Abstract
Objective
Hemodynamic monitoring via right heart catheterization (RHC) is critical for managing acute coronary syndrome (ACS) patients with heart failure or cardiogenic shock. However, the prognostic value of RHC-derived hemodynamic indices in ACS patients with left ventricular systolic dysfunction (LVSD) but without heart failure or shock remains uncertain.
Methods
A retrospective cohort study included 1151 consecutive ACS patients who underwent RHC during hospitalization from 2007 to 2016. After excluding patients with shock, pulmonary edema, and severe valvular disease, 750 ACS patients with LVSD and ejection fraction < 50% were analyzed. Major adverse cardiovascular events (MACEs), including myocardial infarction and all-cause mortality, were followed for five years. Cox regression identified predictors of MACEs, adjusting for comorbidities, treatments, and hemodynamic indices, including pulmonary arterial capacitance (PAC).
Results
After a mean follow-up of 4.0 ± 1.7 years, 113 (15.1%) patients experienced MACEs. Multivariate analysis showed that independent predictors included prior stroke, calcified coronary lesions, and PAC. Patients in the lowest PAC tertile (≤2.89 ml/mmHg) had significantly higher risks of myocardial infarction (adjusted hazard ratio [HR]: 3.74; 95% confidence interval [CI]: 1.55–9.07; p = .003), all-cause mortality (adjusted HR: 2.55; 95% CI: 1.27–5.10; p = .008), and MACEs (adjusted HR: 2.35; 95% CI: 1.25–4.42; p = .008) compared to those in the highest tertile (>4.43 ml/mmHg).
Discussion
The study demonstrated that PAC is a notably strong hemodynamic parameter with independent long-term prognostic value in ACS patients with LVSD, who do not present with shock or heart failure. This is the first study to establish the prognostic significance of hemodynamic indices obtained from RHC in this population, extending the clinical relevance of RHC from high-risk to intermediate-risk ACS populations.
Conclusions
The use of RHC to assess hemodynamic indices, including PAC, during index hospitalization in this population may enhance long-term risk stratification and improve outcome prediction.
Keywords
Introduction
Early and aggressive invasive therapy has improved prognosis and survival rates in patients with acute coronary syndrome (ACS) over the past two decades.1–3 Patients with left ventricular systolic dysfunction (LVSD) and cardiac remodeling post-ACS remain particularly vulnerable, often experiencing poorer long-term outcomes. The Swedish SWEDEHEART registry reports LVSD prevalence in ACS patients to be between 13% and 32%, correlating with a two- to threefold increase in mortality and heart failure hospitalizations. 4 The presentations of ACS can vary widely among patients with left ventricular systolic dysfunction (LVSD), spanning from asymptomatic compensatory stages and mild hemodynamic instability to severe heart failure and profound cardiogenic shock. Naturally, critical, and high-risk ACS patients receive more attention. Effective and early identification of ACS with LVSD patients, initially presenting at intermediate risk but with a poor long-term prognosis, is crucial for optimizing clinical care. 5
Right heart catheterization (RHC) assumes a pivotal role in managing these critical ACS patients, particularly those experiencing cardiogenic shock or heart failure. 6 Continuous monitoring of comprehensive hemodynamic profiles through this technique may prompt early use of mechanical circulatory support, potentially enhancing outcomes in critical situations. 7 Despite advancements in noninvasive or minimally invasive examinations, RHC remains the gold standard for hemodynamic monitoring in intensive care. Hemodynamic indices from RHC—including cardiac output, pulmonary artery pressure (PAP), pulmonary artery wedge pressure (PAWP), and pulmonary artery pulsatility index (PAPI)—provide crucial insights into myocardial stunning and overall pulmonary and cardiac function post-ACS.8–12 Pulmonary arterial capacitance (PAC), defined as the ratio of stroke volume to pulmonary pulse pressure, serves as an index of right ventricular afterload, response to therapy, and is a strong predictor of long-term survival in patients with heart failure and pulmonary hypertension.13–16 However, the prognostic relevance of hemodynamic indices, including PAC, in ACS patients with LVSD who do not present with shock or heart failure, remains to be clarified. This study aims to determine the prognostic value of hemodynamic indices derived from RHC, including PAC, in ACS patients with moderate risk.
