Abstract
Background
Tetralogy of Fallot (ToF) is a life-threatening congenital cardiovascular disorder. Currently, the most effective therapeutic intervention for pediatric ToF remains corrective surgery with cardiopulmonary bypass (CPB). Ferroptosis is an iron-dependent form of regulated cell death, driven by an accumulation of lipid peroxides to levels sufficient to trigger cell death. Ferroptosis was recently linked to cardiac ischemia and reperfusion injury. However, few studies have examined CPB-associated ferroptosis.
Method
In the current study, pediatric ToF patient pre- and post-CPB atrial biopsy gene expression profiles were downloaded from a public database, and 117 differentially expressed genes (DEGs) were identified using the Wilcoxon rank-sum test and weighted gene correlation network analysis. These were screened for ferroptosis-associated genes using the FerrDb database, thereby identifying ten genes. Finally, the construction of gene-microRNA (miRNA) and gene-transcription factor (TF) networks, in conjunction with gene ontology and biological pathway enrichment analysis, were used to inform hypotheses regarding the molecular mechanisms underlying CPB-associated ferroptosis.
Results
Ten genes involved in CPB-associated ferroptosis(ATF3,TNFAIP3,CDKN1A, ZFP36, JUN,SLC2A3, IL6, CXCL2, PTGS2, and DDIT3). Ferroptosis-associated genes were largely involved in myocardial inflammatory responses and may be regulated by a number of identified miRNAs and TFs, thereby suggesting modulatable pathways potentially involved in CPB-associated ferroptosis.
Conclusions
Results suggest that CPB precipitates ferroptosis within cardiac tissue during corrective Surgery for Pediatric Tetralogy of Fallot. These findings may ultimately help improve outcomes of corrective surgery for pediatric ToF.
Introduction
Tetralogy of Fallot (ToF) is a life-threatening congenital cardiovascular disorder. 1 Currently, the most effective therapeutic intervention for pediatric ToF remains corrective surgery with cardiopulmonary bypass (CPB). 2 However, CPB can damage multiple organ systems, especially during corrective surgery for ToF, which requires prolonged CPB. For example, prolonged extracorporeal circulation is associated with prolonged mechanical ventilation and intensive care unit stays, as well as increased pediatric mortality. 3 Prolonged aortic occlusion often predicts a higher degree of surgical complexity, which increases the risk of postoperative hemodynamic complications (such as low cardiac output syndrome and arrhythmia) and pulmonary infection. 4
Ferroptosis is an iron-dependent form of regulated cell death, driven by an accumulation of lipid peroxides to levels sufficient to trigger cell death 5 (Supplementary Figure 1). Ferroptosis was recently linked to cardiac ischemia and reperfusion injury. 6 However, few studies have examined ferroptosis in the context of CPB.
In the present study, right atrial biopsy mRNA expression profiles were downloaded from a publicly accessible database. Biopsies were derived from pediatric ToF patients undergoing surgical primary defect repair, both pre- and post-CPB. Next, differentially expressed genes (DEGs) were identified and filtered for ferroptosis-associated genes. Finally, functional enrichment analysis and construction of gene-microRNA (miRNA) and gene-transcription factor (TF) networks facilitated the exploration of molecular mechanisms underlying CPB-associated ferroptosis. If validated, findings may contribute to improving outcomes following CPB, thereby decreasing complications following corrective surgery for pediatric ToF surgery.
Methods
Data acquisition
Gene expression dataset GSE132176 (platform: GPL13158) was downloaded from the Gene Expression Omnibus (https://www.ncbi.nlm.nih.gov/geo/) database. 7 This included data deriving from pre- and post-CPB atrial tissue samples of ten pediatric ToF patients undergoing corrective surgery (6 males, 4 females, mean age 1 year). The R software for statistical computing (v3.6) 8 was used in conjunction with the R package limma 9 to normalize raw data.
Identification of differentially expressed genes (DEGs)
The base R function wilcox.test was used to apply the Wilcoxon rank-sum test to identify DEGs. The R packages ggplot2 10 and pheatmap 11 were used to generate a volcano plot and heatmap, respectively. Statistical significance thresholds for differential expression were set at p < 0.05 and |log2 fold-change| > 1.
