Abstract
Gastric adenocarcinoma of the fundic gland type (GA-FG) is a rare gastric neoplasm. We present a unique case of multiple GA-FG that coexisted with the well-differentiated neuroendocrine tumors in a patient with autoimmune gastritis. To our knowledge, this is the first documented instance of such a co-occurrence and the molecular mechanism of their origin has been reviewed systematically. A 47-year-old male presented to our hospital with abdominal distension for over 10 years. Gastroscopy revealed multiple gastric eminence lesions (0.2–1.5 cm). After endoscopic mucosal resection, the pathological morphology showed mixed tumor components infiltrating into the submucosa with puzzling similarity. One with uniform-sized tumor cells arranged in nests or tubes and the other a well-differentiated tubular adenocarcinoma with irregular branching and visible gland fusion. Immunohistochemistry findings revealed the first component expressed typical markers of neuroendocrine tumor, whereas the second component expressed pepsinogen and mucin-6, indicating the presence of oxyntic gland adenocarcinoma. Due to the tumors’ proximity to the surgical margins, the patient underwent laparoscopic subtotal gastrectomy three months after the diagnosis without any tumor residue and showed no recurrence or metastasis occurred in the following regular checkups.
Introduction
Gastric adenocarcinoma of the fundic gland type (GA-FG) is a highly differentiated adenocarcinoma that differentiates into the chief and parietal cells of the fundic gland. As a rare and low-grade malignant subtype of gastric adenocarcinoma; GA-FG accounts for 1% of patients with early gastric cancer who undergo endoscopic mucosal resection. 1 In 2010, Ueyama et al. 2 defined GA-FG based on their investigation of clinicopathological features in 10 patients with gastric adenocarcinoma characterized by chief cell differentiation. According to the latest World Health Organization (WHO) Classification of Digestive System tumors, 3 these lesions are known as oxyntic gland adenoma when confined within the mucosa, whereas those infiltrating into the submucosa or deeper are defined as GA-FG/oxyntic gland adenocarcinoma. GA-FG can be characterized into three subtypes based on different cell compositions: chief cell predominant type, parietal cell dominant type, and mixed type. The chief cell predominant type is the most common, accounting for ∼ 99% of reported cases. 4 Despite the gradual increase in awareness with regard to GA-FG, a few instances still pose a challenge in terms of accurate diagnosis.
GA-FG has typically been considered a solitary tumor originating in the non-atrophic mucosa of the upper or middle part of the stomach. 1 However, we recently encountered a unique case involving a patient with multiple lesions of GA-FG that arose in the context of autoimmune atrophic gastritis. Of particular interest in this case is the presence of both fundus-type adenocarcinoma and well-differentiated neuroendocrine tumor within the same lesion, an extremely rare occurrence that has not been reported previously.
Thus, we present a case report of GA-FG mixed with a well-differentiated neuroendocrine tumor in a Chinese patient. Given the endoscopically and pathological similarity of the mixed components, the diagnosis was highly complex. This case further expands our knowledge of this type of disease and highlights the need for continued research in this area.
Clinical summary
A 47-year-old man with abdominal distension for > 10 years visited our hospital in Dongcheng District, Beijing, on 24 December 2021. Gastroscopy performed at a local hospital four months ago had revealed atrophic gastritis and multiple gastric eminence lesions (0.2–1.5 cm) in the gastric body with infiltration into the submucosa. The largest lesion (1.5 cm in diameter) was located in the upper third of the stomach with branching dilated blood vessels as well as superficial ulceration on the surface (Figure 1A to C). Following the endoscopic mucosal resection (EMR) of these eminence lesions (Figure 1D), a diagnosis of the gastric neuroendocrine tumor was made at the local hospital.
