Abstract
High hardness, low friction coefficient and chemical resistance are only a few of the exceptional mechanical qualities of diamond. Diamonds can be artificially created to have different levels of conductivity, or they can be single, micro or nanocrystalline and highly electrically insulating. It also has high biocompatibility and is famous for being mechanically robust. Due to its high hardness, lack of ductility and difficulty in welding, diamond is a challenging material to construct devices with. Diamonds have experienced a rise in attention as a biological material in recent decades due to new synthesis and fabrication techniques that have eliminated some of these disadvantages. In general, entropic measurements are used for investigating the chemical or biological properties of molecular structures. This study calculates several important
Introduction
Diamond is regarded as a suitable strengthening stage of metal matrix composites because it possesses the maximum heat conductivity of any natural substance, a low rate of thermal expansion, extreme durability and exceptional chemical solidity.1,2 The artificial diamond market has grown recently, and this has culminated in a large drop in price, which encourages the use of artificial diamond in composite materials. 2
Due to its outstanding properties, diamond is a valued material for a wide range of uses. For instance, this carbon allotrope's tetrahedral geometry with covalent carbon bonds allows for the greatest thermal conductivity, the lowest heat expansion coefficient (0.8 × 10−6 K-1), and exceptionally high hardness (98 GPa).1,3–6
Additionally, diamond is biocompatible and has a strong chemical resistance to bases and acids. 7 Carbon is one of the lightest elements and makes up diamonds as well. An isotope effect's size is directly related to the square root of the mass ratio among the different isotopes. Where this ratio is biggest, the lighter elements are where isotope effects are most prominent. The stable isotopes of carbon are 12C (98.9 percentage) and 13C (1.1 percentage), which occur naturally.
Simple cryogenic distillation can be used to separate carbon isotopes due to the comparatively large percentage variation in mass. This has led to the commercial availability of a wide range of carbon compounds with high isotopic purity. 8 The lattice of tightly coupled, extremely symmetric crystals formed by covalent bonds is formed by the lowest mass element, which is diamond. The material is currently attracting enormous interest for a variety of optical applications due to its exceptional qualities that arise from recent advancements in its synthesis.
There is a tantalizing possibility of much increased capability because the optical properties are distinct from those of other materials in many ways. The assistance of optical design greatly depends on having a thorough understanding of how electromagnetic radiation interacts with diamond's bulk and surface. 9 The results of the simulation showed that too much oxygen increases the oxidation of diamond, which raises the substance's mechanical rigidity and roughness rate. The lower rigidity of the oxidized diamond allowed for a larger surface deformation.10–12
Due to its advantageous characteristics, such as its malleability and mechanical workability, the alloy of copper is widely used in diamond instruments. 13 Copper is a typical bond material used for numerous diamond-cutting instruments due to its inexpensive cost and low sintering temperature. 14 The diamond swords can be properly squeezed and self-sharpen when a suitable ratio of diamonds is established and enough cutting edges are scattered across the working surface of the diamond tool. In addition, the cutting life and efficiency of diamond particles is directly affected by their size.15–17
Several methods exist for converting a chemical network into a graph. Examine the types of compounds that are present in the network. 18 These may involve other chemicals, ions or radicals. 19 Find out which species in the network are involved in which chemical processes. This can entail looking over experimental results, chemical equations or other knowledge sources. 20
Al-Raee 21 initiated the formula for calculating the specific bond volume of the Morse potential is universal and can be used for many materials. Shi et al. studied the stability of the interface is determined by the type of interfacial border and bond, as well as the populations and numbers of interfacial bonds. 22 The layer-projected density of states analysis indicates that all the examined contacts display metallic properties. 22 Zhang et al. presents the prediction of a new carbon phase, called pentapeptide diamond, which exhibits orthorhombic crystal structure and sp3 bonding. 23 The prediction was made by integrating the particle swarm optimization method with first-principle calculations. The phonon spectra, total energy and elastic constants calculations of the pentapeptide diamond provide evidence that it is dynamically, thermally and mechanically stable under zero pressure. It has a large bulk modulus of 385 GPa and a Vickers hardness of 72.6 GPa, which is like that of diamond. The electronic band structure simulations indicate that the pentapeptide diamond possesses a direct band gap of 4.18 electron volts (eV). 23
Make an edge connecting the points that represent the reactants and outcomes of each chemical reaction in the graph. 24 Complete the graph by adding any other details, such as rates of reactions or thermodynamic characteristics, that might be essential to the chemical network.25,26 Once the chemical network is represented as a graph, its composition can be examined using a range of graph-theoretical methods and procedures.27–29 For instance, strongly connected node groups can be detected using community detection techniques, and important nodes in the network can be located using centrality metrics.30,31
This method can offer guidance for the development of novel chemical processes and reactions as well as knowledge about the chemistry of intricate chemical structures. 32 Topological indices play an essential role in providing guidance for the treatment of tumours or malignancies.33,34 Experimental or numerical methods can be used to find these indices. Computer analysis offers a time- and money-efficient solution because experimental data, despite their high cost, are important. The eccentricity-based topological indices are a key component of the theory of chemical graphs. 35 Chemist Wiener created the first topological index in 1947. 36
Methodology
Consider a graph with the form
Degree-based topological indices.