Weight gene correlation network analysis (WGCNA)
A weighted correlation network analysis was performed using expression data to find clusters (modules) of highly correlated genes. 12 The best soft threshold power value was automatically selected by the software, then the co-expression networks were constructed and disease-associated modules were identified using the R package WGCNA. 12 To identify key DEGs, The R package VennDiagram 13 was used to construct Venn diagrams demonstrating the intersection of DEGs and key modules.
Screening for ferroptosis-associated genes
A list of ferroptosis-associated genes was downloaded from FerrDb, the world's first database of ferroptosis regulators and markers, as well as ferroptosis-disease associations. 14 The list contained drivers (genes that promote ferroptosis), suppressors (genes that prevent ferroptosis), and markers (genes for which altered expression indicates the occurrence of ferroptosis). The R package VennDiagram 13 was used to construct Venn diagrams demonstrating the intersection of key DEGs and all ferroptosis-associated genes, as well as (separately) the intersection of key DEGs and drivers, suppressors, and markers. In this manner, pre- and post-CPB ferroptosis-associated genes were identified. The R packages beeswarm 15 and ggpubr 16 were used to draw boxplots demonstrating differential expression levels of ferroptosis-associated genes in the pre- and post-CPB groups. Finally, a correlation heatmap of ferroptosis-associated genes was generated using Pearson correlation analysis as provided by Sangerbox tools, a free online platform for data analysis (http://www.sangerbox.com/tool).
