Abstract
Objectives
Guidewire occasionally creates a loop-like appearance between the right subclavian artery and brachiocephalic artery when performing coronary angiography (CAG) with right radial artery (RtRA) access. We called this occurrence a looped guidewire at the brachiocephalic artery (looped GW at BA). It is associated with difficulties in catheter manipulation. This study aimed to assess the predictors of forming a looped GW at the BA.
Methods
We examined 175 (mean age, 71.3 ± 9.5 years; 74.9% men) consecutive patients who underwent elective CAG with the RtRA access. Looped GW at the BA was defined as a loop-like appearance of the 0.035-inch GW between the right subclavian and brachiocephalic artery on a frontal view or left anterior oblique. To identify the predictors of looped GW at BA, patient characteristics and examination data obtained before CAG were compared between patients with and without looped GW at the BA.
Results
The prevalence of looped GW at BA was 10.9%. The cardio-ankle vascular index (CAVI), which reflects arterial stiffness, was significantly different in patients with or without looped GW at BA (9.8 ± 1.0 vs. 8.9 ± 1.5, p = 0.0092). The area under the receiver-operating characteristic curve of CAVI to predict looped GW at BA was 0.745, with 0.94 sensitivity and 0.57 specificity in a cutoff point of CAVI ≥9.0.
Conclusions
Looped GW at BA can be ruled out by CAVI and is associated with high arterial stiffness.
Keywords
Introduction
Trans-radial intervention (TRI) is a common percutaneous coronary intervention procedure that is increasing in importance. Previous studies have revealed a lower incidence of bleeding complications in TRI than transfemoral intervention (TFI). 1 TRI has been recommended in both elective and emergency situations such as acute coronary syndrome (ACS). 2
Coronary angiography (CAG) and subsequent TRI are available via both the right radial artery (RtRA) and left radial artery (LtRA). The choice of RtRA or LtRA access depends on the operator's preference and the situation. We considered shorter procedure time and less radiation exposure and contrast material to be favorable within the same type of procedure.
The LtRA access has the advantage of providing a catheter back-up force similar to the TFI. Because operators must stand on the right side of the patient, the access point is on the far side of the patient's body. Therefore, LtRA access is associated with complex catheter manipulation occasionally depending on the body size of operators and patients: for example, small operators and large patients. Operators must stand closer to the site of the image intensifier, which leads to increased radiation exposure.
In RtRA access, guidewire occasionally draws a loop like appearance between the right subclavian artery and brachiocephalic artery (Figure 1). We called this the occurrence of looped guidewire at the brachiocephalic artery (hereinafter, referred to as looped GW at BA). Looped GW at BA is one of the causes of difficulties in catheter manipulation with RtRA access, such as forward, back, and rotational movements, which are related to prolonged procedure time, increased radiation exposure, and contrast material.
