Abstract
IL-17a is a pro-inflammatory cytokine produced primarily by T helper-17 cells. Several studies have shown that maternal IL-17a, associated with maternal immune activation (MIA), affects the developing brain. However, the mechanisms underlying maternal IL-17a signaling remain partially unknown. This study detected trans-placental IL-17a passage using luminescent activity studies and an in vitro transfer assay. First, the luminescent activity was observed using LiCoR dye-conjugated IL-17a injected into pregnant mice. IL-17a luminescent activity was highly detected in the placenta and isolated fetus, but positive control IgG and negative control IgM showed low or no luminescence in the placenta and fetus, respectively. Next, IL-17a transmission across the placenta was investigated using a transwell experiment with trophoblast BeWo cells and primary trophoblast cells. Significant amounts of IL-17a were detected in the lower compartment. And in various placenta cell lines, IL-17a treatment significantly increased IL-17RA mRNA expression. However, it did not affect IL-17RC mRNA expression.
This study showed that elevated IL-17a increased the IL-17RA expression in the trophoblast and may accumulate in the placenta. Furthermore, these results indicate the molecular basis of an important role in IL-17a/IL-17RA in the maternal placenta.
Introduction
The placenta is involved in the transport of nutrients, various growth factors and gases, including oxygen, to the fetus and the removal of waste products from the fetus. In other words, the placenta is the principal site of maternal-fetal exchange. 1 In addition, it is the first protective barrier between the mother and developing fetus during pregnancy. Moreover, the placenta protects the fetus by suppressing the maternal immune system during infection. Despite the barrier presented by the placenta, some pathogens, including Zika virus,2–5 human immunodeficiency virus (HIV)6,7 and rubella virus8–10 overcome the placental barrier and have devastating consequences on fetal development. Maternal immune activation (MIA) is mediated by activation of inflammatory pathways resulting in increased levels of cytokines that cross the placenta altering fetal-neural development. Thus, MIA is a major risk factor for neurodevelopmental disorder, autism spectrum disorder (ASD), and schizophrenia induced by chronic inflammation associated with infections during pregnancy.11–13 To induce MIA in rodents, influenza virus (PR18 strain), its mimetic viral polyinosine: polycytidylic acid (poly [I:C]), 14 or bacterial lipopolysaccharide (LPS) 15 were used as immunogens during pregnancy. As a result, the major cytokines including interleukin-6 (IL-6), interleukin-17 (IL-17a), interleukin −1β (IL-1β), and tumor necrosis factor α (TNFα) are induced by MIA. 16 MIA induces a chronic inflammatory state in the fetal brain, inducing significantly altered neuronal gene expression and major damage to developing neurons, which in turn affects brain development and behavioral change in the offspring.17,18
List of primers used in RT-PCR.
According to a recent study, exposure to specific microbiota (i.e. Segmented filament bacteria) induces T helper-17 (Th17) differentiation and maternal serum IL-17a levels were increased by injecting poly(I:C) during the second trimester. Elevated IL-17a resulted in ASD-like behavioral changes in offspring. 19 IL-17a/IL-17RA signaling affects neural progenitor cells, neurons, astrocytes, oligodendrocytes, and microglia in the brain.20–22
However, it is poorly studied whether elevated maternal IL-17a levels pass through the placenta and influence fetal brain development. In this study, it was observed that maternal IL-17a accumulated in the placenta through luminescence activity and increased IL-17RA expression.
Materials and methods
Animals
All animals were purchased from Taconic Biosciences (Germantown, NY) and were maintained in a controlled environment with a 12:12 h light/dark cycle, constant temperature/humidity, and free access to food and water. All experiments were performed following the guidelines of Animal Care using protocols approved by the Animal Care Committee of the Harvard University School of Medicine.
