Abstract
Hepatocellular carcinoma is one of the leading causes of malignant tumor related death word wide with poor prognosis. Chemotherapy and TACE are main treatment methods for advanced stage cases. Rapamycin, a macrolide compound that initially used to coat coronary stents, can inhibit the growth of a variety of cancer cells especially hepatocellular carcinoma. Twenty-four healthy adult New Zealand white rabbits underwent CT-guided puncture to prepare a model of VX2 liver xenograft tumor. The rabbits were randomly divided into four groups with six in each group and received the following treatments: APR-TACE1: arterial perfusion of high-dose rapamycin combined with TACE; APR-TACE2: arterial perfusion of low-dose rapamycin combined with TACE; TACE: TACE alone; and IVR-TACE: intravenous injection of rapamycin combined with TACE. Two weeks after TACE treatment, the rabbits received CT scan and DSA angiography examination, and then killed by air embolism. The non-necrotic region and surrounding tissues were obtained from the peripheral tumor for iNOS, HIF-1α, VEGF, Bcl-2, and Bax protein expression analysis. Protein
Introduction
Hepatocellular carcinoma ranks fifth in males and seventh in females in terms of incidence and ranks second in males and sixth in females in the aspect of mortality among all malignant tumors. 1 Transcatheter arterial chemoembolization (TACE) is the preferred treatment for patients with liver cancer who cannot undergo surgical resection or those who have postoperative recurrence disease.2–5 Although TACE demonstrated significant curative effect on hepatocellular carcinoma, the recurrence rate of this disease remains high and interventional treatment is thus required. After multiple interventional treatments, the established collateral circulation often cannot achieve complete embolism because TACE can only embolize the existing tumor to nourish blood vessels but cannot inhibit tumor angiogenesis.6,7 As had been proved that carcinoma angiogenesis was one of the important approach for cancer cells metastasis. Hence, inhibiting tumor angiogenesis after TACE treatment is the key to improve the effectiveness of interventional embolization of hepatocellular carcinoma. Rapamycin is a new type of macrolide immunosuppressant with high immunosuppressive effect. Rapamycin can inhibit the growth of a variety of cancer cells, such as liver cancer,8,9 renal cancer, 10 colorectal cancer, 11 ovarian cancer, 12 lung and breast cancers. 13 Studies have demonstrated that rapamycin had a significant cell proliferation inhibitory effect on hepatocellular carcinoma. 8 Publications also indicated that rapamycin can inhibit tumor angiogenesis and induce apoptosis and autophagy. 14 Therefore, in our present work, we establish rabbit hepatic xenograft tumors and aim to investigate efficacy of arterial instillation of rapamycin+TACE on VEGF, iNOS, HIF-1α, Bcl-2, Bax, and vascular density in rabbit hepatic xenograft tumors.
Material and methods
Ethical approval
This animal experiment was approved by the Ethics Committee of the First affiliated Hospital of Henan University.
Rabbit hepatic xenograft tumors modeling
Healthy male and female New Zealand white rabbits weighing 2.5 ± 0.4 kg were selected, placed in single cages, and fed with pellets. VX2 tumor cells was inoculated into the hind leg muscle tissue to establish a hepatic xenograft tumor model with tumor diameter of 1–2 cm. The xenograft tumor was peeled out from the thigh muscle tissue. The white fresh xenograft tumor tissue was cut into 1 mm3 block and placed into a sterile Petri dish containing 1640 fluid. After general anesthesia, the rabbit was placed in supine position and fixed on the experimental platform. The upper abdomen and the sword in the vicinity of hair removal were disinfected. An 18 G puncture needle was inserted first into a pipe with 2 cm-long tube made of gelatin sponge bar and then into two to four 1 mm3 tumor blocks. The left lobe of the hepatic puncture was confirmed by CT scan. The rabbits were given CT examination 2 weeks after modeling to measure the xenograft tumor diameter (Figure 1).

Rabbit hepatic xenograft tumors model successful made (CT scan of the rabbit liver showed a low-density area of the liver, marked by the red circle).