Methods
Study design
A cross-sectional retrospectively analysis of pooled individual-level data from an institutional database. This analysis follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 17
Patient population
This study retrospectively analyzed consecutive ACS patients underwent the coronary angiography and RHC evaluation at the index intensive care unit hospitalization at a tertiary medical center (Chang Gung Memorial Hospital, Linkou, Taiwan) from January 1, 2007, to December 31, 2016. The diagnosis of ACS was based on the diagnosis at index hospitalization discharge. Patients presenting with cardiogenic shock, pulmonary edema, or clinical shortness of breath on medical record were excluded from the study. Additional exclusions were applied to those with moderate to severe valvular heart disease, hypertrophic or restrictive cardiomyopathy, pericardial diseases, or those undergoing chronic dialysis.
Echocardiographic data obtained at index hospitalization were used to assess left ventricular ejection fraction (LVEF), right ventricular function, and valvular pathology. Left ventricular systolic dysfunction (LVSD) was defined as an LVEF of less than 50%, according to standard echocardiographic measurements. Therapeutic interventions, serum creatine, estimated glomerular filtration rate (eGFR), and the presence of comorbidities such as hypertension, diabetes mellitus, hyperlipidemia, chronic kidney disease, or prior stroke at the time of index intensive care hospitalization, were documented. The Global Registry of Acute Coronary Events (GRACE) risk score was calculated based on baseline characteristics at index hospitalization. The major adverse cardiac events (MACEs) were defined as a composition of recurrent myocardial infarction and all-cause mortality within 5-years follow-up.
Hemodynamic measurement in RHC
In our practice, RHC was performed concurrently with coronary angiography during the index intensive care unit hospitalization for ACS patients with impaired LVEF. Patients were generally maintained in the supine position, and the pressure system was zeroed and referenced at the level of the heart. A balloon wedge pressure catheter (Teleflex, Wayne, Pennsylvania, USA) was inserted through the internal jugular vein or femoral vein, and pressures were recorded at end-expiration. All hemodynamic tracings were reviewed by the authors. Cardiac output was calculated using the Fick formula. Pulmonary vascular resistance (PVR) was calculated as (mean PAP – PCWP)/cardiac output. PAC was calculated as stroke volume/pulmonary pulse pressure. The PAPI was calculated from the ratio of pulmonary artery pulse pressure to right atrial pressure.
Statistical analysis
Continuous variables were presented as means and standard deviations and categorical variables were summarized as frequencies and proportions. Cox regression analyses were performed to explore the variables associated with the MACEs and the results displayed as hazard ratio (HR) and 95% confidence interval (CI) in univariate and multivariate model. Determinants identified through univariate Cox regression analysis with a p value less than 0.1 underwent subsequent testing in multivariate Cox regression analysis, utilizing stepwise subset selection to discern associated factors influencing clinical outcomes. To clarify the association between independent hemodynamic indices and clinical outcomes, patients were grouped into three parts (tertile T1-T3) according to the ordered hemodynamic indices. The Kaplan–Meier method determined the cumulative incidence of clinical outcomes, with differences between tertiles assessed using the log-rank test. Furthermore, two adjusted Cox proportional hazards models were constructed to assess the association between categorical (tertiles) and outcomes, including all-cause mortality, non-fatal myocardial infarction, or MACEs, respectively. In model 1, independent hemodynamic predictors were adjusted for sex, diabetes, calcified lesions, multivessel disease, GRACE score, and complete revascularization. In model 2, all variables in model 1, along with other hemodynamic indices, were adjusted. Stratified subgroup analyses were performed based on age (≥ 65 or < 65), gender and LVEF (> 40 or ≤ 40). Each stratification was adjusted for factors using model 2, except for the stratification factor itself. All analyses were performed using SPSS (version 26.0; IBM, Chicago, IL).
