Abstract
Objective:
The role of hyperoxia in patients with traumatic brain injury (TBI) remains controversial. The objective of this study was to determine the association between hyperoxia and mortality in critically ill TBI patients compared to critically ill trauma patients without TBI.
Design:
Secondary analysis of a multicenter retrospective cohort study.
Setting:
Three regional trauma centers in Colorado, USA, between October 1, 2015, and June 30, 2018.
Patients:
We included 3464 critically injured adults who were admitted to an intensive care unit (ICU) within 24 h of arrival and qualified for inclusion into the state trauma registry. We analyzed all available SpO2 values during the first seven ICU days. The primary outcome was in-hospital mortality. Secondary outcomes included the proportion of time spent in hyperoxia (defined as SpO2 > 96%) and ventilator-free days.
Interventions:
None.
Measurements and Main Results:
In-hospital mortality occurred in 163 patients (10.7%) in the TBI group and 101 patients (5.2%) in the non-TBI group. After adjusting for ICU length of stay, TBI patients spent a significantly greater amount of time in hyperoxia versus non-TBI patients (p = 0.024). TBI status significantly modified the effect of hyperoxia on mortality. At each specific SpO2 level, the risk of mortality increases with increasing FiO2 for both patients with and without TBI. This trend was more pronounced at lower FiO2 and higher SpO2 values, where a greater number of patient observations were obtained. Among patients who required invasive mechanical ventilation, TBI patients required significantly more days of ventilation to day 28 than non-TBI patients.
Conclusions:
Critically ill trauma patients with a TBI spend a greater proportion of time in hyperoxia compared to those without a TBI. TBI status significantly modified the effect of hyperoxia on mortality. Prospective clinical trials are needed to better assess a possible causal relationship.
Keywords
Introduction
Clinical variation in oxygenation for traumatic brain injury (TBI) protection may be associated with both protection and exacerbation of neuronal injury and associated neurological outcomes. TBI is characterized by initial local and/or regional trauma with edema followed by secondary injuries resulting from subsequent inflammatory reactions.
1
Hypoxia, hypotension, and hypothermia are key drivers of such secondary injury and are the targets of many therapeutic interventions.
2
Oxygen administration can be a lifesaving maneuver with undisputed importance to minimize the risk of hypoxia and associated secondary injury in TBI and other non-TBI critically injured patients.3,4 However, the administration of oxygen in excess of physiologic needs is common and frequently leads to hyperoxia (oxygen saturation [SpO2] > 96% or arterial partial pressure of oxygen [Pa
The effects of hyperoxia in TBI patients, however, remain controversial. Recent studies have yielded conflicting conclusions. Although the primary outcomes of interest varied widely (neurological outcomes, mortality, or acute lung injury), some studies supported the avoidance of hypoxia, some supported hyperoxia, and others were neutral.
13
For example, early hyperoxia in severe trauma patients, including those with TBI, may be associated with reduced in-hospital mortality.
14
Hyperbaric hyperoxia may also decrease intracranial pressure, improve cerebral oxygenation, and reduce mortality in severe TBI patients.
15
Similar improvements in 6-month neurological function have been observed with normobaric hyperoxia.
16
However, other studies suggest early hyperoxia may increase mortality in TBI patients depending on the threshold chosen for hyperoxia (Pa
In this study, we report the results of a secondary analysis of a large multicenter cohort of critically injured patients 12 to compare critically ill trauma patients with and without TBI. The objective was to compare time spent in hyperoxia and to determine whether hyperoxia is more strongly associated with mortality in critically ill trauma patients with and without TBI. We hypothesized that supplemental oxygen use and associated hyperoxia would be higher in critically ill trauma patients with TBI compared to non-TBI trauma patients. Furthermore, we hypothesized that time in hyperoxia would be associated with increased mortality in TBI patients to a greater extent than in trauma patients without TBI.