LiCoR-conjugated mIL-17a injection
Female mice (7–10 weeks old) were mated with male mice (7–16 weeks old) overnight. Subsequently, the male mouse was removed, and the females were checked for plug development early in pregnancy. This day was referred to as embryonic day 0.5 (E0.5). At embryonic day 12 (E12.5), pregnant female mice were weighed and injected with 60 μg LiCoR-dye (IRDye 680RD Maleimide; LI-COR Corp., Lincoln, NE) – conjugated recombinant murine IgG, IgM, and IL-17a cytokines (PeproTech, Cranbury, NJ) via the intra-tail vein. After 1 h, placentas and fetuses were isolated from the dam and luminescence activity was measured using the Odyssey DLx imaging system (LI-COR Corp., Lincoln, NE).
Primary mouse trophoblast cell isolation
Primary mouse trophoblasts were isolated as described by Pennington et al. 23 Briefly, placentas were collected at the E8.5-10.5 stage of pregnancy, fetuses, and fetal membranes were separated and all decidual tissue was removed from the placenta. The placenta was treated with 0.25% trypsin-EDTA (ThermoFisher Scientific, Waltham, MA) and 0.2 mg/mL DNAse I (ThermoFisher Scientific) in sterile Hank's balanced salt solution (HBSS) (ThermoFisher Scientific, Gibco) to release cells. The trophoblasts were isolated using Percoll (Sigma-Aldrich, St Louis, MO) gradient centrifugation. Cells were maintained in Dulbecco's modified Eagles medium (DMEM)/F12 medium (ThermoFisher Scientific) supplemented with 10% fetal bovine serum (FBS; Gibco) and antibiotics (penicillin and streptomycin).
Bewo cell cultures and in vitro transwell assay
The BeWo cell line, which originated from human choriocarcinoma was purchased from ATCC and used in the study. BeWo and JEG-3 cells were maintained in DMEM/F12 supplemented with 10% FBS and 1% antibiotics, grown in T-75 flasks (Corning Inc., Corning, NY) and typically passaged after 2–3 days in culture.
The upper surface of the insert membrane was coated with Matrigel solution (ThermoFisher Scientific). 200 μℓ of 0.5 mg Matrigel solution was transferred into 8 μm pore 24-well transwell plates with permeable polycarbonate membranes (Corning Inc.) and incubated at 37°C for more than 1 h. BeWo cells (7.5 × 104 cells) or primary trophoblast cells (1 × 105 cells) were seeded in the upper chamber with Matrigel-coated membrane inserts and culture media in the low – compartment. Cells were grown until they formed a monolayer. To investigate the transmission through the placenta, 10 ng of IL-17a cytokines, IgG, and IgM were added in the upper compartment and kept for 1 h. Subsequently, enzyme-linked immunosorbent assays (ELISA) were performed using the media collected from the upper and lower compartments.
Reverse transcription polymerase chain reaction (RT-PCR)
The BeWo and JEG-3 cell were maintained and treated 100 ng recombinant IL-17a cytokine or 100 μg poly (I:C) for 24 h, and then harvested cell lysates. Total RNA was extracted from the placental cell lines, BeWo cells, and JEG-3 cells (Korean Cell Line Bank) using TRIzol (Invitrogen, Carlsbad, CA). Subsequently, cDNA was synthesized using oligo-dT and SuperScript III Reverse Transcriptase (ThermoFisher Scientific) according to the protocol provided by the manufacturer, and RT-PCR was performed. Primer sequence is summarized in Table 1 below.
ELISA
Media was collected from the upper and lower transwell compartments and cytokine levels were measured according to the protocol provided by the manufacturers (BioLegend, San Diego, CA). The detection limit of the ELISA was 16.8 pg/mL. hIL-17a, hIgG, and hIgM ELISA kits were used for BeWo cells and mIL-17a and mIgG ELISA kits were used for primary trophoblast cells.
Statistics
All data were analyzed using a paired two-tailed Student's t-test, one-way analysis of variance (ANOVA), or two-way ANOVA followed by Tukey's post hoc test. Statistical significance was set at p < 0.05. All data are presented as mean ± standard error of the mean (SEM).
