Abstract
Warthin tumor (WT)-like mucoepidermoid carcinoma resembles the histologic pattern of WT and pathologists unaware of this possibility may misdiagnose it as WT with squamous and mucous epithelium metaplasia or WT malignant transfer into mucoepidermoid carcinoma. The present study reported a case of a 41-year-old Chinese female with a solitary mass in the left parotid gland. In this case, microscopic observation revealed prominent lymph node stroma and multiple cystic structures similar to those seen in WT. However, it lacked the two layers of oncocytic epithelial tissue characteristic of WT. Furthermore, fluorescence in situ hybridization detected MAML2 rearrangement in the case. Considering the histological findings, this case was diagnosed as WT-like mucoepidermoid carcinoma. The present case report provides pathological and clinical features to differentiate it from WT malignant transition into mucoepidermoid carcinoma, WT with squamous and mucous epithelium metaplasia and non-sebaceous lymphadenoma-like mucoepidermoid carcinoma. In conclusion, WT-like mucoepidermoid carcinoma as a special subtype of mucoepidermoid carcinoma has special histological characteristics, which required further observations and more case reports to clearly define this variant.
Introduction
Warthin tumor (WT) is a benign neoplasm of salivary gland origin, the second most common after pleomorphic adenoma. The WTs described to date are characterized by a bilayered glandular epithelium with a dense lymphocytic stroma and overlying acidophilic cytoplasm. Mucoepidermoid carcinoma (MEC), characterized as the most common malignant salivary gland neoplasm, is histologically defined by a mixture of three cell types: Epidermal cells, intermediate cells and mucous cells, which may be present in different proportions. In the diagnosis of salivary gland tumours, the term WT-like MEC was first formally proposed by Ishibashi et al. 1 in 2015. As WT-like MECs are characterized by what is a close resemblance to the histological pattern of WT, this potential pitfall may go unnoticed and the lesion may be misidentified as WT. 2 WT-like MEC is a rare neoplasm, only recently defined, and just a small number of cases have been described in the literature. In the present study, one case of WT-like MEC, a neoplasm arising in the parotid gland, was discussed.