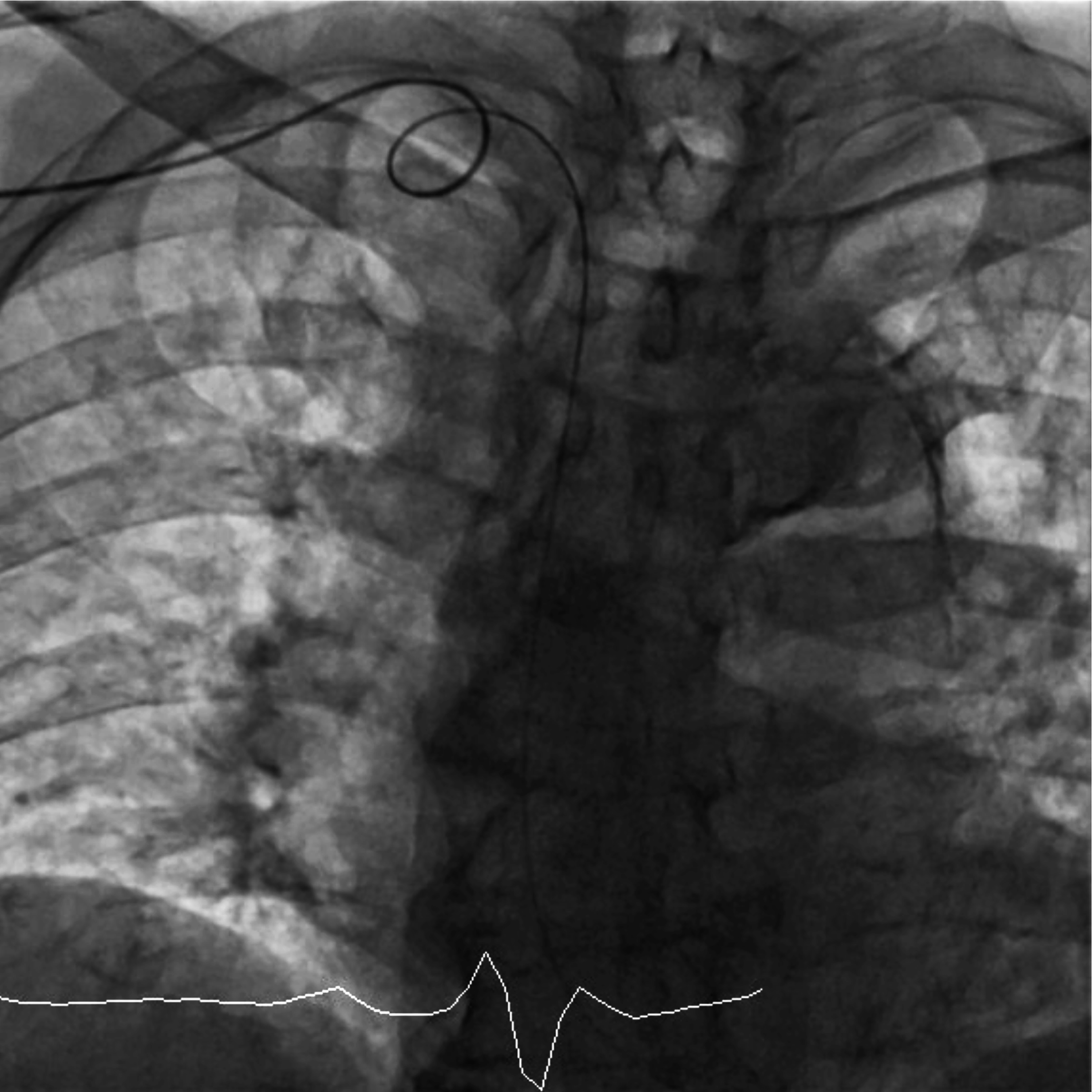
The 0.035-inch Radiforcus® guidewire (Terumo) drew a loop-like appearance between the right subclavian and brachiocephalic artery on a frontal view in one patient who underwent coronary angiography.
If the formation of looped GW at BA is predicted using patient characteristics and examination data obtained before CAG, we can gain additional insight into selecting RtRA or LtRA access. To the best of our knowledge, few studies have focused on looped GW at BA in patients who underwent CAG with RtRA access. This study aimed to assess the predictors of looped GW at BA.
Patients and methods
Study design and patient population
This single-center, retrospective observational study was designed to investigate the predictors of looped GW at BA when performing CAG with RtRA access. This study included 175 patients who met the inclusion and exclusion criteria (mean age, 71.3 ± 9.5 years; 74.9% men). The inclusion criteria were patients who underwent CAG with RtRA access at the Nagano Matstushiro General Hospital from January 2015 to December 2020. Exclusion criteria were emergent CAG, history of peripheral artery disease (PAD) and/or ankle brachial pressure index (ABI) < 0.9, and dialysis. Because LtRA access was suitable for lower extremity angiography subsequent CAG, we excluded the patients with or suspected PAD in this study.
