Abstract

Introduction
Animal behavior relies on the proper functioning and coordination of multiple neural circuits and peripheral organs. Let us consider the example of communication signals such as vocalizations of humans, birds, and crickets. The carrier frequencies of the sounds depend on the morphology or physical characteristics of peripheral effectors. The rhythms of sound production depend on the function of central motor circuits, which determines the activity patterns of motor neurons. Further, communication behavior is not determined by motor aspects alone. Sensory receptors and central circuits must be tuned to conspecific vocalizations to effectively detect behaviorally relevant signals. Thus, changes in behavior during evolution, development, or plasticity may require coordinated changes to multiple neural networks spanning peripheral and central elements of both sensory and motor systems. However, coordinated changes in sensorimotor integration, which links sensory and motor systems, have not been well studied. Our group has exploited the unique advantages of mormyrid electric fish to show that a sensorimotor integration circuit is also involved in behavioral change through evolution and plasticity.1,2
Mormyrid electric fish and corollary discharge
Mormyrid fish are found throughout sub-Saharan Africa and the Nile River basin and are the largest group of Osteoglossiformes, with more than 230 species currently identified. This diversity has occurred against a background of diversification of electrical communication signals. 3 Mormyrid fish generate a pulsed electric signal from the electric organ in their tail, called an electric organ discharge or EOD (Figure 1). The electric organ consists of a series of electrically excitable cells called electrocytes that are derived from muscle. 4 These electrocytes generate synchronized action potentials that create an electric field surrounding the fish's body. The electric field produced by an EOD can be distorted by the presence of surrounding objects, and the fish can sense these distortions to gain information about the environment even in complete darkness. 5 In addition, the electric field is transmitted to other nearby individuals, and thus the EOD is also used for communication. 6

Electric organ discharge and the sensorimotor integration circuit of a mormyrid fish. (a) Outline of a mormyrid fish, Brienomyrus brachyistius. The electric organ is located in the caudal peduncle. (b) Voltage recording of electric signaling behavior. The EOD has a fixed waveform, and changes in the recorded EOD amplitude over time are due to movement of the fish relative to the recording electrode. The intervals between EODs can be flexibly regulated to communicate the fish's behavioral states. (c) Diagram showing electromotor (blue), KO sensory (red), and corollary discharge (purple) pathways. The CN drives the EO to generate each EOD via the MRN and spinal EMN. KO electroreceptors receive the EOD and send time-locked spikes to the nELL via primary afferents. The nELL neurons project their axons to the ELa, which sends its only output to the adjacent ELp. The CN provides another output to the BCA, which in turn projects to the MRN and to the MCA. The MCA sends its output to the slem, which has GABAergic inhibitory neurons projecting to the nELL. Panels (a) and (b) adapted with permission from reference [1], Elsevier. Panel (c) adapted from reference [2]. BCA: bulbar command-associated nucleus; CN: command nucleus; ELa: anterior exterolateral nucleus; ELL: electrosensory lateral line lobe; ELp: posterior exterolateral nucleus; EMN: electromotor neurons; EO: electric organ; EOD: electric organ discharge; GABA: γ-aminobutyric acid; KO: knollenorgan; MCA: mesencephalic command-associated nucleus; MRN: medullary relay nucleus; nELL: nucleus of the ELL; OB: olfactory bulb; OT: optic tectum; slem: sublemniscal nucleus; tel: telencephalon; val: valvula of the cerebellum.
Electrocommunication in mormyrid fish has two components: EOD waveform and interpulse interval (IPI).6,7 EOD waveform is stereotyped within an individual fish and represents the sender's identity including species, sex, and social status (Figure 1(b)). IPI is flexibly varied to communicate behavioral states in real time, and is associated with behaviors such as aggression, submission, territoriality, social cohesion, courtship, and mating (Figure 1(b)).
Encoding of these electrocommunication signals begins at peripheral electroreceptors distributed over the surface of the skin, called knollenorgans (KOs). 7 When processing information during communication, it is important to know whether received signals are from oneself or from other individuals. KOs respond to both self-generated and other-generated EODs, and are therefore unable to make this distinction. Failure of this discrimination would result in dysfunctional communication and social behavior.