Functional enrichment analysis
The R package clusterProfile 17 was used to conduct gene ontology (GO) term 18 and Kyoto Encyclopedia of Genes and Genomes (KEGG) biological pathway 19 enrichment analysis of CPB-altered ferroptosis-associated genes. In addition, a pathway diagram of ferroptosis-associated genes was downloaded from FerrDb.
Construction and analysis of gene-miRNA and gene-TF networks
NetworkAnalyst (https://www.networkanalystca/) was applied to integrate miRTarBase (miRNA) 20 and ENCODE (TF) 21 databases. Nodes with degree ≥ 2 were extracted to construct and visualize ferroptosis-associated gene-miRNA and gene-TF networks using the Cytoscape 22 plugin. An overview of the complete study workflow is provided in Figure 1.

The workflow of the present study.
Results
Identification of ferroptosis-associated DEGs
A total of 119 DEGs were identified (112 up-regulated and 7 down-regulated in the post-CPB group; Figure 2(a), Supplementary Table 1). The 50 most significantly differentially expressed genes were visualized by means of a heatmap (Figure 2(b)).
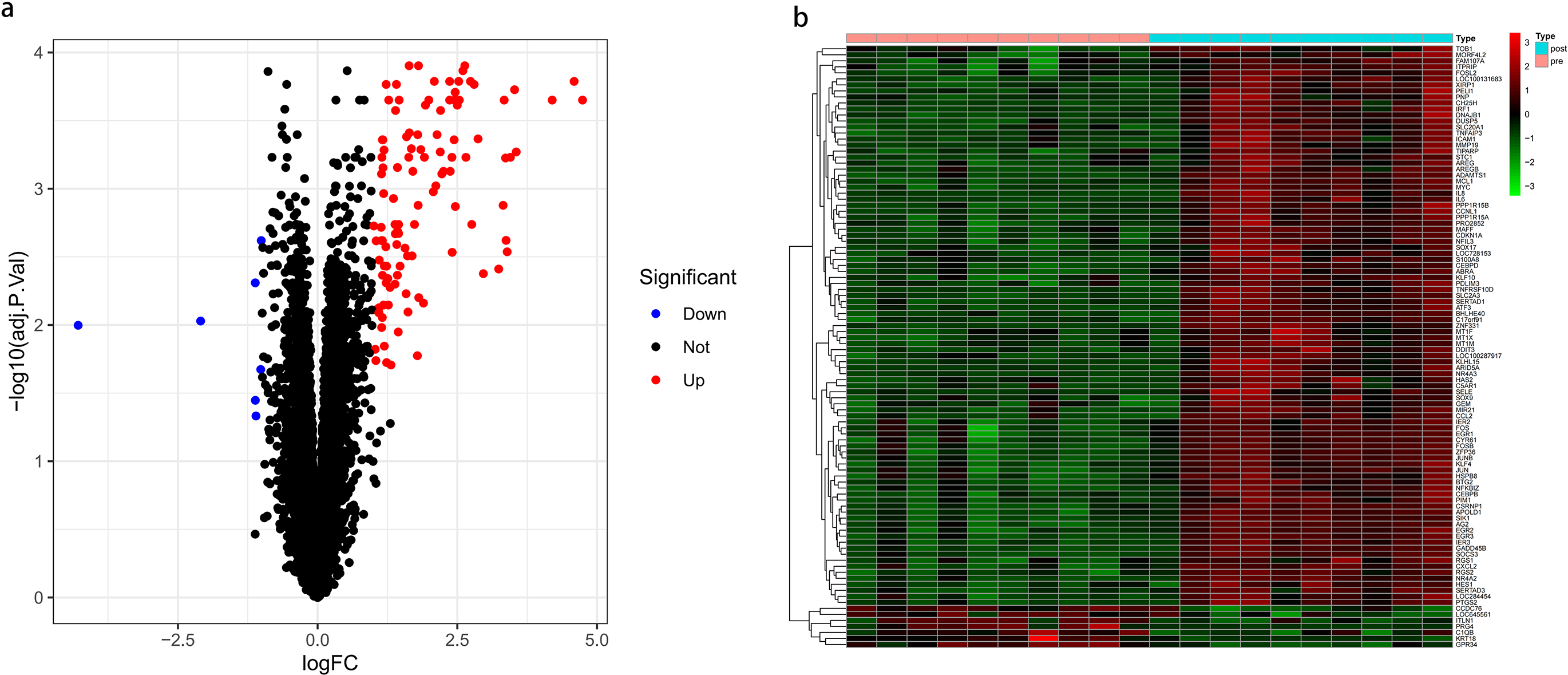
Differentially expressed genes identification (a)volcano plots of DEGs. (b)A heatmap of differentally expressed genes.
WGCNA analysis
Five modules were identified using WGCNA (Figure 3(b)). The turquoise module included 358 genes and was positively correlated with the post-CPB group (correlation = 0.84, p = 4×10−6; Figure 3(b)). We identified 117 key DEGs in the intersection of the turquoise module and DEGs (Figure 3(c)).
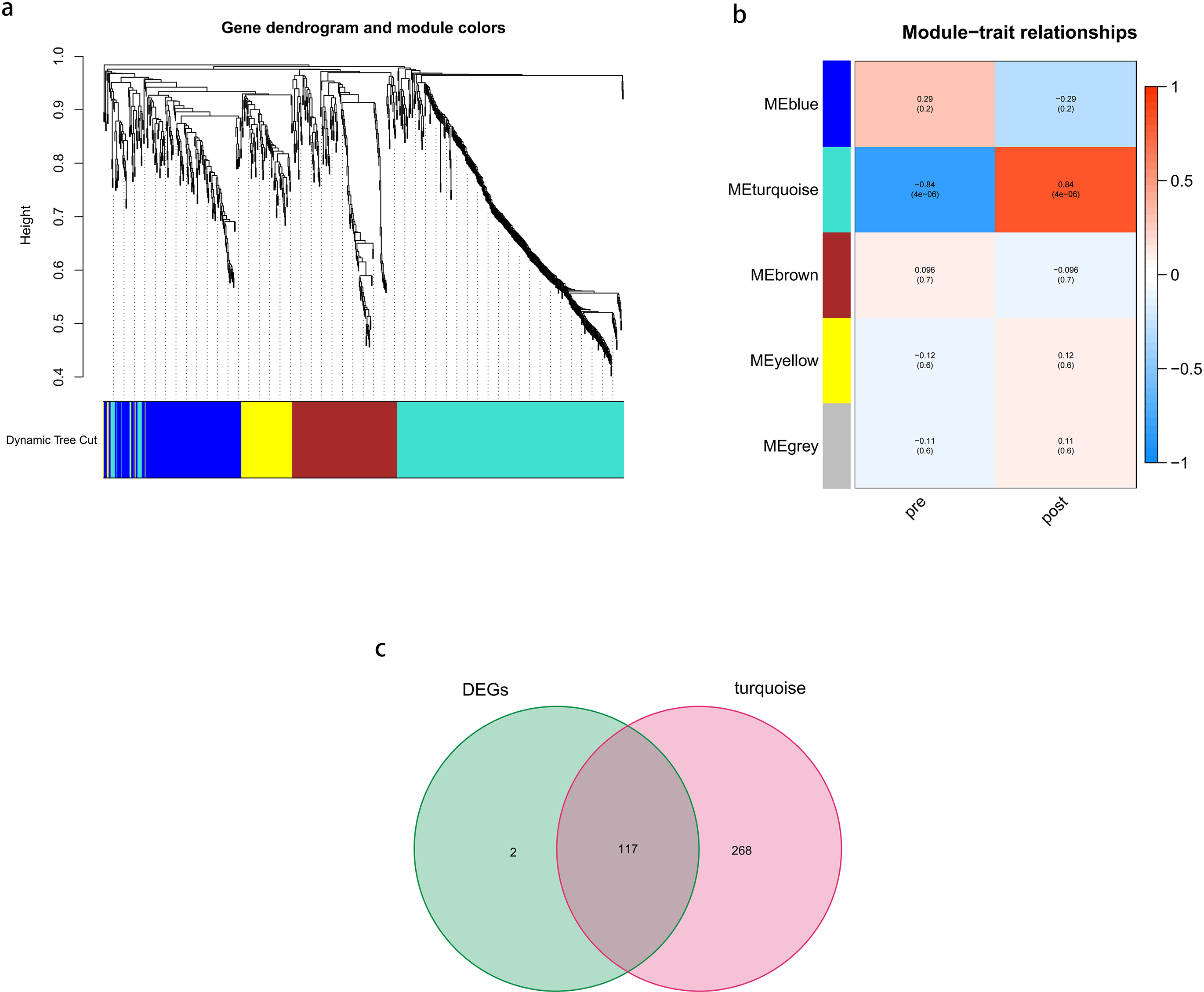