Looped GW at BA was defined as a loop-like appearance of the 0.035-inch Radiforcus® guidewire (Terumo, Tokyo, Japan) between the right subclavian and brachiocephalic artery on a frontal view or left anterior oblique view, when this GW passed to the ascending aorta. This definition does not include a momentary loop-like appearance relieved by guidewire passing.
As usual, we recorded the presence or absence of looped GW at BA in the medical records. We retrospectively reviewed patient records and collected clinical data. Patient characteristics and examination data obtained from medical records were compared between the patients with and without looped GW at BA. Examination data included a blood sample, electrocardiogram, ultrasonic cardiogram, ABI, and cardio-ankle vascular index (CAVI). ABI and CAVI were measured by VaSera VS 1500A® (Fukuda Denshi, Tokyo, Japan). ABI and CAVI values are the means of the right and left sides in this study.
We measured atrial fibrillation and left ventricular hypertrophy with electrocardiography, and ejection fraction, E/e’ (mean of septal and lateral), and moderate or severe aortic stenosis on the ultrasonic cardiogram.
Written informed consent from the participants was waived because the retrospective study was based on existing medical records obtained during routine medical care. The study design was developed in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Nagano Matsushiro General Hospital (approval number: 021-3).
Definition
Body mass index (BMI) was calculated as the weight in kilograms divided by the square of height in meters. Hypertension was defined as systolic blood pressure (BP) of ≥140 mmHg, diastolic BP of ≥90 mmHg at rest, or ongoing antihypertensive therapy. Dyslipidemia was defined as serum total cholesterol concentration of ≥220 mg/dL, low-density lipoprotein cholesterol concentration of ≥140 mg/dL, or ongoing lipid-lowering agents. Diabetes mellitus was defined as HbA1c level ≥6.5%, requiring treatment with oral antidiabetic agents, or requiring insulin injection. CAVI was the index of arterial stiffness derived from the formula mentioned in the literature. 3
Data analysis
All statistical analyses were conducted using EZR version 1.21 (Saitama Medical Center, Jichi Medical University, Saitama, Japan). 4 Continuous variables were presented as mean ± standard deviation. Categorical variables were presented as frequencies and percentages. Continuous and categorical variables in patients with and without looped GW at BA were compared using Student's t-test and Fisher's exact test, respectively. The significance threshold was set at 0.05 with a two-tailed test. For significant categorical variables, the sensitivity and specificity for looped GW at BA were calculated from 2 × 2 contingency tables. For significant continuous variables, sensitivity, specificity, and area under the curve were calculated using the receiver operating characteristic curve (ROC) analysis. Cutoff points were optimal values that maximized the sum of the sensitivity and specificity. The positive predictive value (PPV) and negative predictive value (NPV) were calculated based on the prevalence of looped GW at BA.
Results
Looped GW at BA was observed in 19 of the 175 patients (10.9%). Patient characteristics and examination data obtained before CAG are shown in Tables 1 and 2. There were no significant differences in categorical variables. Patient age, estimated glomerular filtration rate (eGFR), and CAVI were significantly different between patients with and without looped GW at BA.
Patient characteristics.

BMI: body mass index; looped GW at BA: looped guidewire at the brachiocephalic artery.
Examination data.