Corollary discharge plays an essential role in self versus other discrimination. EOD production begins in the EOD command nucleus (CN) in the medulla, which drives the electric organ to produce each EOD through the electromotor command pathway (Figure 1(c)). 8 At the same time, the CN also sends an inhibitory signal to the primary electrosensory center (nucleus of the electrosensory lateral line lobe [nELL]), as an internal prediction of the timing of the EOD, via the corollary discharge pathway (Figure 1(c)). 8 This inhibitory signal precisely blocks the sensory response to self-generated EODs, allowing the downstream sensory pathway to selectively process sensory information from the EODs emitted by other fish. 9 Thus, this KO pathway is thought to be devoted to processing communication signals, while a separate pathway is thought to be devoted to mediating active sensing.
Corollary discharges are not specific to the electrosensory systems of mormyrid fish but are found across various sensory systems and throughout the animal kingdom.10,11 It is thanks to corollary discharges that we do not perceive movement of the world when we shift our gaze, nor do we feel tickled when we tickle ourselves. Mormyrid fish have several advantages for studying neural mechanisms of corollary discharge. 11 First, the EOD is a very simple all-or-none behavior that can be easily reproduced and manipulated, unlike many other behaviors that involve complex interactions between multiple movements. Second, fish that are paralyzed for electrophysiological recording are electrically silenced as well, but they still generate “fictive” EODs that can be recorded from the spinal electromotor neurons innervating the electric organ. Fictive EOD recording is noninvasive, and its site is distant from the brain, which allows for simultaneous electrophysiological recording from the brain. These advantages have fostered a detailed understanding of the neural circuitry of corollary discharges and provided many general principles that are shared among diverse animal species.11,12
Corollary discharge shift underlies evolutionary divergence of behavior
EOD waveform is highly variable across species, especially in its duration, which can vary from 0.1 to more than 10 ms. This variation in EOD duration should lead to variation in the timing of reafference (afferent sensory input caused by one's own behavior). This raises the question of whether or how the corollary discharge circuit adapts to these species differences. We used 7 species of mormyrid fishes having a variety of EOD durations (0.2–8.6 ms; Figure 2(a)) and compared the timing of corollary discharge inhibition measured by recording evoked potentials from midbrain electrosensory nuclei in the KO pathway. 2 We found that species producing longer EODs (e.g. Campylomormyrus numenius) have more delayed corollary discharge inhibition than those producing shorter EODs (e.g. Campylomormyrus compressirostris) (Figure 2(b)). Furthermore, this timing shift was consistent with KO spike timing in response to self-generated, reafferent EODs (Figure 2(c)). These results indicate that the corollary discharge circuit adapts to evolutionary changes in EOD waveform to maintain optimal cancelation of sensory responses to reafferent inputs.