Weighted gene correlation network analysis. (a) Recognition module,each module was given an individual color as identifiers, including 5 different modules. (b) Correlation heat map of gene modules and phenotypes, the red is positively correlated with the phenotype, blue is negatively correlated with the phenotype. (c)Venn diagrams between DEGs and turquoise module.
Screening for ferroptosis-associated genes
The intersection of key DEGs and ferroptosis-associated genes consisted of 10 genes, including 2 drivers: Activating Transcription Factor 3(ATF3) and TNF Alpha Induced Protein 3 (TNFAIP3), 3 suppressors: Cyclin Dependent Kinase Inhibitor 1A (CDKN1A), Zinc Finger Protein 36(ZFP36), and Jun proto-oncogene (JUN), and 6 markers :ATF3, Solute Carrier Family 2 Member 3 (SLC2A3), Interleukin 6(IL6), C-X-C Motif Chemokine Ligand 2(CXCL2), Prostaglandin-Endoperoxide Synthase 2(PTGS2), and DNA Damage Inducible Transcript 3 (DDIT3) (Figure 4). Boxplots demonstrated up-regulation of all of these in the post- CPB group (Figure 5).

Venn diagrams of (a) ferroptosis-related genes (b) driver genes of ferroptosis-related genes (c)suppress genes of ferroptosis-related genes (d)marker genes of ferroptosis-related genes.

Ferroptosis-related genes are up-regulated in post-CPB group.
Correlations between expression patterns of ferroptosis-associated genes
Driver expression levels exhibited a strong positive correlation with expression levels of a number of other ferroptosis-associated genes: ATF3 expression correlated with that of SLC2A3, IL6, CDKN1A, and ZFP36, while TNFAIP3 expression correlated with that of both ATF3 and IL6 (Figure 6(a)). In addition, the expression of the suppressor JUN exhibited a strong positive correlation with that of the suppressor ZFP36.

