Abstract
There are two primary threats to the brain after concussion. The first is a buildup of neurotoxic proteins in the brain. The second, a partial consequence of the first, is a sustained neuroinflammatory response that may lead to central sensitization and the development of persistent post-concussive symptoms. These threats make neurotoxin clearance a high clinical priority in the acute period after injury. The glymphatic system is the brain's primary mechanism for clearing neurotoxic waste. The glymphatic system is intimately tied to the sleep cycle and circadian dynamics. However, glymphatic dysfunction and sleep disturbances are nearly ubiquitous in the acute period after concussion injury. Because of this, sleep optimization via circadian therapy is a time-sensitive and critical tool in acute concussion management.
Introduction
Traumatic brain injury (TBI) cases in the United States approach 2 million annually. 1 Nearly 90% of TBIs are mild, injuries that are commonly referred to as “concussions.” 2 The two most concerning sequelae of concussion injuries are the possibility of early dementia (including manifestations such as chronic traumatic encephalopathy [CTE]) 3 and the development of persistent post-concussive symptoms (PPCS). 4
Because of these concerns, tremendous efforts are being spent to address concussion injuries. Prevention efforts have advanced significantly, including the development of sports equipment technology 5 and changes in the rules of contact sports. 6 Significant resources are being spent increasing education about concussion. 7 Return-to-play laws have been passed for youth athletes and the related protocols are being continually refined. 8 Improved diagnosis via biological and clinical biomarkers is currently underway. 9 Finally, progress is being made on determining who is at risk for PPCS and the optimal treatments for PPCS. 4
