Abstract
Background:
Angiogenesis is essential for the repair process after intracerebral hemorrhage (ICH).
Methods:
Given the importance of the extracellular matrix (ECM) in angiogenesis, we analysed the temporal profile of angiogenesis in rat brains on days 4, 7, and 21 after ICH. To this end, we compared the expression of ECM-related genes between ICH-induced and sham-operated groups using a complementary DNA (cDNA) array. We further measured protein expression using western blot and immunohistochemistry assays. Fluorescein isothiocyanate (FITC)-dextran was injected into the tail vein to examine the angioarchitecture in the perihematomal region.
Results:
Among the 88 ECM-related genes, we identified 42, 50, and 38 genes that were significantly upregulated on days 4, 7, and 21 after ICH, respectively (P < 0.05). Particularly, collagens, integrins, and matrix metalloproteinases (MMPs) were significantly increased on day 4 post-ICH and continued to increase at the other time points. Western blot and immunohistochemistry analyses showed a comparable trend in the upregulation of MMPs. Compared to the sham group, FITC-dextran labelling demonstrated decreased perfusion and increased vascular permeability in the perihematomal region in the ICH group. Doxycycline, an MMP inhibitor, significantly reduced angiogenesis (P < 0.05).
Conclusions:
The results of this study indicate that MMPs are involved in modulating angiogenesis following ICH.
Introduction
Intracerebral hemorrhage (ICH) is a devastating cerebrovascular disease associated with significant mortality and morbidity. 1 Despite basic and clinical research efforts, there are no definitive pharmacological or surgical treatment options for ICH. 2 Therefore, further research is warranted to uncover novel therapeutic approaches for ICH. 3
Angiogenesis is a tightly controlled physiological process that includes endothelial cell (EC) activation, extracellular matrix (ECM) degradation, EC proliferation and migration, lumen formation, and vessel maturation and stabilization.4,5 Following stroke, angiogenesis is usually accompanied by axonal outgrowth and neural stem/progenitor cell (NSC) activation, which can contribute to functional recovery. 6 In the perihematomal region, the new microvasculature can restore blood reperfusion and oxygen supply to the damaged brain tissues and aid in the functional recovery of the blood-brain barrier (BBB). 7 –9 Therefore, modulating angiogenesis could be a promising therapeutic strategy after ICH.
Accumulating evidence has demonstrated that the ECM has a pivotal role in angiogenesis. 10 The ECM creates the physical scaffold necessary to maintain blood vessel organization. Further, the ECM participates in biochemical and biophysical signalling transduction during angiogenesis.9–12 Specifically, collagens and fibronectins stimulate EC tubular morphogenic events; laminins are involved in EC tip formation and sprouting, and are also crucial for maintaining vascular homeostasis, and proteoglycans regulate EC migration to form new vessels. In contrast, thrombospondins have anti-angiogenic properties that maintain angiogenesis balance. 13
During angiogenesis, matrix metalloproteinases (MMPs) degrade collagen and other ECM components, thereby enabling EC migration from preexisting vessels towards angiogenic stimuli. 14 There are six types of MMPs: collagenases (MMP-1, −8, −13), gelatinases (MMP-2, −9), stromelysins (MMP-3, −10, −11), matrilysins (MMP-7, −26), membrane-type MMPs (MT-MMPs), and other non-classified MMPs. Among the MMP family, MMP-2, MMP-9, and MT1-MMP are of particular interest in angiogenesis.11,12,15,16 In this study, we aimed to examine the impact of ICH on ECM-related genes using a DNA microarray.
Materials & methods
Animals and experimental protocol
A total of 125 adults (8 − 9 weeks) male Sprague–Dawley rats (250 − 300 g) were obtained from the Experimental Animal Center of Central South University (n = 5 per experiment for each timepoint). Rats were maintained in a 12-h light-dark cycle with free access to food and water. The study protocol was approved by the Institutional Animal Care and Use Committee of Central South University (201803286). The study was designed and executed in agreement with the guidelines of Central South University and the Guide for the Care and Use of Laboratory Animals (National Institutes of Health).