Results
Baseline characteristics of enrolled patient
A total of 1151 ACS patients who underwent coronary angiography and RHC during the index hospitalization were identified. Exclusions included 217 patients with pulmonary edema or cardiogenic shock (Killip class 3 or higher), 56 patients undergoing dialysis, and 128 patients with moderate to severe valvular heart disease. Ultimately, 750 subjects met the inclusion and exclusion criteria and were enrolled for further analysis. The flow chart of patient enrollment is shown in Figure 1. The baseline characteristics and hemodynamic parameters of the study participants were displayed in Table 1. The mean age of the cohort was 62.6 ± 12 years, with females accounting for 16.8% of the participants. Among them, 36.5% had diabetes mellitus, 12.8% exhibited chronic kidney disease, and 66.5% were diagnosed with multivessel disease, with 64.3% undergoing complete revascularization. The mean eGFR was 113.0 ± 34.8 ml/min/1.73 m2, GRACE score during the index hospitalization stood at 124.2 ± 28.6, while the average LVEF during this period was 41.4%. Hemodynamic assessments revealed a mean PCWP of 19.8 ± 8.0 mmHg, PVR of 119.4 ± 102.2 dynes-s/cm5, and PAC of 3.9 ± 2.5 ml/mmHg.

Flowchart of enrollment.
Baseline clinical characteristics of study cohort, stratified by the tertiles of PAC.

ACEi, angiotensin converting enzyme inhibitors; ARB, angiotensin receptor blockers; eGFR, estimated Glomerular filtration rate; GRACE, Global Registry of Acute Coronary Events; LVEDV, left ventricular end-diastolic volume; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; PAC, pulmonary arterial capacitance; PAP, pulmonary artery pressure; PAPI, pulmonary artery pulsatility index; PCWP, pulmonary capillary wedge pressure; PVR, pulmonary vascular resistance.
Independent predictors of MACEs
After a mean follow-up duration of 4.0 ± 1.7 years, 113 (15.1%) patients suffered from MACEs during the study period. As Table 2 illustrates, significant variables in univariate analysis included age (per 10 years), diabetes mellitus, previous stroke, chronic kidney disease, calcified coronary lesion, multivessel disease, GRACE score, complete revascularization, LVEF, PVR, cardiac output, and PAC. However, after adjusting with covariates and hemodynamic indices in multivariate analysis, the independent predictors of MACEs were previous stroke (HR: 2.31; 95% CI: 1.25–4.29, p = .008), calcified coronary lesion (HR: 1.55; 95% CI: 1.01–2.37, p = .043), and PAC (HR: 0.81; 95% CI: 0.67–0.97, p = .021).
Univariate and multivariable cox regression analysis for 5-year major adverse cardiovascular events.

ACEi, angiotensin-converting enzyme inhibitors; ACS, acute coronary syndrome; ARB, angiotensin receptor blockers; LVEF, left ventricular ejection fraction; PAC, pulmonary arterial capacitance; PAPI, pulmonary artery pulsatility index; PCWP, pulmonary capillary wedge pressure; PVR, pulmonary vascular resistance.
* Indicates p < 0.05.
Characteristics and hemodynamic parameters by tertiles of PAC
To evaluate the prognostic significance of the independent predictor, PAC, patients were stratified into tertiles according to their PAC values (Table 1). T1 represented the lowest range (PAC ≤ 2.89 ml/mmHg), T3 signified the highest (PAC > 4.43 ml/mmHg), and T2 denoted the middle range (2.89 < PAC ≤ 4.43 ml/mmHg). Patients in T1 tended to be older, with a higher prevalence of diabetes mellitus, chronic kidney disease, calcified lesion, and higher GRACE scores, but they were less likely to have hyperlipidemia. Hemodynamically, T1 group showed lower LVEF, higher PCWP, higher PVR, and reduced cardiac output compared to those in the higher PAC tertiles.