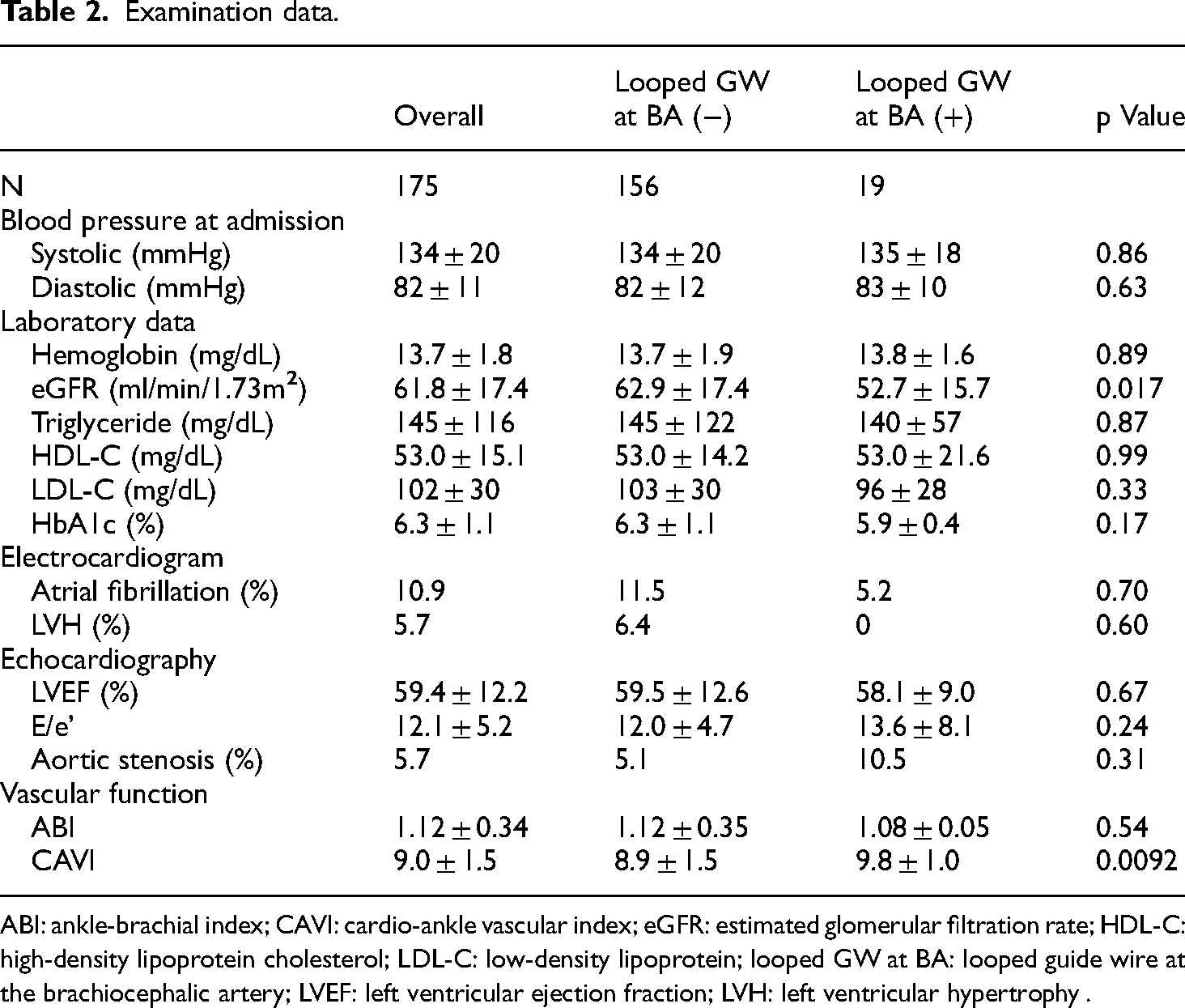
ABI: ankle-brachial index; CAVI: cardio-ankle vascular index; eGFR: estimated glomerular filtration rate; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein; looped GW at BA: looped guide wire at the brachiocephalic artery; LVEF: left ventricular ejection fraction; LVH: left ventricular hypertrophy .
In the ROC analysis, CAVI had a higher area under the curve than patient age and eGFR (Figure 2). Considering a cutoff point of CAVI ≥9.0, the sensitivity and specificity for looped GW at BA were 0.95 and 0.57, respectively. As a result, PPV and NPV were calculated as 0.21 and 0.99 at this cutoff point, respectively.

Receiver operating characteristic curve of CAVI, age, and eGFR to predict looped GW at BA. CAVI had the highest AUC. Considering a cutoff point of CAVI ≥9.0, the sensitivity and specificity were 0.94 and 0.57, respectively. AUC: area under the curve; CAVI: cardio-ankle vascular Index; CI: confidence interval; looped GW at BA: looped guidewire at the brachiocephalic artery.