Materials and methods
Study design
We performed a secondary analysis of a large multicenter cohort study of critically injured trauma patients 12 to evaluate the association between hyperoxia and supplemental oxygen use and in-hospital mortality in critically injured patients both with and without TBI. Subjects were identified from three regional trauma centers affiliated with the University of Colorado Health (UCHealth) system. These included two level I trauma centers (University of Colorado Hospital, Aurora, CO, and Memorial Hospital Center, Colorado Springs, CO) and one level II trauma center (Medical Center of the Rockies, Loveland, CO). All share a common reporting structure to the Colorado Trauma Registry and use the same electronic health record system (Epic, Verona, WI). Our study was reviewed and approved by the Colorado Multiple Institutional Review Board (COMIRB) #17-1359.
Study sample
Critically ill trauma patients admitted to the ICU within 24 h of admission at a participating hospital between October 1, 2015, and June 30, 2018, were included in this study if they also had an ICD-10 code for acute injury/trauma and met criteria for entry into the Colorado state trauma registry. Patients without an injury code from the trauma registry definitions were excluded. Subjects were further divided into those with a TBI and those without a TBI according to ICD-10 codes (Supplementary Table 1). ICD-10 codes for TBI screenings (Z13.850) or personal history of TBI (Z87.820) were not included in the definition of TBI.
Outcomes and variables collected
Details of individual variable definitions and data collection methods are described in our previously published cohort study. 12 Briefly, we collected all measurements of fraction of inspired oxygen (FiO2) and SpO2 from the time of presentation in the emergency department through the first 7 days in the ICU. Measured covariates included: age, race/ethnicity, sex, smoking status, Acute Physiology and Chronic Health Evaluation (APACHE) II score, mechanism of injury, patient comorbidities (presence of a cardiovascular comorbidity, presence of a chronic pulmonary comorbidity, and the number of other comorbidities), and time of intubation and extubation (if applicable). Cardiovascular comorbidities included congestive heart failure, valvular disorders, pulmonary circulation disorders, peripheral vascular disease, and coronary artery disease. Chronic pulmonary disorders included chronic obstructive pulmonary disorder, asthma, interstitial lung disease, and fibrotic lung disease (Supplemental Table 2). We categorized oxygenation status based on a modified Delphi approach agreed upon by a panel of 31 international military and civilian experts in trauma surgery, emergency medicine, critical care, and military operational medicine as hypoxia (SpO2 < 88%), borderline (SpO2 88–89%), normoxia (SpO2 90–96%), and hyperoxia (SpO2 > 96%). 21 The primary outcome was in-hospital mortality. Secondary outcomes included: the proportion of time spent in hyperoxia (defined as SpO2 > 96% and either on supplemental oxygen or with FiO2 > 21%) and ventilator-free days (VFD) to day 28.
Statistical analysis
We calculated summary statistics for all variables overall and stratified by TBI status. Differences in patient characteristics were analyzed using chi-squared tests for categorical data and either unequal variance t-tests or Mann–Whitney tests for continuous data, as appropriate. The primary outcome, in-hospital mortality, was analyzed using a Cox proportional hazards regression model with time-dependent covariates. TBI status was the primary predictor of interest. All available oxygenation variables (SpO2 and FiO2) were evaluated longitudinally for the first seven days of the ICU stay. Linear and quadratic terms were used for both SpO2 and FiO2, along with their interaction, to accommodate a nonlinear relationship between these variables and the risk of mortality. 12 Because of the difficulty of interpreting coefficients in a nonlinear interaction model, graphical summaries of the relationship between the oxygenation variables and risk of mortality were assessed using risk scores. These risk scores are calculated as the ratio of the exponentiated linear predictor with specified values of the oxygenation variables and reference values for all other covariates to the exponentiated linear predictor for a hypothetical patient with mean values for all predictors included in the model.
