Abstract
Objective
Acute limb ischemia (ALI) is a rapid decrease in lower limb blood flow due to acute occlusion of peripheral arteries or bypass grafts. This study aimed to establish an ALI model using microsized gelatin beads and to investigate the pathophysiological conditions.
Methods
Male Sprague-Dawley rats were anesthetized, and a low or high dose of microsized gelatin beads was administered into the left femoral artery on days 0 and 7. A control, that is, normal saline (NS) group in which NS was administered in the left femoral artery, a femoral artery cut (FAC) group in which the left femoral artery was cut, and a sciatic nerve cut (SNC) group in which the left sciatic nerve was cut were prepared. After 21 days, the temperature changes and the muscle weights in the lower limbs were measured. To assess nerve damage, the L1–6 sympathetic ganglia were immunostained with activating transcription factor 3 (ATF3) antibody.
Results
In the Low-dose, High-dose, and FAC groups, a decrease in temperature was predominantly observed in the left limb. In the High-dose and SNC groups, the weight of the soleus muscle and extensor digitorum longus in the left limb decreased; however, no weight changes were observed in the Low-dose and FAC groups. Conversely, the weight of the gastrocnemius muscle significantly decreased in the Low-dose, High-dose, FAC, and SNC groups. In the High-dose and SNC groups, the number of ATF3-positive cells in the sympathetic ganglia significantly increased, and in the Low-dose, a small number of ATF3-positive cells were observed. However, ATF3-positive cells were rarely observed in the FAC and NS groups.
Conclusion
We established an ALI rat model using microsized gelatin beads. The results of this study suggest that autonomic neuropathy in ALI is related to both muscle damage and peripheral neuropathy.
Introduction
Acute limb ischemia (ALI) is defined as a rapid decrease in lower limb blood flow due to acute occlusion of peripheral arteries. The main reason is an embolism caused by an underlying disease or iatrogenic bypass grafts. 1 The incidence of ALI is between 10 and 15 persons per 100,000 per year in the United States. 1 The associated morbidity and mortality are extremely high, with 1-year mortality rates at more than 40%. 2 ALI is 20-fold more common in the lower extremities than in the upper extremities. 3 The causes of ALI, excluding trauma, are broadly divided into embolism and thrombosis. A recent report from England revealed that the incidence rates of embolism, thrombosis due to an occlusive atherosclerotic lesion, complex factors, and stent-related or graft-related thrombosis are 46%, 24%, 20%, and 10%, respectively. 4 Small vessel rather than large vessel occlusion is the usual cause of blue toe syndrome, which is usually due to embolic occlusion of digital arteries with microembolization, such as atherothrombotic material from proximal arterial sources. 5
Among animal experiments aimed at elucidating the pathology and treatment of ALI or peripheral arterial disease (PAD), the unilateral femoral artery transection model and the femoral/saphenous artery excision model are the two commonly used murine models. 6 The femoral artery transection model produces only a mild-to-moderate amount of ischemia that induces arteriogenesis mainly in the thigh, with minimal calf angiogenesis. 6 The femoral/saphenous artery excision model produces more severe ischemia that induces both thigh arteriogenesis and calf angiogenesis. 6 In addition, a rabbit model using an injection of microspheres (15 μm in diameter) into the left common iliac artery and then ligation of the common iliac artery has been established. 2 This model seems to be a combination of PAD and ALI. The abovementioned animal models have been used for investigating both PAD and ALI, yet no ALI-specific model has been established. Therefore, a model without ligating or cutting of blood vessels is needed.
Gelatin beads are clinically used as drug-induced carriers, and chemoembolization therapy uses gelatin beads containing an anticancer drug that are injected into the tumor site via a catheter. 7 We hypothesized that microsized gelatin beads could be useful to establish an ALI rat model. This study aims to establish an ALI rat model using microsized gelatin beads and to investigate the pathophysiological conditions. In addition, to elucidate the mechanism of this model, we compared it with the femoral artery cut (FAC) and sciatic nerve cut (SNC) models, a well-known model of peripheral artery and peripheral nerve injures.
Materials and methods
Experimental animals
A total of 29 male Sprague-Dawley rats (9–13 weeks, mean weight, 230 g; range, 220–245 g; SLC, Hamamatsu, Japan) were used in this study. All rats were housed in individual cages with food and water available ad libitum under a 12 h light-dark cycle at 23 ± 1 °C and 50% relative humidity. All rats were acclimatized for at least 1 week prior to the experiment and were maintained in accordance with the Guide for the Care and Use of Laboratory Animals. The study was approved by the local animal ethics committee in accordance with the regulations for animal experiments at Aichi Medical University (authorization numbers: 2018-47, 2019-39, 2020-83), the Animal Protection and Management Law of Japan (No. 105), and the Ethical Issues of the International Association for the Study of Pain. All efforts were made to minimize animal suffering.