Conspicuously lacking in this coordinated effort is elucidation of optimal acute treatment protocols. Increased attention is needed in acute care protocols, principally because the two primary pathophysiological forces that occur in the acute period after concussion, neurotoxin build-up and neuroinflammation, peak in the first week after injury. 10 In this paper, we focus on the importance of glymphatic system dysfunction in acute concussion pathophysiology, its relationship to near-ubiquitous sleep disturbances in the acute period, and the importance of circadian therapy as a critical early intervention tool for acute concussion management (Table 1).
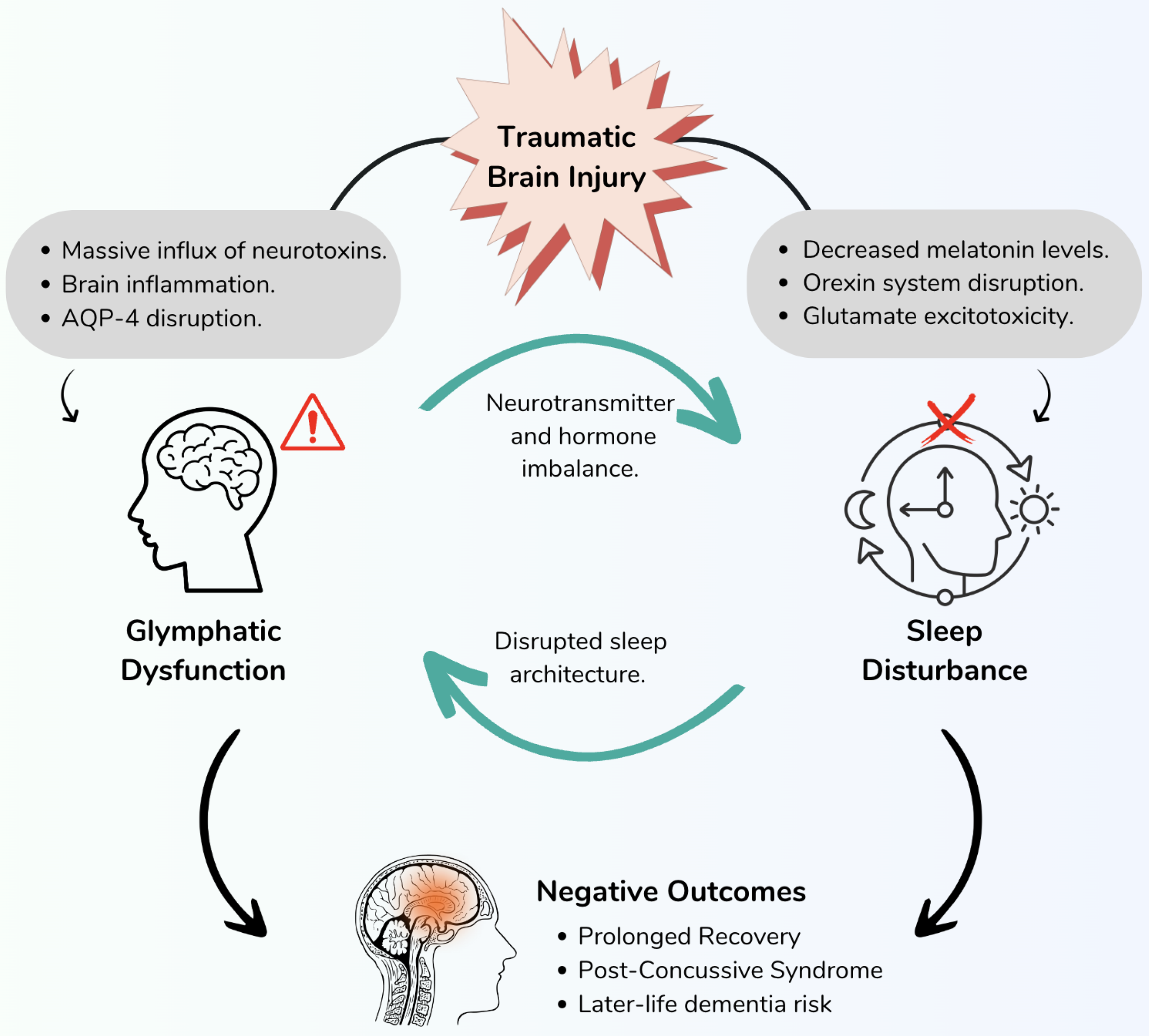
There is a reticulum of causation between TBI, glymphatic dysfunction, and sleep disturbances. The pathological consequences of TBI initiate a cascade of factors that disrupt both sleep and glymphatic clearance processes. These interconnected systems, once impaired, contribute to a cycle of dysfunction, exacerbating the negative outcomes following the injury. As a result, individuals may experience compromised recovery and suboptimal outcomes.
Selected studies that support circadian therapy for concussion management.
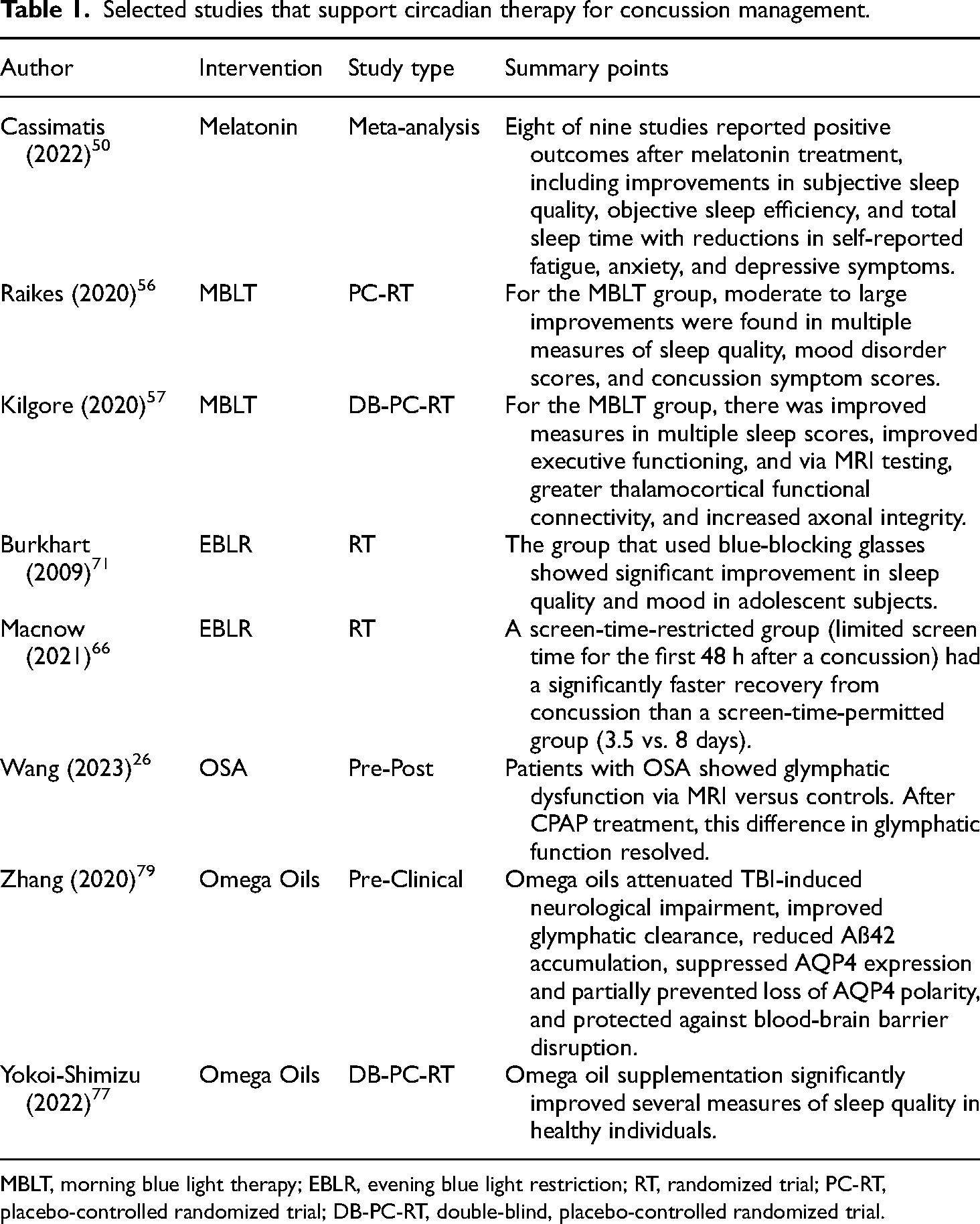
MBLT, morning blue light therapy; EBLR, evening blue light restriction; RT, randomized trial; PC-RT, placebo-controlled randomized trial; DB-PC-RT, double-blind, placebo-controlled randomized trial.
Neurotoxic proteins in concussion