Following acclimatization, rats were randomly assigned to a sham control group or an ICH group (n = 5 per group per time point). Induction of ICH was performed as previously described. 17 Briefly, rats in the ICH group received 2.5 μl of 0.5 U collagenase (type VII) in 0.9% sterile saline via the right globus pallidus using the coordinates 1.4 mm posterior, 5.6 mm ventral, and 3.2 mm lateral relative to the bregma. Rats in the sham group received an intracerebral injection of 0.9% sterile saline at the same location. On postoperative days 4, 7, and 21, rats were sacrificed by cervical dislocation and perfused. In a separate experiment, rats were divided into sham, ICH, and doxycycline groups. Doxycycline was administered (30 mg/kg, intraperitoneal injection) starting from day 7 to day 14 following ICH induction and rats were sacrificed on postoperative day 21 (n = 5). Animals in the ICH group were sacrificed on days 4, 7, 14, 21, and 28 post-ICH induction (n = 5 per time point). Rats in the sham group were sacrificed on day 21 post-ICH induction.
Sample preparation
Following chloral hydrate (800 mg/kg) anesthesia, rats underwent transcardial perfusion with ice cold 0.9% saline. Brain tissues surrounding the hematoma were dissected and stored in liquid nitrogen for subsequent experiments. Alternatively, rats were perfused with normal saline followed by ice-cold 4% paraformaldehyde, and the brains were post-fixed in the same fixative for 4 h before further manipulation.
Complementary DNA (cDNA) array
Brain tissues were homogenized, and total RNA was extracted using the E.Z.N.A. Total RNA Kit (Omega, USA) according to the manufacturer's instructions. A total of 3 ug RNA was reverse transcribed with biotin-16-dUTP (1093070; Roche, Germany), M-MLV reverse transcriptase (M1701; Promega, USA), and a primer mix from the cDNA array kit (MM-009N; SuperArray, USA) at 42°C for 90 min according to the manufacturer's protocol. Labelling, hybridization, and membrane development were performed according to standard protocols. Briefly, ECM-related genes and housekeeping genes prehybridized with salmon sperm DNA (15632–011; Invitrogen) were immobilized on a nylon membrane (MM-009N; SuperArray) and hybridized with the prepared cDNA solution. Following hybridization (12 h at 60°C), the membrane was rinsed, blocked, and incubated with a streptavidin alkaline phosphatase conjugate. Following treatment with CDP-star (MS050R; Applied Biosystems, USA), membranes were exposed on an x-ray film. Densitometric analysis was carried out using a Multi-Analyst software (ST32151N; Bio-Rad, USA), and values were normalized to the housekeeping genes (β-actin and RPL13A) as described previously. 18 The obtained relative ratio of each gene spot in the ICH group was compared with those from the sham-operated brains at the same point. The relative ratio of the two ratios (ratio of each time point to the control ratio) was calculated and presented.
Fluorescein isothiocyanate (FITC)-dextran labelling
To analyse the angioarchitecture and blood perfusion in the perihematomal region, we injected 1 ml of FITC-dextran (2 × 106 molecular weight, Sigma-Aldrich, 50 mg/ml) into the tail veins, and the animals were decapitated at 1 min after injection. The brains were quickly dissected and fixed in 4% paraformaldehyde at 4°C for 48 h. Free-floating axial sections (100 μm thickness) were cut on a vibratome and visualized on a laser confocal microscope (TCS SP8 X & MP, Leica). The total number of vessels in the entire area of the left cerebral cortex or caudate to the middle cerebral artery territory were counted and divided by the size of the contralateral cerebral cortex. Results are expressed as number/mm2. The vascular area was compared between rats in the sham and ICH groups
Immunohistochemical analysis
Endogenous peroxidase was quenched using 3% hydrogen peroxide and nonspecific binding was blocked using 2% bovine serum albumin (BSA) for 1 h. Tissue sections were incubated overnight at 4°C with either goat anti-MMP-9 (1:200, Santa Cruz, USA), mouse anti-MMP-2 (1:200, Santa Cruz, USA), or mouse anti-MT1-MMP (1:400, Santa Cruz, USA) antibodies. To detect cerebral microvascular EC or neural proliferation, sections were incubated with mouse anti-PCNA (1:1400, Cell Signaling Technology) or rabbit anti-vWF (von Willebrand factor; 1:400, Dako, Denmark) for 1 h at 37°C. Subsequently, sections were incubated with biotinylated anti-mouse IgG (1:800, Santa Cruz) or anti-goat IgG (1:800, Santa Cruz) secondary antibodies for 1 h at 37°C followed by the avidin-biotin-peroxidase complex (1:100, Vector Laboratories, USA). Immunoreactivity was detected with diaminobenzidine (Boster Biotech Co., P.R. China).
