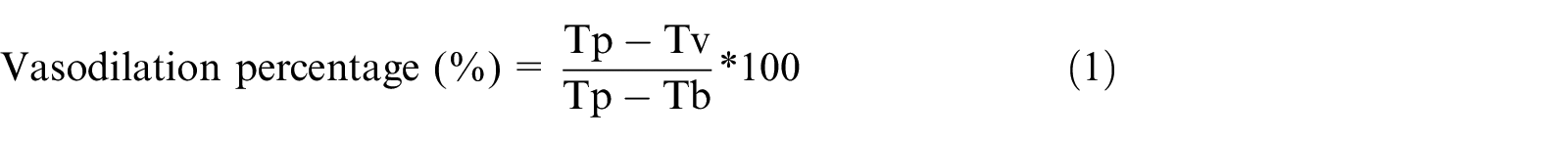Abstract
Regular transient limb ischemia (RTLI) can prevent atherosclerosis in hypercholesterolemic rabbits. As endothelial dysfunction is the initial factor leading to atherosclerosis, we investigated the effect of RTLI on endothelial function in hypercholesterolemic rabbits. We randomly allocated 15 New Zealand white rabbits to three groups, five animals per group: the hypercholesterolemic group (Group H), the sham RTLI group (Group S), and the RTLI group (Group L). All rabbits received hypercholesterolemic fodder daily. No intervention was performed on the rabbits in Group H. Rabbits in Group S were kept in hutches, with a deflated cuff applied to their left hind limb for 60 min every day. For rabbits in Group L, RTLI (six cycles of 5-min ischemia and 5-min reperfusion of the left hind limb) was applied once daily for 12 weeks. At the end of week 12, a segment of the abdominal aorta was isolated from each rabbit for in vitro measurement of the endothelium-dependent vasodilation (EDV) response to different concentrations of acetylcholine and the endothelium-independent vasodilation (EIV) response to sodium nitroprusside. The EDV response was significantly higher in Group L than in Groups S and H (p < 0.05), with no significant difference between Groups S and H (p > 0.05). There was no difference in the EIV response among the three groups. RTLI could improve the EDV response, protecting endothelial function against hypercholesterolemic damage.
Keywords
Introduction
Ischemic heart disease and stroke have emerged as leading global causes of morbidity and mortality over the past 15 years. 1 Endothelial cells exert important functions in this regard, including releasing bioactive substances and maintaining a dynamic balance of inflammatory cells within the vascular wall. 2 Endothelial dysfunction (ED), characterized by impairment of nitric oxide (NO) bioavailability and endothelial-dependent vasodilation (EDV), is the initial factor leading to atherosclerosis.3,4 Oxidative stress caused by an imbalance of oxidant/anti-oxidant mechanisms decreases endothelial NO production and contributes to resultant changes in vascular reactivity.5,6 ED and oxidative stress interact with each other to cause progressive pathophysiological changes apparent in all stages of atherogenesis, which includes pro-inflammatory, pro-oxidant, proliferative, pro-coagulation, and pro-vascular adhesion features.5–8 As the major pathophysiological link between cardiovascular risk factors and atherosclerosis,9–16 ED can be used as a marker of risk factors for cardiovascular disease. Moreover, the prevention of ED would be an attractive therapeutic target to lower the risk of cardiovascular disease.
Przyklenk et al. 17 reported that brief episodes of ischemia of one of the coronary arteries had a protective effect on the remote myocardium against ischemia reperfusion injury caused by subsequent sustained coronary-artery occlusion in a canine model, a phenomenon known as remote ischemic preconditioning (rIPC). Based on this finding, Kharbanda et al. 18 established a model of limb ischemia preconditioning, induced by three cycles of ischemia-reperfusion applied to one arm, against ED induced by subsequent long-lasting ischemia in the contralateral arm. Other studies have similarly demonstrated a protective effect of remote limb ischemia preconditioning against ED induced by ischemia reperfusion in humans.19–24
In a previous study, we demonstrated that regular transient limb ischemia (RTLI), induced by six cycles of 5-min ischemia and 5-min reperfusion of the hind limb, implemented daily for 12 weeks could prevent atherosclerosis in hypercholesterolemic rabbits. 25 However, the mechanism by which RTLI protects against atherosclerosis remains unknown. We hypothesized that RTLI could protect endothelial function against the damage induced by hypercholesterolemia. Accordingly, our aim in this study was to measure EDV and endothelium-independent vasodilation (EIV) in aortic segments from rabbits who underwent RTLI while being fed a high-cholesterol chow.
Materials and methods
Ethics and informed consent
This study was based on hypercholesterolemic rabbit model, approved by the Institutional Animal Care and Use Committee (IACUC) of Sun Yat-sen University, Guangzhou, China (No. LACE-2010-0901, in September 2010) and conformed to the guidelines on animal welfare set by the National Committee for Animal Experiments.