After concussive injury, whether through axonal strain or tearing, there is a release of structural proteins into the interstitial fluid of the brain. These include tau proteins and beta-amyloid proteins, notorious for their role in neurodegenerative diseases. When these proteins are outside of their intracellular physiological context, they tend to propagate in a prion-like fashion, forming plaques and tangles. 11
The tau protein is of particular importance in concussion pathophysiology. 12 The tau protein is a microtubule associated protein. In pathological contexts, tau protein forms neurofibrillary tangles, one of the well-known hallmarks of neurogenerative diseases. While intracellular tauopathy was initially thought to be causative of neuronal damage, new research suggests that extracellular tau is also a primary threat to brain health. 13 Extracellular tau is not only directly toxic to neurons, it also triggers a sustained neuroinflammatory response via microglial activation. 12
After concussion injury, there is a large release of tau proteins into the brain. This has been recorded in the interstitial fluid in animal models and in the cerebrospinal fluid and serum in human subjects.10,14,15 Beyond being a biomarker of acute neuronal injury, serum tau is an indicator of slower recovery and the development of neurobehavioral deficits.
15
Several important findings were revealed regarding the relationship between serum tau levels in the acute period and clinical outcomes in a recent study of 229 TBI patients:
Lower levels of acute period serum tau are associated with favorable outcomes. Early serum tau is negatively correlated with the presenting Glasgow Coma Score (GCS; i.e., high serum tau is positively correlated with injury severity). Tau protein levels at days 1, 3, and 5 after TBI are significantly correlated with cognitive dysfunction 6 months after injury.