Grouping
Twenty-four of the model rabbits were divided into four groups on average (n = 6), and received the following treatments: APR-TACE1group: rapamycin+TACE (arterial perfusion of 2 mg/kg rapamycin+1 mg/kg epirubicin and 0.2 mg/kg mitomycin and Lipiodol emulsion embolization; arterial perfusion of rapamycin and TACE); APR-TACE2 group: rapamycin+TACE (arterial perfusion of 1 mg/kg rapamycin+1 mg/kg epirubicin and 0.2 mg/kg mitomycin and Lipiodol emulsion embolization; arterial perfusion of rapamycin and TACE); TACE group (1 mg/kg epirubicin, 0.2 mg/kg mitomycin, iodized oil made of emulsion embolism; also known as TACE group); and IVR-TACE group: intravenous injection of rapamycin+TACE (1 mg/kg rapamycin+1 mg/kg epirubicin and 0.2 mg/kg mitomycin and Lipiodol emulsion embolization; intravenous injection of rapamycin and TACE). After intraperitoneal injection of general anesthesia with 5% pentobarbital, the exposed femoral artery of the rabbit was incised and successfully punctured with a 21 G needle. A 0.014# micro guide wire was introduced, the puncture needle was withdrawn, and a 2.7 F microcatheter was placed (Terumo, Japan). Micro-guide wire was conducted to super select the artery that nutrition the tumor, and the microcatheter guide angiography was performed to analyze vascular staining, location, size, and other conditions of the hepatic xenograft tumor. A micro catheter ultra was inserted into the main tumor to nourish blood vessels. Perfusion and embolization was performed. 2 The degree of embolism was set as the standard to achieve complete embolization of hepatic xenograft tumor vessels (Figure 2). After the TACE, the rabbit was given intramuscular injection of 40 (million U) penicillin for 3 days regularly.
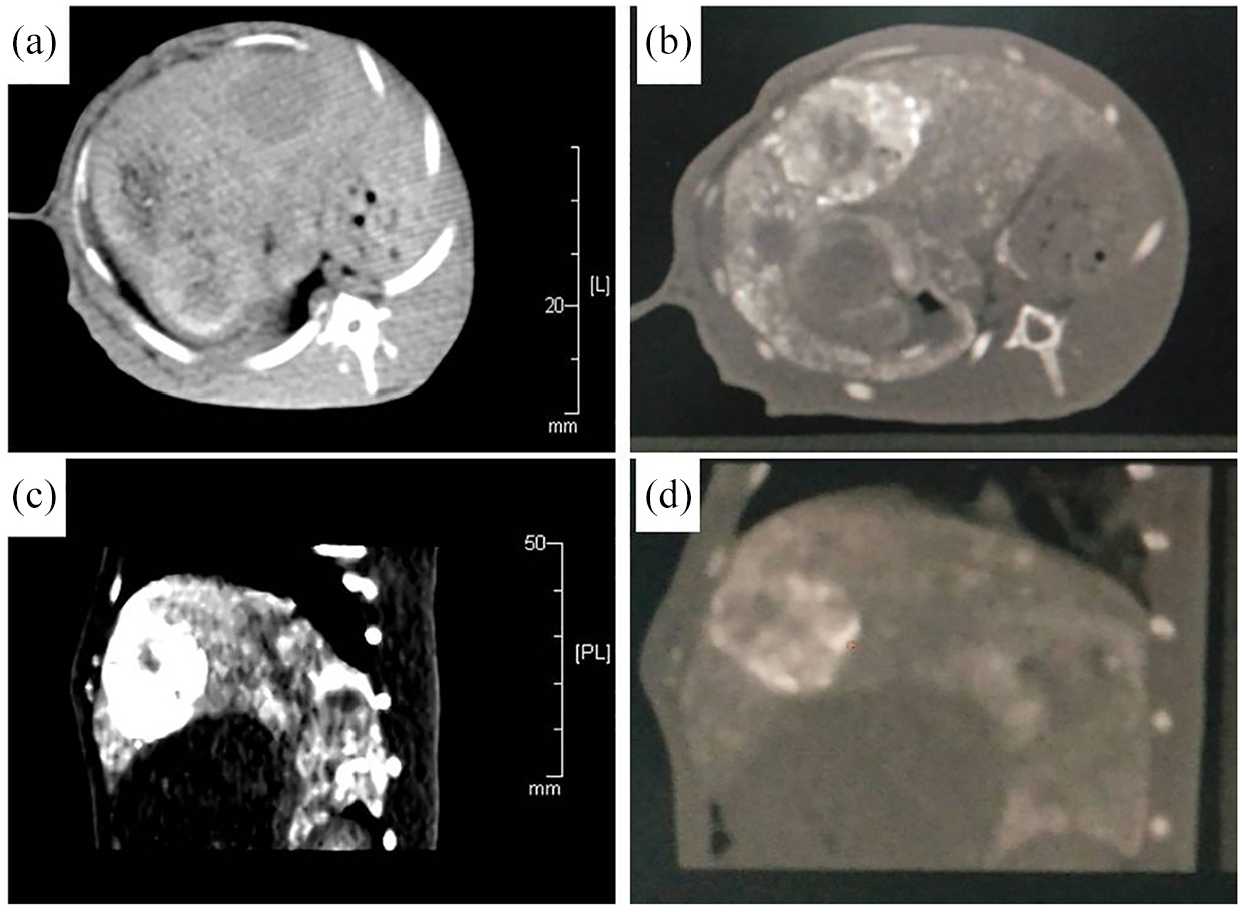
Rabbit hepatic xenograft tumors after arterial instillation of rapamycin combined with TACE treatment: (a) CT scan of the hepatic xenograft tumors before treatment showed a round low density area in live, (b) CT scan with the axial position after treatment showed an highlight round area in the live, (c and d) CT scan of the rabbit live with sagittal position after treatment demonstrated highlight round area in the live.
Observation index