Experimental design
The 29 rats were divided into 5 groups: (1) the normal saline (NS) group in which 200 μl of NS was administered into the left femoral artery (n = 5), (2) the Low-dose group in which a solution of 10 μl of 10% (weight/volume) microsized gelatin beads mixed with 190 μl of NS was administered into the left femoral artery (n = 7), (3) the High-dose group in which a solution of 50 μl of 10% W/V microsized gelatin beads mixed with 150 μl of NS was administered into the left femoral artery (n = 6), (4) the FAC group in which the left femoral artery was cut at the center of the left femoral region (n = 5), and (5) the SNC group in which the left sciatic nerve was cut (n = 6) (shown in Figure 1(a)).

Experimental design. (a) A scheme of experimental design. The normal saline (NS), low-dose (Low-dose), and high-dose (High-dose) groups underwent the same surgery on days 0 and 7. The femoral artery cut (FAC) and sciatic nerve cut (SNC) groups underwent surgery on day 0 and sham surgery on day 7. Skin temperature, objective findings (swelling, pallor, and nail color), limb movement (ankle dorsiflexion and toe abduction), weight of the lower limb muscle (extensor digitorum longus, soleus, and gastrocnemius), histopathological assessment and activating transcription factor 3 (ATF3)-positive cells of the L1–6 sympathetic nerve ganglia were evaluated. (b) An image of the microsized gelatin beads.
Each group underwent the same surgery on days 0 and 7. The FAC and SNC groups underwent surgery on day 0 and sham surgery on day 7. Skin temperature, objective findings (swelling, pallor, and nail color), and limb movement (ankle dorsiflexion and toe abduction) were evaluated every week. After 21 days of observation, 1 ml of sterile saline with medetomidine hydrochloride (0.15 mg/kg), midazolam (2.0 mg/kg), and butorphanol (2.5 mg/kg) was intraperitoneally administered. Under deep anesthesia, the rats were perfused via the aorta with saline and thereafter with Zamboni solution (2% paraformaldehyde with 0.2% picric acid in 0.1 M phosphate buffer). Leg muscles (extensor digitorum longus, soleus, and gastrocnemius) were collected and weighed. In addition, the histopathological assessment of the gastrocnemius muscles was performed and the left and right L1–6 sympathetic ganglia were collected and immunostained with a neuropathy marker (an activating transcription factor 3 (ATF3) antibody).
Microsized gelatin beads
Gelatin beads were prepared according to the method by Tabata. 8 Gelatin (0.2 g; Type No. GLS250, Lot No. 170221, Nitta Gelatin Co., Ltd., Osaka) was placed in 2.0 ml of distilled water and dissolved in a hot water bath. A gelatin solution was added dropwise to 35 ml of olive oil (Wako, Osaka, Lot: WDN5532) at approximately 40 °C, and the mixture was stirred with a stirrer for 1 h to prepare a water/oil emulsion. Finally, glycine was replaced with 0.1 M phosphate-buffered saline, and the size was controlled by passing through a 100 μm filter (cell strainer 100 μm, Falcon, USA). The number of prepared beads was calculated using a hemocytometer (Erma Burker-Turk deep 1/10 mm 03-303-4), and the size was measured using a microscope (Olympus IMT-2, Tokyo, Japan). As a result, the average diameter of microsized gelatin beads was 33.69 ± 26.47 μm, and the median value was 22.14 μm (shown in Figure 1(b)).
Surgical procedure
Rat was administered in subcutaneous injection of the mixture of medetomidine, midazolam, and butorphanol (0.15, 2.0, and 2.5 mg/kg, respectively). Under deep anesthesia, the skin was incised with scissors parallel to the inguinal ligament, and the arterial pulsation was confirmed in the femoral triangle. The left femoral vein and left femoral nerve were separated from the left femoral artery using a cotton swab and tweezers under a stereomicroscope (Olympus SZH10, Japan). In order for the microsized gelatin beads to reach the peripheral blood vessels of the foot, the injections were performed into the central site of the left femoral artery which bifurcates into the left saphenous artery and left popliteal artery.