The potential mechanism of ferroptosis in CPB.(a) The correlation between ferroptosis-related genes, blue indicates negative correlation and red indicates positive correlation.(b)GO and KEGG enrichment analysis.(c)Pathway of ferroptosis-related genes.
Functional enrichment analysis
The identified ferroptosis-associated genes were significantly enriched for multiple GO biological process terms indicating smooth muscle cell-related processes (Figure 6(b)), as well as for various GO molecular function terms including, but not limited to, DNA-binding transcription activator activity, RNA polymerase II-specific, cytokine activity, transcription corepressor activity, glucose binding, and leucine zipper domain binding (Figure 6(b)). Enriched KEGG pathways suggest an involvement of ferroptosis-associated genes in many inflammatory-like response pathways (Figure 6(b)). Pathway maps for drivers and suppressors are shown in Figure 6(c).
Gene-miRNA and gene-TF network analysis
The gene-miRNA network demonstrated that 2 drivers, 1 suppressor, and 1 marker are simultaneously regulated by has-mir-26b-5p and that 2 drivers and 1 marker are simultaneously regulated by has-mir-24-3p. Therefore, we speculate that has-mir-26b-5p and has-mir-24-3p are involved in the regulation of ferroptosis in the context of CPB (Figure 7). The gene-TF network suggested that 2 drivers and multiple suppressors and markers are regulated by Zinc Finger Protein 2(ZFP2), General Transcription Factor IIA Subunit 2(GTF2A2), Forkhead Box J2(FOXJ2), TGFB Induced Factor Homeobox 2(TGIF2), Zinc Finger Protein 589(ZNF589), Interferon Regulatory Factor 1(IRF1), and SMAD Family Member 5 (SMAD5) (Figure 8).

Gene-miRNA network. Red indicates driver gene,blue indicates suppress gene,green indicates marker gene,purple indicates miRNA,pink indicates key miRNA.
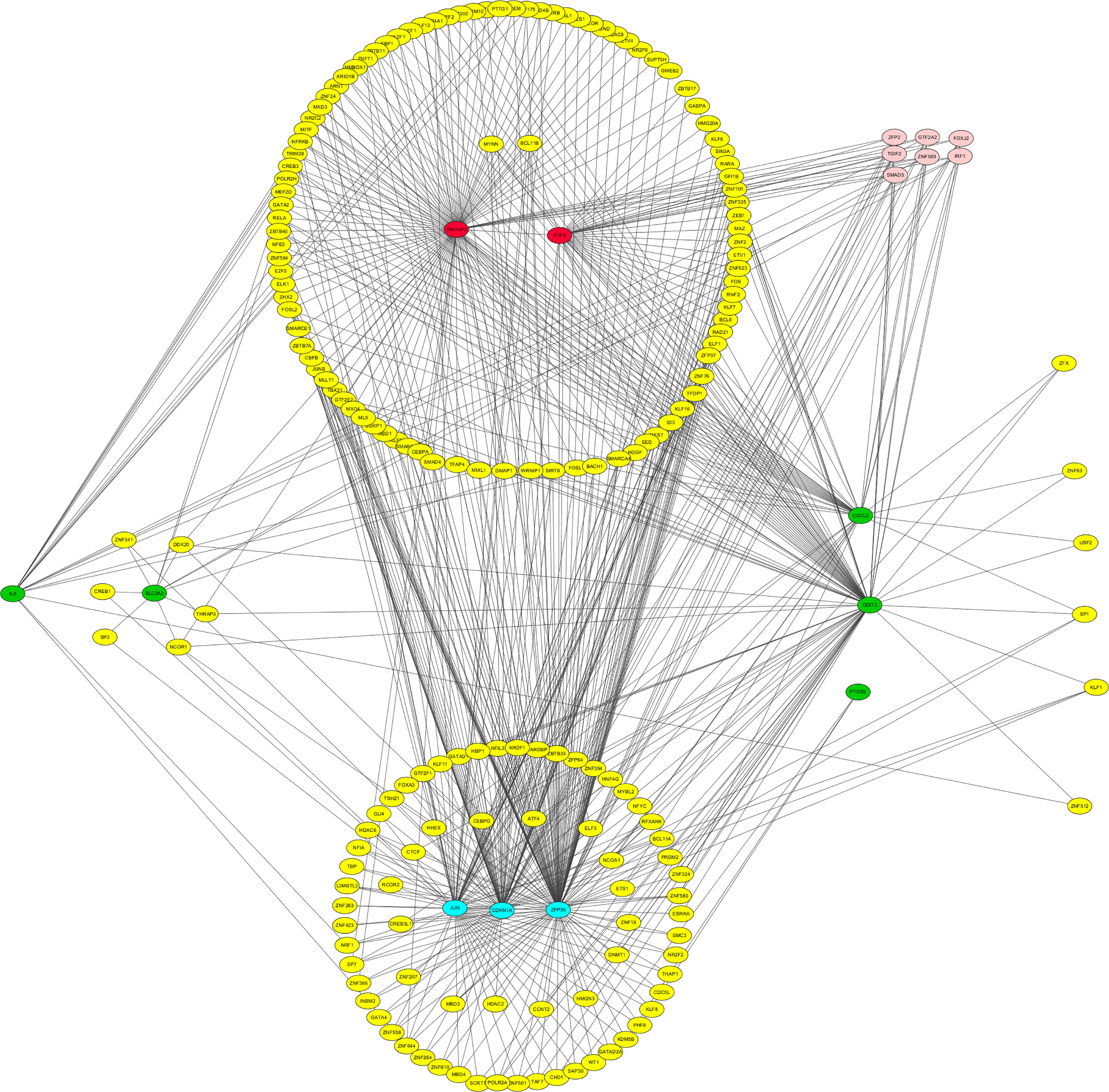
Gene-TF network. Red indicates driver gene,blue indicates suppress gene,green indicates marker gene,yellow indicates transcription factor,pink indicates key transcription factor.
Discussion