Results
To investigate whether maternal IL-17a migrated into the placenta, LiCoR-dye-conjugated mouse IL-17a was intravenously injected through the maternal tail at E12.5. After 1 h, the mouse was sacrificed, and the luminescence of LiCoR dye in the placenta and fetus was observed using Odyssey. Significant amounts of mouse IgG, IgM, and IL-17a were detected in maternal blood using the luminescence assay. It is well known that placenta transfer of maternal IgG antibodies is mediated by FcRn receptor to the fetus. So, IgG was used as a positive control as it selectively passes through the maternal placenta and is transmitted to the fetus. In addition, IgM was used as a negative control and could not cross the placenta. IgG luminescence activity was observed in the placenta and fetus, but IgM was not. [Figure 1(a)]. Moreover, luminescent activities were significantly increased in the placenta and fetus obtained from the IL-17a injected dam [Figure 1(b)]. These visual outcomes indicate that maternal IL-17a accumulated in the placenta and passed through the placenta affecting the fetus. To confirm whether IL-17a passes through placental cells, transwell experiments were performed using human placental cell lines, BeWo cells, and primary mouse trophoblast cells. BeWo cells were cultured on a Matrigel-coated upper compartment until monolayer formation, and then IgG, IgM, or IL-17a cytokines were added to the upper supernatants and incubated for 30 min. IgG was detected in the lower compartment, but IgM was not. IL-17a was detected prominently in the lower compartment [Figure 2(a)]. While analyzing the transit of IL-17a in a time-dependent manner, it was noticed that the amount of IL-17a in the lower compartment increased with the time up to 24 h [Figure 2(b)]. Moreover, these results were confirmed using primary mouse trophoblast cells, mouse IL-17a and IgG passed through the primary mouse trophoblast cells [Figure 2(c)]. Thus, these data suggest that maternal IL-17a levels passing through the placenta can affect fetal health and brain development.

Detection of IL-17a cytokine in the placenta.
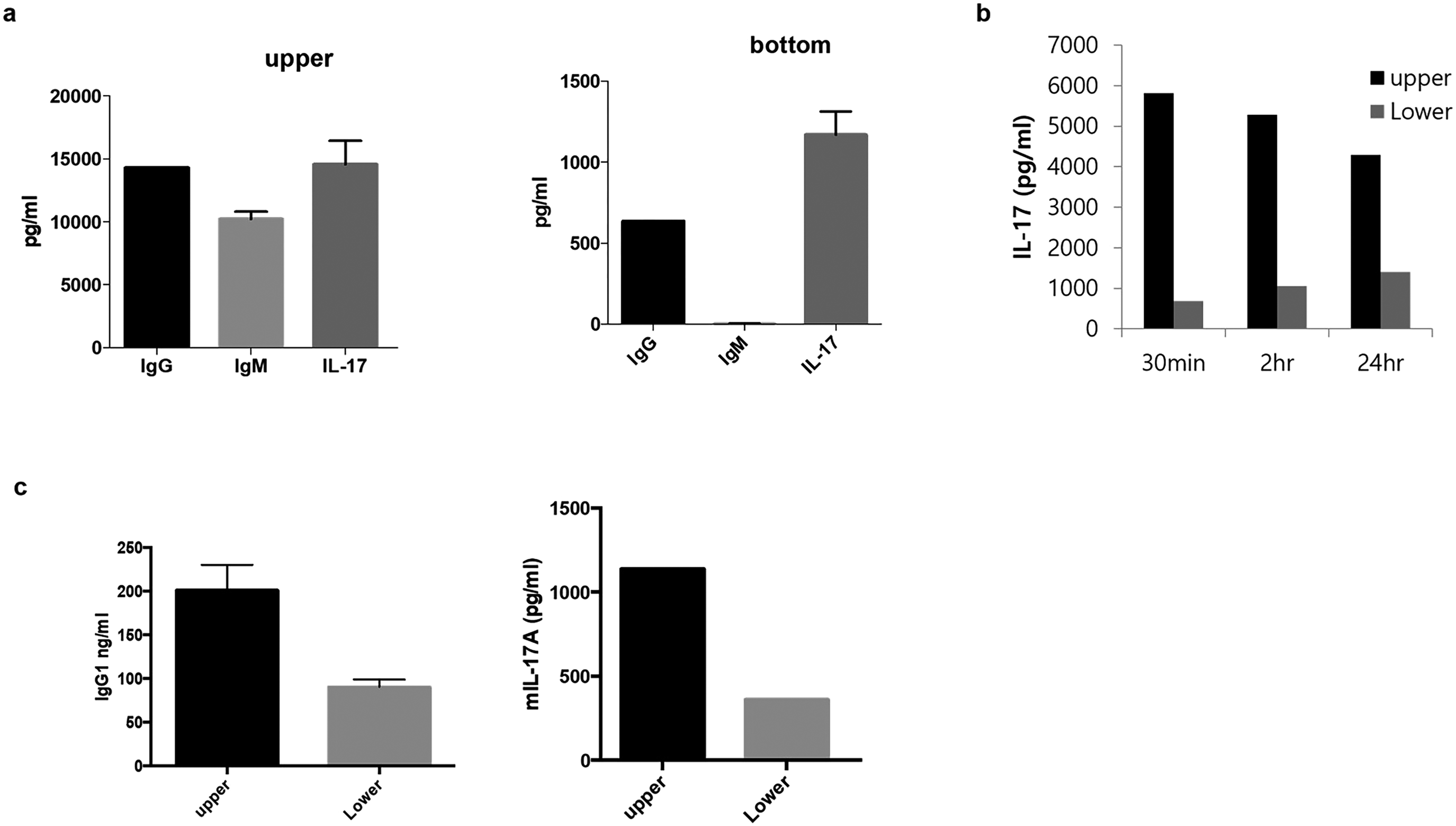