Animals and groupings
The overall experimental protocol is shown in Figure 1. The experiment was performed using 15 male New Zealand white rabbits, 3–4 months of age, obtained from the Medical Experimental Animal Center of Guangdong Province, Guangzhou, China. After a 1-week period of acclimatization, rabbits were randomly allocated to the following three groups, with five animals in each group: the hypercholesterolemic group (Group H), the sham RTLI group (Group S), and the RTLI group (Group L). The preparation of the hypercholesterolemic fodder and the feeding protocol was adopted in accordance with our previous study. 25 In brief, all the rabbits were fed with 70 g of normal fodder in the morning and 80 g of hypercholesterolemic fodder, which included 1.5 g of cholesterol, 9 g of corn oil, and 69.5 g of normal chow, in the evening. Cholesterol powder (Sinopharm Chemical Reagent Co., Ltd, Shanghai, China) was dissolved in heated corn oil (Wilmar International Co., Ltd, Shanghai, China) and mixed with the normal rabbit chow to prepare the hypercholesterolemic fodder, and this fodder was used after cooling down to room temperature. The rabbits were fed in individual stainless-steel cages for 12 weeks. All animals had free access to water during the experimental period.

Experimental protocol.
RTLI procedures
RTLI was induced by six cycles of left hind-limb ischemia and reperfusion. A 4.5-cm wide blood pressure cuff was placed around the base of the left hind limb and inflated to a pressure of 200 mmHg for 5 min to occlude arterial blood flow and then deflated to allow reperfusion for 5 min for each cycle. 26 Rabbits in Group H did not receive any intervention. Those in Group S were placed in hutches with a deflated cuff placed on the left hind limb for 60 min daily as the sham intervention. Rabbits in Group L were kept in hutches and underwent RTLI every afternoon for 12 weeks. All procedures were performed without anesthesia but with caressing to maintain the animal’s calm. 26
Preparation and connection of aortic segments
At the end of the 12 weeks of the study period, animals were euthanized using an intravenous overdose of sodium pentobarbital (120 mg/kg; Sigma-Aldrich, Saint Louis, Missouri, USA). A segment of about 4 cm was isolated from the abdominal aorta, just above the bifurcation of the common iliac artery. The abdominal aorta segment was immediately immersed in a petri dish filled with cooled Krebs buffer (pH 7.4) composed of (in mM) 118.0 NaCl, 4.7 KCl, 1.2 KH2PO4, 1.2 MgSO4, 15.0 NaHCO3, 5.5 glucose, and 2.5 CaCl2 (Guangzhou Chemical Reagent Factory, Guangzhou, China). The Krebs buffer was cooled by placing the petri dish on a bed of ice and continuously oxygenated with mixed gas (95% O2 and 5% CO2; Guangzhou Junqi Gas Co., Ltd., Guangzhou, China). After removal of loose connective tissue, the vessel was cut transversely into four segments, each about 4-mm in length, using microscissors. We left the endothelium intact in two of these segments (endothelium-preserved segments) and completely removed the endothelium, using absorbent cotton, in the other two segments (endothelium-deprived segments). Two stiff stainless-steel hooks were threaded through the segments. One hook was coupled to a force transducer (JZJ01H; Chengdu Instrument Factory, Sichuan, China), via a surgical suture, and the other fixed to a plastic support located near the bottom of the single-organ bath (SQG-4; Chengdu Instrument Factory). We adjusted the relative position of the two hooks by changing the distance between the transducer and the plastic support to equilibrate the segments for 90 min at a resting tension of 2.0 g. 27 Subsequently, each segment was mounted in a separate single-organ bath (SQG-4; Chengdu Instrument Factory) filled with thermostatic Krebs buffer and connected to a force transducer and a polygraph (BL-420E+; Chengdu Taimeng Software Co., Ltd., Sichuan, China) for real-time measurement and recording of within-segment changes in tension. The Krebs buffer in the organ baths was infused using a mixed gas (95% O2 and 5% CO2), with continuous thermostatic control, via a high-precision water cabinet (DKB-501A; Shanghai Sumsung Laboratory Instrument Co., Ltd., Shanghai, China), to maintain a temperature of 37°C. The standard fixation of the aortic segments during testing is shown in Figure 2.

Standard fixation of the aortic segments for testing. Two stiff stainless-steel hooks were threaded through the segments. One hook was coupled to a force transducer, via a surgical suture, and the other to a plastic support located near the bottom of the single-organ bath.