We conducted a secondary analysis of VFD. Subjects who died were assigned to a different category than those with zero VFD. Our first analysis employed a Wilcoxon test to determine whether a significant difference in the distribution of VFD exists between TBI and non-TBI patients due to the highly non-normal nature of this outcome variable. For covariate-adjusted modeling, we analyzed ventilator days (i.e. number of days on a ventilator while in hospital for each patient who survived hospital discharge) among survivors using a hurdle model with negative binomial outcome distribution. Hurdle models treat zero responses differently from nonzero responses and involve specifying a logistic model for the probability of response exceeding zero and a separate truncated count model for those responses that do exceed zero. 22 TBI status was included as a predictor in these models, as well as primary exposure variables, percentage of time in hyperoxia (SpO2 > 96%), and percentage of time on room air (FiO2 = 21%). Interactions between TBI status and these exposure variables were included to test for differential effects between TBI and non-TBI patients of oxygenation on mean ventilator days.
Another secondary analysis was performed to assess how the percentage of time spent in hyperoxia in the first 7 days of hospitalization might depend on TBI status. For these models, the data were reformatted by ventilation status so that each patient contributed up to two rows of data, one for time spent mechanically ventilated and one for time spent not mechanically ventilated. However, if a patient spent the entirety of their first 7 days in the hospital in one or the other of these states, there would only be a single row of data for that patient. We used a generalized estimating equations approach to fit regression models for the dependence of the proportion of time spent in hyperoxia on explanatory variables, including TBI status and ventilation status. This approach was adopted to address issues with the outcome distribution, which is a proportion (bounded between 0 and 1), and with correlation within patients (due to having repeated observations for those who spent time both on and off of mechanical ventilation). The identity link function was used in order to maintain interpretations of regression coefficients as mean differences in the proportion of time spent hyperoxic. TBI status was again the primary predictor of interest, while ventilation status was included in the model as a covariate.
We adjusted all regression models for age, race/ethnicity, sex, smoking status, APACHE II score, presence of cardiopulmonary comorbidities, number of other comorbidities, and MOI based on the results of univariate analyses (Supplemental Tables 1–3). Analyses were performed in R (Vienna, Austria) 23 with the exception of the proportion of time hyperoxia analysis, with was performed with SAS software (v 9.4 SAS Institute, Cary, NC) and PROC GENMOD. Estimation of all Cox models was performed with the survival package v.2.44.1.1 in R v3.5.2. 24 Marginal risk scores were calculated using the ggeffects package v.0.9.0 in R v3.5.2. 25 Hurdle models were estimated using the pscl package (v.1.5.5 (32,33).
Results
Patient characteristics
Out of 3464 individuals, 1524 (44%) were admitted with either a primary or secondary diagnosis of TBI (Table 1). No statistically significant differences in age, sex, race, or ethnicity were noted between TBI and non-TBI patients. The median APACHE II scores were similar between TBI and non-TBI groups. Patients with a TBI were more likely to have a fall (49.6% vs. 24.8%) or motor vehicle collision (36.9% vs. 26.1%) as the mechanism of injury and less likely to have penetrating trauma (3.9% vs. 10.2%). The proportion of patients requiring mechanical ventilation outside of the operating room at any point during their hospitalization (40.9% vs. 41.2%) was similar between groups.
Characteristics of critically ill trauma patients stratified by TBI status.

APACHE II, Acute Physiology and Chronic Health Evaluation II; IQR, interquartile range; LOS, length of stay in days; SD, standard deviation; TBI, traumatic brain injury.
Primary outcome
The primary outcome (in-hospital mortality) occurred in 163 patients (10.7%) in the TBI group and 101 patients (5.2%) in the non-TBI group. Cox regression modeling revealed a significant effect modification of TBI status for the nonlinear interaction between SpO2 and FiO2 (p = 0.005) (Figure 1). The risk for mortality was higher for TBI patients with hyperoxia than non-TBI patients with hyperoxia, particularly at lower FiO2. At each specific SpO2 level, both for patients with and without TBI, the risk of mortality increases with increasing FiO2. This trend was observed at all FiO2 and SpO2 levels but was more pronounced at lower FiO2 and higher SpO2 values where a greater number of patient observations were available.