Incidence and relative risk of outcomes in tertile of PAC
The Kaplan-Meier curves demonstrate the rates of myocardial infarction (Figure 2), all-cause mortality (Figure 3), and MACEs (Figure 4) over a period of five years for patients stratified into three groups based on their PAC levels. Analyses of these outcomes showed that patients in the lowest PAC tertile (≤2.89 ml/mmHg) exhibited the highest rate of clinical events over time. The differences between the tertiles were statistically significant for all outcomes (log-rank tests, p < .00).
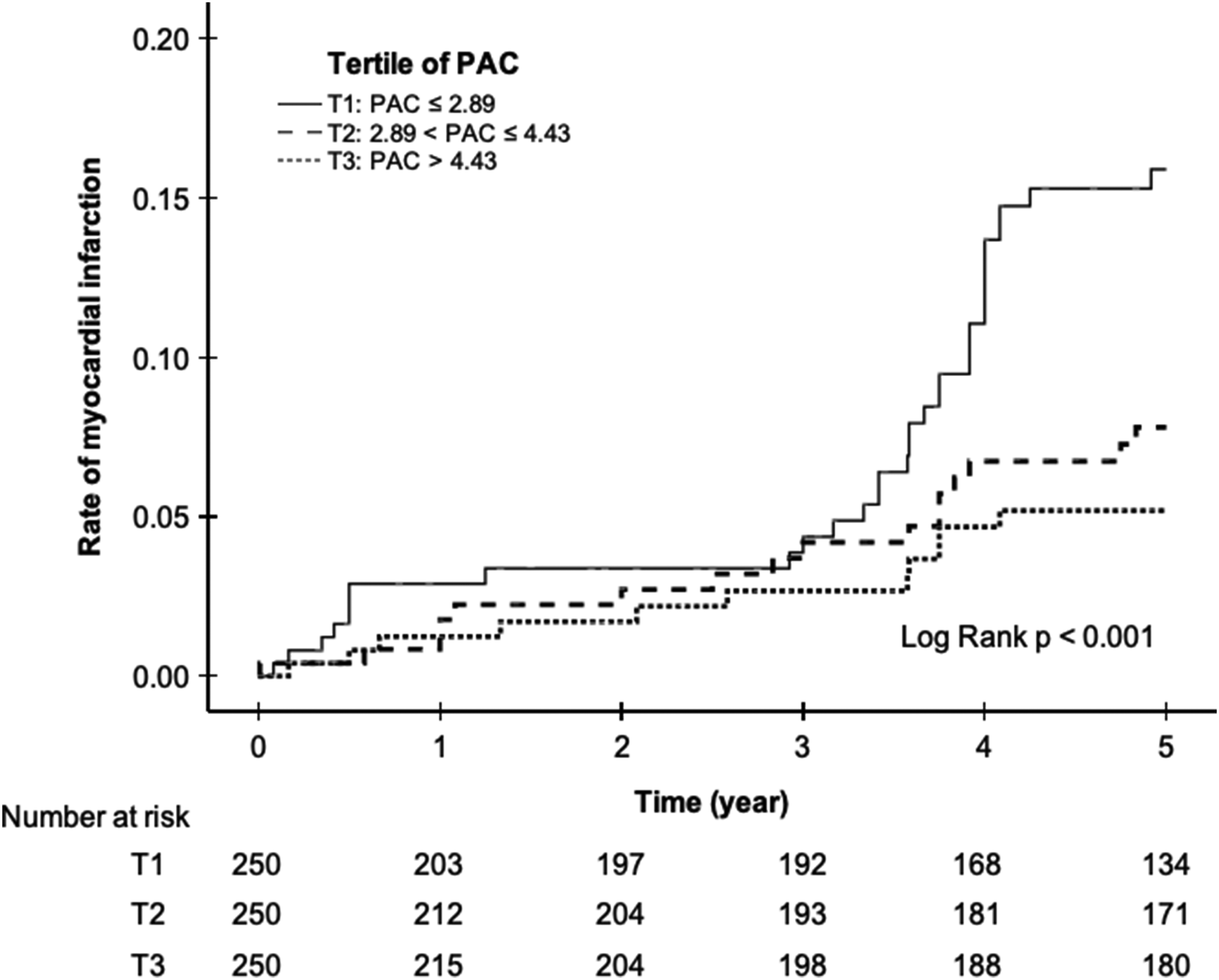
Kaplan–Meier curves of myocardial infarction according to tertile of PAC.