Case report
In April 2021, a 41-year-old Chinese female who had found a painless mass behind her left ear for three days visited Tangshan Gongren Hospital (Tangshan, China). Clinical examination revealed a 2.5 × 2 × 2 cm solitary mass arising from the left parotid gland. CT scan (Figure 1) revealed a 1.8 × 2.4 cm smooth rounded nodule with varying density and a well-defined border. A lumpectomy of the left parotid gland was performed.
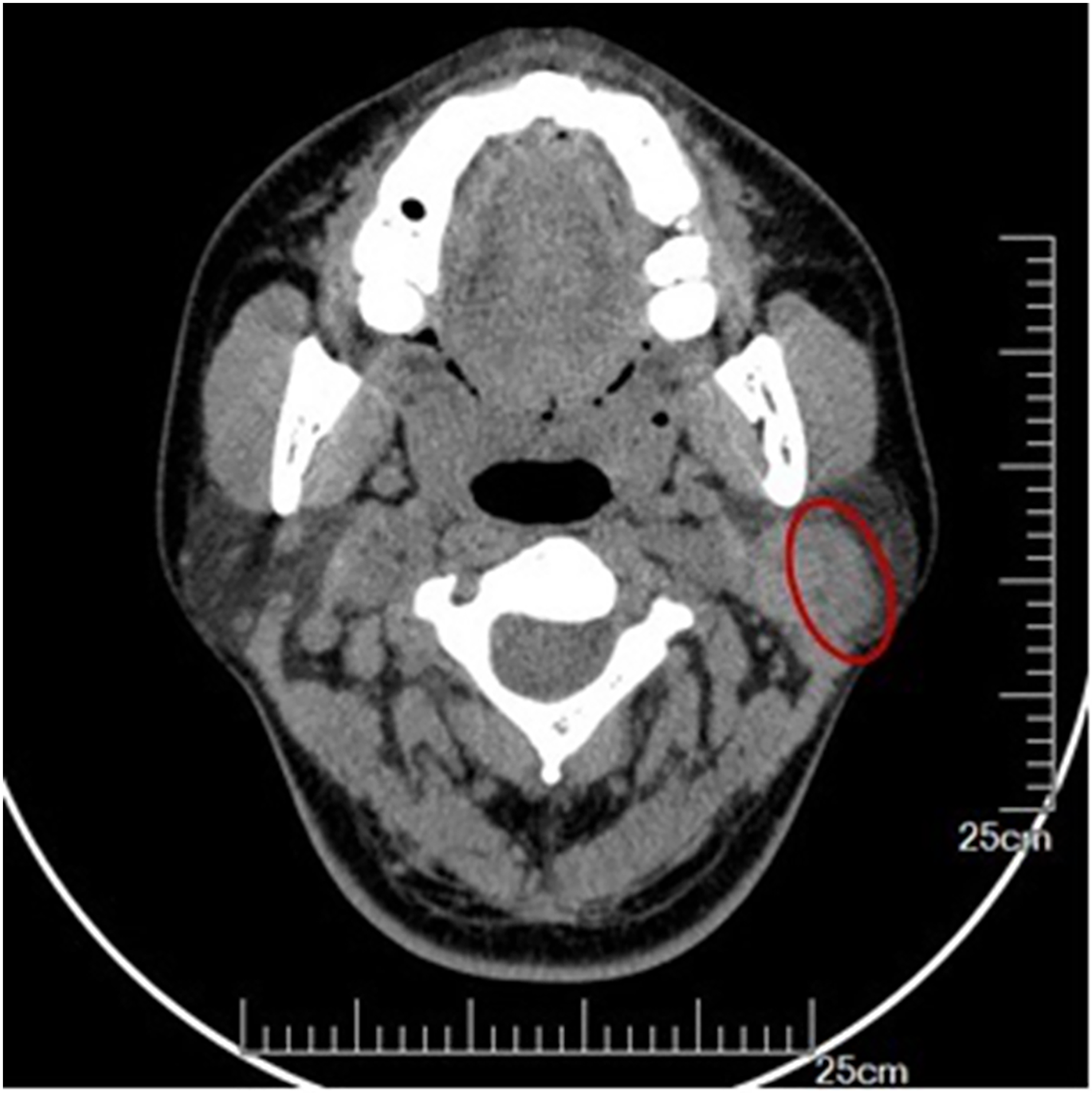
CT scan revealed a smooth rounded nodule with varying density and well-defined border in the left parotid gland (red circle).
The excised tissue measured 3 × 2.5 × 2 cm and, according to gross findings, was an oval nodule with a smooth gray-brown surface (Figure 2A). Microcapsule structures were also observed within the tumour, and the cross-section of the tumour was shiny (Figure 2B). Microscopic examination revealed multiple cystic structures of varying size and shape filled with a protein-like substance within the tumour (Figure 3A). There is a complete and thin fibrous connective tissue around the tumour (Figure 3B). Infiltration of lymphocytes with the formation of lymph follicles was evident around the cysts (Figure 3C). The epithelium was multilayered and oncocytic but without any malignant characteristics, containing single scattered mucous cells (Figure 3D and E). Certain cysts were lined by only a single mucinous columnar epithelium (Figure 3F). Of note, there is no lymph node sinus structure in the lymphoid stroma (Figure 3G). Only a few normal salivary duct structures were found in the internal peripheral area of the tumour, but no salivary acini were discovered (Figure 3H). On the other hand, no necrotic, keratinized squamous epithelium or bilayered oncocytes were observed.

Gross examination. (A) Oval nodule with smooth surface; (B) the cross-section of the tumour was shiny and with microcapsule structures.