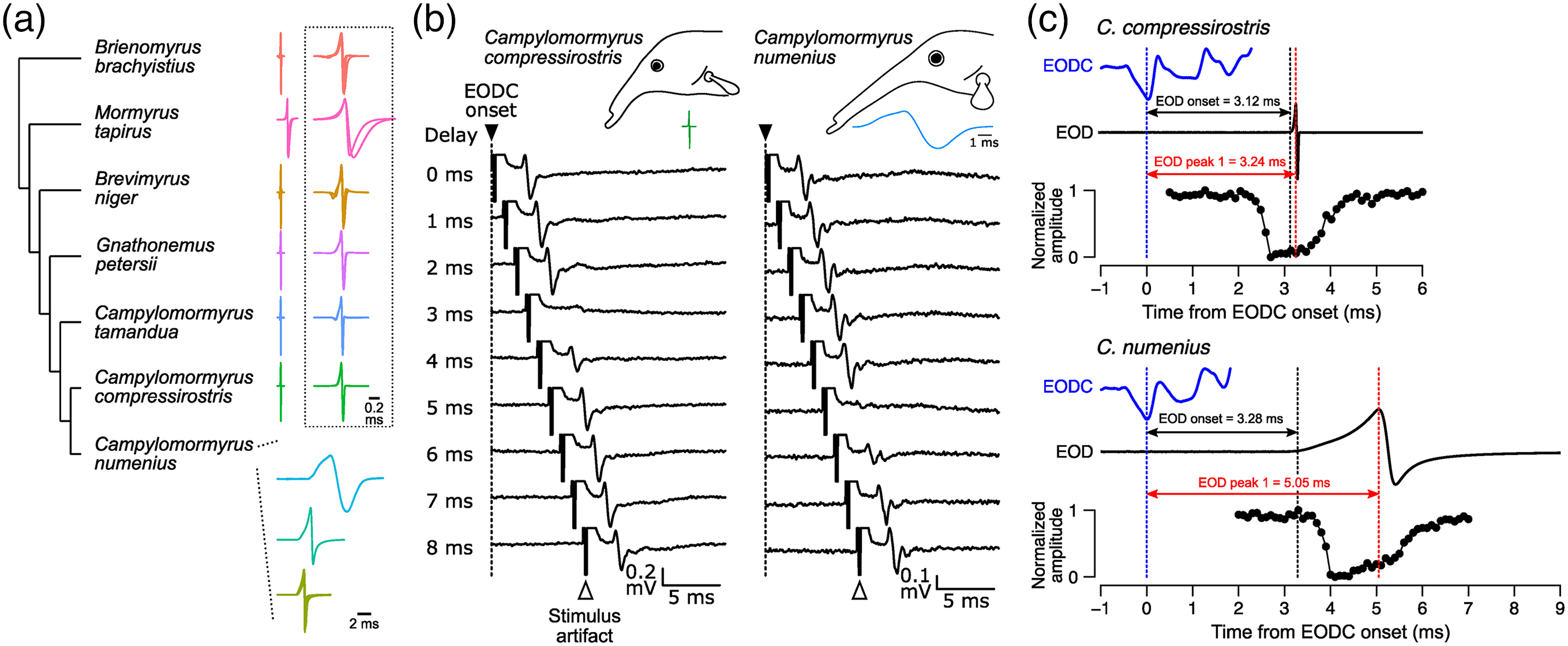
Corollary discharge shift matches EOD evolutionary divergence. (a) Diversity of EOD waveform across the seven species. The EODs in Campylomormyrus numenius are displayed in three categories with distinct EOD durations. The dotted box to the right displays EODs on an expanded timescale for the other six species. (b) Comparison of evoked potentials in the ELa between Campylomormyrus compressirostris and C. numenius. In this example, C. compressirostris generates an EOD of 0.2 ms duration while C. numenius generates an EOD of 8.6 ms duration. Evoked potentials were recorded in response to electrosensory stimuli at varying delays following EODC onsets. In C. compressirostris, the sensory responses to stimuli with a 3 ms delay are blocked by the corollary discharge while, in C. numenius, the sensory responses to stimuli with 5 ms delay are blocked by the corollary discharge. (c) Precise matching between reafferent timing and corollary discharge inhibition. We compare the time courses of the EODC (top blue trace), the EOD (middle trace), and the inhibition curve of the corollary discharge recorded in ELa (bottom trace). The inhibition curve is plotted as a function of normalized amplitudes versus stimulus delay, which were calculated using the maximum and minimum peak-to-peak amplitude across all stimulus delays. Vertical dotted lines indicate EODC onset (blue), EOD onset (black), and peak 1 of the EOD (red). Peak 1 of the EOD is strongly correlated with the spike timing of knollenorgans in response to a self-generated EOD. Thus, each inhibition window optimally blocks sensory responses to reafferent input. Note that individuals displayed in (c) are different from ones in (b). All panels adapted from reference [2]. ELa: anterior exterolateral nucleus; EOD: electric organ discharge; EODC: EOD command.
Behavioral changes through evolution have been associated with changes in peripheral effectors, motor circuits, and sensory systems. Species differences in EOD waveform reflect differences in the biophysical characteristics of electrocytes in the electric organs. 13 On the sensory side, the frequency tuning of KOs is correlated with the frequency spectrum of conspecific EODs. 14 Our study provides further evidence that behavioral evolution is accompanied by evolutionary change in the neural circuitry underlying sensorimotor integration. 2 Taken together, we suggest that behavioral evolution requires coordinated changes throughout the sensorimotor network.