Mortality risk at given SpO2 and FiO2 by TBI status. (A) Heatmap of risk scores for mortality at each pulse oximetry (SpO2) and fraction of inspired oxygen (FiO2) level stratified by TBI status. For each SpO2 level, greater FiO2 increases risk of mortality. Greater FiO2 increased the risk for mortality in TBI patients to a greater extent than in non-TBI trauma patients. (B) Heatmap of risk differences between patients with and without TBI. Circles in the bottom right panel represent the number of patient-hour observations associated with each SpO2 and FiO2 level. The greatest proportion of observations was obtained at higher SpO2 and lower FiO2 values. All risk scores are relative to a patient with average values of all covariates. Therefore, a score of 1 implies an equal risk to the average, a score greater than 1 implies elevated risk, and a score less than 1 implies reduced risk.
Secondary outcomes
Of the 171,295 patient-hours recorded for TBI patients, 84,277 (49.2%) were spent in hyperoxia (SpO2 > 96%) and 84,812 (49.5%) in normoxia (SpO2 90–96%) (Table 2). Non-TBI patients spent 101,139 h (44.0%) in hyperoxia and 124,814 h (54.3%) in normoxia (Table 2). Very little time was spent in either borderline (SpO2 88–89%) or hypoxia (SpO2 < 88%) categories for both groups. After adjusting for ICU length of stay and mechanical ventilation status, critically ill TBI patients spent significantly more time in hyperoxia than critically ill non-TBI patients (p = 0.024).
Outcomes by TBI status.a
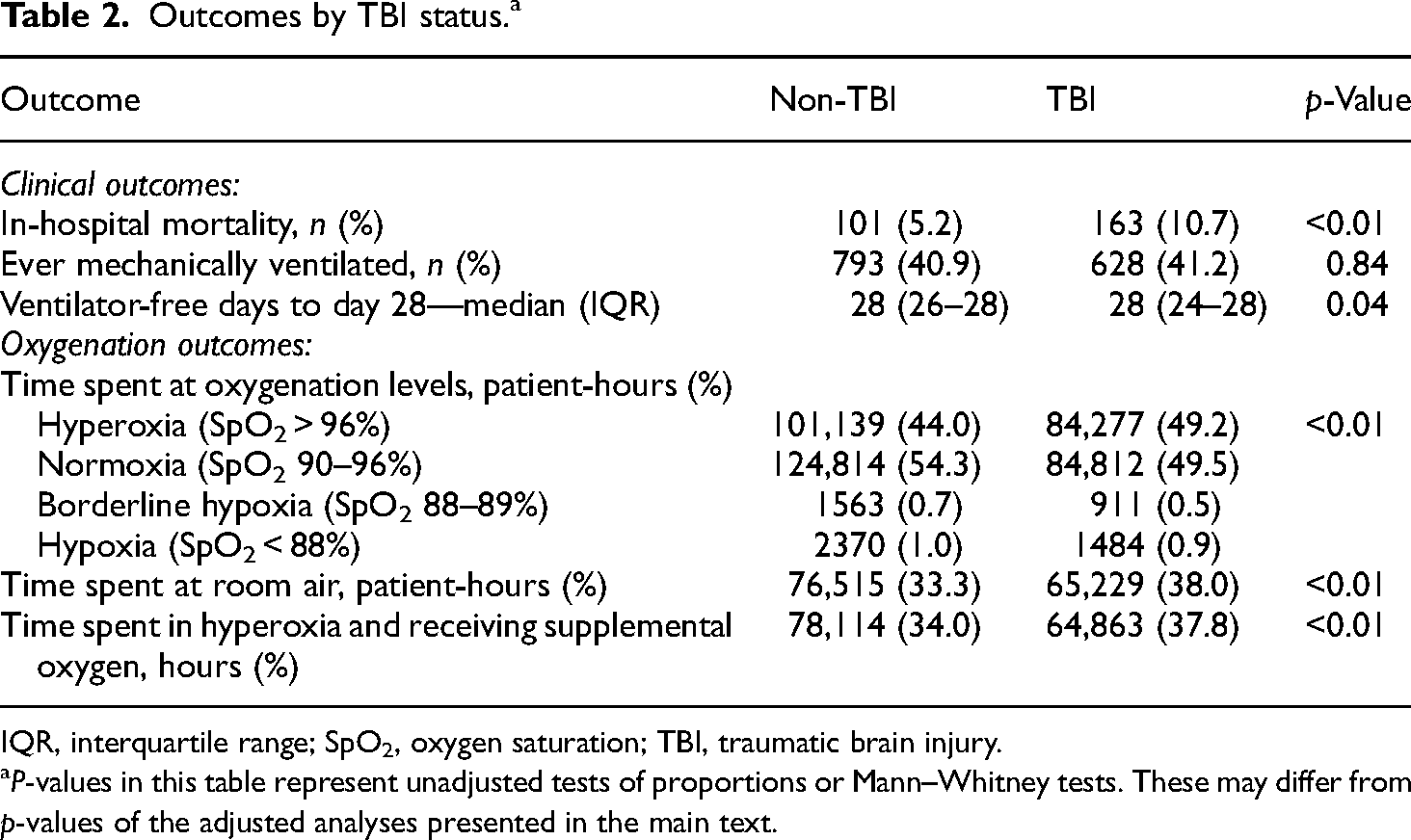
IQR, interquartile range; SpO2, oxygen saturation; TBI, traumatic brain injury.