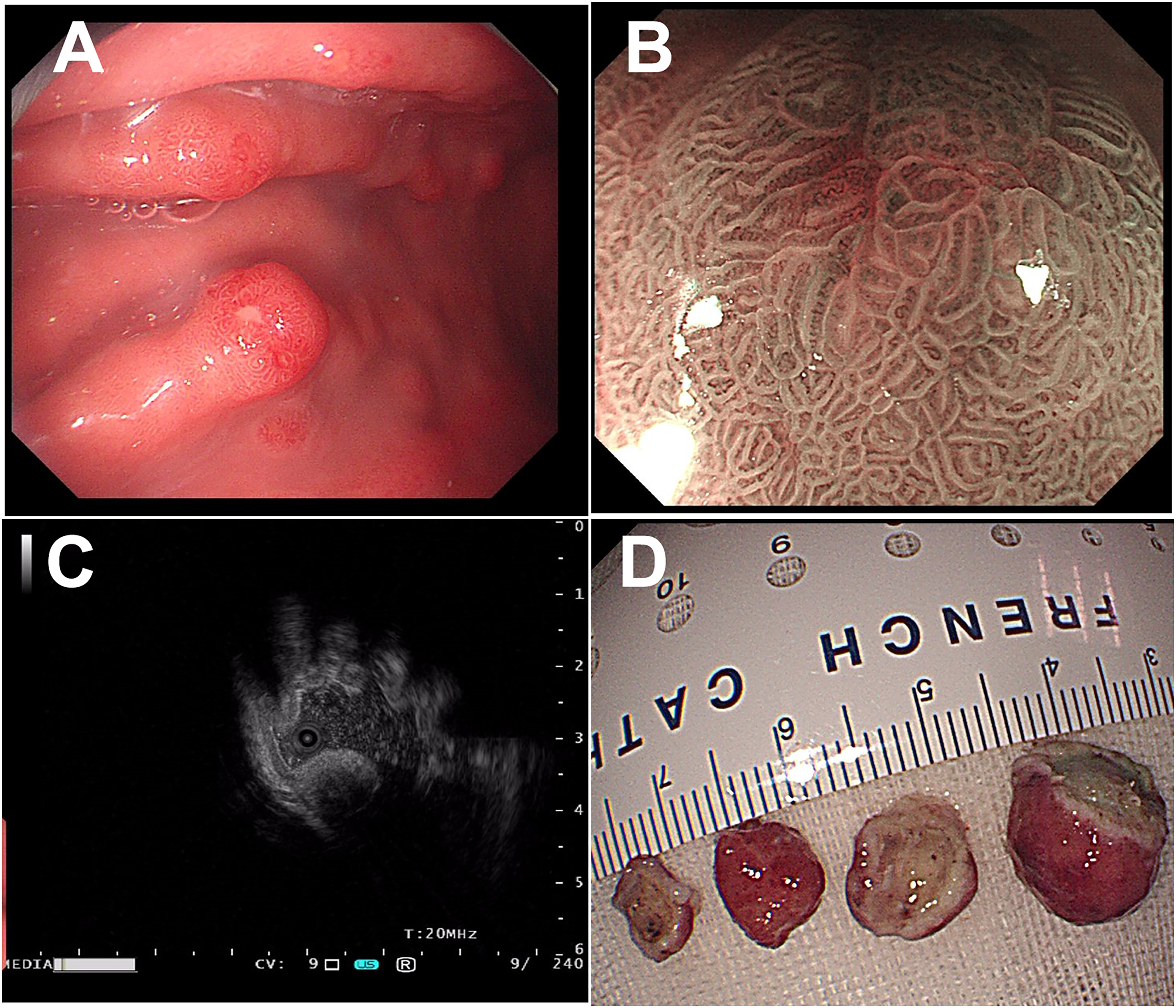
Gastroscopy manifestations of the gastric eminence lesions. (A) Gastroscopy revealed atrophic gastritis and multiple gastric eminence lesions. (B) Magnifying gastroscope revealed the typical lesions with superficial hyperemia and superficial ulceration. (C) The ultrasound gastroscope showed that the lesion was infiltrated into the submucosa. (D) Endoscopic mucosal resection was performed.
Due to persistent symptoms of abdominal distension and concerns about the tumor, the patient sought a consultation at our hospital. He denied any medication assumptions including proton pump inhibitor (PPI) and had never received noninvasive testing of Helicobacter pylori (H. pylori). The results of the laboratory tests conducted at our hospital revealed a significant elevation in gastrin levels (895 pg/mL) and the presence of anti-gastric parietal cell antibody (+) 1:640. Based on these findings, it is highly indicative of autoimmune atrophic gastritis.
Pathological findings
The pathological examination revealed that these tumors were composed of two distinct components (Figure 2A). The first component was a well-differentiated neuroendocrine tumor, microscopically characterized by uniformly sized tumor cells arranged in nests or tubes and surrounded by fibrovascular tissue (Figure 2B). The tumor cells displayed round or oval-shaped nuclei, some with small nucleoli, and a “salt-and-pepper” chromatin pattern. This component extended into the lamina propria and muscularis mucosa and invaded the submucosa. The mitotic rate was 0–1 per 10 HPF (Figure 2C).

