the patients were usually offered another injection, unless patients refused the procedure.
Data collection
We utilized Multi-Dimensional Voice Program (MDVP, Model 4500, Kay, NJ, USA) software for acoustic analysis. The patient was directed to sit in a sound-proof room and wear a microphone positioned 3 cm in front of their mouth. Phonation time was computed by measuring the waveform signal with the MDVP software. Maximum phonation time (MPT) was obtained by instructing the patient to produce the vowel /a/ for as long as possible three times consecutively, and recording the longest phonation time on the chart. For perceptual analysis, a single experienced speech pathologist evaluated the recorded sentences and scored them on the GRBAS scale. 15 The researchers were blinded to all voice data collection and analysis.
Patients’ clinical information, postinjection GRBAS, maximal phonation time (MPT), jitter, shimmer, and noise-to-harmonics ratio (NHR) data were obtained from electronic medical records. The data included age, sex, body mass index, cause of vocal cord paralysis, dates of each injection, voice therapy history, and prior radiation exposure. Patients were further stratified into three groups based on the time interval between the first and second injections: < 3 months, 3–6 months, and >6 months. The voice parameters were measured approximately 1 month after the first and second IL.
Postinjection GRBAS scales were used to determine if the additional IL had been successful instead of preprocedure voice parameters. Success of the second IL was defined as a postinjection grade of dysphonia score of 0 or 1, with a grade reduction from the preinjection grade. The enrolled patients were classified into two groups according to the success of the additional injection, and their clinical characteristics were compared to determine the prognostic indicators of satisfactory results.
Statistical analysis
Statistical analyses were performed using the Statistical Package for Social Sciences ver.26 (SPSS, Chicago, IL). The level of significance was set at 0.05. Data are presented as mean and standard deviation for continuous variables and as numbers and percentages for categorical variables. Procedure vocal parameters after the first and second IL were compared using Student's t-test. Logistic regression analysis was used to determine relevant clinical factors associated with additional IL. The significance of the regression model was reported as odds ratios (ORs) and 95% confidence intervals (CIs).
Results
Baseline characteristics
Table 1 shows the baseline characteristics of all included patients. The mean age of the patients was 61.6 (± 14.2) years, with a male preponderance (72.5%). The causes of UVFP were categorized into thoracic surgery, neck surgery, metastatic lesion, skull base surgery, and idiopathic factors. The most common cause of UVFP was metastatic lesion (47.1%), followed by thoracic surgery (31.4%). Approximately 74.5% of patients received voice therapy, and 13.7% were exposed to radiation. The mean time interval between the first and second injections was 8.2 (± 6.6) months (Table 1).
Baseline characteristics of the patients (N = 51).

Data are expressed as number (percentage), or mean ± standard deviation.
Out of 51 patients who underwent the second injection laryngoplasty (IL), 15 experienced failure during the first IL, as indicated by their post-injection grade of dysphonia score. The remaining 36 patients initially had success with the first IL but required a second injection due to the gradual reduction in its effectiveness over time (Figure 2).

A flowchart depicts the number of patients in each of the success and failure groups after the first and second injection of laryngoplasty.
The efficacy of the second injection laryngoplasty was evaluated by comparing the vocal parameters after the first and second IL. Overall, all voice parameters appeared to improve after the second IL. Compared with post-first IL, parameters such as roughness, breathiness, jitter, and shimmer significantly improved after the second IL (Table 2). MPT increased from 5.0 to 6.7 s, although nonsignificant (p = 0.383). NHR decreased from 1.1 to 0.2 but was also nonsignificant (p = 0.916). The patients were divided into the success (n = 23) and failure (n = 28) groups to compare the voice parameters of the first and second post-IL (Tables 3 and 4). Compared with the failure group, the success group showed better grades of dysphonia, breathiness, and asthenia for the perceptual parameters post the second IL (p < 0.001) (Table 3). MPT was more prolonged in the success group (8.4 s) than in the failure group after the second IL (5.1 s, p < 0.03). However, the initial MPT (after the first IL) was longer in the success group (6.1 vs. 4.0 s, p = 0.04). Jitter was lower in the success group (1.6%) than the failure group after the second IL (5.6%, p < 0.001), and its initial value was also lower in the success group (2.6% vs. 5.9%, p < 0.01). Shimmer was lower in the success group (4.6%) than the failure group after the second IL (10.7%, p < 0.001), and its initial value was also lower in the success group (6.0 vs. 10.4, p = 0.008). NHR was lower in the success group (0.1) than the failure group (0.3, p < 0.001), with the initial value of NHR also being lower in the success group (0.16 vs. 0.19, p = 0.054).
Comparison of voice parameters of the first and second postinjection laryngoplasty.

Data are expressed as mean ± standard deviation. P-values with statistical significance are shown in bold text. MPT, maximum phonation time; NHR, nose-to-harmonic ratio.