Before inserting the needle, two threads were pulled at the central and peripheral sides of the injection site to temporarily stop the blood flow. The needle tip of a 1 ml syringe with a 27G needle (Terumo, Tokyo, Japan) was slightly bent and inserted into the left femoral artery to inject each solution. After the injection, hemostasis was firmly performed with a cotton swab. This process was performed twice, on days 0 and 7. Furthermore, the FAC group was prepared according to Couffinhal. 9 Briefly, the left femoral artery was ligated with two threads and in between cut with scissors. The SNC group was prepared according to well-established protocol. 10 ,11 A sham operation was performed 7 days after the artery excision, which was subsequently confirmed.
Skin temperature of the limb
The skin temperature of the sole was measured using thermography (Flir i7 Infrared, Compact Camera, FLIR, USA). The temperature was measured by placing the rat under 1.5–2.0% isoflurane (Pfizer, Tokyo, Japan) inhalation anesthesia in the prone position. After waiting for the temperature to stabilize in a stable position, the temperature was acquired three times, and the mean value was estimated.
Appearance of the limb
The swelling of the sole and the color of the sole and nail were observed, and images were acquired with a camera. The evaluation was performed by comparing the right and left limbs and scored according to the criteria in Table 1.
Criteria for sole swelling and sole and nail color.

Using the acquired images, the left limb was scored in four stages with respect to the right limb (control side).
Limb movement
Ankle dorsiflexion and toe abduction were observed during spontaneous joint movements. Normal rats, when their backs are held and their feet lifted vertically from the ground, strongly bend their ankle joints, which simultaneously causes their toes to abduct significantly. The evaluation was performed by comparing the left and right limbs and scored according to the criteria in Table 2.
Criteria for ankle dorsiflexion and abduction.

Using the acquired movie, the left limb was scored in three stages with respect to the right limb (control side).
Muscle weight
Right and left lower limb muscles (extensor digitorum longus, soleus, and gastrocnemius) were collected, imaged, and weighed, and the average value of the left-to-right muscle weight ratio was evaluated.
Histopathological assessment
The fixed medial gastrocnemius was divided into three parts; the proximal part was de-alcoholized with xylene and wrapped in a paraffin film. The specimens were cut into 5 μm sections. Hematoxylin and eosin (HE) staining was performed to assess myofibril cross-sectional area. The sections were observed and the images were captured under a light microscope. 11
Detection of damaged/hyperactivated neurons in sympathetic ganglia using ATF3
ATF3 belongs to the ATF/cyclic adenosine monophosphate basal reactive binding family of transcription factors and is called an adaptive-response gene whose activity is usually regulated by stress stimuli. ATF3 expression is rarely expressed normally 12 but is strongly expressed in damaged/stressed sympathetic ganglion neurons and glial cells.13,14 Therefore, the expression of ATF3 in sympathetic ganglia was examined.
Sympathetic ganglion immunofluorescence analysis was performed as previously published. 15 To investigate changes in the expression of ATF3 during stress loading, the total number of ATF3-positive cells was calculated. We analyzed an average of 6–9 sections per each ganglion. Dots were added into the positive cells on the microscopic images using Photoshop CC (Adobe, San Jose, CA, USA), and the number of cells was counted using ImageJ 1.50i (NIH, Bethesda, MD, USA).
Statistical analysis
Data are expressed as mean values ± standard deviation of the mean. Statistical analysis was performed using GraphPad Prism version 7 (GraphPad Software, Inc., San Diego, CA). Data were analyzed by one-way analysis of variance (ANOVA) with the post hoc Tukey's multiple comparison test or by the Kruskal–Wallis test with Dunn's multiple comparison test. The datasets analyzed during the present study are available from the corresponding author upon reasonable request.
Results
Changes in limb skin temperature
On day 0, there were no significant differences in temperature between the right and left limbs in each group. However, in the Low-dose, High-dose, and FAC groups, the skin temperature of the left limb significantly decreased compared with the right limb. In the NS and SNC groups, no significant differences were observed throughout (Figure 2).