The transmission of IL-17a in trophoblast BeWo and primary trophoblast cells.
Next, to identify the mechanism of IL-17a transmission into the placenta, it was investigated whether tight junction protein expression changed upon IL-17a treatment. ZO-1 and occludin protein expression were not observed significantly upon IL-17a treatment in BeWo and JEG-3 cells (data not shown). Next, IL-17 receptor expression was observed, which is involved in the pro-inflammatory response through binding with IL-17a. IL-17RA mRNA expression in BeWo and JEG-3 cells was significantly increased by IL-17a cytokine, but IL-17RC expression was not changed at the transcript level [Figure 3(c)]. These data suggest that, despite the predominance of IL-17RC in the placental cell line, IL-17RA plays a dominant role in the placenta upon inflammatory response [Figure 3(a) and (b)]. Hence, elevated IL-17a does not affect placental permeability, but binds to the placenta IL-17RA receptor and moves across the placenta.

IL-17 receptor expression in the human trophoblast cells.
Discussion
IL-17a, a key cytokine, is involved in antimicrobial defense and inflammatory response in autoimmune diseases. Moreover, IL-17a has a protective effect against extracellular microorganisms in normal pregnancy.24,25 Although there is no significant difference between IL-17a found in sera of non-pregnant and pregnant women, 26 IL-17a levels are increased in pathological conditions and elevated maternal IL-17a levels affect fetal brain development in the MIA rodent model.17,19 However, several studies reported that maternal IL-17a passes through the placenta and affects fetal health nevertheless mechanistic studies are poor. Therefore, in this study, it was visually confirmed that IL-17a can pass the placenta using in vivo experiments using biological imaging of pregnancy dams. IL-17a cytokine conjugated with fluorescence dye was injected into the pregnant dam (E12.5) and was observed in the placenta and fetus; moreover, it increased in maternal blood (data not shown). Systemic IL-17a circulates through the maternal blood and affects the placenta.