Comparison of various voice parameters between the success and failure groups after injection laryngoplasty.

Data are expressed as mean ± standard deviation, median [25%, 75%]. P-values with statistical significance are shown in bold text. P-value compares patients in the success and failure groups. MPT, maximum phonation time; NHR, nose-to-harmonic ratio
Comparison of the clinical factors between the success and failure groups.
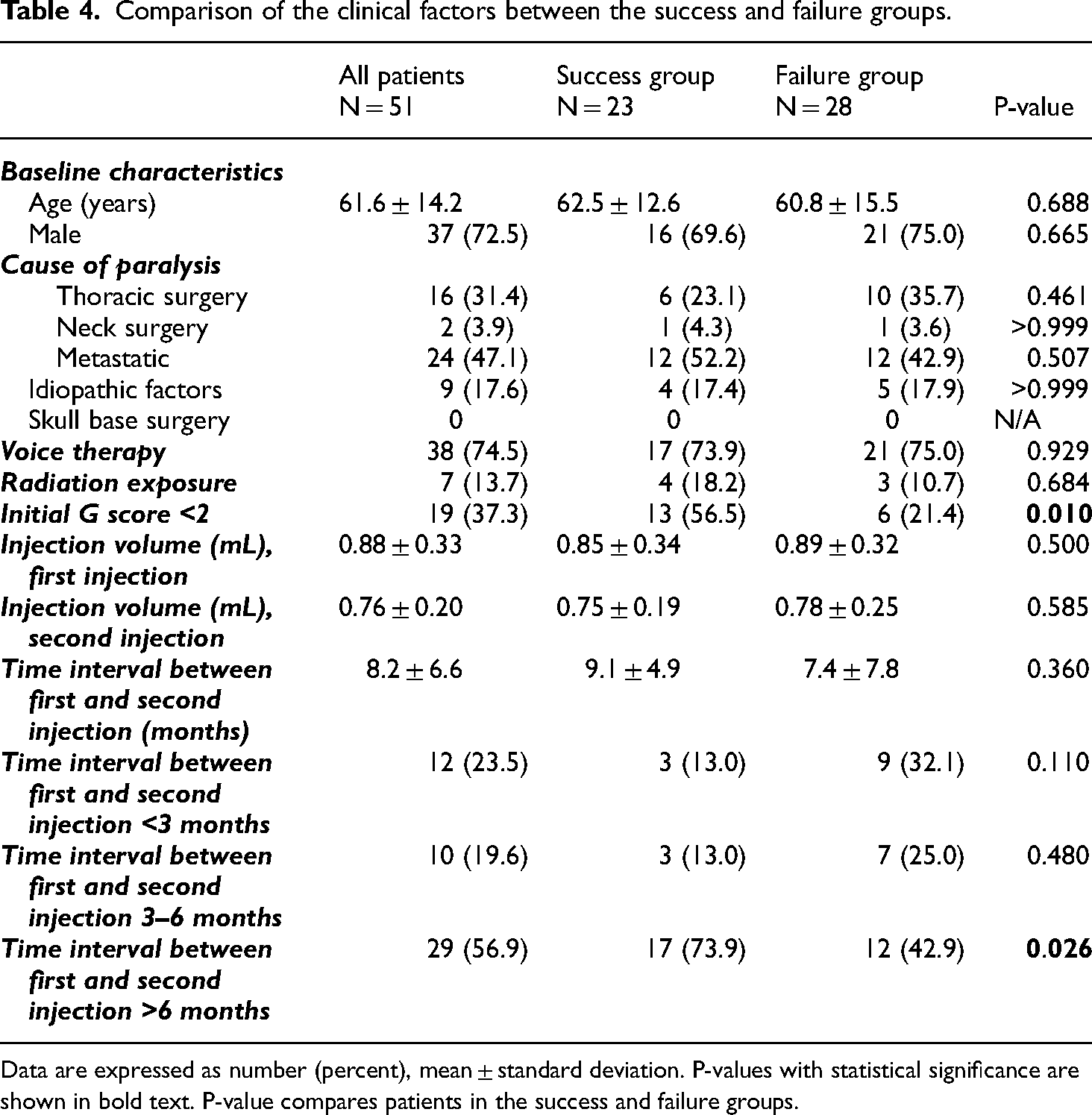
Data are expressed as number (percent), mean ± standard deviation. P-values with statistical significance are shown in bold text. P-value compares patients in the success and failure groups.