P-values in this table represent unadjusted tests of proportions or Mann–Whitney tests. These may differ from p-values of the adjusted analyses presented in the main text.
Critically ill TBI patients experienced a median of 28 VFD (IQR 24–28) to day 28, while critically ill non-TBI trauma patients experienced a median of 28 VFD (IQR 26–28) to day 28 (Table 2). Unadjusted analysis indicated the distribution of VFD differed between TBI and non-TBI patients (p = 0.04) (Figure 2). Adjusted analyses with the hurdle model revealed patients with TBI were not significantly more likely than patients without TBI ever to require invasive mechanical ventilation (p = 0.086) (Supplemental Table 3). However, among patients who did require invasive mechanical ventilation, TBI patients experienced a significantly greater duration of ventilation to day 28 than non-TBI patients (p < 0.001).

Proportion of patients with a given number of VFD. Proportion of patients with a given number of VFD among TBI (blue) and non-TBI (red) patients. VFDs for TBI and non-TBI patients were significantly different (p = 0.039).
Discussion
In this analysis of a large multicenter cohort of critically injured patients, hyperoxia was more common in TBI patients, and the association between hyperoxia and risk for mortality is significantly modified by TBI status. After adjusting for time spent in hyperoxia, critically ill TBI patients also experienced a greater duration of invasive mechanical ventilation to day 28 than their non-TBI counterparts.
Exposure to supraphysiologic levels of oxygen remains common for both non-TBI and TBI patients. However, in our cohort, patients with TBI spent a greater proportion of their time in hyperoxia when compared to patients without TBI. The clinical reasoning and decision-making processes that lead to these differences remain unclear. Clinicians may wish to minimize exposure to hypoxia in TBI patients and thereby mitigate the potentially detrimental effects of cerebral ischemia on brain recovery. 26 The use of cerebral oxygenation monitoring, such as near-infrared spectroscopy (NIRS) and continuous brain tissue oxygenation (PbtO2), may guide clinical decisions regarding oxygen titration. However, cerebral oxygenation monitoring was used infrequently at the three included institutions during the study period. Additionally, clinicians may be actively targeting higher SpO2 based on ongoing clinical trials evaluating the role of hyperbaric hyperoxia. 27 Further data are required to elucidate the source of the observed difference in the proportion of time spent in hyperoxia.
Of note, the risk for mortality was greater in non-TBI patients than in TBI patients at high FiO2 and low SpO2 values (Figure 1B). However, very few patient observations were recorded within these parameters (FiO2 > 80% and SpO2 < 90%), likely because such a clinical scenario occurs infrequently. We, therefore, caution against overinterpretation and dissemination of the risk difference between TBI and non-TBI patients in this setting (i.e. the right lower corner of Figure 1B).