Assessment of vasodilation function
After the 90-min period of equilibration, we recorded the basal tension (Tb) of the aortic segments. Next, phenylephrine (1 × 10−6 mol/L; Sigma-Aldrich) was used to induce a preconstruction of the aortic segments. After obtaining a stable plateau of contraction, the maximum contraction tension (Tp) was recorded. Then, acetylcholine (ACH; Sigma-Aldrich), added at sequential cumulative doses of 1 × 10−9, 1 × 10−8, 1 × 10−7, 5 × 10−7, 1 × 10−6, and 5 × 10−6 mol/L to the two organ baths containing the endothelium-preserved segments, was used to assess the EDV response. Similarly, we tested the tension response to sodium nitroprusside (SNP; Sigma-Aldrich), added at sequential cumulative doses of 1 × 10−9, 1 × 10−8, 3.3 × 10−8, 1 × 10−7, 3.3 × 10−7, and 1 × 10−6 mol/L to the other two organ baths containing the endothelium-deprived segments, to assess the EIV response. The vasodilation tension (Tv) response of the aortic segments was recorded at every cumulative concentration of ACH or SNP to obtain the dose-response curves. The percent of vasodilation was calculated using equation (1), where Tp is the maximum contraction tension, Tv, the vasodilation tension, and Tb, the basal tension. This study was blinded by using sample numbers to analyze the vasodilation function and disclosing it after analysis.
Statistical analysis
The normality of the data distribution was evaluated using the Shapiro-Wilk test, with a p > 0.05 indicating a normal distribution. All of the data were expressed as the mean ± standard deviation (SD). Differences in the EDV and EIV response among the three groups were evaluated using a repeated-measures analysis of variance, with post hoc multiple comparisons performed using the Bonferroni test. Between-group differences in the EDV and EIV response at each ACH/SNP concentration were evaluated using a multivariate general linear model, with post hoc multiple comparisons performed using Bonferroni test. All statistical analyses were performed using SPSS statistical (version 16.0; IBM Corp., Armonk New York, US). Statistical significance was accepted at a p-value <0.05.
Results
General data
There was no dropout of experimental animals; however, the data from two aortic segments from one rabbit in Group L were excluded due to the incorrect concentration of the mixed gas infused in the buffer solution during the experiment. The percentage vasodilation data of the aortic segments in response to different cumulative concentrations of ACH were normally distributed (p > 0.05) for the three experimental groups. The percentage vasodilation data of the aortic segments in response to different cumulative concentrations of SNP were also considered to have an approximately normal distribution among the three groups as non-normal distribution (p = 0.019) was identified only at one concentration (1 × 10−7 mol/L) in Group S.
Endothelium-dependent vasodilation
Differences in the EDV response among the three groups were significant (p < 0.05). The percentage of vasodilation was significantly higher in Group L than in Group H (p < 0.05) at cumulative doses of 1 × 10−7, 5 × 10−7, and 1 × 10−6 mol/L. The percentage of vasodilation in Group L was also significantly higher than in Group S (p < 0.05) at cumulative doses of 1 × 10−7 and 5 × 10−7 mol/L. There was no difference in the percentage of vasodilation between Groups S and H (p > 0.05). The maximal vasodilation response was induced at an ACH concentration of 1 × 10−6 mol/L; further increases in the dose of ACH did not result in an increase in the percentage of vasodilation (Figure 3).

Percentage of vasodilation of the abdominal aortic segments in response to ACH application for the three experimental groups. Two aortic segments were obtained from each animal: Group H (10 segments), Group S (10 segments), and Group L (8 segments).
Endothelium-independent vasodilation
There was no difference in the percentage of the EIV response among the three groups (p > 0.05). The dose–response curves of the EIV response, induced by SNP, in the three groups almost overlapped (Figure 4).

Percentage vasodilation of the abdominal aortic segments in response to SNP. Two aortic segments were obtained from each animal: Group H (10 segments), Group S (10 segments), and Group L (8 segments).
Discussion
Our study found that RTLI provided a protective effect of the EDV response in hypercholesterolemic rabbits, indicating that RTLI could protect endothelial function against hypercholesterolemic damage.
There are two types of vasodilation, EDV and EIV.2,28,29 EDV depends on the endothelium to produce vascular dilation substances.2,30 Specifically, ACH, bradykinins, and adenosine can increase the production of NO from the endothelium to induce vasodilation.31,32 By contrast, the substances inducing vascular dilation are not produced by the endothelium in EIV, such as SNP, which offers exogenous NO to induce vascular dilation. 33 In accordance with these classic principles, we used ACH and SNP respectively to test EDV and EIV of the aortic segments.