Prediction of success and failure
No difference in age, sex, cause of paralysis, voice therapy history, and radiation exposure was observed in both groups (Table 4). No significant difference in the injection volume (in mL) was observed between the success and failure groups after the first IL (0.85 vs 0.89 mL, p = 0.5) and the second IL (0.75 vs. 0.78 mL, p = 0.59). The mean interval between the first and the second injection was longer in the success group (9.1 months) than in the failure group (7.4 months, p = 0.360). The patients were divided into three groups based on the interval between the first and the second IL. The number of patients who received the second IL in <3 months, 3–6 months, and >6 months were 12, 10, and 29 patients, respectively. A larger proportion of patients received the second injection, > 6 months after the first injection (73.9% vs. 42.9%, p = 0.026) (Figure 3).

Time interval between the first and second injection laryngoplasty in the success and failure groups.
Univariate analysis was performed using the significant perceptual voice parameters (Table 3) and clinical factors (Table 4). The injection time interval between the first and second IL (> 6 months), low breathiness score, low asthenia score, jitter, and shimmer were determinants of the success of the second IL (Table 5). In the multivariate analysis, only shimmer (OR = 0.84, 95% CI: 0.73–0.98, p < 0.023) was the significant determinant of the success of IL (Table 5). Among the clinical factors, the time interval of >6 months between first and second injection and the initial grade of <2 for dysphonia were associated with the success of the second IL (p = 0.01 and 0.026, respectively) (Table 4).
Predictive factors of success of additional injection laryngoplasty.

P-values with statistical significance are highlighted in bold. OR, odds ratio; CI, confidence interval; MPT, maximum phonation time; NHR, nose-to-harmonic ratio
Discussion
Injection laryngoplasty has been widely used for UVFP owing to its simplicity and convenience for both patients and surgeons. Additionally, compared with laryngeal framework surgeries, the main advantages of IL are the acceptable lasting effects of the currently developed biocompatible injection materials and the low likelihood of postprocedure complications.10,16 However, IL also has disadvantages, including unpredictable resorption of injection material over time and insufficient vocal fold medialization. 6 A previous study investigated the effects of additional IL following the unsatisfactory results after the first IL, 12 showing improvement in voice outcomes in few patients with UVFP. However, the predictive factors of successful additional IL for patients who were dissatisfied with the initial IL are unknown. This study showed that most of the patients who received additional IL presented with improved voice parameters. Compared with the failure group, the success group presented with significant improvement in voice parameters, including time interval, breathiness, asthenia, MPT, jitter, and shimmer. Although only shimmer was a significant predictive factor, the results of the other parameters would help in selecting patients who will benefit from an additional injection.
Failure is closely associated with persistent or recurrent vocal cord gap during the closing phases even after injection laryngoplasty (IL). There is no clear reason for the failure of IL despite injecting enough materials to medialize the lateralized vocal fold and confirming the obliteration of the gap between the vocal cords after the procedure. Some mechanisms are suggested. The first cause of failure is injection into an inadequate site or depth within the vocal fold. The optimal injection site is considered to be the lateral side of the vocal process of the arytenoid cartilage. Subglottic or medial injection could induce superficial injection, decreasing the mucosal wave and deteriorating voice quality even though the vocal cord gap is diminished. The second cause is early resorption and migration of the injected material. This may be related to injection into an inadequate site or depth. Migration into the paraglottic space hinders the injected material from maintaining the decreased vocal cord gap. The third cause is decreased movement and volume of the opposite vocal cord, which reduces compensatory movement to ameliorate the vocal cord gap. Thus, maintenance of voice quality for more than 6 months may suggest that the injected material resides at an adequate position without migration or resorption, and compensatory movement is effective. Conversely, failure cases within 6 months may imply that at least one of the failure mechanisms underlies. If the underlying problems are not resolved, other treatment options including laryngeal framework surgery can be considered.
For initial injection laryngoplasty, we used temporary injection materials, such as HA, with the effect expected to last up to 4–6 months. Inadequate injection, early resorption, or migration are the primary causes for unsatisfactory results after the initial IL. Hypothetically, owing to the biochemical properties of HA, an additional IL is typically administered a few months after the initial procedure. Few patients experienced efficacy that lasted >1 year. The anticipated duration of the effect of cross-linked hyaluronic acid (HA) administered at a concentration of 20 mg/mL into the vocal fold is approximately four months. Subsequently, patients may require additional injection laryngoplasty (IL) as a salvage therapy due to the finite longevity of the product. While the efficacy of cross-linked HA may extend beyond four months, eventual retreatment with IL or alternative procedures is inevitable, as it does not constitute a permanent injection material. Especially in the success group, the average time interval between the first and the second IL was approximately 8.2 months, ranging from 1 to 35 months. This might imply that temporary materials last longer than expected and compensatory vocal fold movement ameliorates voice quality. Postoperative voice therapy may improve voice quality. 11 Among the enrolled patients, those with unsatisfactory voice quality usually received voice therapy. Thus, in the present study, it was unlikely that voice therapy was associated with satisfactory results.