Shannon introduced the concept of entropy in 1948,41–43 which measures the molecular disorganization of the system as well.19,44 The measurement of edge-weighted graph entropy was initially presented in 2009
18
for an edge-weighted graph 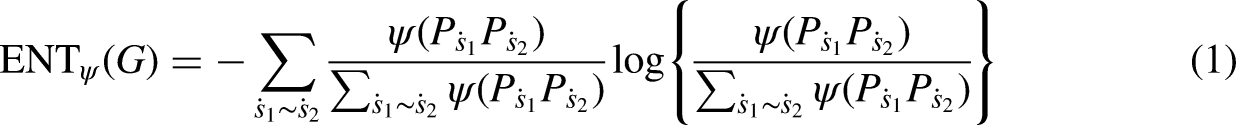
Results
In this section, we determine exact values of all the degree-based topological indices and entropies tabulated in Tables 1 and 2. For this, first we provide the edge partition of diamond crystal
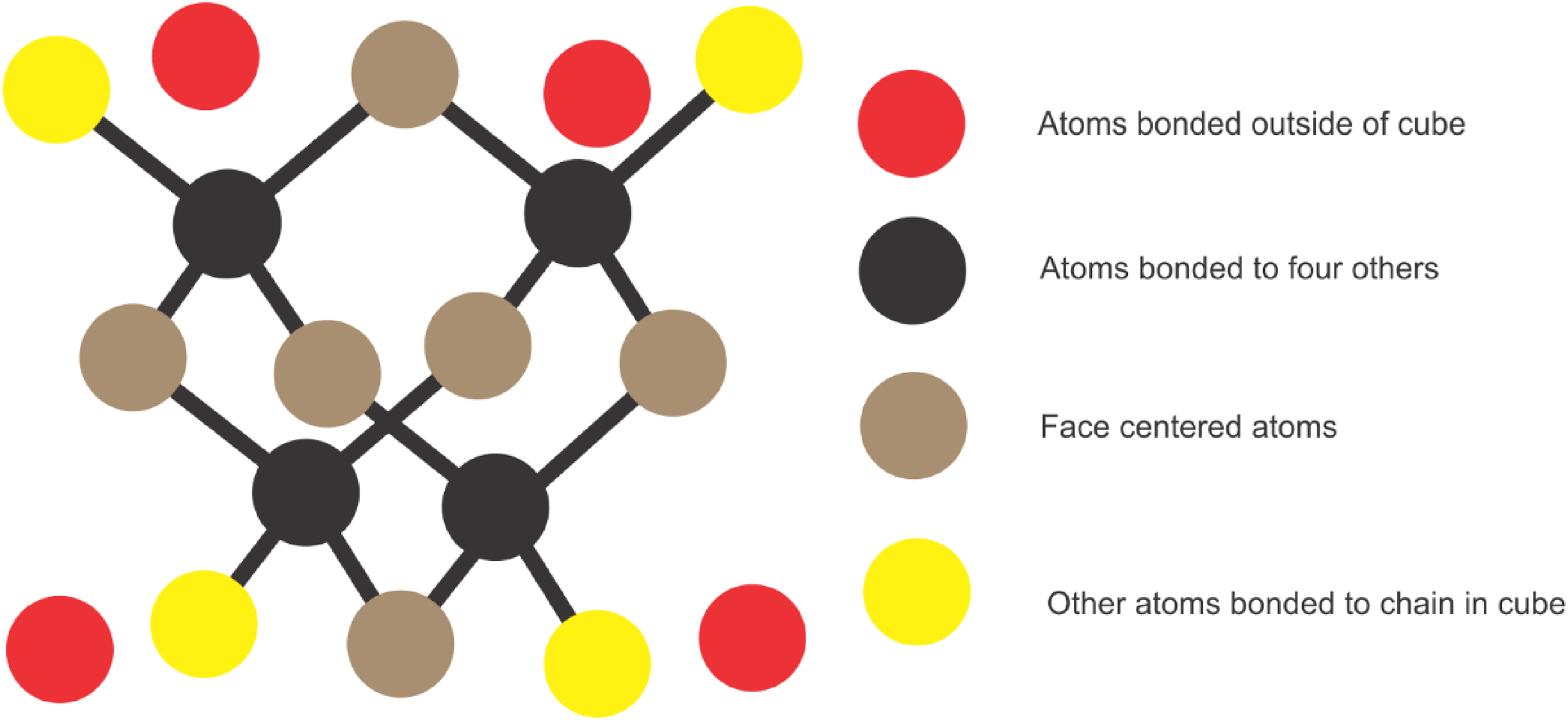
Step-by-step structure of a diamond.
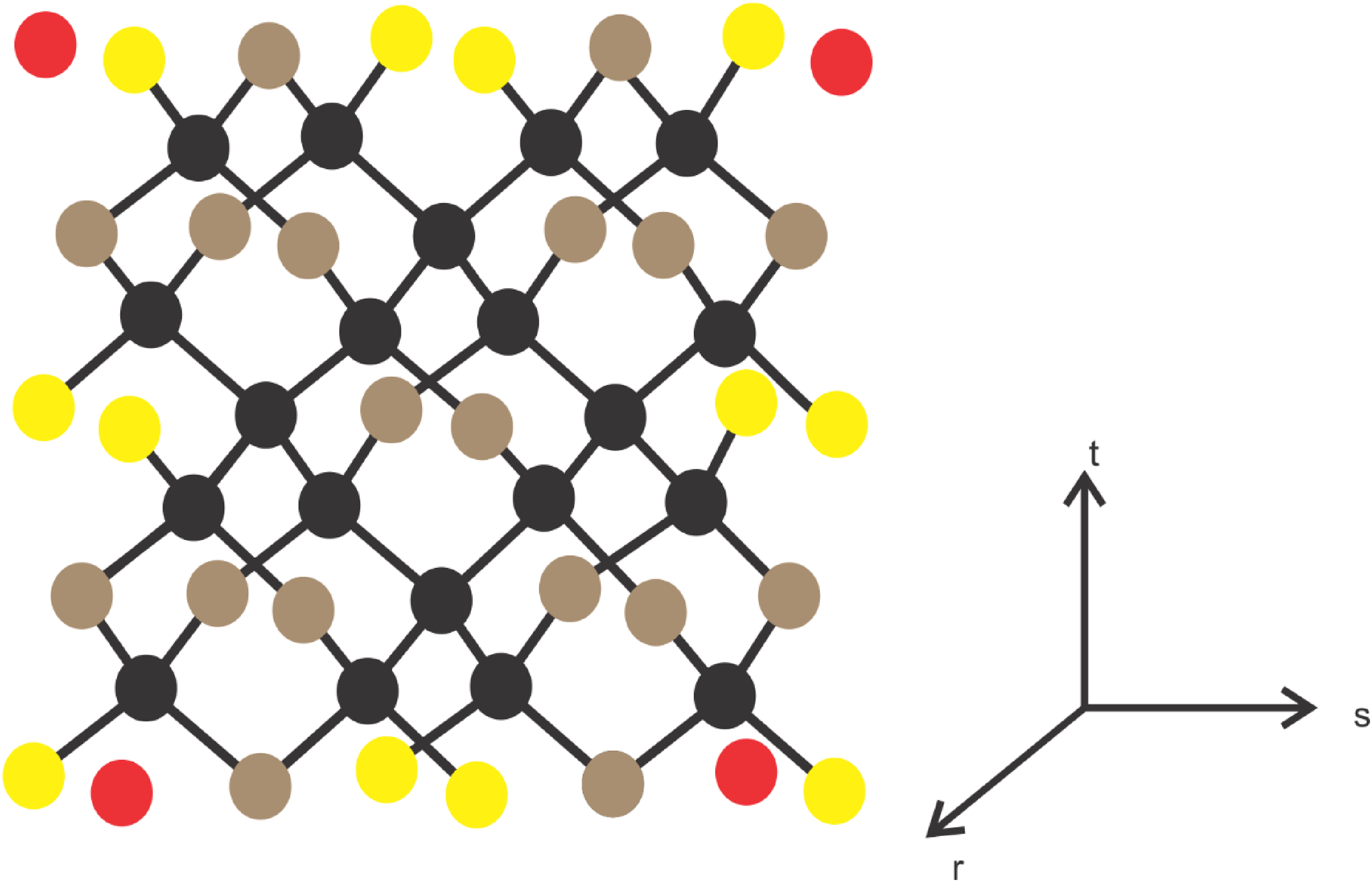
Tetrahedral structure of a diamond.
Formulae to compute the entropy on the base of indices tabulated in Table 1.