Injury to endothelium cells is the vital inducing factor of atherosclerosis and plays an important role in the occurrence and progression of the disease.15,16 Although the manifestations of endothelial injury vary, the key feature is impairment of EDV function.3,4 Over the past few years, several studies have suggested that rIPC can improve endothelial function in healthy humans.20,23,34 However, the long-term effect of preconditioning in models of atherosclerosis has remained unknown. For this reason, we emphasized the early, regular, and long-term nature of RTLI as an interventional strategy. Our results indicated that RLTI preserved the EDV response to ACH, confirming our hypothesis that RTLI might protect EDV against hypercholesterolemic damage in rabbits. Our findings also provided preliminary evidence of the mechanism underlying the preventive effect of RTLI on atherosclerosis, which we had detected in our previous study. 25
Various pharmacological interventions improve EDV, including the use of antioxidant vitamins and endothelial nitric oxide synthase (eNOS) cofactor tetrahydrobiopterin (BH4).35–37 Endothelial function can also be improved by lifestyle changes, including physical exercise and weight loss.38–40 However, these interventions do not exert a direct anti-atherosclerotic effect. Our previous study verified that RTLI, provided once per day over a 12-week period of hypercholesterolemic-fodder feeding, could reduce the percentage of plaque area in the aorta in hypercholesterolemic rabbits. 25 As an early, long-term, feasible and non-pharmacological intervention to slow down the progression of atherosclerosis, further investigation of RTLI was warranted. And our present finding may have the potential for further investigation of preventing atherosclerosis.
The main limitations of this study are the level of protein biomarkers of ED such as endothelin-1, C-reactive protein, Willebrand factor were not investigated. Also, nitric oxide production in the aorta, plasma levels of nitrite/nitrate, endothelial proliferation, susceptibility to apoptosis, permeability, and leukocyte adhesion were neither tested. Moreover, whether ACH induced endothelium-dependent relaxation via releasing NO or prostacyclin and/or by hyperpolarizing cells in this study was not confirmed. We are planning to do experiments to investigate the above limitations.
However, we did not specifically investigate the mechanism by which RTLI protected endothelial function from hypercholesterolemia, which might be a limitation of our study. Previous studies have explored a similar question. One study reported that ischemic preconditioning (IPC) might provide sustained protection to ischemia reperfusion induced ED in humans through a cyclooxygenase-2-independent mechanism. 21 Another study indicated that repeated IPC improved EDV in humans by increasing NO production and a number of endothelial progenitor cells. 20 A recent study showed that in patients with coronary heart disease, IPC might improve endothelial function via activation of signal transducer and activator of transcription-3, resulting in an increase in endothelial progenitor cells. 24 This will be the focus of our future studies.
As well, our study design was based on the finding of a previous study that confirmed that RTLI could prevent the progression of atherosclerosis in hypercholesterolemic rabbits. 25 As such, we did not compare endothelial function between rabbits fed with normal and hypercholesterolemic fodders. Although this might be the other limitation of our study design, we do note previous studies which reported that hypercholesterolemia impairs EDV but not EIV.38,41–44
Conclusion
RTLI conferred protection against ED induced by hypercholesterolemia in rabbits. This might be one key pathway by which RTLI prevents the progression of atherosclerosis in hypercholesterolemic rabbits. Based on our findings, RTLI might be an ancillary therapeutic technique for people with a high risk for atherosclerosis.
Footnotes
Acknowledgements
We thank Baofeng Lv from the department of anesthesia at the First Affiliated Hospital of Zhengzhou University, Yi Yang from the department of urology at the First Affiliated Hospital of Shenzhen University, and Kai Wang from the department of anesthesia at the Sixth Affiliated Hospital of Sun Yat-sen University for their assistance in rabbit feeding and tissue collection. We are also grateful to Cheng Li from the School of Pharmaceutical Science of Sun Yat-sen University and Zhihui Wu from Hefei Zhihui Excellence Education Consulting Co., Ltd. for their technical assistance. We would like to thank Editage (![]() ) for English language editing.
) for English language editing.
Authors contributions
Sanqing Jin proposed and supervised the experiment and revised the draft. Zhinan Zheng took part in the study design, data analysis, and revision of the draft. Hongli Wang, Nanrong Zhang, and Yan Zhou performed the experiment. Hongli Wang wrote the draft. All of the authors have reviewed and approved the final version of this paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the provincial Science and Technique Project [No. 2011B080701029] and provincial Natural Science Foundation [No. S2012010009396, 2016A030313303] from the Department of Science and Technology of Guangdong Province.
Ethical statement
This study was based on hypercholesterolemic rabbit model, approved by the Institutional Animal Care and Use Committee (IACUC) of Sun Yat-sen University, Guangzhou, China (No. LACE-2010-0901) and conformed to the guidelines on animal welfare set by the National Committee for Animal Experiments.
Data availability
The data of vasodilation responses of rabbits’ abdominal aortic segments to ACH or SNP, used to support the conclusions of this study, are available from the corresponding author upon request.