We often used long-lasting injectable materials, such as Artesense,® for the second IL because they are less likely to cause foreign body reactions and provide a prolonged effect than temporary injection materials. 14 As long-lasting injectable materials were generally administered to patients with good tolerance of their first HA IL, they were more likely to benefit from additional IL. A previous study on IL with Artesense® compared various voice outcomes between the initial and additional IL. Multiple voice parameters (grade of dysphonia, MPT, jitter, shimmer, and NHR) significantly improved following both procedures. 12 It was difficult to determine the factors associated with successful additional IL after the initial procedure.
In our study, comparison of the success and failure groups using the first and second postprocedure voice parameters showed that the grade of dysphonia, breathiness, asthenia, MPT, jitter, shimmer, and NHR scores significantly improved after the second IL. Patients with UVFP often complain of breathiness and asthenic voice change; thus, no significant changes in the roughness and strain scores were observed. For patients dissatisfied after the first IL, open framework surgery could benefit a few patients with UVFP having a large posterior gap or vertical height difference in the vocal folds. 17 Few patients underwent open framework surgeries in our study; two received type 1 thyroplasty and three received arytenoid adduction after the second IL.
Our study has several limitations. First, patient characteristics may have contributed to the success of the additional IL. The only statistically significant factors for determining the success of the additional IL were the time interval between the first and second IL of >6 months and the initial grade of dysphonia <2. However, we cannot definitely confirm that they are factors involved in the outcome of successful additional IL because patients who maintained the effect of the initial IL for >6 months were more likely to respond to additional IL than those who were unable to maintain the effect of initial IL. Further studies are necessary to determine if other factors are involved in successful additional IL. Second, our study did not have preinjection voice lab results; therefore, the immediate improvement in voice outcomes cannot be assessed. The comparison of the pre- and postinjection voice parameters will accurately measure the effect of each IL, thereby providing clues to other factors involved in successful additional IL. Third, long-term follow-up data on voice parameters in patients who experienced successful outcomes after the second IL are lacking. If patients present with successful voice outcomes after an IL, they are usually scheduled for a follow-up exam after 6–12 months, during which several patients are lost to follow-up. Furthermore, glottal gap was not evaluated in this study. 18 Considering that better voice outcome is not always associated with decreased glottal gap upon the closing phase, perceptual analysis could replace the laryngoscopic findings. Fourth, the efficacy of injection laryngoplasty (IL) may exhibit variability dependent on the concentration of hyaluronic acid (HA) employed during the initial procedure. Notably, a higher concentration of injected HA may prolong the therapeutic effect. It is imperative to note that the criterion of >6 months’ duration post-IL solely signifies an improvement subsequent to the initial application. This observation is particularly pertinent given the uniformity in HA concentration and minimal variance in injected volume across all patients. Further investigation, incorporating meticulous pre- and post-assessments for each IL session, is warranted to elucidate these nuances comprehensively. Fifth, The calculation and justification of the sample size selected for this study were not conducted. Future investigations will necessitate a prospective study with an appropriate sample size to attain statistical power.
Conclusion
Injection laryngoplasty (IL) has been commonly employed as a primary treatment modality. However, there remains controversy regarding the optimal therapeutic approach when a gap re-emerges post-IL, necessitating further surgical intervention for vocal fold (VF) palsy. While many surgoens resort to additional IL, this technique does not benefit all patients uniformly. Therefore, this study aimed to identify factors that distinguish patients who may benefit from a second IL from those who may not. Although the precise mechanisms remain unclear, patients who exhibited sustained long-term effects from the initial IL were likely to experience favorable outcomes with subsequent IL procedures. Additional IL can benefit patients who present with prolonged IL treatment efficacy of >6 months with improved voice parameters after initial IL. Early failure of IL could be an indication to consider alternate treatment options for patients with UVFP, such as voice therapy or open framework surgery.
The authors confirm contribution to the paper as follows: study conception and design: Yoon Se Lee; data collection: Taegyeong Kim, Go Eun Jung; funding acquisition: Yoon Se Lee; analysis and interpretation of results: X. Taegyeong Kim, Yoon Se Lee; resources: Minsu Kwon, Young Ho Jung, Seung-Ho Choi, Soon Yuhl Nam; draft manuscript preparation: Taegyeong Kim, Yoon Se Lee. All authors reviewed the results and approved the final version of the manuscript
Footnotes
Acknowledgements
We thank the staff and patients involved in this study.
Consent
The requirement for patients’ informed consent was waived considering the low risk associated with the retrospective review of the data.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
This study was approved by the institutional review board of Asan Medical Center (approval no.: S2022-1735-0001) awarded to Y.S.Lee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Asan Institute for Life Sciences and Elimination of Cancer Project Fund, National Research Foundation of Korea, (grant number 2023IP0093-1, 2021R1F1A1061438).