Western blot analysis
Brain tissues were homogenized in RIPA lysis buffer and cleared by centrifugation at 12,000 rpm for 30 min at 4°C. Proteins were separated using SDS-PAGE and transferred onto PVDF membranes. Following blocking, membranes were incubated with either mouse anti-MMP-2 (1:400, Santa Cruz, USA), goat anti-MMP-9 (1:400, Santa Cruz, USA), mouse anti-MT1-MMP (1:400, Santa Cruz, USA), or mouse anti-actin (1:5,000, Abcam, Cambridge, UK) antibodies overnight at 4°C. The next day, membranes were incubated with horseradish peroxidase-conjugated anti-mouse IgG or anti-goat IgG (1:5,000, Promega, USA) secondary antibodies for 2 h at room temperature, and the positive signal was detected with an enhanced chemiluminescent substrate (Thermo Fisher) on a Bio-Rad ChemiDoc XRS digital documentation system. The relative protein expression was compared among rats of different experimental groups.
Statistical analysis
Results are expressed as mean ± standard deviation (SD). A Student's t-test was used to compare results between the ICH and the sham groups. One-way ANOVA was used to compare results among the ICH, sham, and doxycycline groups. A P value < 0.05 was considered statistically significant.
Results
cDNA array analysis
We analysed a total of 88 ECM-related genes using the cDNA array in the sham and ICH groups on postoperative days 4, 7, and 21. Compared to the sham group, there were 42 (47.7%) up-regulated and 14 (15.9%) significantly down-regulated genes following ICH on day 4 (Table 1, P < 0.05). Specifically, collagen (collagen І, III, IV, V, VI, and VIII), laminin (LAMA-1, LAMC-1), fibronectin (FN-1), thrombin sensitive protein (THBS-1, THBS-2), other glycoproteins (ECM-1, EMILIN-1, FBLN-1, SPP-1), proteoglycan (CSPG-2), MMPs (MMP-2, −7, −10, −12, −13, −14, and −15), TIMP-1, depolymerized metalloproteinases (ADAMTS-8, ADAMTS-2), hyaluronic acid mucins (CD44, ENTPD-1), immunoglobulin superfamily (ICAM-1), selectins (SELP), and integrins (integrins αd, αm, αl, αe, α4, α5, β2, β3) were all up-regulated at day 4 in the ICH group.
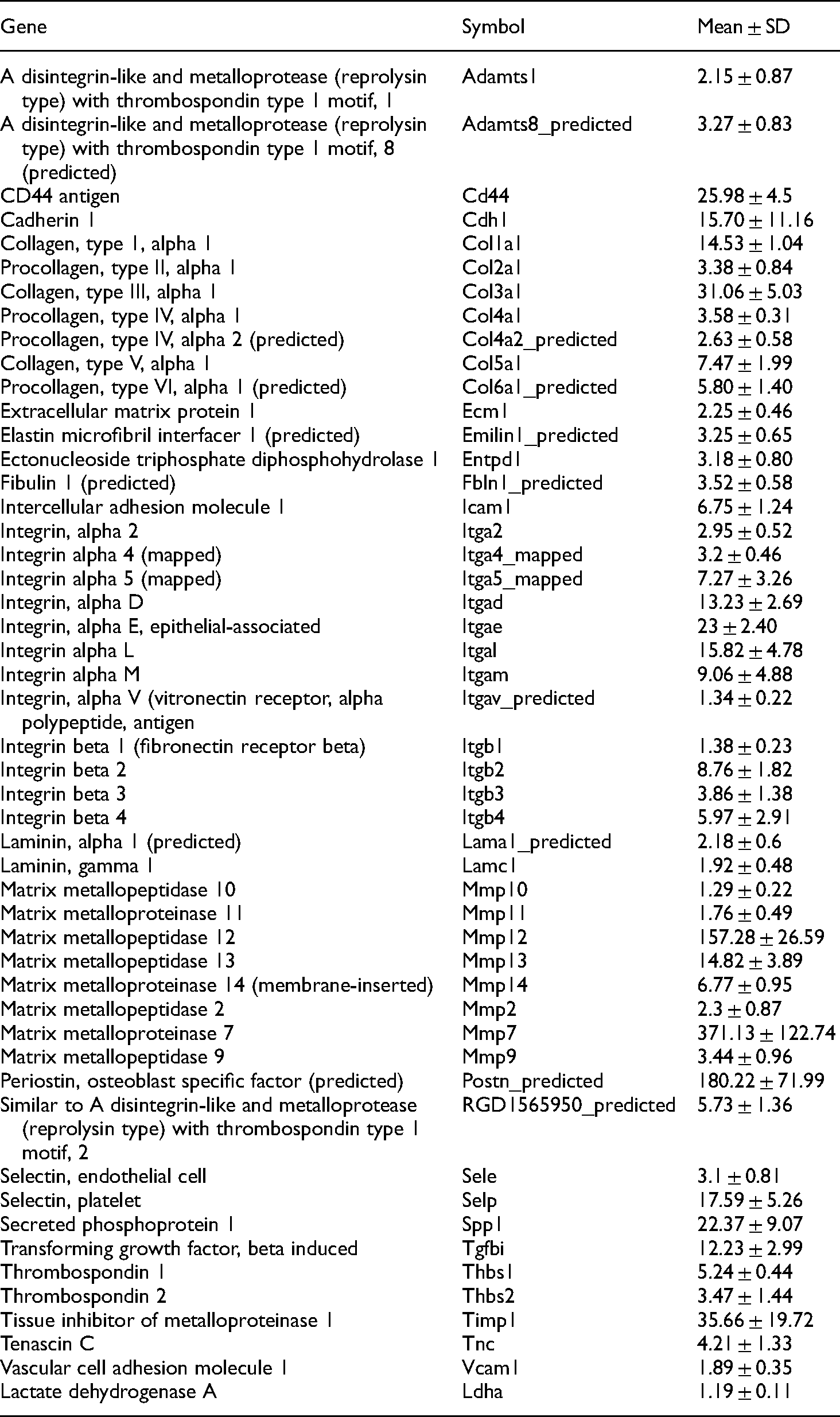
List of upregulated genes 4 days after ICH (n = 5 per group).