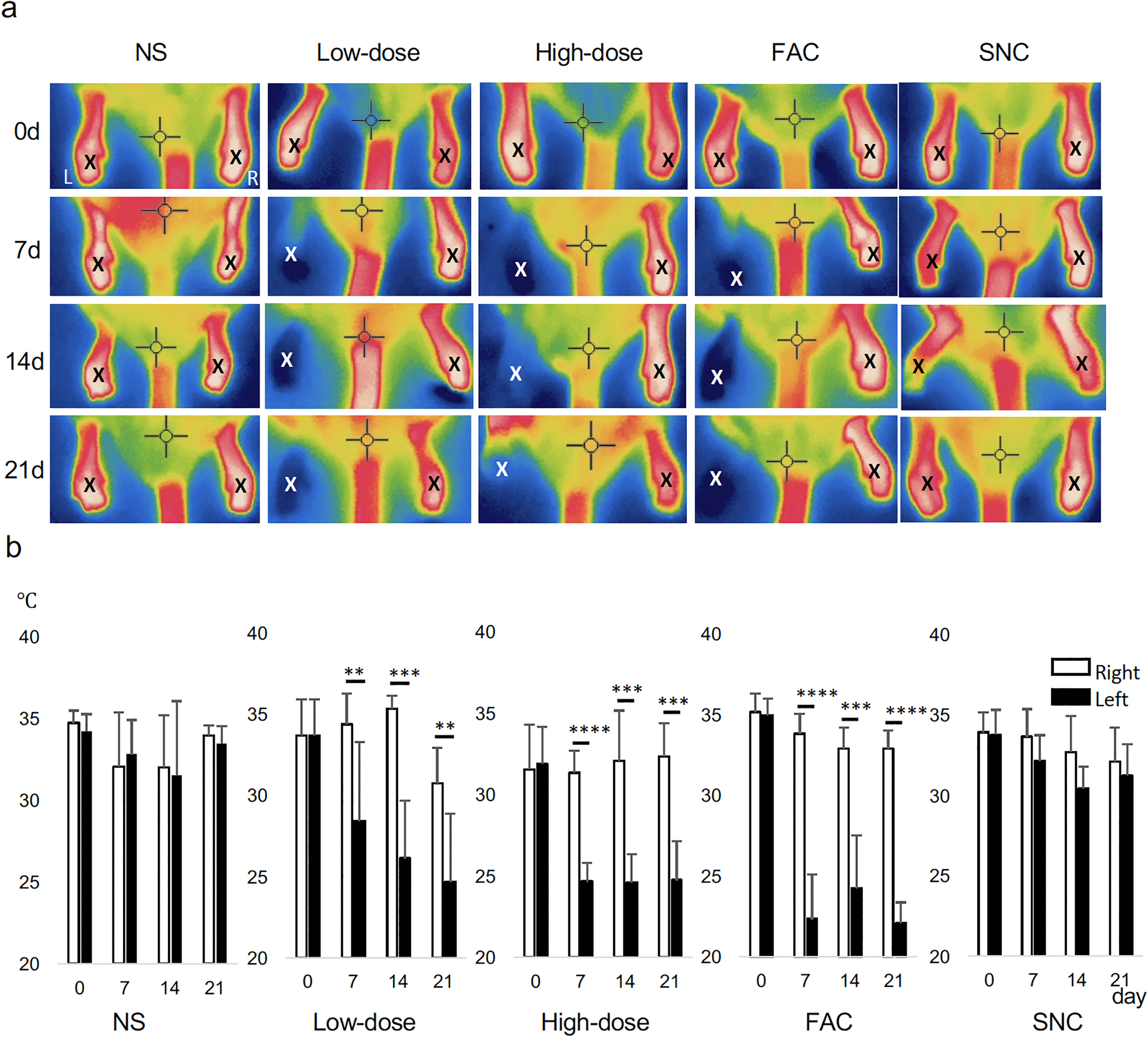
The limb skin temperature in each group. (a) A thermography image in the prone position. The temperature is displayed on the thermography monitor as a color image in seven levels ranging from −20°C to 250 °C. Low temperatures are displayed in blue, and high temperatures are displayed in red. (X) shows the point adjusted to the sole of the limb. (b) The temperature changes in each group every week. The data was acquired three times, and the mean value was calculated. A significant decrease was observed in the Low-dose, High-dose, and FAC groups. Data are expressed as mean values ± standard deviation of the mean. One-way analysis of variance with post hoc Tukey's multiple comparison test (*p < 0.05, ** p < 0.01, ***p < 0.001, ****p < 0.0001).
Changes in limb appearance
No change was observed in the swelling of soles between the NS, FAC, and SNC groups at all times. In the Low-dose group, a little swelling was observed compared to the NS group at 14 days, but there was no significant difference, and after 21 days, it was not swollen at all. In the High-dose group, swelling of the soles was observed after the first injection, and it continued on the 21st day (Figure 3).