Kaplan–Meier curves of all-cause mortality according to tertile of PAC.

Kaplan–Meier curves of MACEs according to tertile of PAC.
Table 3 illustrates the relationship between levels of PAC and patient outcomes post-ACS. Patients in the lowest tertile of PAC (T1) exhibited the highest incidence rates of clinical events. Specifically, the incidence of myocardial infarction in T1 was 33.6 events per 1000 person-years, significantly surpassing those in T2 (15.9) and T3 (10.7). Regarding all-cause mortality, T1 reported an incidence of 49.5 per 1000 person-years, again markedly higher than T2 (19.1) and T3 (14.9). For MACEs, the incidence in T1 was 61.9 per 1000 person-years, compared to 29.9 in T2 and 23.4 in T3.
Clinical outcomes and relative risk according to tertile of PAC.

Model 1: Adjusted with sex, diabetes mellitus, calcified lesion, multivessel disease, complete revascularization, and GRACE score in the Cox-proportional hazard model.
Model 2: Adjusted with variables in model 1 plus LVEF, cardiac output, PCWP, PVR, and PAPI in the Cox-proportional hazard model.
Compared to T3, T1 exhibited significantly higher crude risks for myocardial infarction (HR 3.13, 95% CI: 1.58–6.21, p = .001), all-cause mortality (HR 3.33, 95% CI: 1.96–5.67, p < .001), and MACEs (HR 2.64, 95% CI: 1.64–4.24, p < .001). Even after adjusting for clinical variables and the GRACE score in model 1, T1 retained higher adjusted risks compared to T3 for myocardial infarction (HR 2.89, 95% CI: 1.41–5.91, p = .004), all-cause mortality (HR: 2.49, 95% CI: 1.42–4.36, p = .001), and MACEs (HR: 2.08, 95% CI: 1.26–3.44, p = .004), respectively. Further adjustments for other hemodynamic indices in model 2 revealed significantly higher risks in T1 compared to T3 for myocardial infarction (HR 3.74, 95% CI: 1.55–9.07, p = .003), all-cause mortality (HR: 2.55, 95% CI: 1.28–5.10, p = .008), and MACEs (HR: 2.35, 95% CI: 1.25–4.42, p = .008). Notably, there were no significant differences observed between the middle and highest tertiles in both crude and adjusted models of clinical outcomes.
Subgroup stratification analysis
Subgroup and interaction analyses were conducted to assess the robustness of the association between PAC tertiles and clinical outcomes. Table 4 presents the associations between CPO and the three outcomes—myocardial infarction, all-cause mortality, and MACEs—across various subgroups, including age (≥65 or <65 years old), gender (male or female), and LVEF (>40% or ≤40%). No significant interactions were found between PAC tertiles and any subgroups with respect to clinical endpoints (all interaction p-values > .05). Additionally, the association between the lowest tertile (T1) and an increased risk of clinical events remained consistent across all subgroups.
Subgroup analysis of myocardial infarction, all-cause mortality, and MACEs. Each stratification was adjusted for factors using model 2 in Table 3, except for the stratification factor itself.

Discussion
The primary finding of this observational study is that PAC is a notably strong hemodynamic parameter derived from RHC with independent long-term prognostic predictive value in patients with ACS and LVSD, who do not present with shock or heart failure. This contrasts with other hemodynamic parameters assessed via RHC. Notably, patients in the lowest tertile of PAC (≤2.89 ml/mmHg) demonstrated a markedly elevated risk of cardiovascular events over a five-year follow-up period compared to those in higher PAC tertiles.