10
The fact that acute serum tau levels are predictive of poor outcomes is important because it highlights the importance of decreasing the burden of brain tau levels in the first week of recovery. As we will outline below, the primary mechanism for clearing extracellular tau proteins is the glymphatic system.
Importantly, tau accumulation after concussion is a precursor state to CTE. CTE is defined histologically as a perivascular accumulation of phosphorylated tau proteins (perivascular tauopathy). 16 The proximate cause of CTE is the high extracellular burden of tau proteins after each concussive event. Regardless of whether a person develops PPCS, each concussive is a step closer to the development of CTE. This makes tau clearance in the acute period an important clinical priority.
The glymphatic system
In 2012, Maiken Nedergaard and colleagues first outlined the function of the glymphatic system in brain waste metabolism.17,18 The glymphatic system is now central to our understanding of brain physiology and neuropathology. In its essence, the glymphatic system cleans the interstitial space of brain waste products through the convective flow of water (interstitial and cerebral spinal fluids [CSFs]) from intracerebral arteries to veins. This waste transport is facilitated by water channels (specifically aquaporin-4) located in astrocyte processes that surround cerebral veins. 19
Impairment of glymphatic function is central to neurodegenerative pathogenesis. In fact, some researchers now propose that neurodegenerative diseases such as Alzheimer's disease and CTE be recategorized as “interstitial fluidopathies.” 20 When neurotoxic substances like amyloid and tau cannot be sufficiently cleared, neuropathology ensues through a downward spiral; impaired perivascular clearance leads to tau accumulation, neurodegeneration, and neuroinflammation, all of which in turn further impair waste clearance. 21
The glymphatic system is inexorably linked to sleep and the circadian cycle because the convective flow of CSF occurs during deep sleep. Importantly, glymphatic clearance is 80–90% higher during sleep relative to awake states. 22 During slow wave sleep (SWS), the extracellular space expands in brain by more than 60%. 23 This is partially explained by the synchronous firing of brain waves during SWS, which may orchestrate ion-driven fluid influx into the interstitial space. 24 The transition to asynchronous firing in REM sleep then facilitates fluid efflux. Amazingly, the principal biological purpose of dreaming may be to facilitate this important brain-cleaning process.
Given that glymphatic function is so highly dependent on sleep, it is not surprising that sleep disturbances are causally linked to glymphatic dysfunction. For instance, people with obstructive sleep apnea have impaired glymphatic function as determined by diffusion tensor imaging, 25 and this dysfunction reverses with continuous positive airway pressure (CPAP) treatment. 26 In the case of dementia, the appearance of glymphatic dysfunction may coincide with the onset of sleep disturbances, acting in a bidirectional manner. On this point, Nedergaard and Goldman note that “the glymphatic system degrades with age, suggesting a causal relationship between sleep disturbance and symptomatic progression in the neurodegenerative dementias.” 27 In the case of TBI, the appearance of glymphatic dysfunction and sleep disturbances also coincides in a temporal fashion.
Overall, impaired glymphatic function is now considered the “missing link” between sleep disorders and multiple neurological disorders. 28 Many researchers now argue that neurotoxin clearance in the brain through glymphatic system optimization should be a central strategy to treat neurodegenerative diseases. 29 The principal way to do this is through optimization of deep sleep, as this is when the glymphatic system is most active. Sleep, not rest, is what is important.