At 14th day post-operation, CT scan and DSA angiography were performed and air embolism was executed. The non-necrotic region and the surrounding hepatic tissues were obtained from the peripheral tumor. HIF-1α, VEGF, iNOS, BCL-2, Bax, and CD34 protein expression were detected by Western blot and immunohistochemical assay. Microvessel density (MVD) of the hepatic carcinoma was also calculated.
Western blot and MVD measurement
The hepatic xenograft tumor tissues were crushed in liquid nitrogen and transferred to a tube with cold RIPA buffer. The lysate were incubated on ice for 0.5 h. The supernatants were resolved on 6% or 11% gradient SDS-PAGE and transferred NC membranes. Membranes were blocked for 1 h at room temperature in 5% milk in PBST and incubated with the antibodies at 4°C overnight. The membranes were then incubated with appropriate HRP-conjugated secondary antibody. The densitometry of immunoblotting results were measured by using ImageJ1.8.0 software (https://imagej.nih.gov/ij/download.html). MVD was measured according to the CD34 protein expression by immunohistochemistry assay. The MVD was calculated by counting the number of microvessels in three areas of maximum microvessel density within (400×). The MVD was calculated by taking the average value, and the results were expressed as mean±sd.
Statistics analysis
STATA11.0 (http://www.stata.com) statistical software was applied for data analysis. The measurement data of xenograft tumor diameter was expressed by
Results
Xenograft tumor diameter
After 14 days of the treatment, the xenograft tumor diameter was re-measure by the CT scan. The xenograft tumor diameters were not changed obviously before and after treatment in the four groups (p > 0.05), Table 1.
The xenograft tumor diameter for each group before and after treatment (

Immunohistochemistry assay
iNOS was mainly expressed in the cytoplasm, and diffuse brown–yellow particles were detected. iNOS in APR-TACE1 was lower than those of groups APR-TACE1, TACE, IVR-TACE with statistical difference (p < 0.05). HIF-1α was mainly expressed in the cytoplasm, and the positive staining revealed an even distribution of brown–yellow or chocolate brown particles. The positive staining of VEGF was uniformly diffused in the cytoplasm and showed brown–yellow particles. Bcl-2 was positively stained in the cell membrane, cytoplasm, and/or nuclear membrane of the cancer cells, and chocolate-brown particles were noted. Bax-positive staining was localized in the perinuclear cytoplasm of hepatocellular carcinoma cells, and chocolate brown particles were observed (Figure 3).