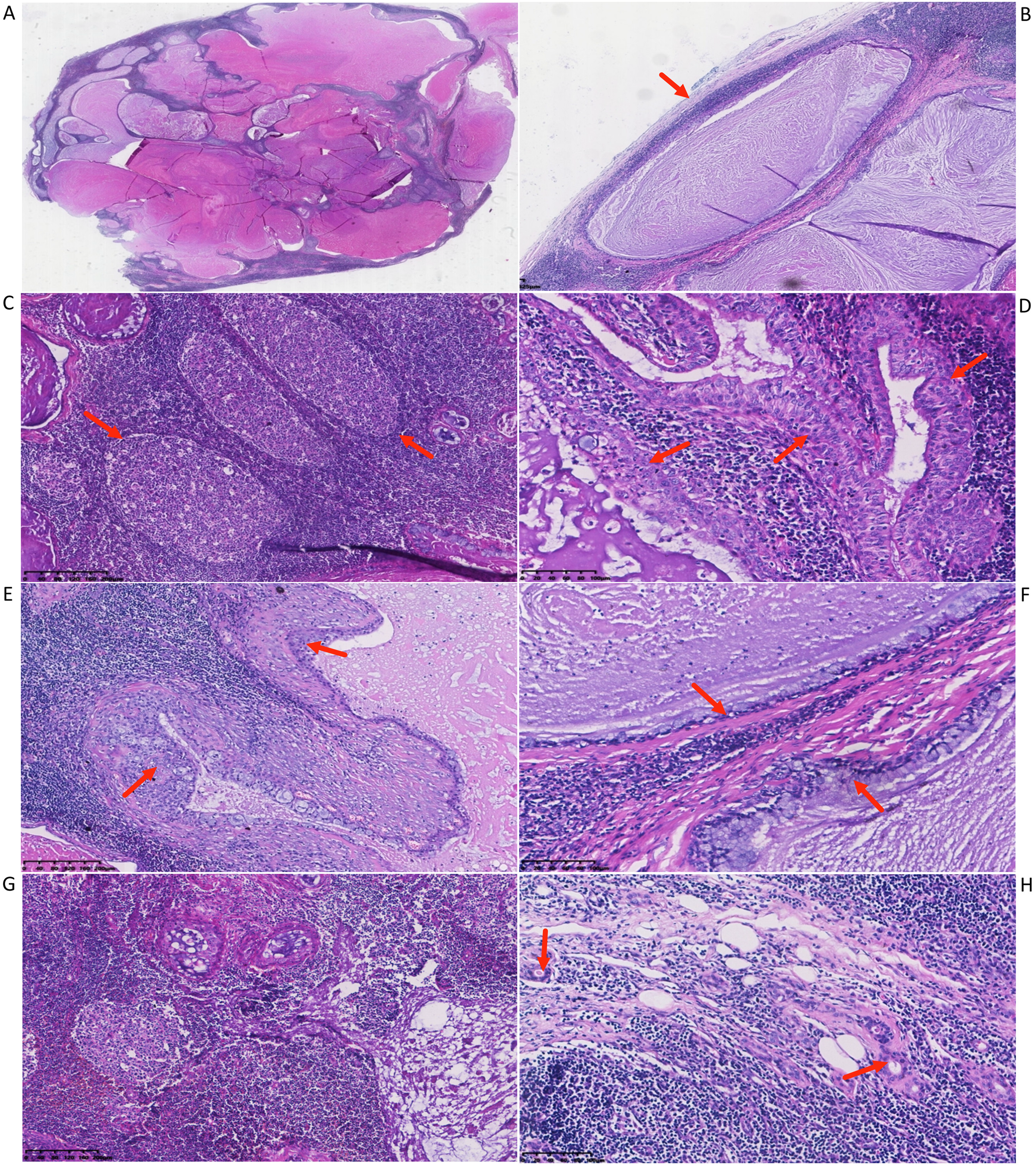
Microscopic examination. (A) Multiple cystic structures of varying size and shape were present within the tumour (H&E; magnification, ×5). (B) A thin fibrous connective tissue around the tumour (H&E, magnification, ×40). (C) Prominent infiltration of lymphocytes around the cysts (red arrows; H&E; magnification, ×100, scale length was 200 µm); (D and E) the epithelium was multilayered and oncocytic, containing single scattered mucous cells [red arrows; H&E; magnification, (D) ×200, scale length was 100 µm and (E) ×100, scale length was 200 µm]. (F) Cysts lined by only a single mucinous columnar epithelium (red arrows; H&E; magnification, ×200, scale length was 100 µm); (G) there is no sinus structure in the lymphoid stroma (H&E; magnification, ×100, scale length was 200 µm); (H) a few normal parotid ducts appeared in the internal peripheral area of the tumour (red arrows; H&E; magnification, ×200, scale length was 100 µm).