Microscopic manifestations of mixed neuroendocrine tumor-gastric adenocarcinoma of the fundic gland type. (A) Light micrograph of a hematoxylin–eosin-stained resected tissue specimen of the nodular tumor, involving the lamina propria and muscularis mucosa, invading the submucosa; most of the surface area was covered by the gastric mucosa (scale bar = 6 mm). (B) Well-differentiated neuroendocrine tumor components were present in the tumor tissue (scale bar = 500 µm). (C) At high magnification, the tumor cells were composed of uniform-sized neuroendocrine tumor cells arranged in nests or glands. The nests were rich in fibrovascular tissue. The tumor cells had round or oval-shaped nuclei with small nucleoli and spotty, salt-and-pepper chromatin (scale bar = 50 µm). (D) In the local area of the tumor tissue, the adenocarcinoma component of the fundic gland was seen, arising from the deep mucosa and showing irregular branching or glandular fusion (scale bar = 500 µm). (E) The tumor cells in the adenocarcinoma of the fundic gland showed no obvious atypia; the cytoplasm was basophilic (black star), similar to the fundic gland cells; and neuroendocrine tumor cells (triangle) were seen among them (scale bar = 100 µm). (F) Mucosal atrophy, chronic inflammatory cell infiltration, and intestinal metaplasia were observed in the lamina propria of the surface mucosa (scale bar = 100 µm).
The second component was a well-differentiated tubular adenocarcinoma, resembling normal fundic glands but larger, with irregular branching and visible gland fusion. Chief cells were the predominant cell type, and most tumor cells had visible nucleoli. Mitoses occurred at 1–2 per 10 HPF without necrosis (Figure 2D and E). This component invaded the submucosa and was covered by normal gastric pit epithelium.
The neuroendocrine tumor component accounted for 70% of the main tumor, whereas the adenocarcinoma component contributed to the remaining 30%. In some areas, there was a mixed growth of these components (Figure 2E), making their distinction challenging. It was noted that the tumor component was near surgical margins but no definitive neural or vascular invasion was observed. Besides, the significant presence of lymphocytes and plasma cells was noticed in the surface lamina propria of the gastric mucosa, especially in the deeper region near the lamina propria. The oxyntic gland was significantly reduced, accompanied by moderate intestinal metaplasia and nodular hyperplasia of neuroendocrine cells (Figure 2F).
Immunohistochemical testing showed positive expression of cytokeratin in both tumor components (Figure 3A). Chromogranin A (CgA) (Figure 3B) and somatostatin receptor 2 (Figure 3C) displayed diffuse positive in the tumor cells of the neuroendocrine tumor component; while pepsinogen was negative (Figure 3D). The Ki-67 proliferation index of this component was 1% (Figure 3E). In contrast, the well-differentiated adenocarcinoma component expressed pepsinogen and mucin-6 (MUC6) (Figure 3F and G). CgA was nearly negative (Figure 3H), and the Ki-67 proliferation index was 5% (Figure 3I). Linear and nodular hyperplasia of neuroendocrine cells in the surface gastric mucosa were evident with CgA staining (Figure 3J). Besides, the staining for H. pylori in multiple biopsies was negative. The final pathological diagnosis was gastric mixed neuroendocrine–non-neuroendocrine neoplasm (MiNEN), with the neuroendocrine component identified as the neuroendocrine tumor (NET, Grade 1) and the non-neuroendocrine component as fundus adenocarcinoma (highly differentiated, Lauren type, intestinal type). Additionally, the diagnosis of autoimmune atrophic gastritis was confirmed through serologic and histologic analysis.

The results of immunohistochemical staining. (A) Cytokeratin is positive in the neuroendocrine tumor cells. (B) Chromogranin A and (C) somatostatin receptor 2 is positive in the typical cell of a neuroendocrine tumor. (D) Pepsinogen is negative in the neuroendocrine tumor cells. (E) The Ki-67 proliferation index in the neuroendocrine tumor was 1%. (F) Pepsinogen and (G) mucin-6 (MUC6) were highly expressed in the fundic adenocarcinoma component of the tumor. (H) There was almost no expression of chromogranin A in the fundic adenocarcinoma tumor cells. (I) The Ki-67 proliferation index was 5% in the fundic adenocarcinoma component. (J) Chromogranin A was positive at the site of endocrine cell proliferation in the gastric mucosa.