Immunohistochemistry assay examined the iNOS, HIF-1α, VEGF, Bcl-2, and Bax expression in rabbit hepatic xenograft tumors of different groups, ×200.
Protein expression of HIF-1\alpha , VEGF, and iNOS
The protein expression levels of iNOS, HIF-1α, and VEGF in APR-TACE1 were significantly lower than those in groups APR-TACE1, TACE, and IVR-TACE (p < 0.05). The expression levels of iNOS, HIF-1α, and VEGF in APR-TACE2 were lower than those in TACE, and the difference was statistically significant (p < 0.05). The expression levels of iNOS and VEGF in APR-TACE2 were significantly lower than those in IVR-TACE (p < 0.05). The expression of HIF-1α was not significantly different between groups APR-TACE2 and IVR-TACE (p > 0.05). Moreover, the expression levels of VEGF, iNOS, and HIF-1α differed between groups TACE and IVR-TACE. The difference in the expression of iNOS was significant (p < 0.05), whereas that in the expression levels of VEGF and HIF-1α was not statistically significant between the two groups (p > 0.05), Figure 4.

HIF-1α, VEGF, and iNOS protein expression of different groups: (a) western blotting assay in detection of HIF-1α, VEGF, and iNOS protein expression and (b) bar plot of HIF-1α, VEGF, and iNOS different expression in different groups normalization to β-actin.
Analysis of MVD of tumor tissues
The MVD in APR-TACE1 was significantly lower than that in groups APR-TACE2, TACE, and IVR-TACE (p < 0.05). The MVD in APR-TACE2 was lower than that in groups TACE and IVR-TACE, and the difference was statistically significant (p < 0.05). MVD was also compared between groups TACE and IVR-TACE, and the difference was not statistically significant (p > 0.05), Figure 5.

Immunohistochemistry assay examined the CD34 expression: (a) APR-TACE1, (b) APR-TACE2, (c) TACE, (d) IVR-TACE, and (e) scatter plot of CD34 expression in different groups, ×400.
Protein expression of BCL-2 and Bax
The protein expression of BCL-2 in APR-TACE1 was significantly lower than that in groups APR-TACE2, TACE, and IVR-TACE (p < 0.05). The Bax expression was not significantly different between groups APR-TACE1 and APR-TACE2 (p > 0.05). By contrast, the Bax expression in APR-TACE1 was significantly different from that in groups TACE and IVR-TACE (p > 0.05). The expression levels of BCL-2 and Bax were significantly different between groups APR-TACE2 and TACE (p < 0.05) but were not significantly different between groups APR-TACE1 and IVR-TACE (p > 0.05). Moreover, the expression levels of BCL-2 and Bax were not significantly different between groups TACE and IVR-TACE (p > 0.05), Figure 6.

Bcl-2 and Bax protein expression of different groups: (a) western blotting assay in detection of Bcl-2 and Bax protein expression and (b) bar plot of Bcl-2 and Bax protein different expression in different groups.
Discussion
In the present work, we found that arterial instillation of rapamycin combined with TACE in treatment of rabbit hepatic xenograft tumors can reduce tumor neovascularization and inhibit the expression levels of NOS, HIF-1α, VEGF, Bcl-2, and Bax. This findings may provide new prospective for hepatocellular carcinoma through combination of rapamycin and TACE. Rapamycin inhibits the expression of iNOS, HIF-1α, and VEGF protein, which can promote the angiogenesis, therefore inhibiting the tumor angiogenesis. The results also showed that the MVD value of the tumor surrounding tissue in the artery perfusion rapamycin group was significantly lower than that in the TACE only group, which demonstrated that rapamycin could inhibit the formation of tumor vessels after embolization. Therefore, arterial infusion of rapamycin can improve the therapeutic effects of TACE.