List of upregulated genes 4 days after ICH (n = 5 per group).

Compared to the sham group, there were 56 differentially expressed genes on postoperative day 7 (Table 2). Among them, 50 genes were significantly upregulated, and 6 genes were significantly downregulated in the ICH group (P < 0.05). In addition to the above-mentioned genes (day 4; Table 1), we observed the upregulation of several ECM-related genes including ADAMTS-1, collagen II, integrin αv and α2, integrin β1 and β4, MMP-9 and −11, TNC, and LDHA. Interestingly, VCAM-1 expression was downregulated on day 4 but was upregulated on day 7 in the ICH group.
List of upregulated genes 7 days after ICH (n = 5 per group).
Compared to the sham group, 38 genes were upregulated and 6 genes that were downregulated in the ICH group at postoperative day 21 (Table 3, P < 0.05). Specifically, we observed consistent upregulation of collagen genes (collagen I, II, III, IV, V, and VI), members of the MMP family (MMP- 3, 7, 10, 11, 12, 13, 14) and integrin family of genes (integrins αd, αm, αl, αe, αv, α4, α5, β2, β3, β4), as well as genes associated with cell adhesion and angiogenesis (CD44, EMILIN-1, ADAMTS-8, THBS-1 and −2). However, MMP-9 expression was decreased.
List of upregulated genes 21 days after ICH (n = 5 per group).

Next, we measured MMP protein expression in the rat brains of the ICH and sham groups. In the sham animals, immunohistochemical analysis revealed no MMP-2, MMP-9, or MT1-MMP expression (Figure 1A-C). In contrast, MMP-2, MMP-9, and MT1-MMP staining were observed in the micro-vessels in the perihematomal region of the ICH group 4 days post-ICH (Figure 1D-F). Compared to the sham group, western blot analysis confirmed the significant upregulation of MMP-2, MMP-9, and MT1-MMP in the ICH group 4 days post-ICH (Figure 1G-J; P < 0.05). Interestingly, protein expression of MMP-2 and MT1-MMP gradually increased and peaked at day 14 but started to decline at 21- and 28- days post-ICH (Figure 1G and J; P < 0.01). In contrast, peak MMP-9 protein expression was observed on day 7 post-ICH, but expression declined thereafter (Figure 1I, P < 0.05).