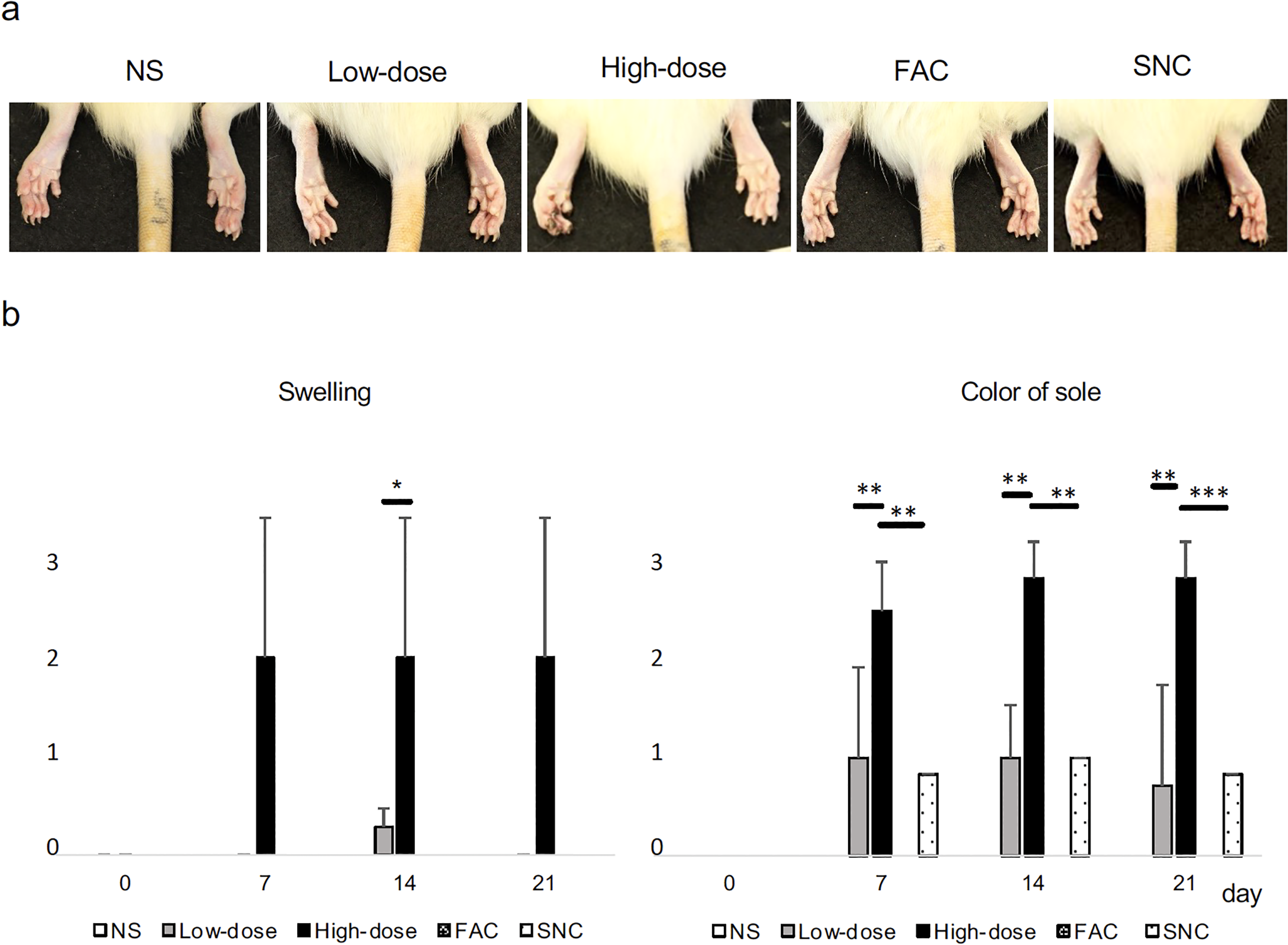
Swelling and color of sole in each group. (a) Images of the sole on day 21. (b) The scores of swellings and color of sole in each group. The appearance of the left limbs was evaluated in comparison with the right limbs according to the criteria in Table 1. Data are expressed as mean values ± standard deviation of the mean. One-way analysis of variance with post hoc Tukey's multiple comparison test; *, **, and *** indicate p < 0.05, p < 0.01, and p < 0.001, respectively.