The glymphatic system and concussion
As noted above, there is a flood of neurotoxic proteins into the interstitial space after concussion injuries. The glymphatic system is the primary mode of clearance for these toxic substances, yet experimental evidence suggests that in the acute phase, the glymphatic system experiences significant dysfunction. In animal models, the glymphatic system becomes impaired by up to 60% after experimental TBI. 21 These deficits are detected in the acute period, 30 but can last for weeks after the injury.21,31
Radiological studies provide evidence of glymphatic dysfunction after TBI in human subjects. Expansion of the perivascular space can be detected by MRI and is used as a proxy for glymphatic dysfunction. Several studies have documented a correlation between an expanded perivascular space and a history of TBI.32,33 Recent studies that employ diffusion tensor imaging also record glymphatic impairment in patients with a history of TBI. 34
Aquaporin-4 knock-out models also provide evidence of the centrality of the glymphatic system after TBI. Multiple studies show that genetic elimination of aquaporin-4 channels leads to glymphatic dysfunction, and subsequently to the accumulation of tau and other biomarkers of neurotrauma.35,36 This corresponds to preclinical studies that show that in the acute phase after TBI, fluid influx is intact but fluid efflux is impaired. 30 The expanded perivascular space studies mentioned above further demonstrate that the impairment is on the efflux, not influx, side of the system. 32 Importantly, impaired efflux is permissive of the perivenous tauopathy seen in CTE. 37
Sleep disturbances and concussion
Sleep disturbances are exceedingly common after concussion injuries, with up to 70% of cases being affected.38,39 These disturbances can include hypersomnia, insomnia, sleep–wake disorders, and sleep apnea. Fatigue is also reported in up to 70% of cases. 40 There is strong clinical evidence that sleep disturbances are both a symptom of mTBI and a causative factor for poor clinical outcomes. 41 Several studies document that sleep disturbance in the acute phase of concussion is predictive of both PPCS incidence and severity.42,43
There are several mechanistic causes of sleep disturbances after concussion. Several recent reports implicate glymphatic disruption as the putative link between concussion and sleep disturbances.28,33 Additionally, neuroinflammation and sleep disorders appear to have a bi-directional relationship in the context of TBI, each reinforcing and propagating the other. 44 Finally, so-called “clock genes,” that is, genes directly connected to circadian physiology, are shown to be altered in preclinical mTBI models. 38 Interestingly, acute sleep disturbances after concussion appear to be worse in female patients. 45 This corresponds to preclinical findings that clock gene expression after mTBI is more significantly altered in females than males. 38
Melatonin regulation is also affected after concussion injury. In fact, the entire neuroendocrine and neuroimmune system becomes imbalanced in the acute phase after concussions, including melatonin regulation. 46 Cerebral melatonin levels are significantly decreased after acute concussion in animal models. 47 In humans, several studies document a decrease in salivary melatonin levels in acutely concussed patients.48,49 Hence, reduced melatonin levels, increased sleep disturbances, and glymphatic dysfunction are all convergent and interconnected phenomena in the acute phase of concussion.
Circadian therapy for acute concussion
Summarizing the information above, acute concussion results in a significant deposition of neurotoxic proteins into the extracellular space. The primary mechanism for bulk removal of these toxins is the glymphatic system. However, this system apparently becomes overwhelmed in the acute period. Simultaneously, sleep disturbances arise in the acute period, affecting the ability of the glymphatic system to clear toxins, presumably through a bidirectional dynamic. These forces induce neuroinflammation which in turn negatively reinforce the impaired waste clearance and sleep disturbances. In short, there is a reticulum of causation between TBI, glymphatic impairment, and sleep disturbances. These dynamics are illustrated in Figure 1.
Based on the above, it can be inferred that sleep optimization through circadian therapy will help restore glymphatic function, improve clearance of neurotoxins, reduce neuroinflammation, and improve outcomes. We will now examine evidence that supports this inference.
The circadian system regulates various bodily functions, including sleep–wake cycles, hormone production, body temperature, metabolism, and cognitive performance. It helps optimize physiological processes to align with the demands of the external environment. Disruptions to the circadian system can lead to disturbances in sleep patterns, mood, alertness, and overall health. Given the significant impact of sleep on concussion recovery and considering that sleep is primarily regulated by the circadian system, it is reasonable to explore therapeutic interventions that support and reinstate healthy circadian rhythms.
Circadian therapy is a general term that describes tools that are used to optimize sleep. Melatonin and light therapy are typical interventions for circadian pathology. However, other treatments that optimize sleep can be considered circadian therapy as they help normalize circadian sleep architecture. Below is a list of interventions that we will discuss in more detail:
Melatonin therapy Morning blue light therapy Evening blue light restriction Sleep apnea treatment Omega oil supplementation
Other important circadian therapy interventions include sleep hygiene education, cognitive behavioral therapy, adoption of a circadian diet, and prescribed physical activity.