Discussion
The coexistence of a well-differentiated neuroendocrine tumor (type-1 gastric neuroendocrine tumors) with GA-FG is an exceedingly rare phenomenon. The accurate diagnosis was achieved by complete resection of the lesions and considering the combined clinical as well as serological manifestations. Several distinctive features were noted in our case: First, the simultaneous occurrence of mixed growth of fundic gland type adenocarcinoma and the neuroendocrine tumor. Second, the presence of multiple GA-FG in the context of autoimmune gastritis. Third, the well-differentiated tendency of both components.
GA-FG is already relatively rare, but its co-occurrence with neuroendocrine tumors is even rarer. Besides, GA-FG and neuroendocrine tumors present similar endoscopic and histological features. On endoscopy, both tumors appeared as submucosal tumors with dilated vessels in the gastric body.1,5 Histologically, both tumors are composed of small round tumor cells and originate from deep layers. Moreover, recent studies have shed light on the fact that certain GA-FG tumors may exhibit varying degrees of positivity for CD56 and synaptophysin immunohistochemical staining, which have traditionally been employed as the markers of neuroendocrine tumors. 6 Notably, MiNEN with adenocarcinoma components can present a significant challenge to physicians and pathologists due to the invasive potential. Accurately distinguishing between these two types of tumors is crucial for effective treatment strategy and evaluation of therapeutic effect. In this case, the specific expression of pepsinogen and MUC6 in GA-FG, coupled with the absence of CgA provides a clear distinction. These immunohistochemical features were consistent with previous literature.1,7
Another prominent feature of this case was the coexistence of neuroendocrine tumor and GA-FG in the context of autoimmune atrophic gastritis. Chronic autoimmune atrophic gastritis (CAAG) has been linked to type-1 gastric neuroendocrine tumors. 8 However, GA-FG is not common in cases of CAAG. 9 Previous literature has shown that no chronic gastritis, atrophic changes, or intestinal metaplasia were seen in the surrounding mucosa of 31 cases of GA-FG. 10 Interestingly, recent research has revealed that CAAG is associated with gastric adenocarcinomas due to moderate to severe atrophic gastritis and the presence of intestinal metaplasia.11,12 Mechanically, mature gastric chief cells have progenitor cell potential and can regain their proliferation capacity under specific conditions (such as mucosal injury or inflammation). 13 The loss of parietal cells is sufficient to activate progenitor cell potential and induce the trans-differentiation of chief cells, 14 which is a process considered to be closely related to the pathogenesis of gastric cancer. Therefore, it is worth exploring whether the development of fundic adenocarcinoma in CAAG is a direct consequence of parietal cell loss or a matter of coincidence. It is noteworthy that our case did not involve H. pylori infection and PPI assumption, despite their recognition as risk factors for aggravating gastric mucosal atrophy and cancerization.15–18
The origin of tumor tissue differentiation in MiNEN-containing GA-FG components remains unclear. MiNEN is believed to stem from pluripotent stem cells with bidirectional differentiation potential, 19 although it may have an independent origin. The fundic gland is considered to originate from the normal gastric mucosa in the fundic gland region. GA-FG development is associated with the Wnt/β-catenin signaling pathway and GNAS mutations. 10 While the dedifferentiation of GA-FG is presently controversial. GA-FG with signet ring cell carcinoma components can express MUC5A, a marker of foveal epithelium, suggesting that cell differentiation from the fundic gland type to the foveal type during the progression of the disease, thus supporting the potential of GA-FG transformation. 20 Moreover, mixed adenoneuroendocrine carcinoma combined with GA-FG and poorly differentiated neuroendocrine carcinoma has an overlapping gene mutation spectrum of the two components, including FGFR3, EGFR, MET, and TP53 mutations, suggesting that the two components are simultaneously differentiated from monoclonal pluripotent epithelial stem cells. 21
Given the rarity of GA-FG mixed with neuroendocrine tumors, there is limited experience surrounding prognosis and treatment. A case reported by Nassereddine et al. 21 in 2017 described poorly differentiated neuroendocrine carcinoma coexisting with GA-FG in a 69-year-old man with a history of chronic smoking, alcohol abuse, myocardial infarction, and peripheral arterial occlusion. Gastroscopy revealed a 12-mm sessile polypoid lesion located at the greater curvature of the stomach. The patient's prognosis was unfavorable, and he succumbed to sepsis and multiple organ failure two months after the diagnosis. Our report, on the other hand, represents the first instance of a well-differentiated neuroendocrine tumor mixed with GA-FG in the available literature. Despite the typically indolent clinical behavior of these tumors, 22 aggressive type 1 gastric carcinoid has been reported (2% die of metastases). 23 In addition, while large case reports of GA-FG have generally suggested a favorable prognosis,6,10 6% of GA-FGs invade the serosal membrane and spread to blood vessels. 4 Therefore, identifying patients with poor prognosis is a critical task that requires large cohort studies. In the present case, given the tumors’ proximity to the surgical margins and the presence of multiple lesions, it is imperative to proceed with caution and take additional measures to ensure complete removal of the tumors. Consequently, the patient underwent laparoscopic subtotal gastrectomy three months after the diagnosis. Fortunately, no residual tumor was found in the patient's surgical specimen, and there were no signs of recurrence or metastasis during subsequent regular checkups.