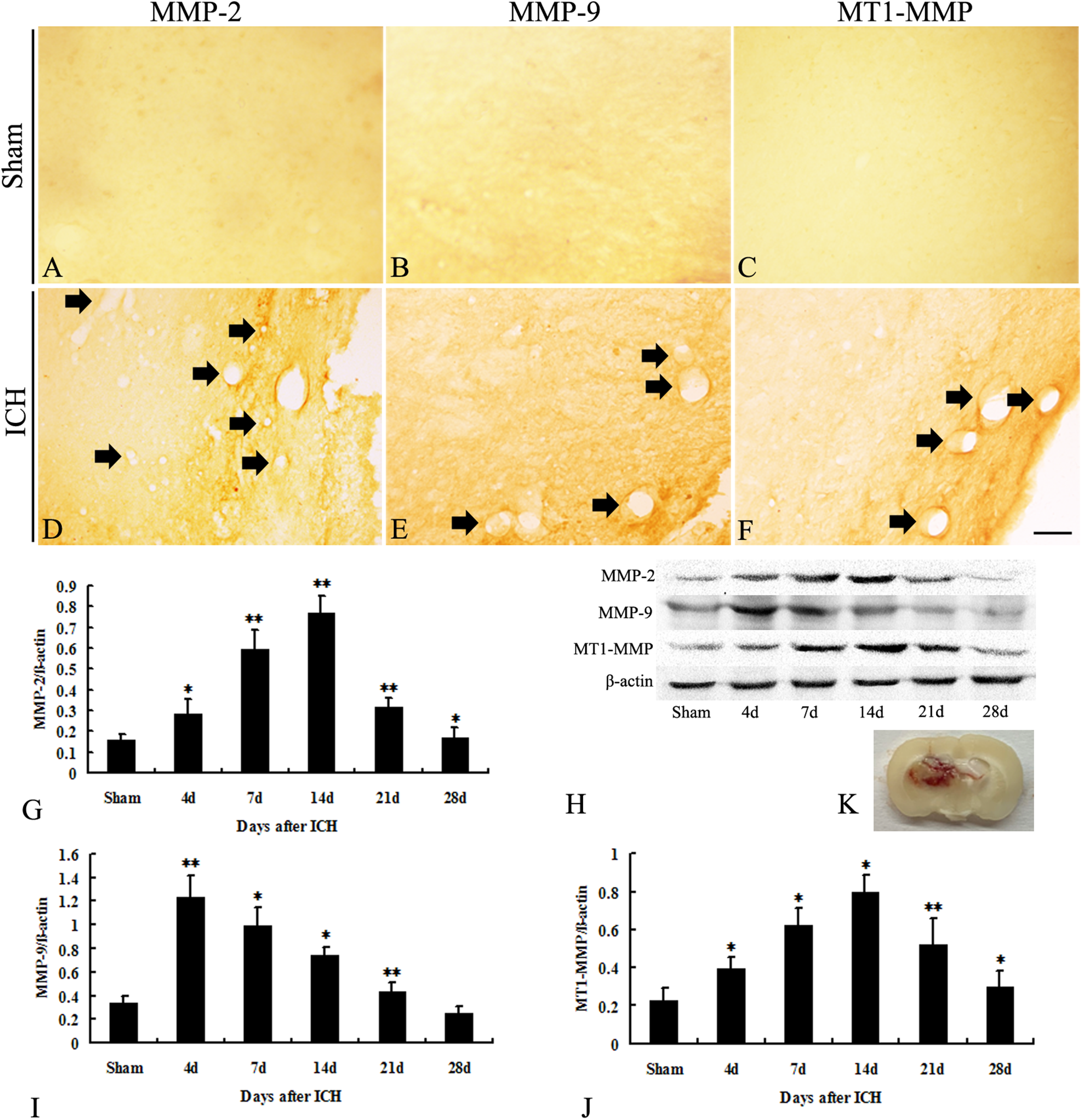
MMP-2, MMP-9, and MT1-MMP expression in the sham and ICH groups. Absence of MMP-2, MMP-9, and MT1-MMP immunostaining in the sham group on day 4 (A-C). MMP-2, MMP-9, and MT1-MMP staining were observed in the micro-vessels in the perihematomal region on day 4 in the IHC group. These micro-vessels were dilated closer to the hematoma region in the ICH group (DF). Western blot analysis demonstrating the relative protein expression of MMP-2, MMP-9, and MT1-MMP in the sham and ICH groups (G - J). MMP-2, MMP-9, and MT1-MMP expression was higher on day 4 in the ICH group compared to the sham group (P < 0.05). MMP-2 and MT1-MMP protein expression increased gradually from days 7 to 14 and decreased on day 21 in the IHC group (P < 0.01). MMP-9 protein expression decreased on day 7 in the ICH group (P < 0.05). (n = 5 animals per group at each time point). ICH, intracerebral hemorrhage; MMP, matrix metalloproteinase. *P < 0.05, ** P < 0.01. Panel K is a representative image to present the location of the hematoma. Bar = 50 µm.
