Abstract
Case summary
A patient who underwent mechanical aortic and mitral valve replacement developed three paravalvular leaks 10 months later. We located the tracks by puncturing the apex cordis under transoesophageal echocardiography guidance alone and puncturing the femoral artery guided by fluoroscopy. Three paravalvular leaks were occluded with a hybridization method simultaneously. The patient was followed up for 24 months and maintained a good condition.
Conclusion
Multiple paravalvular leaks after double valve replacement can be occluded in patients by the use of different approaches under echocardiographic guidance alone.
Introduction
Paravalvular leak (PVL) is a special complication after cardiac valve replacement, with an incidence of 5% – 17%; PVL is the main reason for a secondary surgery after valve replacement.1–3 PVL mainly occurs after aortic valve replacement (APVL), followed by mitral valve replacement (MPVL). Patients with PVL often have symptoms, such as cardiac insufficiency and haemolysis, which require surgical treatment. 4 We report a case of multiple PVLs after mechanical valve replacement of the aortic and mitral valves. Puncture of the left ventricular apex performed under transoesophageal echocardiography (TEE) guidance and puncture of the femoral artery performed under fluoroscopy guidance allowed three PVLs to be occluded with hybrid intervention at the same time.
Case presentation
A 60-year-old woman underwent mechanical aortic and mitral valve replacement 10 months prior and then developed symptoms of cardiac insufficiencies, such as shortness of breath and chest tightness, for 10 days. Transthoracic echocardiography showed a 4.1 mm fissure at the 8 o’clock position of the mechanical aortic valve position. A 3.8 mm fissure was detected at the 2 o’clock position, and a 4.1 mm fissure was detected at the 8 o’clock position around the mechanical mitral valve. Considering the (1) age, (2) cardiac insufficiency, (3) haemodynamic significance of PVL, (4) double mechanical valve replacement status and (5) PVLs on both the inner and outer sides of the mitral valve, ultimately, the cardiac diagnosis and treatment team decided to implement a hybrid interventional closure scheme for the patient. The ventricular apex was punctured solely under TEE guidance, and a track was established to close the three PVLs. If the occlusion did not go well, the occlusion was prepared with a percutaneous transcatheter under the guidance of fluoroscopy.
The patient underwent general anaesthesia intubation in the compound operating room. A 2 cm incision was made on the left thoracotomy (the fifth intercostal space on the left side). The tissue was separated, and part of the pericardium was cut and suspended. After heparinization, a pouch was sewn at the left ventricular apex and then punctured directly with a trocar under TEE guidance alone (see Figure 1). On the fissure at the 2 o’clock positions of the mitral valve, a 7 Fr occlusion delivery system was used to establish the track, and a 10 mm equilateral ventricular septal defect (VSD) occluder was used to close the PVL. On the fissure at the 8 o’clock position of the mitral valve, a 7 Fr occlusion delivery system was used to feed an 8 mm equilateral VSD occluder. TEE showed that the occluders were in a good position and did not affect the movement of the mechanical mitral valve, and no residual shunt was seen. Closure of the APVL via the apical cordis approach was unsuccessful in multiple attempts to establish tracks. Then, we punctured the right femoral artery, inserted a 6 Fr vascular sheath, and retrogradely sent a 5 Fr pigtail catheter to the root of the aorta. Angiography showed paravalvular regurgitation from the aortic root to the left ventricle, and the APVL measured ∼3 mm (see Figure 2). A 6 Fr right heart catheter was used to establish the track through the (1) femoral artery, (2) descending aorta, (3) ascending aorta, (4) APVL and (5) left ventricle. Next, 260 cm hard guidewire was exchanged in the left ventricle, and the 5 Fr delivery sheath was guided to follow the hard guidewire to the left ventricle. The sheath-core and guidewire were withdrawn, and an 8 mm (non-membrane double-lumen) VSD occluder was sent to the left ventricle along the delivery sheath to complete the closure. All the occluder devices were provided by Amplatzer. Angiography of the aortic root after occlusion showed that the occlusion was well fixed and did not affect the opening of the mechanical aortic valve or coronary blood flow, and there was no residual shunt (see Figure 3).