The color of the sole was normal in the NS and FAC groups at all times. In the Low-dose group was whiter than the unaffected side after the first bead injection, but improved on the 21st day. In the High-dose group, a clear paleness was observed at the first injection, and necrosis was observed at the fingertips on the 21st day. The SNC group had a slightly white sole, 67% and white sole was 17% observed. Regarding nail color, the High-dose group had the most damage, followed by the Low-dose and SNC groups. Again, NS and FAC groups were no change observed (Figure 4).

Color of nail in each group. (a) Images of nail on day 21. (b) The scores of appearances in each group. Arrows indicate black nails. The appearance of the left limbs was evaluated in comparison with the right limbs according to the criteria in Table 1. Data are expressed as mean values ± standard deviation of the mean. Kruskal–Wallis test with Dunn's multiple comparison test. **, ***, and **** indicate p < 0.01, p < 0.001, and p < 0.0001, respectively.
Changes in ankle dorsiflexion and toe abduction
Foot dorsiflexion and toe abduction were normal at all times in the NS and FAC groups. There were only a few rats with weak dorsiflexion in the Low-dose group, but there was no difference from the FAC group. In the High-dose group, foot drop was observed in all cases after the first injection, and it continued 21 days later, which was different from the Low-dose group at any time. In the SNC group, ankle dorsiflexion and toe abduction were weaker at any time (Figure 5).

Limb movement in each group. (a) Images of the limbs on day 21. (b) The limb movement scores of in each group. The movement of the left limbs was evaluated in comparison with the right limbs according to the criteria in Table 2. Data are expressed as mean values ± standard deviation of the mean. Kruskal–Wallis test with Dunn's multiple comparison test. ** and *** indicate p < 0.01 and p < 0.001, respectively.
Changes in muscle weight
In the NS group, there were no significant differences in muscle weight between the left and right limbs, and the ratios were 0.97 ± 0.06, 1.01 ± 0.11, and 1.05 ± 0.02 for the extensor digitorum longus, soleus, and gastrocnemius muscle, respectively. There were no statistically significant differences in the weight ratio of the extensor digitorum longus and soleus among the NS, Low-dose, and FAC groups. The weight ratios of the extensor digitorum longus and soleus in the High-dose group were 0.66 ± 0.15 and 0.67 ± 0.08, respectively; these ratios were significantly decreased compared with those of the NS group (p < 0.01). The gastrocnemius weight ratios in the Low-dose, High-dose, and FAC groups were 0.82 ± 0.10, 0.58 ± 0.08, and 0.87 ± 0.03, respectively; these ratios were significantly lower compared with those of the NS group (p < 0.001, p < 0.0001, and p < 0.01, respectively). The ratio of three muscles of SNC was most decreased 0.54 ± 0.02, 0.44 ± 0.07, 0.46 ± 0.03 (p < 0.0001, p < 0.0001, and p < 0.0001, respectively).
As a result of HE staining, atrophy of muscle cells was observed in the Low-dose, High-dose, and SNC groups. When observing the entire tissue, the atrophy of the High-dose group was worse than that of the Low-dose group. However, it was difficult to quantify because it varies from place to place. No obvious muscle cell atrophy was observed in the FAC group, but the intercellular space was enlarged compared to the NS group. In the SNC group, expansion of intercellular spaces due to muscle cell atrophy was observed, and no nuclear abnormality was observed (Figure 6).

The limb muscles in each group. (a) Images of limb muscles. (b) The weight of each muscle. The left-to-right muscle weight ratio was evaluated. Data are expressed as mean values ± standard deviation of the mean. One-way analysis of variance with post hoc Tukey's multiple comparison test; **p < 0.01, ***p < 0.001, and ****p < 0.0001. (c) Hematoxylin and eosin staining of gastrocnemius muscle sections from each group at day 21. Bar:40 μm.