Immunohistochemical analysis indicated that the mucosa cells were positive for keratin 7 and keratin 8/18, and the neoplastic epidermal cells were positive for keratin 5/6, keratin 8/18 and P40; the cell proliferation index due to ki67 positivity was 5% for both the neoplastic epidermal and mucosal cells; in the area of lymphoid stroma, CAM5.2 was negative, while D2-40 was positive for lymph vessels (Figure 4). Observe under the fluorescence microscope, red and green fluorescent label was separated, and MAML2 rearrangement was detected (Figure 5).

Immunohistochemical staining of the tumour. (A) The neoplastic epidermal and mucosa cells with H&E staining. (B and C) Neoplastic epidermal cells were positive for (B) P40 and (C) keratin 5/6. (D and E) Neoplastic epidermal cells and mucous cells were (D) both positive for keratin 7 and both weakly positive for (E) keratin 8/18. (F) The proliferation index due to ki67 positivity was 5%. (G) CAM5.2 was negative in lymphoid stroma. (H) D2-40 was positive for lymph-vessels. (A-F: magnification, ×200, scale length was 100 µm; G, H: magnification, ×100, scale length was 200 µm).

Fluorescence in situ hybridization study detected MAML2 rearrangement (red fluorescent label, 5′-MAML2 11q21; green fluorescent label, 3′-MAML2; red arrows indicate split signals; magnification, ×1,000).
Based on these results, the tumour was diagnosed as WT-like MEC. Based on the final histopathological diagnosis, a parotidectomy and supraomohyoid neck dissection was performed. Postoperative pathology revealed no residual mass or lymph node metastasis. After 22 months of postoperative follow-up by ultrasound examination and CT scan, there was no evidence of recurrence or metastasis and no additional treatment was performed.
Discussion
In 2012, Rotellini et al. 3 reported on WT with myxoid and squamous metaplasia. In the same year, Mohapatra and Satyanarayana 4 reported MEC with numerous eosinophilic epithelia lacking typical epidermal, intermediate cells and mucous cells and with extensive lymphocytic infiltration, which was considered WT-like MEC in terms of diagnosis. The term WT-like MEC was first formally proposed in the diagnosis of salivary gland tumours by Ishibashi et al. 1 in 2015. Subsequently, several cases of mutant MEC, consisting of multilayered cuboidal eosinophils and abundant lymphoid tissue with germinal centers similar to the WT form, were reported.5,6 In salivary gland pathology, WT-like MEC is a rare tumour and some of the tumours have been misdiagnosed as WT in the past due to the significant histologic similarities that exist between WT-like MEC and WT. 7
WT and MEC are neoplasms of salivary gland origin and may be characterized as two distinct neoplasms with different histologic diagnostic criteria and clinical features: WT is a clearly demarcated parotid tumour with a cystic structure and variable papillary shape histologically composed of i) prominent lymphoid stroma and oncocytic epithelial elements; and ii) oncocytic epithelium consists of two layers of outer cuboidal basal cells with nuclei at the base and luminal epithelial cells with nuclei predominantly on the luminal side.8,9 On the other hand, MEC, the most common salivary gland malignancy, particularly in young adults, typically aggregates epidermal cells, intermediate cells and mucous cells, forming a solid or cystic pattern.8,10
When analyzed histologically, WT-like MECs observed under low power exhibit a cystic structure of oncocytic epithelial cells with lymphoid interstitium, and their cellular arrangement and characteristics are strongly reminiscent of WTs. The most important finding is that WT-like MECs lack the well-arranged, bilayered oncocytic epithelial tissue characteristic of WTs. 11 In rare cases, perineural invasion and necrosis are also observed. 12 In the study of McLean-Holden et al., 13 extrafollicular reticulum cells were identified via low molecular weight keratin CAM 5.2 immunohistochemistry in all of the WTs and most of the controlled lymph nodes. Then they considered that WTs arise within intraparotid lymph nodes, presumably from salivary gland inclusions entrapped during embryonic development. But in our case, immunohistochemical analysis of CAM5.2 was negative, which was absent extrafollicular reticulum cells in lymphoid stroma. In addition, D2-40 marked only lymph vessels but was absent in the structure of lymph node sinuses. The above finding strongly supported that lymphoid stroma in WT-like MECs was tumour-associated lymphoid proliferation, which was different from WTs. In the internal peripheral area of the tumour, a few normal salivary duct structures were found, and we think these salivary ducts mimicked salivary inclusions of the lymphoid stroma of WT.