It has been reported that lymphocytes including CD4+, CD8+, gamma-delta (γδ T cells), invariant NKT and innate lymphoid cells (ILCs) are the primary sources of IL-17a. 27 Non-T cells such as neutrophils, have also been reported to produce IL-17a under certain circumstances. 28 It is also produced in placental macrophages and is affected by autocrine IL-17a. 29 In this study, to confirm whether the amount of IL-17a released from primary trophoblast cells affects fetal health, IL-17a released from trophoblast cells was measured in a culture medium; however, IL-17a was not detected. Therefore, IL-17a produced in the placenta would not be sufficient to affect fetal brain development (data not shown). To measure the permeability of IL-17a, BeWo cells were cultured as a monolayer and investigated microscopically. The formation of BeWo cell monolayer with tight junctions was previously characterized.30,31 Enhanced permeability of IL-17a was detected compared to that of IgM used as a negative control. Furthermore, an increase in the amount of IL-17a did not affect the gene expression of gap junctional proteins such as ZO-1, occluding, dextran-FITC permeability (data not shown). These results indicated that IL-17a was not passively transmitted by leakage through intercellular junctions. Additionally, it was confirmed that the used recombinant IL-17a cytokine was in the dimer form that binds to IL-17R, and its molecular weight was 31.3 kDa through silver staining (data not shown).
IgG transfer from the mother to the fetus occurs across the syncytiotrophoblasts of present in the placenta during pregnancy. Maternal IgG bound to FcRn is protected from degradation by lysosomal enzymes and transcytoses.32,33 To observe the transmission of IL-17a through IL-17 receptors, the expression of IL-17 receptors (IL-17RA and IL-17RC) was examined in trophoblast cell lines, which confirmed the specifically increased expression of IL-17RA induced by elevated IL-17A. Although IL-17RC is predominantly expressed in trophoblast cells, the expression of IL-17RA significantly increased after IL17a treatment. These results suggest that IL-17a transmission is regulated by IL-17A/IL-17RA binding. Moreover, these outcomes may contribute to further research on the function of the placenta in immune activation and the development of the fetus through the IL-17A/IL-17R signal transduction mechanism.
A total of 60 μg of LiCoR-dye conjugated mouse recombinant IL-17a cytokine, mIgG, or mIgM were intravenously injected through the maternal tail at embryonic day 12.5 (E12.5) and the specimen was isolated after 1 h. (a) The luminescence of LiCOR dye was observed using Odyssey DLx imaging system. Mouse IgG and IgM were used as a positive and negative control, respectively. (b) Moreover, luminescence was observed in the fetus and placenta.
BeWo cells and primary trophoblast cells were cultured for 5 days to form a monolayer on the Matrigel-coated upper inner compartment, and then 10 ng of IgG, IgM, or IL-17a for BeWo cells was added to the upper compartment for 30 min. The culture medium was harvested from the upper and lower compartments of the Transwell and ELISA was performed to detect IL-17a transmission [a]. In addition, IL-17a was treated for 30 min, 2 h and 24 h, and ELISA was performed using the harvested culture medium [b]. Primary trophoblast cells were isolated, treated with 10 ng of murine IgG or murine IL-17a for 30 min and verified using ELISA [c]. All experiment was repeated three times, and the presented data are the mean ± standard error of the mean (SEM). * p < 0.05 compared with IgG positive control.
BeWo (a) and JEG-3 (b) cells were cultured with 100 ng IL-17a cytokine for 24 h. RNA was extracted, and the mRNA expressions of IL-17a, IL-17RA, and IL-17RC were investigated using RT-PCR with specific primers. β-actin was used as the loading control. NT means no treatment
Footnotes
Acknowledgments
I thank Jun R Huh at Harvard medical school and Sangdoo Kim for valuable discussion and helping for experiment design.
Data availability
The data supporting the findings of this study are included with the article.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea, (grant number no.2022R1F1A106825211)
Research ethics
All experiments were performed following the guideline of Animal Care using protocols approved by the Animal Care Committee of the Harvard University School of Medicine.
Author biography
Hyunju Kim is assistant professor in the department of Medical Biotechnology at Dong-A university. She received her MS at Pusan National university and degree of Philosophy at Dong-A university of Republic of Korea and did postdoc at Harvard medical school. Her research focused on neurodevelopmental disorders, epigenetics modulation, and immune system.