Sympathetic neuropathy marker
In both the NS and FAC groups, ATF3 expression was rarely observed in the L1–6 sympathetic ganglia (Figure 7). The number of ATF3-positive cells/section in the NS group was 0.37 ± 0.34, 0.14 ± 0.22, 0.22 ± 0.16, 0.16 ± 0.11, 0.23 ± 0.25, and 0.12 ± 0.18, respectively. The number of ATF3-positive cells/section in the FAC group was 0.58 ± 0.56, 0.16 ± 0.12, 0.21 ± 0.13, 0.41 ± 0.42, 0.27 ± 0.29, and 0.08 ± 0.08, respectively. Conversely, in the Low-dose group, the number of ATF3-positive cells/section in the L1–6 sympathetic ganglia was 1.36 ± 1.30, 0.60 ± 0.90, 1.38 ± 2.09, 0.39 ± 0.34, 0.13 ± 0.13, and 0.10 ± 0.10, respectively. There were no significant differences between the NS and Low-dose groups; however, the numbers slightly increased in the L1 and L3 sympathetic ganglia. In the High-dose group, the number of ATF3-positive cells/section in the L1–6 sympathetic ganglia was 3.52 ± 1.57, 10.08 ± 4.05, 37.65 ± 9.86, 10.71 ± 2.67, 0.40 ± 0.28, and 0.14 ± 0.14, respectively; there were significant increases in the L2, L3, and L4 sympathetic ganglia compared with the NS group (p < 0.05, p < 0.0005, and p < 0.0005, respectively). The SNC group was 3.05 ± 2.28, 1.15 ± 0.67, 30.12 ± 10.52, 25.85 ± 13.23, 2.23 ± 2.12, and 0.45 ± 0.35 (p < 0.05, p < 0.0005, and p < 0.0005, respectively).

The appearance of an activating transcription factor 3 (ATF3)-positive signal in the sympathetic ganglia of the L1–6 of each group. (a) Images of ATF3 immunoreactivity in the L3 sympathetic ganglia of each group. Almost no expression of ATF3 was observed in the normal saline (NS) and femoral artery cut (FAC) groups, but significant expression was observed at the sympathetic ganglion level in the Low-dose and High-dose groups. White arrowheads indicate ATF3-positive cells. The square-shaped enlarged photograph is attached to the figures. (b) The number of ATF3-positive cells in each group. Data are expressed as mean values ± standard deviation of the mean. One-way analysis of variance with post hoc Tukey's multiple comparison test; *p < 0.05 and ***p < 0.005.
Discussion
In this study, we established a rat model of ALI using microsized gelatin beads. This model showed that the degree of ALI symptoms can be adjusted by adjusting the number of beads. The ALI model with microsized gelatin beads and SNC model caused sympathetic ganglion damage, however, the FAC model did not. This is the first report comparing the FAC and SNC models with the ALI model.
Previous FAC mouse and rat models, which have been mainly used as models for PAD, have shown that the lower extremities do not become necrotic despite ligating and cutting the femoral artery.9,16,17 According to a detailed investigation of the blood flow in the lower limbs of the mouse, it has been clarified that the blood flow in the lower limbs of the mouse is nourished by the iliac artery in addition to the femoral artery. 18 Therefore, even if the femoral artery is ligated or cut the iliac artery can nourish the lower limbs. Unthank et al. 19 demonstrated that considerable adaptation occurred in collateral pathways during the first week after femoral artery ligation. It was also found that vascular endothelial growth factor expression augmented from day 4 after femoral artery ligation and the peak was on day 7. 9 In fact, our preliminary experiment revealed that only one injection of microsized gelatin beads does not cause stable symptoms. Therefore, it is presumed that the second administration after 1 week is important for stabilizing the degree of ALI damage.
Gelatin sponge is made by processing gelatin extracted from bovine or porcine skin or ligaments into a porous structure; this was developed as a surgical hemostatic material for medical applications, and its use began in 1945. 20 In 1967, the use of gelatin sponge as an intravascular embolic substance for carotid cavernous fistula was reported for the first time. 21 Since then, it has been widely used for hematological malignancies such as hepatocellular carcinoma 22 and trauma for vascular rupture lesions. 23 Tokunaga et al. 24 described establishing a rabbit ALI model by injecting 15 μm gelatin microspheres into the common iliac artery and then ligating and cutting the femoral artery. Recently, Smriti et al. (2020) reported that an ALI model was induced in male apolipoprotein E-deficient mice by a two-stage procedure of initial gradual femoral artery occlusion by ameroid constrictors for 14 days and subsequent excision of the femoral artery. 25 In our study, an ALI model was established by injecting microsized gelatin beads twice on days 0 and 7 without ligating and cutting the femoral artery. Our model has the advantage that the degree of ALI damage can be adjusted by changing the amount of gelatin beads injected. It is known that ALI in clinical patients is recurrent and gradually shifts to PAD. 26 Therefore, our model is considered to more closely mimic the clinical state of ALI compared with the previously reported models.