Patients may present with a broad spectrum of ACS severity, ranging from those in cardiogenic shock to individuals experiencing silent myocardial infarctions without symptoms, as well as patients who remain normotensive despite ongoing hypoperfusion.18,19 The definition of cardiogenic shock categorization significantly affects patient survival, emphasizing the importance of timely and accurate risk assessment in patients with ACS. 20 Patients with ACS at risk of or in the early stages of cardiogenic shock are often not recognized, potentially missing the critical window for optimal intervention. Moreover, symptomatic heart failure events in patients following ACS are influenced by several key factors, including the extent and severity of myocardial damage, hemodynamic abnormalities, and neurohormonal activation. Comorbidities such as diabetes, hypertension, and chronic kidney disease further predispose patients to heart failure. Delays and incomplete revascularization can worsen myocardial recovery and increase susceptibility to heart failure. 21 Managing these factors is crucial to reducing heart failure risk in ACS patients.
Invasive hemodynamic measurements obtained through RHC, which provide data on fluid status, right heart filling pressures, pulmonary pressures, and vascular resistance, have proven survival benefits in the management protocol for ACS patients with cardiogenic shock.8,9 Despite these advances, the clinical utilization of RHC has been declining in real-world practice. One possible explanation for this trend is the uncertain prognosis significance of hemodynamic indices obtained from RHC. In this study, all participants diagnosed with ACS had LVSD without concurrent shock or heart failure. Most patients in this cohort were classified as intermediate risk, as evidenced by a mean GRACE score of 124.2 ± 28.6. Nevertheless, the potential utility of hemodynamic indices derived from RHC in this intermediate-risk population has not been previously established. To our knowledge, this study is the first to demonstrate the prognostic value of hemodynamic indices obtained from RHC in such patients. The findings of this study extend the clinical relevance of RHC from high-risk to intermediate-risk ACS populations. Additionally, PAC, independent of the GRACE risk score, emerges as a robust hemodynamic index for predicting MACEs.
PAC has emerged as a significant prognostic tool in clinical settings. Defined as the ratio of stroke volume to pulmonary pulse pressure, PAC integrates the effects of PVR and left-sided filling pressures on right ventricular afterload. Previous studies have demonstrated that PAC emerges as a strong prognostic predictor for patients with pulmonary hypertension arising from various etiologies.14,22–24 Pellegrini et al. proved the predictive capability of PAC for patients with chronic heart failure and normal PVR without pulmonary hypertension. 25 In relation to heart failure subtypes, Dupont et al. conducted a study on patients with heart failure with reduced ejection fraction. They found that PAC was strongly associated with right ventricular dysfunction, poor long-term prognosis, and response to therapy. 13 Al-Naamani et al. highlighted the prognostic significance of invasively measured PAC in patients with heart failure with preserved ejection fraction. 26 Similarly, Nakagawa et al. investigated a larger cohort of patients with acute decompensated heart failure with preserved ejection fraction and found that impaired PAC was linked to adverse outcomes. 27 Bayram et al. focused on patients with end-stage heart failure and found that ischemic cardiomyopathy was associated with lower levels of both PAC and PAPI. 28 Given the findings from these studies, it is plausible to consider that PAC holds a significant role in the prognosis of cardiovascular diseases.