Development of motor output and corollary discharge
EOD duration can also vary within species. It is known that EOD duration in C. numenius changes with growth, and this correlates with ontogenetic changes in electric organ anatomy. 15 Within this species, we also found that the timing of corollary discharge inhibition correlates with individual differences in EOD waveform. 2 This result suggests that developmental changes in behavior also involve coordinated changes in the sensorimotor network.
This raises a fundamental question: is the mechanism underlying the precise matching of corollary discharge and reafferent timing within individuals the same as the mechanism underlying matching during evolutionary divergence between species? One promising hypothesis is that altered sensory feedback resulting from changes in EOD waveform alters the timing of corollary discharge inhibition through activity-dependent plasticity.
A steroid hormone independently regulates motor output and corollary discharge in a coordinated manner
EOD waveform shows seasonal plasticity in some mormyrid species, which is mediated by steroid hormones. 16 Male Brienomyrus brachyistius elongate their EOD waveform two-fold to three-fold during the breeding season. This plasticity is mediated by increasing levels of circulating testosterone, and is easily reproduced in the laboratory, even in juvenile and female individuals (Figure 3(a)). We tracked changes in the timing of corollary discharge inhibition up to 17 days after hormone treatment and found that it is precisely matched to the shift in reafferent timing due to the elongated EOD waveform (Figure 3(b) and (c)). 1 Unlike the evolutionary and developmental changes, the hormonal shift in corollary discharge showed a substantially elongated inhibition window. This longer inhibition may help to cancel multiple spikes in KOs, which were more common in testosterone-treated fish.
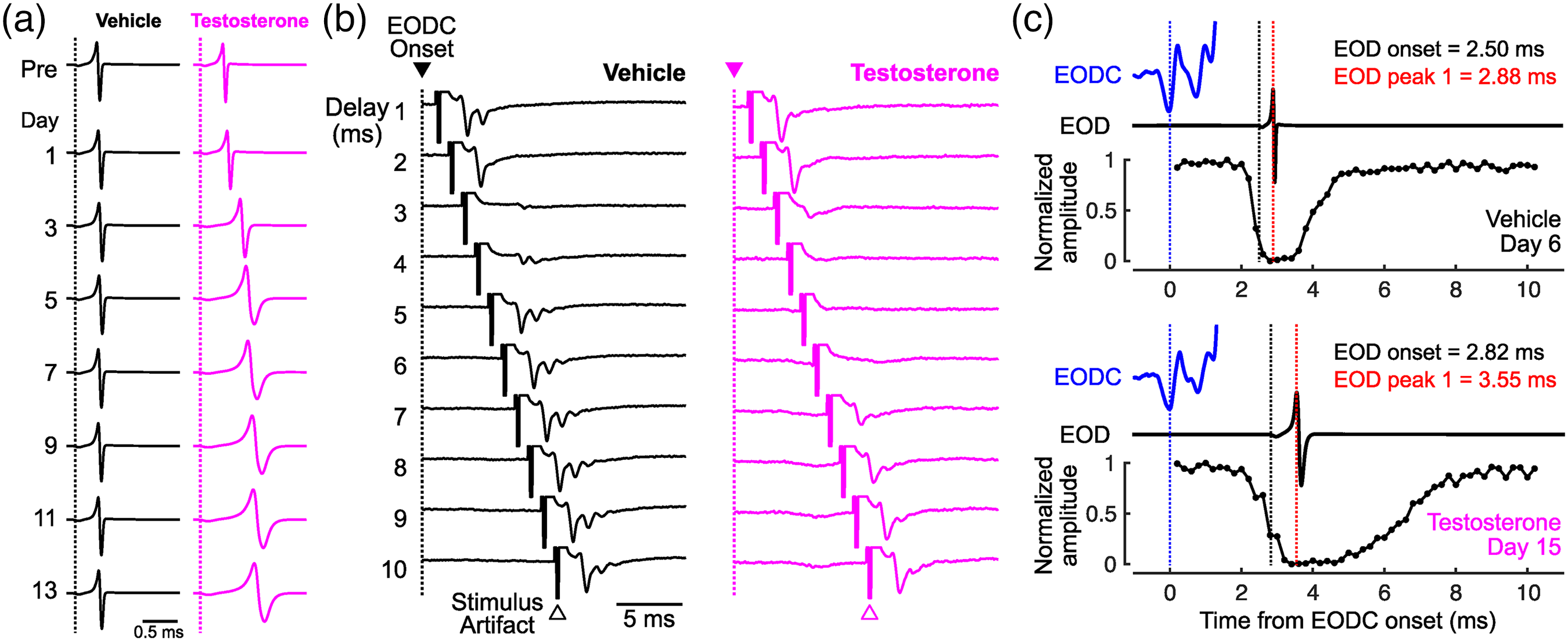
Corollary discharge shift matches hormonal elongation of EOD. (a) Daily changes in EOD waveform in response to vehicle (black) and testosterone (magenta) treatment. Dotted line indicates EOD onset. (b) Comparison of evoked potentials in the ELa between vehicle-treated and testosterone-treated fish. Similar to Figure 2, evoked potentials were recorded in response to electrosensory stimuli at varying delays following EODC onset. (c) Precise matching between reafferent input and corollary discharge inhibition. EODC, EOD, and inhibition curve are presented as in Figure 2. All panels adapted with permission from reference [1], Elsevier. ELa: anterior exterolateral nucleus; EOD: electric organ discharge; EODC: EOD command.