The present study systematically investigated functions and regulation of ten ferroptosis-associated genes demonstrating significantly altered expression in response to CPB during corrective surgery for pediatric ToF. Expression of all ten ferroptosis-associated genes was up-regulated in atrial tissue post-CPB, including six markers. This strongly suggests that CPB precipitates ferroptosis within cardiac tissue. It has previously been found that ferroptosis is involved in myocyte loss during heart failure, 23 and heart failure as well as low cardiac output syndrome are the most serious complications following ToF-corrective surgery. 1
Regarding potential regulatory mechanisms controlling CPB-associated ferroptosis, the up-regulated expression of three suppressor genes post- CPB suggests that promotion and restraint of ferroptosis coexist, a phenomenon common to many biological responses (e.g. inflammation). Additionally, all ten ferroptosis-associated genes exhibit positively correlated expression patterns, suggesting possible co-regulation of these genes. That expression of the driver ATF3 correlates particularly strongly with expression of the suppressors CDKN1A and ZFP36 further support the coexistence of ferroptosis initiation and regulation. However, based on the up-regulated expression of multiple marker genes, ferroptosis was activated overall.
Regarding potential molecular mechanisms underlying CPB -associated ferroptosis, enrichment analysis results suggest the involvement of ferroptosis-associated genes in smooth muscle inflammatory pathways. Therefore, we speculate that CPB -associated ferroptosis is associated with myocardial inflammatory responses. Indeed, it has previously been demonstrated that CPB induces a systemic inflammatory response24,25 linked to adverse clinical outcomes ranging from mild adverse effects to severe complications and death. 26 Therefore, limiting the inflammatory response has been an important myocardioprotective intervention during CPB. 26 Clearly, an improved understanding of the molecular mechanism underlying CPB-associated ferroptosis is key to improving myocardioprotective strategies.