Edge partition with edge degree of

By using the edge partition of Table 3 in each formula of Table 1, we have the following equations:




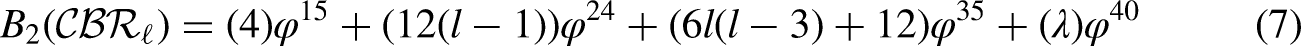

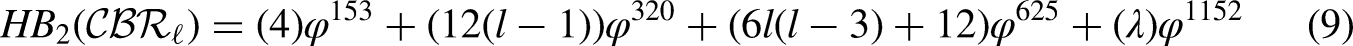




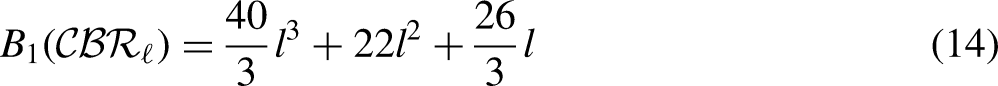

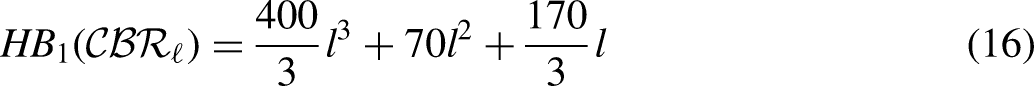

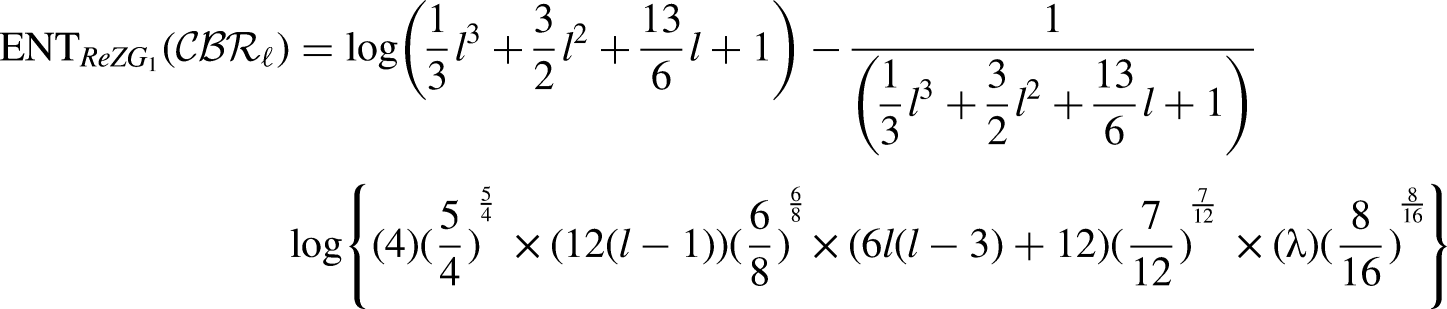
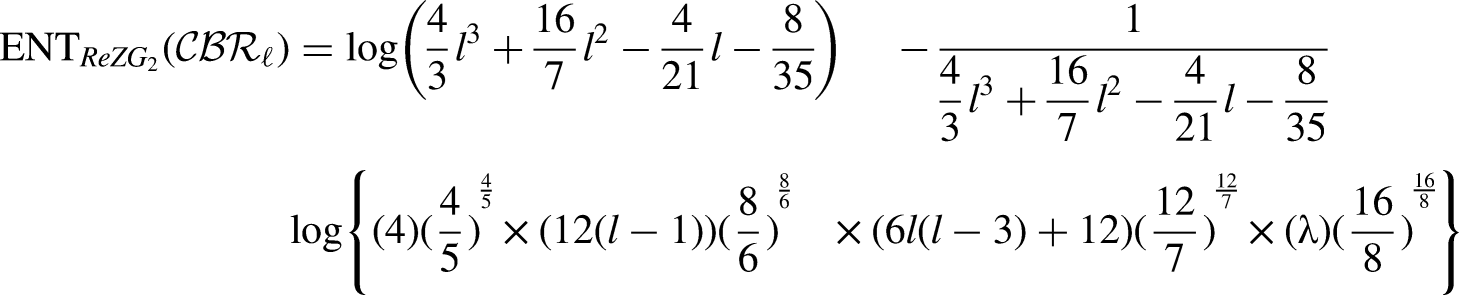






Comparison
This part presents numeric and graphical comparisons of the computed results as presented in Table 4 and their graphical analysis as shown in Figure 3. For the sake of comparison of results, we take values

Graphical expression of TIs of
Numeric expressions of TIs of

Concluding remarks
This manuscript presents key findings regarding the entropy of eight degree-based topological indices for a molecular structure of diamonds. By the reason of rich conception, we presented numeric and graphical analysis of obtained results of toplogical indices for few initial values of the used parameter as shown in Table 4 and Figure 3. From obtained results and their provided comparisons, each of the calculated topological indices can be regarded as a finest/moral or nastiest/immoral descriptor for a topological characterization for the molecular structure of diamond according to the following values-based hierarchy (this pyramid is just based upon our mathematical computations, numeric and graphical analysis. However, the novelty of each topological index can be followed after investigating that which index is best correlating with which of the chemical property):
Footnotes
Acknowledgements
This research was supported by the researchers Supporting Project Number (RSP2024R401), King Saud University, Riyadh, Saudi Arabia.
Authors’ note
Future work: We intend to characterize the molecular structure of a diamond crystal via distance-based topological indices and entropies.
Authors contribution
This work was equally contributed by all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