Discussion
The main findings of this study are as follows. First, looped GW at BA is not a rare occurrence (approximately 10%). Second, CAVI, which reflected arterial stiffness, could be a predictor for ruling out the formation of looped GW at BA before performing CAG.
A previous study reported that the duration of catheter manipulation was longer in RtRA than in LtRA access. 5 This report noted severe tortuosity of the right subclavian artery, consistent with the looped GW at BA in our study, as one of the causes for prolonged operation time. This occurrence was observed in approximately 10% of the participants, which was similar to the rate observed in our study.
CAVI and pulse wave velocity (PWV) are arterial stiffness parameters commonly used in clinical practice. The clinical utility of arterial stiffness has been reported widely. A previous meta-analysis showed that arterial stiffness predicted future cardiovascular events and all-cause mortality. 6 CAVI is independent of blood pressure, and has advantages in predicting arterial stiffness and arteriosclerotic diseases compared to PWV.7,8
CAVI can be measured with ABI easily and noninvasively using one piece of equipment, with good reproducibility. 3 ABI measurement is recommended in cases with suspected peripheral artery (PAD) disease and risk factors of arteriosclerosis without symptoms of suspected PAD. 9 Measurement of ABI with CAVI is considered reasonable as a routine examination in patients undergoing CAG.
We considered a possible mechanism of looped GW at BA. In one of the patients with looped GW at BA, contrast computed tomography (CT) images taken for an unrelated matter showed a severe bend between the right subclavian and brachiocephalic artery (Figure 3). A high CAVI (10.0) was observed for this patent. In another patient, a loop-like appearance was momentarily formed by the Radifocus ® guidewire and disappeared when the wire tip reached the ostium of the ascending aorta, without catheter manipulation difficulty during subsequent CAG. This patient did not have a high CAVI (8.1) and was categorized as being absent of looped GW at BA, according to the definition of this study. These findings show that looped GW at BA occurs when the guidewire passes the bended zone between the right subclavian and brachiocephalic artery, which is not extended due to high arterial stiffness. Looped GW at BA is attributed to the coexistence of structural and functional conditions, such as arterial bend and high arterial stiffness, respectively (Figure 4). High arterial stiffness is considered necessary for forming looped GW at BA.

Three-dimensional CT image of one patient with looped GW at BA showing a severe bend between the subclavian and brachiocephalic artery (white dashed line). CT: computed tomography; looped GW at BA: looped guidewire at the brachiocephalic artery.
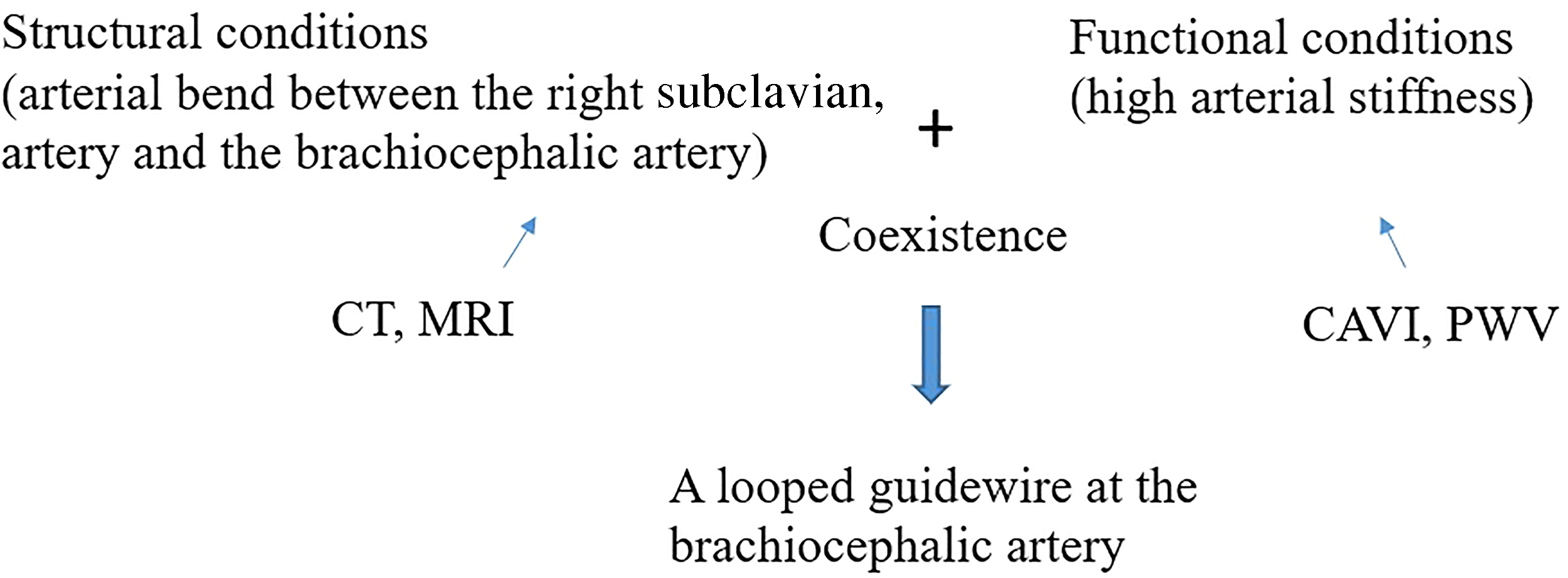