Cellular accumulation of oxidant-induced lipid peroxidation is the major mechanism causing iron-dependent ferroptosis.5,27 Reduced glutathione (GSH) is the most abundant human antioxidant and a major contributor to the maintenance of cellular redox homeostasis. 28 Depletion of GSH is an important feature of ferroptosis.29,30 Indeed, it has even been suggested that induction of ferroptosis by means of inhibiting GSH generation could be used as a strategy to promote tumor cell death. 31 In contrast, it may be possible to suppress ferroptosis (and perhaps protect the myocardium) by promoting the generation of GSH or by supplementation with GSH, although this hypothesis requires additional study. In the present study, the driver-suppressor pathway map demonstrates that ATF3 and CDKN1A promote the generation of GSH, likely reflecting a drive to neutralize the excessive lipid reactive oxygen species (ROS) responsible for ferroptosis. Additionally, TNFAIP3 and JUN favor lipid ROS neutralization and GSH generation, respectively.
Although miRNAs play important roles in diverse fundamental biological processes,32,33 few reports discuss miRNA-mediated regulation of ferroptosis. One such study found that miR-137 negatively regulates ferroptosis via direct targeting of the glutamine transporter SLC1A5 in melanoma cells. 34 The gene-miRNA network constructed by the present study suggests that has-mir-26b-5p and has-mir-24-3p simultaneously regulate multiple ferroptosis-associated genes in the context of CPB. In addition, TFs are key regulators of cellular gene expression, 35 and have been implicated in the pathogenesis of various diseases. For example, 164 TFs have been identified as being directly involved in 277 diseases. 36 The gene-TF network constructed by the present study indicates that TFs ZFP2, GTF2A2, FOXJ2, TGIF2, ZNF589, IRF1, and SMAD5 control the expression of multiple ferroptosis-associated genes exhibiting CPB-altered expression, suggesting that these TFs may be the major elements regulating ferroptosis-associated gene expression in this context. Several studies have focused on TFs as targets for myocardial protection.37–40
Taken together, findings suggest that CPB initiates atrial tissue ferroptosis, which may be associated with myocardial inflammatory responses, and it is likely subject to modulatable regulation by the identified miRNAs and TFs. From this perspective, it may be feasible to inhibit CPB-associated ferroptosis via modulation of inflammation, miRNAs, and/or TFs. Despite certain limitations [e.g., the FerrDb knowledgebase reflects mRNA levels in a relatively small dataset (n = 10)], the present study provides initial evidence supporting this novel direction for myocardial protection during CPB. Although the appropriate pharmaceutical compounds may not exist yet, inhibition of ferroptosis in this context may improve outcomes of corrective surgery for pediatric ToF. In future studies, we would like to validate these findings at the protein expression level in a larger cohort, provided such data are available or can be generated, and to confirm that ferroptosis indeed occurs in vivo. We intend to examine peripheral blood for an increase in circulating markers of ferroptosis (end products of lipid peroxidation; reactive aldehydes, such as malondialdehyde and 4-hydroxynonenal) in patients prior to and following CPB.
Supplemental Material
sj-xls-1-sci-10.1177_00368504211050275 - Supplemental material for Identification of ferroptosis-associated genes exhibiting altered expression in response to cardiopulmonary bypass during corrective surgery for pediatric tetralogy of fallot
Supplemental material, sj-xls-1-sci-10.1177_00368504211050275 for Identification of ferroptosis-associated genes exhibiting altered expression in response to cardiopulmonary bypass during corrective surgery for pediatric tetralogy of fallot by Hongtao Liu, Bingyong Zhang, Shaofeng Chen, Yun Zhang, Xin Ye, Yi Wei, Guihong Zhong and Liangqing Zhang in Science Progress
Supplemental Material
sj-jfif-2-sci-10.1177_00368504211050275 - Supplemental material for Identification of ferroptosis-associated genes exhibiting altered expression in response to cardiopulmonary bypass during corrective surgery for pediatric tetralogy of fallot
Supplemental material, sj-jfif-2-sci-10.1177_00368504211050275 for Identification of ferroptosis-associated genes exhibiting altered expression in response to cardiopulmonary bypass during corrective surgery for pediatric tetralogy of fallot by Hongtao Liu, Bingyong Zhang, Shaofeng Chen, Yun Zhang, Xin Ye, Yi Wei, Guihong Zhong and Liangqing Zhang in Science Progress
Footnotes
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request
Ethics statement
The research does not involve ethics, ethics approval was not required.
Consent statement
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