In our experiments, the temperature of limb skin decreased in the Low-dose, High-dose, and FAC groups. The sympathetic nerves are known to innervate blood vessels 27 and muscles, 2 and a decrease in skin temperature is thought to be associated with dysfunction of the muscles and sympathetic nerves. Tokunaga et al. 2 implanted dialysis probes in the adductor muscle of anesthetized rabbits and measured dialysate norepinephrine levels as an index of muscle sympathetic nerve activity in an ALI rabbit model. As a result, the ALI caused a decrease in norepinephrine levels. Therefore, our ALI model also may impair axonal conduction and/or the norepinephrine-releasing function at sympathetic nerve endings in skeletal muscle. A previous study retrogradely labeled the skeletal muscle of the rat hindlimb with horseradish peroxidase to elucidate segmental distributions of sympathetic neurons projecting in hindlimb nerves and it was found that the sympathetic chain ganglia either in L2 and L3 or in L3 and L4 distributed to the gastrocnemius and soleus muscles. 28 From these facts, it is possible that some sympathetic nerve damage is caused by the decrease in sole temperature and the decrease in gastrocnemius muscle weight in the three groups shown in our results, which causes expression of ATF3. In the High-dose group of our ALI model, the muscle weight of the extensor digitorum longus, soleus, and gastrocnemius decreased with abnormal ankle movement compared to the NS group. Histologically, a similar tendency was observed in the gastrocnemius. The number of ATF3-positive cells in the sympathetic ganglia significantly increased in the High-dose and SNC groups compared to the NS group. In the Low-dose group of this study, a few ATF3-positive cells were found, but there was no statistically significant difference. On the other hand, FAC group had also gastrocnemius damage, but no ATF3-positive cells were observed. These findings suggest that autonomic neuropathy in ALI is related to both muscle damage and peripheral neuropathy.
This study revealed that the ALI, FAC, and SNC models had different characteristics in skin temperature, physical activity, muscle weight, and sympathetic dysfunction, respectively. These results suggest that the developing mechanism of the ALI model using microsized gelatin beads is completely different from the FAC and SNC models. Clinically, lumbar sympathectomy has been shown to reduce symptoms of critical limb ischemia in patients who suffer from nonreconstructable PAD. Nevertheless, it has also been suggested that it may incur more adverse effects. 29 Furthermore, one study has reported that acupuncture and hyperthermia are effective in stimulating the autonomic nerves in PAD. 27 However, the current evidence is insufficient to determine the relationship between sympathetic neuropathy and PAD. We suppose that the pathology of human PAD is likely to be a combination of ALI and chronic ischemia, and the process leading to ischemia is closely related to sympathetic ganglion damage. Further studies are needed to better understand the relationship between sympathetic ganglion damage and ischemia.
Although this study showed a rat model of ALI using microsized gelatin beads for the first time, there are some limitations. According to the limited use of animals in the intervention experiment, sample size analysis was not performed for this study. However, post hoc power analysis demonstrated that the sample size of our main experiment is almost appropriate (Power (1-β err prob) = 0.88–1.0). Also, different results may be obtained by varying the size and number of beads administered in the experiments. More studies using this ALI model will help to elucidate the pathogenesis of ALI.
Conclusion
We have developed a novel ALI rat model using microsized gelatin beads twice without cutting the femoral artery. This model adequately represents clinical symptoms and exhibits sympathetic ganglion damage. The results of this study suggest that autonomic neuropathy in ALI is related to both muscle damage and peripheral neuropathy.
Footnotes
Acknowledgement
The authors wish to thank Mr Junji Goto and Ms. Yoshiko Kunita for their excellent secretarial and technical assistance.
Author contributions
YM, MN, KF, and MY were involved in study conception; YM and NH in data collection; MY, KF, and NY in experimental skill; YM, KF, and TY in analysis; YM and MN in writing. MN in funding acquisition; and MN in critical review and revision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants-in-aid for Scientific Research (17H04274, 22H03980) from Japan Society for the Promotion of Science (JSPS), KAKENHI.
Author biographies
Yuki Menjo is a student at Graduate School of Medicine. Her area of research is clinical anatomy.
Masaya Yasui is an assistant professor in Integrated Studies of Human Development and Clinical Psychology. His area of research is clinical anatomy. He holds a PhD in Medicine.
Kaori Fukushige is an assistant professor in Anatomy. His area of research is clinical anatomy. She holds a PhD in Pharmacy.
Naoyuki Hatayama is an assistant professor in Anatomy. His area of research is clinical anatomy. He holds a PhD in Medicine.
Tomiko Yakura is an assistant professor in Anatomy. His area of research is clinical anatomy. She holds a PhD in Medicine.
Munekazu Naito is a professor in Anatomy. His area of research is clinical anatomy. He holds a PhD in Medicine.