In conclusion, we present the first documented case of the well-differentiated neuroendocrine tumors mixed with the GA-FG component in the Chinese population. The patient has been closely followed up till now, and the prognosis is good. As more cases accumulate, further analysis of patient prognosis and prognostic factors will provide valuable insights.
Supplemental Material
sj-pdf-2-sci-10.1177_00368504231220765 - Supplemental material for A puzzling case report of well-differentiated neuroendocrine tumor mixed with gastric adenocarcinoma of the fundic gland type associated with autoimmune gastritis
Supplemental material, sj-pdf-2-sci-10.1177_00368504231220765 for A puzzling case report of well-differentiated neuroendocrine tumor mixed with gastric adenocarcinoma of the fundic gland type associated with autoimmune gastritis by Zheng Wang, Weixun Zhou, Jingnan Li, Wenlong Wen, Zhiyong Liang and Zhen Huo in Science Progress
Supplemental Material
sj-pdf-3-sci-10.1177_00368504231220765 - Supplemental material for A puzzling case report of well-differentiated neuroendocrine tumor mixed with gastric adenocarcinoma of the fundic gland type associated with autoimmune gastritis
Supplemental material, sj-pdf-3-sci-10.1177_00368504231220765 for A puzzling case report of well-differentiated neuroendocrine tumor mixed with gastric adenocarcinoma of the fundic gland type associated with autoimmune gastritis by Zheng Wang, Weixun Zhou, Jingnan Li, Wenlong Wen, Zhiyong Liang and Zhen Huo in Science Progress
Footnotes
Acknowledgements
The authors would like to express our appreciation to the patient and the Department of Gastroenterology at Liaocheng People's Hospital for their collaboration in the current report. In addition, the authors thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Authors’ contributions
ZW collected the data and pictures, completed the follow-up information, and wrote the main manuscript. ZH finished the pathological diagnosis of the patient and revised the manuscript. WXZ, JNL, ZYL, and WLW participated in the diagnosis or treatment of the patient and provided valuable suggestions for the manuscript. The author(s) read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The studies were approved by the Ethics Review Committee of the Peking Union Medical College Hospital (Ethics Certificate No. I-23ZM0025). Written informed consent was obtained from the subject as well as a legally authorized representative(s) for anonymized patient information to be published in this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National High-Level Hospital (grant number PUMCH-B-063).
Supplemental material
Supplemental material for this article is available online.
Author biographies
Zheng Wang holds an MD in Gastroenterology. Her area of research is the pathology of gastrointestinal tumors and inflammatory diseases.
Weixun Zhou is a professor of Pathology. Her area of research is the Pathology of digestive system illnesses.
Jingnan Li is a professor of Gastroenterology. His area of research is the screening and prevention of colorectal cancer, inflammatory bowel disease (IBD), and the diagnosis and treatment of neuroendocrine tumors.
Wenlong Wen is an attending physician in Gastroenterology. Her area of research is inflammatory bowel disease (IBD) and Gastrointestinal tumor.
Zhiyong Liang is a professor of Pathology. His area of research is tumor pathology and molecular pathology, particularly for breast and endocrine system illnesses.
Zhen Huo is a professor of Pathology. Her area of research is tumor pathology, particularly for central nervous system illnesses.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