A possible mechanism of looped guidewire at the brachiocephalic artery. Coexistence of structural and functional conditions, such as arterial bend and high arterial stiffness, is necessary. Imaging modalities like CT and MRI predict the arterial bend between the right subclavian and brachiocephalic artery. CAVI and PWV predict arterial stiffness.
Other than CAVI, age and estimated glomerular filtration rate (eGFR) were significantly different between patients with and without looped GW at BA. Age and eGFR are factors related to arterial stiffness.10,11 This result also shows that looped GW at BA is associated with high arterial stiffness.
CAVI and PWV can predict arterial stiffness. Imaging modality, such as CT and magnetic resonance images, can predict the arterial bend between the right subclavian and brachiocephalic artery (Figure 4). A low CAVI indicates the absence of high arterial stiffness and at least the absence of coexistence of structural and functional conditions. As a result, the CAVI has high sensitivity and NPV. Because CAVI cannot predict the presence of arterial bend (a structural condition), high specificity and PPV were not obtained by CAVI alone. Imaging modalities such as contrast CT provide structural evaluations between the right subclavian and brachiocephalic artery; however, these are not reasonable routine examinations before CAG. On the other hand, CAVI is a reasonable routine examination before CAG, as described previously.
If we select other access sites instead of RtRA in patients with high CAVI, we likely can perform CAG without experiencing looped GW at BA. Predictors with a high NPV fulfill the principal objective of not performing CAG with looped GW at BA.
This study had several limitations. First, this study had a small sample size. There were no significant factors among the categorical variables in patients with or without looped GW at BA. If this study involved a higher number of subjects, another significant factor may have been identified. Second, the results included a risk of bias inherent to the retrospective design and selection bias due to the single-center design. The evaluations of looped GW at BA were qualitative and therefore affected by subjective views. Finally, we cannot generalize the findings to different populations owing to the single-center retrospective design. Large numbers of CAG are performed worldwide, and evaluating looped GW at BA is simple. Multicenter studies with large sample sizes are required.
In conclusion, when performing CAG with RtRA access, looped GW at BA is not rare and can be ruled out by CAVI. Our study suggests that high arterial stiffness is necessary for looped GW at BA to occur.
Footnotes
Acknowledgements
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author biographies
Hiroyuki Nakajima is a physician specializing in Cardiology. His research is focused on interventional cardiology and arteriosclerotic disease.
Tomoyasu Momose is a physician specializing in Cardiology.
Takuo Misawa is a physician specializing in Cardiology.