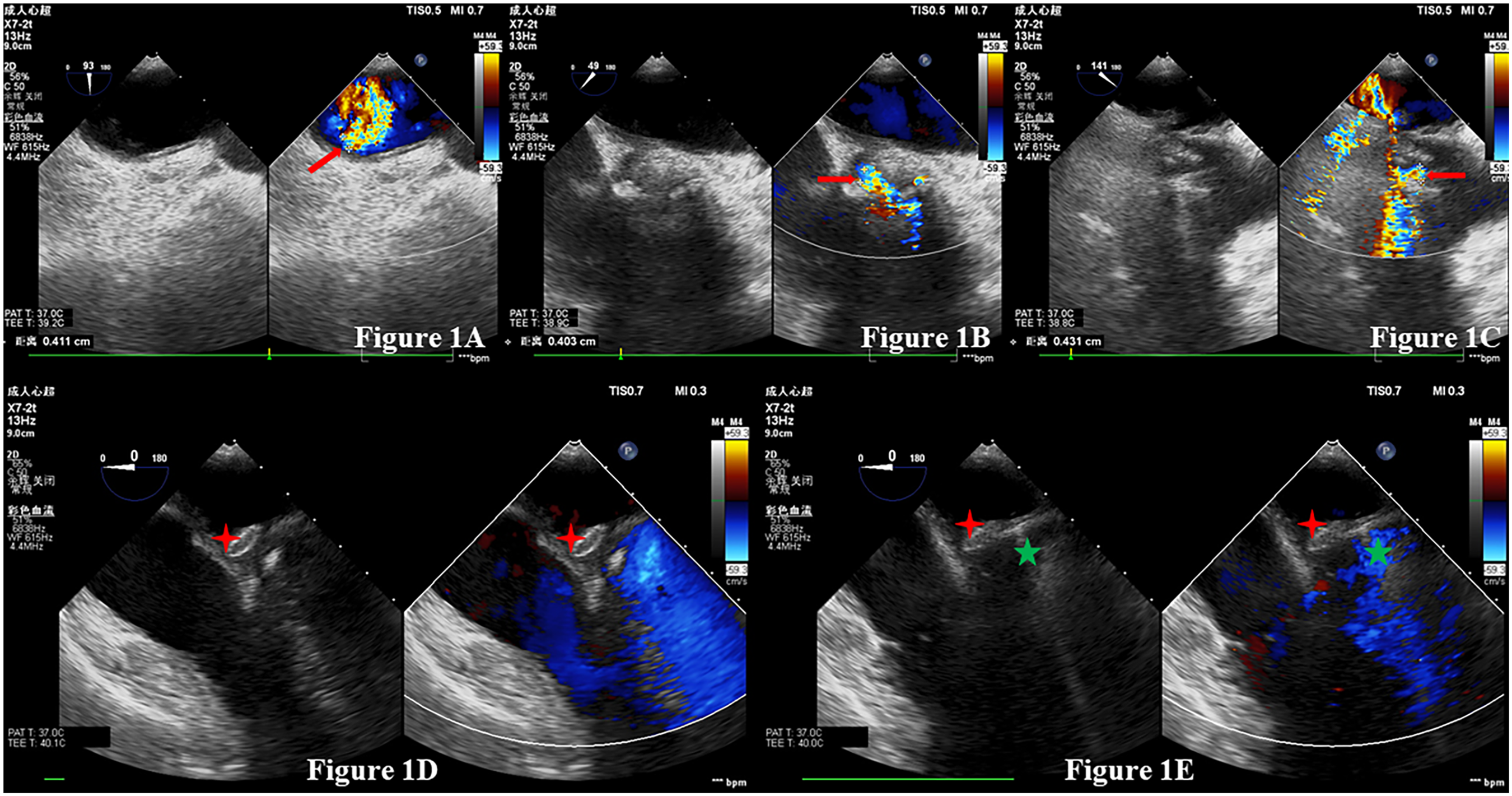
(A) to (C) The aortic paravalvular leak, the paravalvular leak on the inner side of the mitral valve, and the paravalvular leak on the outer side of the mitral valve, respectively; these leaks were measured by intraoperative transoesophageal ultrasound. The red arrow in the figure points to paravalvular regurgitation. (D) and (E) The occlusion process of two paravalvular leaks of the mitral valve via the transapical approach under the guidance of transoesophageal echocardiography. The red four-pointed star in the picture refers to the inner occluder, and the green five-pointed star denotes the outer occluder.

(A) and (B) The regurgitation of the aortic paravalvular leak before and after closure. At this time, the paravalvular leaks at the mitral valve were occluded. The red arrow points to paravalvular regurgitation at the aortic valve, and the blue arrow points to mitral valve paravalvular leak occluders.

(A) and (B) The paravalvular leak occluder (shown by the white arrow) in frontal and lateral photographs, respectively.
The patient developed a pneumothorax after surgery and recovered after 11 days of care. Following up for 3, 6, 12 and 24 months, the patient had no related complications, and cardiac function recovered from Grade IV to Grade II (NYHA classification).
Discussion
The 2014 AHA/ACC guideline of patients with valvular heart disease 5 pointed out that transcatheter PVL closure is reasonable in patients with prosthetic heart valves and intractable haemolysis or cardiac insufficiency who are at high risk for surgical intervention and have anatomic features suitable for catheter-based therapy (Class IIa).
APVL is one of the more common and serious complications after surgical or transcatheter aortic valve replacement, and the clinical symptoms of patients are often severe. When treating this patient, the transapical approach to establish a track for APVL was attempted multiple times and failed. Then, the femoral artery was punctured to establish a retrograde route, and the APVL was occluded under fluoroscopy and echocardiography guidance. Compared with APVL, transcatheter repair of MPVL is more difficult. There are three main approaches to occlusion 6 : (1) the retrograde left ventricular approach; (2) the antegrade left atrium approach, in which the femoral vein is punctured or the right atrium is punctured directly, then the atrial septum is punctured and the sheath is placed via the left atrium side; and (3) the transapical approach, which was the route ultimately chosen for this patient. The transapical approach provided the most direct and shortest path, which could be adjusted to close the APVL and MPVL at the same time. We should also note that the transapical approach is not always an easier way to close the PVL, as illustrated for aortic PVL in this patient with the failed attempts; also, it has higher intraoperative requirements for surgeons. In this case, the delayed recovery caused by pneumothorax should also be considered as a possible complication caused by the transapical approach. In some heart centres with expertise in this procedure, this approach can become the preferred reliable repair plan for patients with PVLs. The conventional transapical approach needs to be completed under the dual guidance of fluoroscopy and echocardiography. 7 For this patient, we tried to complete the closure of the MPVL under TEE guidance, which suggested that under the guidance of TEE alone, it is feasible to complete apical puncture, establish a track and place an appropriate occluder.
Conclusion
Through the apical/artery/vein approach, under the guidance of TEE and/or fluoroscopy, patients with PVLs after cardiac valve replacement can undergo personalized transcatheter repair procedures.
Footnotes
Ethical approval
Ethical approval to report this case was obtained from Gansu Provincial Hospital of medical ethics committee (2021-206).
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