Melatonin therapy
Because melatonin dynamics are acutely dysregulated in concussion, melatonin supplementation has become a well-studied treatment strategy in concussion management. 46 In a recent meta-analysis, eight out of nine studies showed that melatonin supplementation for concussed patients resulted in positive outcomes. 50 It is reasonable to infer that the positive effects of melatonin are due to its effect on glymphatic clearance, as melatonin improves glymphatic clearance of neurotoxins in preclinical models,51,52 and neurotoxin burden is directly related to clinical outcomes. 15 Importantly, preclinical models show that early administration of melatonin is needed in order to see its beneficial actions. 53 Melatonin also has powerful antioxidant and cytoprotective effects. 53 Various dosages of melatonin have been studied, ranging from 1–10 mg, typically given 30 min to one hour before bedtime. 54 Interestingly, several studies found that a mid-range dose (around 3–5 mg) showed the most efficacy. 55
Morning blue light therapy
There have been a series of promising studies looking at the effect of morning blue light therapy on concussion patients. These studies show that morning blue light exposure can improve a range of sleep-related outcomes after concussion, including daytime sleepiness, sleep quality, and quality of life. 56 The typical prescription in these studies calls for 30 min of exposure to an LED blue light panel within 2 hours of awakening, with the device at an arm's length. 57
To avoid exacerbating any underlying photophobia symptoms, patients are advised to not look directly into the light. Photophobia is common after TBIs, affecting around 40% of individuals by some estimates. 58 New research suggests that while bright light is commonly reported as a trigger for migraine attacks, this response is likely due to false attribution 59 and light therapy is actually being investigated as a treatment for migraines with photophobia. 60 Nevertheless, photophobia should be considered a relative contraindication to blue light therapy and patients should be advised to stop treatment if it is triggering adverse symptoms.
The effectiveness of blue light therapy has to do with mammalian physiology and how it responds to different light wavelengths. Melanopsin, a light-sensitive protein in human retinal ganglion cells, has its peak absorption around 480 nm, primarily responding to blue light. 61 This maximal absorption influences non-visual responses, including circadian rhythm regulation, melatonin suppression, alertness modulation, and pupil size regulation. Understanding melanopsin's characteristics helps optimize lighting design, chronobiology research, and clinical interventions for improving sleep, circadian rhythms, and overall well-being, including morning blue light therapy.
As for clinical efficacy, morning blue light therapy reduced daytime sleepiness, increased cognitive functioning, and improved phase-advanced sleep timing in a randomized trial. 57 Beyond this, functional neuroimaging show that these clinical responses correspond to increases in grey matter volume, 62 increased functional connectivity, 62 increased white matter diffusion, 63 and increased resting state effective connectivity. 64 Together, these studies provide solid empirical evidence for the value of morning blue light therapy as treatment strategy for concussion.
Evening light restriction
Light restriction is important in acute concussion management, presumably through its effect on circadian physiology. The effects of screen time on concussion in the acute period have received considerable attention. 65 In a randomized clinical trial, screen time restriction in the first 2 days after injury resulted in a significant reduction in recovery times (3.5 vs. 8 days). 66 In fact, the global recommendation is that >2 h of evening use and all night-time use of screens should be avoided. 67 Incidentally, while the blue light component of screens is particularly implicated for causing sleep disturbances, studies show that when “night shift” mode (a blue light filter mode) is used during evening screen time, it has a negligible effect on sleep parameters versus normal mode. 68
Evening blue light is particularly disruptive of normal sleep architecture. 69 This is because evening blue light can disrupt the circadian rhythm by delaying the onset of melatonin secretion and shifting the timing of the biological clock. This can lead to difficulties falling asleep, reduced sleep quality, and overall disruption of the natural sleep–wake cycle.
Evening blue light restriction has proven effective in promoting circadian normalization. 70 In clinical studies, blue-blocking glasses improve subjective sleep quality, 71 reduce sleep onset latency, 72 and normalize melatonin levels. 73 To the best of our knowledge, blue-blocking glasses have not been studied as an adjuvant treatment for acute concussion.