Testosterone acts directly on the electric organ to elongate EOD waveform, while indirectly altering the frequency tuning of KOs through altered sensory feedback. 17 It is possible that testosterone also indirectly modulates the corollary discharge circuit: altered sensory feedback provides the temporal basis for adaptation through plasticity or learning. We surgically silenced the electric organ of fish to remove the effects of sensory feedback and examined the effect of testosterone on corollary discharge timing. 1 Surprisingly, we found similar hormonal modulation of corollary discharge timing as in intact fish, suggesting that sensory feedback was not required for this timing shift. Thus, our results provide evidence that testosterone directly and independently adjusts peripheral motor output and corollary discharge inhibition in a coordinated manner.
Perspectives
Our series of studies shows that, in mormyrid fish, the corollary discharge that filters out responses to self-generated stimuli is also precisely regulated in response to evolutionary, developmental, and seasonal behavioral changes.1,2 We emphasize that changes in even relatively simple behaviors, such as EOD production, involve coordinated changes in the entire sensorimotor network. In particular, coordinated changes between motor output and its corollary discharge are essential for all animals. If they were mismatched, then it would be difficult to distinguish between self and others. Indeed, corollary discharge dysfunction may be associated with hallucinations and delusion in schizophrenic patients,18–20 as well as sensory prediction deficits in autism.21,22 Steroid hormone levels may also be disrupted in these conditions.23–25 Our results suggest that testosterone modulates the timing of corollary discharge. 1 Of course, this is only evidence in one species, but it may prove to be more common among a wider range of animals, including humans, and might provide therapeutic clues for reducing the mismatch between internal predictions and the sensory consequences of one's behavior in the future.
The key remaining question is whether changes in the neural mechanisms that determine the timing of corollary discharge are the same between evolutionary, developmental, and seasonal hormonal changes. In mormyrid fish, the nuclei that comprise the corollary discharge pathway have been identified, but it remains unknown which nucleus (or nuclei) contributes to the shift in timing. There are many possible mechanisms that could shift the timing of corollary discharge. For example, changes in γ-aminobutyric acid receptor expression in nELL neurons could alter the kinetics of inhibitory synaptic currents. Axonal elongation, decreasing myelination or smaller axonal diameter could increase transmission delays. Passive or active electrical properties of neurons in the corollary discharge pathway could also alter their excitability to shift timing. Future studies will seek to identify the source of the timing shift, the cellular mechanisms underlying the precise matching and whether these are shared among evolutionary, developmental, and seasonal changes in behavior.
Footnotes
Author contributions
MF wrote the first draft of the article and BAC edited the article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Division of Integrative Organismal Systems (grant number IOS-2203122).