In the sham group, FITC-dextran labelling demonstrated evenly distributed blood vessels with unobstructed blood perfusion (Figure 2A). In the ICH group, blood perfusion around the injury was significantly reduced compared to the sham group on day 4 post-ICH (Figure 2B, P < 0.01). Vascular permeability increased in the ICH group, resulting in the leakage of FITC-dextran and its dispersion in the brain parenchyma (Figure 2B). The perfused area increased sharply from day 7 through day 21 (Figure 2C, day 21). Doxycycline treatment significantly decreased perfusion at day 21 post-ICH (Figure 2D).
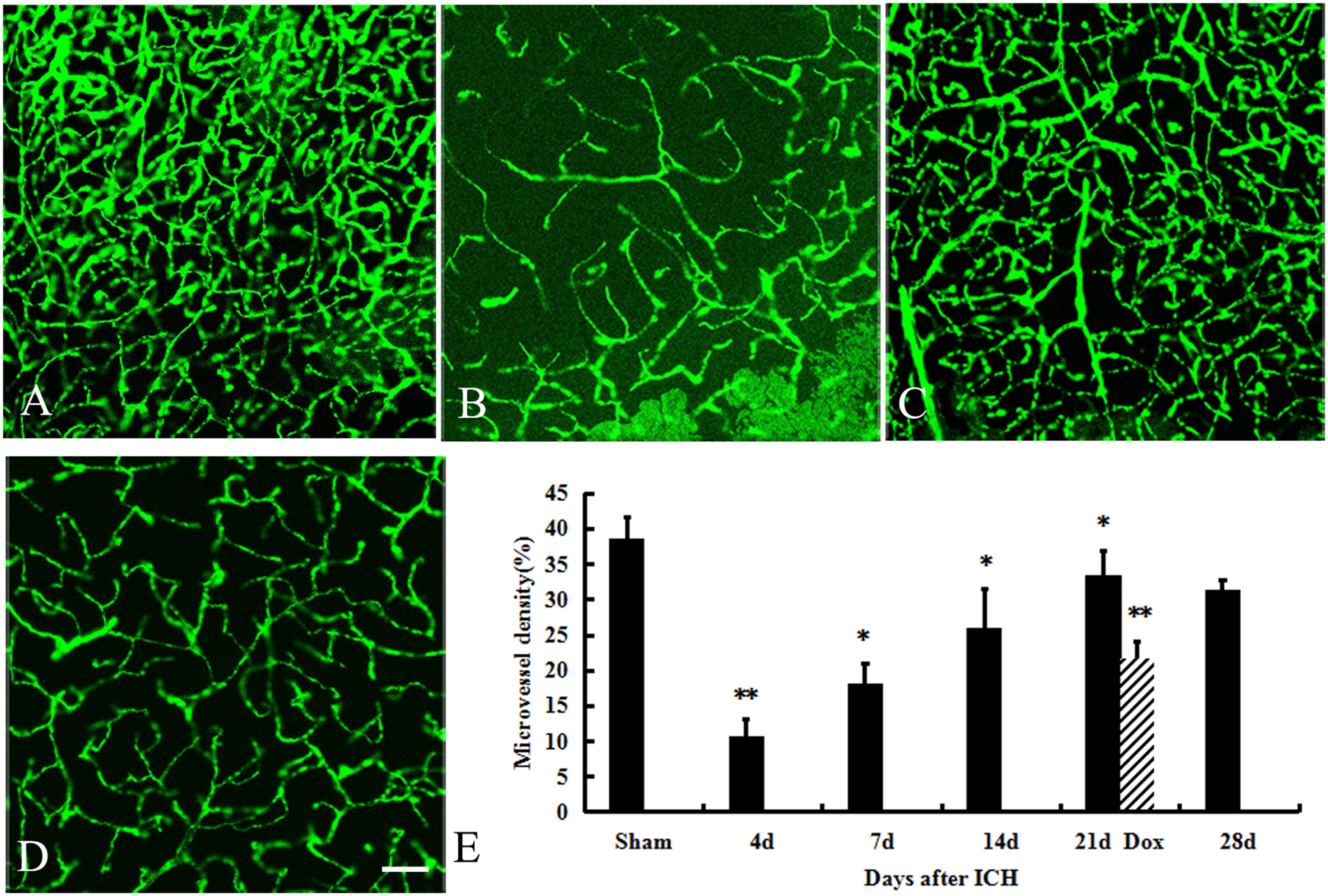
FITC-dextran staining of blood vessels. The blood vessels in the sham group were evenly and orderly distributed with unobstructed blood perfusion (A). Four days post-ICH, perfusion around the injury was significantly reduced compared to the sham group (P < 0.01; B). Increased permeability of vascular walls was observed, resulting in FITC-dextran leakage and its dispersion in the brain parenchyma (B). The perfused area increased sharply from day 7 till day 21 in the ICH group (C, day 21). Doxycycline significantly reversed the effect of ICH at 21 days (D). (n = 5 animals per group at each time point). *P < 0.05, ** P < 0.01. Bar = 100 µm.