Sleep apnea treatment
As noted above, sleep apnea is a common finding in concussed patients, affecting 35–60% of patients. 74 The presence of sleep apnea is predictive of PPCS severity. 75 Sleep apnea also significantly alters circadian physiology. 76 Importantly, sleep apnea impairs glymphatic clearance, whereas positive airway pressure treatment improves glymphatic function. 26 For these reasons, sleep apnea screening and treatment should occur early after injury should be considered an integral part of circadian therapy in acute concussion management.
Omega oil supplementation
Omega oils are commonly prescribed supplements to help with concussion recovery. Relevant here is the impact of omega oils on the circadian system. Multiple studies show that omega oils have a positive effect on sleep quality. 77 Omega oils also influence circadian variations in blood pressure. 78 Putative mechanisms include a direct effect on melatonin release from the pineal gland and the regulation of norepinephrine. 77
Omega oils also have a direct effect on the glymphatic system. In a TBI animal model, researchers found that omega oils positively influenced glymphatic drainage and reduced neurological impairment after simulated TBI injury. 79 Interestingly, when the aquaporin-4 gene was knocked out, the positive neurological effect was eliminated, indicating that the beneficial effect of the omega oils on neurological outcomes was directly related to improved glymphatic clearance.
Conclusion
Circadian therapy is a non-obvious intervention for concussion, but the clinical and preclinical evidence supporting it is sound. If the clues about ubiquitous sleep disturbances after concussion are followed and the importance of impaired sleep on glymphatic function is understood, the argument for circadian therapy in acute concussion management becomes clear.
Circadian therapy can positively affect the two primary threats after concussion; neurotoxin buildup in the brain and the neuroinflammation that ensues. Further, circadian therapy is a non-toxic, conservative, and affordable intervention. We hope that this scoping review provides support for increased translation of circadian therapy science into the clinical management of acute concussion.
In conducting this review, it is crucial to acknowledge the inherent limitations that shape the scope and depth of our findings. Firstly, as this is a fairly new approach to treatment, identifying an ample number of studies specifically focusing on circadian therapy for concussions is challenging. Furthermore, the heterogeneity of study designs, participant populations, treatment protocols, and outcome measures across the existing literature pose a challenge in drawing definitive conclusions or establishing standardized guidelines for circadian therapy in concussion management.
We recognize that our review represents a snapshot of the literature at a particular point in time, and it may not include the most recent developments in this rapidly evolving field. As the understanding of circadian therapy in concussion management continues to expand, it is essential to acknowledge that our findings may not encompass the most up-to-date research.
Despite these limitations, our review paper provides valuable insights, identifies knowledge gaps, and highlights the need for further research in the intersection of circadian therapy and concussion management. We hope that our work serves as a foundation for future investigations and stimulates discussions that ultimately contribute to advancements in the field, enhancing the effectiveness of circadian-based interventions for individuals recovering from concussions.
Footnotes
Acknowledgments
We would like to acknowledge Brian McGuire, Chief Operating Officer of TBI Virtual, for his general support and encouragement of the research team that put this review together.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors of this manuscript are affiliated with TBI Virtual, a for-profit telemedicine clinic whose primary focus is the treatment of concussion injuries. This manuscript reflects the treatment philosophy and practices of TBI Virtual and as such, represents for-profit services offered by the clinic.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author biographies
Sohaib Kureshi is a practicing Neurosurgeon. His clinical practice includes the treatment of traumatic brain injuries and their sequelae.
Caroline Stowe holds a Doctorate of Nursing Practice (DNP) with additional qualifications in headache medicine (AQH). She specializes in the evaluation and treatment of migraine disorders.
John Francis is a medically trained business consultant (MD, MBA). His consulting focuses on translational medicine, applied clinical research, and organizational management.
Hamid Djalilian is a Professor of Otology/Neurotology. His clinical research focuses on chronic brain sensitivity disorders, such as migraines, vestibular disorders, tinnitus, and post-concussion syndrome.