Since the first report by Ishibashi et al. 1 in 2015, only 28 cases (Table 1) of WT-like MECs have been reported in the literature, including the present case.1,2,5–7,11,12,14–18 The tumour preferentially affect females and all cases originate in the parotid gland. It is generally thought that this tumour appears in adults, but it may also occur in teenagers, as the broad age range is 13–67 years. The main pathologic differential diagnoses of WT-like MEC are WT with squamous and mucous epithelium metaplasia and malignant transformation of benign WT to MEC (MEC ex-WT).
Summary of the WT-like MECs had been reported in the literature.

F: Female; M: Male; WT: Warthin tumor; MEC: mucoepidermoid carcinoma.
In WTs, the formation of squamous and mucous epithelium metaplasia may occur; transformation of WT is usually localized, with an increase in the epithelial layer and the formation of cystic structures covered with mucous and nonkeratinized squamous epithelial cells. The presence of double oncocytic epithelium (mucous and squamous epithelium metaplasia) is one of the most reliable histologic findings that distinguish metaplastic WT from WT-like MEC. In addition, while the rate of transformation of WT to squamous epithelium is 7.5%, the presence of both mucous and squamous epithelium metaplasia in WT is rare (0.2%). 5 In addition, since MEC is more common in adults or teenage females but WT is more common in males in their 50s to 60s who are smokers, 8 a differential diagnosis may be possible based on these clinical features. The most important finding is that WT, which has both mucous and squamous epithelium metaplasia, lacks the MAML2 rearrangement that is frequently present in WT-like MEC and conventional MEC. 16 The present case lacked bilayers of oncocytic epithelial tissue, and the epithelium was multilayered and contained single scattered mucous cells. Thus, based on this pathologic morphology, the present case was diagnosed as WT-like MEC, but not as WT with mucous and squamous epithelium metaplasia.
The most common malignant transformation from the epithelial component of WT may result in squamous cell carcinoma. Other malignant metastatic tumours from WT include oncocytic carcinoma, adenocarcinoma and MEC. 19 However, it is important to note that they may occur simultaneously in the salivary glands as collision tumours. 20 To support the diagnosis of MEC ex-WT, Seifert et al. 21 proposed four criteria: i) The presence of a WT background; ii) a transition zone between the epithelial components of WT and MEC, MEC ex-WT; iii) invasive growth into surrounding tissues; and iv) exclusion of metastasis of the extra-salivary primary tumour to lymphatic stromal components. It is noteworthy that this may also occur in the presence of MAML2 rearrangement. In addition, WT generally occurs more frequently in older males; therefore, it is highly unlikely that MEC ex-WT occurs in children or teenagers. Clinically, signs of the sudden rapid growth of the mass, as well as pain or facial nerve palsy, indicate malignant transformation of WT. Rarely, the epithelial portion of WT tumours may become malignant but the cause of this remains to be fully elucidated. Stimulation by chronic local inflammation, hypoxia and ischemia is thought to have an important role in the transformation from benign epithelium to atypical hyperplasia. 4 In the present case, there was no WT background or no transition zone between the epithelial components of WT and MEC. Therefore, it may be concluded that the final diagnosis in the present case is WT-like MEC, not MEC ex-WT.