One distinguishing feature of our study is the continuous assessment of oxygen in terms of both dose and duration of oxygen exposure. Prior studies have sought to establish a safe “upper limit” of hyperoxia for TBI patients. Rincon et al. demonstrated that mechanically ventilated TBI patients with a Pa
Duration of exposure likely modifies the effects of hyperoxia in TBI patients, as it does in other populations. 12 However, hypoxia in TBI is also associated with increased morbidity and mortality.1,2 The results of our study and others may leave clinicians feeling a need to “thread the needle” with regard to oxygenation in TBI patients. That is, strictly avoid both hypoxia and hyperoxia simultaneously. Our group demonstrated in a pilot study that targeted education and best practice advisories could significantly improve supplemental oxygen titration in critically ill trauma patients. 29 Critical care providers were responsive to these changes and readily accepted the down-titration of oxygen in patient care. Recent awareness of the potential harms associated with hyperoxia in critically ill patient populations made this paradigm shift easier to attain. Future work may focus on provider views on oxygenation in TBI patients and the potential factors that may limit willingness to reduce supplemental oxygen use in these patients.
This study should be interpreted in the context of several important limitations due to the nature of the disease of interest and the selected primary outcome. First, the study was retrospective in nature. The primary outcome was uncommon—only 7.6% of patients died during hospitalization. Our large cohort, almost 3500 patients, partially mitigates this limitation, as power in survival analyses of the type we performed derives from the number of deaths rather than the proportion. We did not control for prehospital care and oxygenation practices which may have influenced the primary outcome. Establishing a causal connection between FiO2/SO2 and mortality is statistically challenging because they both influence each other via different pathways. We accounted for this by employing well-validated methods for FiO2/SO2 previously used by our group.12,29,30 We used a previously identified, optimal range of SpO2 (90–96%) to define normoxia, with hyperoxia defined as SpO2 > 96%. However, the precise definition of normoxia and hyperoxia in critically ill trauma patients and, even more specifically, TBI patients is unknown. This study provides a basis to suggest that targeting SpO2 90–96% may improve patient outcomes in the TBI population. We are evaluating this approach in an ongoing multicenter trial to identify precise optimal oxygenation targets in critically ill trauma patients (NCT #04534959). 30 Finally, higher elevation may affect oxygen delivery and oxygenation targets. All three of the included hospitals are located more than 1500 m above sea level. Therefore, it is possible our findings may not be replicated at lower elevations.
In conclusion, this large multicenter cohort study demonstrated that hyperoxia is both more common and associated with a greater risk for mortality in critically ill TBI patients compared to critically ill non-TBI trauma patients. Critically ill patients with TBI spent a significantly greater proportion of time in hyperoxia compared to critically ill non-TBI trauma patients. Prospective studies are needed to elucidate the mechanisms underlying these differences, inform the clinical decision-making processes driving the increased time TBI patients spend in hyperoxia, and create interventions to target normoxia in this patient population.
Supplemental Material
sj-docx-1-sci-10.1177_00368504231160416 - Supplemental material for Hyperoxia is associated with a greater risk for mortality in critically ill traumatic brain injury patients than in critically ill trauma patients without brain injury
Supplemental material, sj-docx-1-sci-10.1177_00368504231160416 for Hyperoxia is associated with a greater risk for mortality in critically ill traumatic brain injury patients than in critically ill trauma patients without brain injury by David J Douin, Layne Dylla, Erin L Anderson, John D Rice, Conner L Jackson, Vikhyat S Bebarta, Robert T Neumann, Steven G Schauer and Adit A Ginde in Science Progress
Footnotes
Acknowledgments
The authors thank Caroline M. Ledbetter, MPH for her work with preliminary data analysis.
Author contributions
Study concept and design: DJD, LD, and AAG; Acquisition of data: ELA, AAG; Statistical analysis: JDR, CLJ; Interpretation of data: All authors; Drafting of the manuscript: DJD, LD; Critical revision of the manuscript for important intellectual content: All authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. This manuscript expresses the authors’ opinions and does not reflect the policy or opinions of the Department of the Army, Department of the Air Force, Department of Defense, or US Government.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the U.S. Department of Defense/SOCOM (grant number W81XWH-17-C-0241).