To examine the number of newly formed blood vessels, brain sections were immunostained with anti-PCNA and anti-vWF antibodies. PCNA-immunoreactive nuclei were scarce in the control group (Figure 3A). In contrast, the number of PCNA + /vWF + dilated blood vessels were detected in the perihematomal region on day 4 post-ICH and this number continued to increase through day 14 post-ICH (Figure 3B and C; P < 0.01). Compared to the ICH group, doxycycline treatment significantly decreased the number of newly formed vessels on day 21 post-ICH (Figure 3C; P < 0.05).

PCNA expression in the sham and ICH groups. PCNA-immunoreactive nuclei were absent in the sham group (A), and PCNA + /vWF + dilated blood vessels were detected around the hematoma on day 4 in the ICH group (B). Bar chart demonstrating the PCNA positivity at different time points (D). PCNA positive signal was more prevalent on day 7 (P < 0.01) and peaked at day 14 in the ICH group. Doxycycline administration decreased the number of newly formed vessels in the ICH group on day 21(C). (n = 5 animals per group at each time point). *P < 0.05, ** P < 0.01. Bar = 50 µm.
In this study, we examined the impact of ICH on ECM-related gene expression and angiogenesis. Our findings indicate that ICH leads to dynamic changes in ECM-related genes and consequently increases angiogenesis. The crucial role of the ECM in angiogenesis has been previously demonstrated. 6 Collagen constitutes the ECM skeleton and forms semi-crystalline fibres in the ECM. 19 Collagen I and III are the main ECM components in the arterial wall.20,21 Collagen IV is mainly synthesized by epithelial cells and some mesenchymal cells and is the main collagen of the basement membrane that is crucial for cell differentiation and proliferation. Collagen IV is also important for maintaining vessel wall stability and cell morphology. 22 During angiogenesis, collagen synthesis and proteolysis at the ECM-vascular cell interphase are tightly regulated. Initially, MMPs degrade the local matrix to promote the invasion of pre-existing vascular cells. Later, fibrin and collagen regulate vascular cell migration and proliferation. 23 In agreement, our microarray results demonstrated the persistent upregulation of collagen genes following ICH.
The integrin family is comprised of 24 different heterodimeric integrins with noncovalently-linked α and β glycoprotein subunits. 24 Integrins mediate cell-to-cell as well as cell-to-ECM interactions and play critical roles in various physiological and pathological processes. 25 During angiogenesis, ECs interact with different integrins to promote cell migration. 26 The partnership between integrins and growth factor receptors is vital for the proper regulation of EC signalling. For example, the crosstalk between vascular endothelial growth factor receptor 2 (VEGFR2) and αvβ3 integrin controls EC adhesion and migration during VEGF-induced angiogenesis. 27 Our microarray results demonstrated the upregulation of integrins following ICH. Likewise, we observed an increase in the number of newly formed blood vessels following ICH. Similar observations were also reported in previous transient cerebral ischemia and central hypoxic models. 28
Mounting evidence indicates that MMPs are major players in vascular formation and remodelling through degrading the vascular basement membrane and ECM proteins. Among the MMP family, MMP-2, MMP-9, and MT1-MMP are of particular interest in angiogenesis.15,16 Under quiescent conditions, MMP expression is not observed in tissues or in circulation.29,30 Following central nervous system injury, MMP-9 and MMP-2 are upregulated to facilitate repair and regeneration processes through the dynamic remodelling of the ECM.23,31 Following activation and release of angiogenesis-related factors, MMP-2 and MMP-9 promote EC migration and neovascularization by degrading the subendothelial basement membrane, increasing BBB permeability, and allowing plasma extravasation.16,32 Interestingly, blocking MMP-9 can impair the angiogenic function of endothelial progenitor cells, suggesting the importance of MMP-9 in de novo vascularization. 33 Results obtained from this study confirmed the importance of MMPs in ICH recovery. Microarray data demonstrated the upregulation of different MMPs (- 2, - 3, - 7, - 9, - 10, - 11, - 12, - 13, - 14) following ICH. Noticeably, MMP-7 and MMP-12 expression continued to rise. MMP-7 has a small molecular weight, is less negatively regulated by TIMP, and has a robust matrix degradation activity. Meanwhile, MMP-12 is a key regulator of macrophage infiltration and inflammation. 34 The findings presented here suggest that matrix degradation and inflammation play an ongoing role in damage and repair after ICH. The lack of upregulated MMP-9 gene expression on day 4 may indicate that MMP-9 was only required at the very early stage of ICH. Consistent with previous reports, we observed that the protein expression of MMP-2 and MMP-9 was significantly upregulated after ICH.35,36 We also observed peak MMP-2 expression on day 7 post-ICH, which persisted through day 14 post-ICH; MMP-9 expression peaked on day 4 post-ICH and started to decline on day 7 post-ICH. Therefore, it is plausible to speculate that MMP-9 is implicated in the early acute phase and MMP-2 is involved in the chronic phase of ICH recovery.