The most important diagnostic clue for WT-like MEC is MAML2 rearrangement determined by FISH analysis, 1 which is specific for MEC and correlates with low- and intermediate-grade histology and improved prognosis. 22 Cytogenetic studies by Citak et al., 23 which characterize both MECs, have identified a specific chromosomal translocation: t(11::19) (q21::p13), resulting in a new fusion oncogene, CRTC1-MAML2. Most recent studies have not reported MAML2 rearrangements in WT; in the study of Bieńkowski et al., 16 MAML2 rearrangements were not observed in 114 WT cases, including WT with squamous and mucous epithelium metaplasia. In the present case, the diagnosis of WT-like MEC was accurate according to the typical histological features and MAML2 rearrangement.
Recently, Sakamoto et al. 24 reported a case of non-sebaceous lymphadenoma (NSLA)-like MEC, which was composed of tubular-cystic and solid nests of epithelial cells equally distributed in the lymphoid tissue. Although the histomorphology was not consistent with MEC, a diagnosis of NSLA-like MEC was made based on the presence of the MAML2 rearrangements. Compared with WT-like MEC, they both consisted of a well-bounded and predominant lymphocytic component with germinal centers. Although the epithelial component of NSLA-like could be distinguished, it was absent in mucous and oncocytic cells and formed solid, glandular or cystic epithelial nests.
Current guidelines state that the treatment strategy for MEC is frequently to perform a total parotidectomy with a certain degree of neck dissection. In cases of high-grade tissue or positive resection margins, adjuvant radiation therapy should be considered. 25 To date, few cases have been reported with follow-up information, but with regard to WT-like MEC in a 17-year-old female, there was one case of recurrence as a conventional MEC 4 years later. 12 However, like low-grade conventional MEC, WT-like MEC appears to have a slow biological behavior, 12 and no metastasis of WT-like MEC has been reported to date. In addition, most cases of WT-like MEC have been successfully treated with complete local excision. In the present clinical report, since the resection margins and lymph node metastasis were negative, postoperative adjuvant therapy was not performed, and no recurrence or metastasis was observed within 22 months of follow-up.
The present clinical report described a rare case of WT-like MEC and the previously published literature on this tumour was reviewed. Clinical and pathologic features may help in the differential diagnosis between WT-like MEC, WT with squamous and mucous epithelium metaplasia and WT MEC ex-WT. Detection of MAML2 rearrangements may also lead to an accurate pathology diagnosis. WT-like MEC as a special subtype of MEC has special histological characteristics, which have been included in the 2022 World Health Organization classification of salivary tumours. However, further observations and more case reports are required to clearly define this variant.
Footnotes
Authors’ contributions
All authors contributed to the study conception and design. LY performed pathological examination, collected clinical data and drafted the manuscript. Images were collected and the layout was designed by JX. Immunohistochemistry was performed by LL. XL is the supervisor of the study and was responsible for the funding, design and quality control of the paper. All authors read and approved the final manuscript. Data authentication is not applicable.
Availability of data and materials
All data generated and/or analysed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient consent for publication
The patient provided oral informed consent for the case information and images in the article to be published.
Author biographies
Limin Yan is a professor in pathology. Her area of research is clinical pathological diagnosis and molecular biology of tumor.
Jinheng Xu is a professor in pathology. Her area of research is clinical pathological diagnosis of tumor.
Liyun Liu is a professor in pathology. Her area of research is clinical pathological diagnosis of lymphoma.
Xin Li is a professor in neurology. Her area of research is neurological diseases of the head.