VEGF is an early vascular permeability factor and a vascular endothelial cell-specific heparin-binding growth factor that stimulates the division and proliferation of vascular endothelial cells and induces neovascularization in vivo; 15 VEGF is currently the most powerful protein factor that promotes angiogenesis and plays a key role in the formation of tumor blood vessels and the proliferation of tumors. 16 The expression of iNOS in tissues is related to the proliferation and growth of tumors. 17 On the one hand, iNOS mediates the proliferation and up-regulation of VEGF through its product of NO, which promotes the growth of tumors.18,19 On the other hand, iNOS promotes tumor angiogenesis, proliferation, differentiation, and growth by up-regulating MMPs and P53 oncogene. 20 HIF-α is an important reverting factor that maintains the oxygen balance in the body and is a gene subunit that regulates the oxygen balance of tissues. 21 HIF-α protein considerably affects biological processes such as differentiation, growth, survival, and apoptosis of tissue cells.22–24 The transitional expression of HIF-α in tissues can induce the transitional proliferation and growth of tissues, which mutate to form tumors. Studies have revealed the presence of HIF-1α transient expression in multiple tumor tissues,25–27 and this phenomenon is correlated with the invasion and metastasis of tumors and affects the sensitivity of tumor tissues to chemotherapy.23,24
Different dosage of rapamycin (1 and 2 mg/kg) in arterial infusion exert varied inhibitory effects on angiogenesis after TACE treatment. Arterial perfusion of 2 mg/kg rapamycin evidently inhibits the expression of iNOS, HIF-1α, and VEGF and decreased the MVD in the tumor peripheral tissue. Arterial instillation of rapamycin also inhibits the expression of the BCL-2 protein, which inhibits cell apoptosis and the effect is dose dependent. Rapamycin treatment promotes the expression of the Bax protein, which can promote apoptosis and the effect is not directly proportional to the dose of rapamycin infused. In addition, arterial administration of rapamycin has stronger inhibitory effect on promoting the expression of cytokines in angiogenesis and the apoptosis of cells in hepatic xenograft tumors compared with intravenous rapamycin treatment. Furthermore, arterial instillation of high doses of rapamycin increases the value of MVD in the surrounding tissue of the xenograft tumor and can inhibit neovascularization of the tumor. However, this experiment only designed few arterial infusion dose groups. Increasing the dose of rapamycin did not significantly increase the expression of the Bax protein but significantly increased that of the BCL-2 protein. Arterial instillation of high doses of rapamycin could lead to strong inhibitory effects on xenograft tumor angiogenesis and promotes cell apoptosis. Rapamycin is an immunosuppressive agent, the greater its dosage is, the stronger the immune inhibition of the organism will be, and the more unfavorable the body to the hepatic cancer. Further experiments must be conducted to determine the effects of arterial instillation of rapamycin on the body’s immunosuppression and hepatic and kidney toxicity.
Rapamycin inhibits cancer angiogenesis mainly through inhibiting iNOS, VEGF expression and pathway. 28 Rapamycin in combination with the FKBP12 protein (FK506-binding protein12) form the RAPAMYCIN/FKBP12 complex, which combines with the mTOR FRB structure domain.29,30 The formed complex suppresses the phosphorylation of mTOR downstream molecules, namely, 4e-bp131,32 and S6K1. 33
Conclusion
Arterial instillation of rapamycin combined with TACE in treatment of rabbit hepatic xenograft tumors can reduce tumor neovascularization and inhibit the expression levels of NOS, HIF-1α, VEGF, Bcl-2, and Bax. This findings may provide new prospective for hepatocellular carcinoma through the combination of rapamycin TACE. However there were also limitations in the present work.,First, whether rapamycin can improve the prognosis of patients with hepatocellular carcinoma was not clear yet. Second, the tumor treatment response was evaluated by CT not MRI. Therefore, the exact anti-cancer efficacy of rapamycin need further validation by well-designed prospective clinical trials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Fund Youth Fund Project (No. 81501567).