MMP-14 (MT1-MMP) has also been implicated in angiogenesis. MT1-MMP degrades a wide variety of ECM components, upregulates angiogenic factors like VEGF, degrades anti-angiogenic factors, and interacts with cell surface molecules including CD44 and sphingosine 1-phosphate receptor 1 (S1P1) to promote EC migration. 37 In addition, MT1-MMP was suggested to aid in vessel maturation and angiogenesis. 38 Moreover, MT1-MMP can activate downstream MMPs, including MMP-2.39,40 Our data support the crucial function of MT1-MMP in regulating angiogenesis, as we observed a trend in MT1-MMP expression consistent with that of MMP-2, indicating that MT1-MMP can act as a regulator for MMP-2. 41
Following stroke, the administration of MMP inhibitors may reduce brain edema and inflammation. Previous studies suggested the importance of inflammation in neuronal recovery and remodelling after ICH. 42 However, the use of MMP inhibitors does not promote the recovery of neurological function in acute stroke because MMP inhibitors can reduce endogenous VEGF signals and inhibit neovascularization. 43 Furthermore, long-term use of MMP inhibitors after stroke may lead to increased brain damage due to a reduction in neovascularization and neural recovery. 44 The accurate timing of therapeutic intervention may be critical to achieve the beneficial response of MMP inhibitors. Doxycycline was previously shown to inhibit MMPs at a sub-antimicrobial dosage. 45 This was attributed to its ability to chelate calcium and zinc ions, as well as its ability to directly inhibit the latent pro-MMPs activation. 46 Further, doxycycline exhibits an inhibitory effect on angiogenesis associated with ECM degradation in several diseases.47,48 In agreement, our results indicate that doxycycline inhibited angiogenesis49,50 in a rat ICH model.
Conclusions
To the best of our knowledge, this is the first study to demonstrate the temporal change in cerebral ECM-related gene expression after ICH. Following ICH, members of the MMP, collagen, and integrin families were among the most prominent upregulated genes. Our results also showed that MMPs are required for ICH-induced angiogenesis. Therefore, the wide and long-term use of MMP inhibitors can delay functional recovery following ICH. Our study suggests that fine-tuning the MMP response may be needed to optimize inhibition during the acute stages of injury without interfering with the favourable outcome of neurovascular remodelling. Limited sample size and the use of brain homogenates are considered the main limitation of our study. Therefore, future studies will be designed to confirm our results and explore the detailed molecular mechanism related to the changes in gene expression following ICH.
Footnotes
Abbreviations
Authors’ contributions
ALY, HJZ, and HJC designed/performed most of the research and data analysis and wrote the manuscript. TT and JKL contributed to interpretation and analyses of the data. All authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the National Natural Science Foundation of China, (grant number 81603414 and 81904078).
Ethics approval and consent to participate
The study protocol was approved by the Institutional Animal Care and Use Committee of Central South University (201803286). The study was designed and executed in agreement with the guidelines of Central South University and the Guide for the Care and Use of Laboratory Animals (National Institutes of Health).
Availability of data and materials
The datasets generated and analysed during the present study are available from the corresponding author upon reasonable request