Supplemental material
Supplemental material for this article is available online.
Author biographies
David J Douin, MD is an assistant professor of anesthesiology at the University of Colorado School of Medicine. His research focuses on epidemiological and clinical trials of critically ill patients, particularly on oxygen delivery, transfusion, and injured/trauma patients. He has been the site Co-PI of two trials assessing the effects of hyperoxemia in critically injured trauma (NCT04534959) and burn (NCT04534972) patients. National leadership in clinical trials networks includes the NIH Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV)-3/4 and the CDC-funded Investigating Respiratory Viruses in the Acutely Ill (IVY). Funding for his research includes NIH (NIGMS), DoD, and CDC, and he has published over 45 original research articles in leading medical journals.
Layne Dylla, MD, PhD is an assistant professor of emergency medicine at the University of Colorado School of Medicine. Her research interests include the emergency and pre-hospital treatment of acute ischemic stroke. She has published over 15 original research articles in leading medical journals.
Erin L Anderson, RN is a research services program manager in the department of emergency medicine at the University of Colorado School of Medicine. She is the lead program manager for the SAVE-O2 clinical trials and related projects.
John D Rice, PhD is a research assistant professor in the Department of Biostatistics in the University of Michigan School of Public Health. He received his MSPH in Biostatistics from Emory University in 2010, and his PhD in Biostatistics from the University of Michigan in 2015, where his dissertation focused on statistical methods for cancer research. He completed postdoctoral training at the University of Rochester in 2017, where he worked in the areas of HIV testing behavior and cardiovascular outcomes. Prior to joining the University of Michigan, Dr. Rice was an assistant professor in the Department of Biostatistics and Informatics in the Colorado School of Public Health from 2018–2022. His research interests include longitudinal data analysis, recurrent events, and semiparametric regression methods for binary and semicontinuous outcomes data.
Conner L Jackson, MS is an instructor of biostatistics and informatics at the University of Colorado Center for Innovative Design and Analysis (CIDA)
Vikhyat S Bebarta, MD is a professor of emergency medicine at the University of Colorado School of Medicine and director of the CU Anschutz Center for Medicine and Battlefield (COMBAT) Research.
Robert T Neumann, MD is an associate professor of neurosurgery and director of the Neuro-Intensive Care Unit at the University of Colorado School of Medicine. His research interests include traumatic brain injury, and the care of critically ill patients with neurologic injuries or disorders.
Steven G Schauer, DO, MS is an emergency medicine physician at the US Army Institute of Surgical Research, JBSA Fort Sam Houston, in San Antonio TX. His research interests include traumatic injury, critical illness, and oxygenation targets.
Adit A Ginde, MD, MPH is a professor of emergency medicine at the University of Colorado School of Medicine. His research focuses on epidemiological and clinical trials in the acute care setting, with a particularly focus on early care of critically ill and injured patients. Specifically, he has extensive experience with designing, overseeing, and implementing clinical trials in a variety of clinical environments including emergency department, inpatient (including intensive care unit), perioperative, and outpatient settings, particularly in the areas of acute respiratory failure and sepsis/infection (including COVID-19). He has been the lead investigator for 6 large multicenter clinical trials and participated in the design (as protocol committee member and co-investigator) and implementation (as site PI) of over a dozen other clinical trials. National leadership in clinical trials networks include the NHLBI Prevention and Early Treatment of Acute Lung Injury (PETAL) network executive and steering committees and steering committees for the NHLBI Collaborating Network of Networks for Evaluating COVID-19 and Therapeutic Strategies (CONNECTS), the trans-NIH Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV)-3, the CDC-funded Influenza and Other Viruses in the Acutely Ill (IVY), the DoD-funded Pragmatic Critical Care Research Group (PCCRG), and the NIAID Strategies and Treatments for Respiratory & Viral Diseases (STRIVE). Funding for his research includes NIH (including NHLBI, NCATS, NIAID, NINDS, NIA), DoD, CDC, foundation, and industry sponsors, and I have published over 240 original research articles in leading medical journals.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