The proposed pathophysiology of varying levels of PAC in ACS reflects the complex interplay between cardiac output, the pulmonary vasculature's ability to accommodate blood flow, and right heart function. In ACS, particularly with LVSD, left ventricular end-diastolic pressure and PCWP can elevated. This elevated pressure is transmitted back to the pulmonary vasculature, compromising its capacity to act as a blood reservoir. Subsequently, various mechanisms such as vascular remodeling, increased vascular tone, and interstitial edema within the lung parenchyma may lead to increased stiffness and reduced compliance of the pulmonary vessels. Over time, this reduced pulmonary vascular capacitance can increase right ventricular afterload, potentially leading to right heart dysfunction and reduced cardiac output, which can exacerbate myocardial ischemia and lead to a worse prognosis for ACS patients. Conversely, higher PAC levels, indicating more compliant vessels, may mitigate these effects. Understanding these mechanisms is essential for assessing risk and guiding treatment in ACS patients. In this study, we established that PAC is a reliable prognostic predictor in patients with ACS and LVSD. Importantly, the prognostic significance of PAC becomes apparent early in the course of the disease, underscoring its clinical relevance from the onset of ACS. These findings enhance our understanding of the pathophysiological mechanisms underlying coronary artery disease and provide clinicians with a valuable tool for early identification of high-risk patients. For healthcare providers, recognizing the value of PAC as an early prognostic marker can focus their attention on this parameter when managing patients with ACS, leading to more tailored treatment approaches. Regular assessments of PAC in ACS patients allow physicians to anticipate potential cardiovascular complications sooner and modify treatment plans more effectively, thus improving overall patient outcomes and prognosis.
This study is subject to several limitations that merit attention. First, it was designed as a retrospective, single-center study, which may limit the generalizability of the findings. Second, the relatively small sample size could compromise the robustness of our conclusions. Third, the study primarily involves ACS patients with LVSD who are in relatively stable hemodynamic conditions. Fourth, the lack of routine quantitative assessment of right ventricular function. There is currently no single practice parameter universally accepted as the standard for representing right ventricular systolic function. Commonly used measures such as tricuspid annular plane systolic excursion, fractional area change, and tissue Doppler S’, each have their own limitations. Due to the lack of a standardized approach, we did not routinely measure these parameters in our cohort. Consequently, we were unable to account for the potential impact of right ventricular function on PAC. This omission could influence the interpretation of PAC's prognostic role in ACS patients with LVSD.
It is important to note that the association of PAC with heart failure incidence was not part of the predefined endpoints in our study. Therefore, we are unable to provide specific data on the prognostic value of PAC for heart failure development within our ACS cohort. Additionally, the study only documented a single cross-sectional cut-off value of PAC at index hospitalization, leaving the prognostic value of PAC changes before and after long-term treatment unexplored. Future studies specifically designed to evaluate this relationship would be valuable in clarifying PAC's role in heart failure risk stratification among ACS patients and should integrate comprehensive right ventricular function assessments to elucidate the interplay between right ventricular function and PAC in predicting long-term outcomes.
Conclusions
PAC is a valuable and independent prognostic predictor for long-term outcomes in patients with ACS and LVSD, who do not present with shock or heart failure. The application of RHC to assess hemodynamic indices, including PAC, during index hospitalization in this population may aid in the stratification of long-term outcome risks.
Footnotes
Author contributions
Conceptualization: Ming-Jer Hsieh, Jih-Kai Yeh; Writing and editing: Shu-Yun Hsieh, Jih-Kai Yeh, Ming-Jer Hsieh; Funding acquisition: Ming-Jer Hsieh, Jih-Kai Yeh, Yu-Chang Huang; Investigation: Yu-Chang Huang, Dong-Yi Chen, Ming-Yun Ho, Chun-Chi Chen, I-Chang Hsieh; Supervision: Ming-Jer Hsieh and I-Chang Hsieh
Consent to participate
This is a retrospective database analysis study and the consent to participate is waived by the institutional review board.
Data availability
The data supporting the findings of this study are available from the Chang Gung Medical Database, but restrictions apply to their availability. These data were used under license for the current study and are not publicly accessible.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval statement
This study adhered to ethical standards, including the Declaration of Helsinki (1975, revised 2013). The use of de-identified patient data ensured privacy and confidentiality. The study design and data analysis were approved by the Institutional Review Board of Chang Gung Memorial Hospital (Approval No. 202301046B0).
Funding
This work was supported by grants from the Chang Gung Medical Research Program (Number: CORPG3N0461 & CORPG3N0481).
