Abstract
To analyze the clinical characteristics and therapeutic effects of transoral paraquat poisoning combined with Esophagitis dissecans superficialis (EDS). A retrospective observational study was conducted on paraquat poisoning patients between January 1, 2011 and August 30, 2016 in Qilu hospital. Fifteen patients with EDS were enrolled in this study. The clinical characteristics, prognosis, and pathological features of esophageal necrosis mucosa of these patients were retrospectively analyzed and summarized. Esophageal mucosal dissection occurs mainly within 3–8 days after transoral paraquat poisoning in 15 patients. Dosage of paraquat is range from 50 to 100 ml. Most patients have physical problems with swallowing before the intramural esophageal dissection occurred. And there are other symptoms, including sore throat or dysphagia (100%), nausea and vomiting (86.7%), heartburn or upper abdominal pain (73.3%), hematemesis (60%), abdominal distension (20%) and cough frequently (6.7%). In death group, most patients demonstrate features of the multiple organ failure when the esophageal mucosal stripping happened, including lung injury, renal failure, and hepatic failure. The shape of esophageal dissection was tubular in 60%, irregular in 40%, and they vary in size. Pathological examination showed extensive injury, necrosis and hemorrhage of digestive tract epithelium, and obvious inflammatory reaction of epithelial tissue. Transoral paraquat poisoning has certain damage to the patient’s esophageal mucosa, and some may be complicated with EDS, and the prognosis is poor, especially when combined with multiple organ dysfunction. Esophageal damage is mainly located in the esophageal mucosa and have different degrees. Special attention should be paid on such patients.
Introduction
Paraquat (1,1′-dimethyl-4,4′-bipyridylium dichloride; PQ) is a nonselective contact herbicide that is widely used in China. 1 It is not harmful to the environment due to its rapid deactivation after absorption in soil, however, PQ is highly toxic to humans when ingested. 2 PQ-exposure generates iNOS-mediated nitric oxide production, which causes lipid peroxidation and oxidative stress, finally leading to organ damage. 3 PQ also massively accumulates in lung epithelial cells and causes cellular damage by damaging the mitochondria directly and generating reactive oxygen species. 4
Oral herbicide PQ often causes serious damage to multiple organs, such as the lungs, kidneys, and liver. Respiratory failure caused by lung injury is the most common cause of death. 5 In addition, PQ poisoning through oral ingestion is often accompanied by corrosive damage of the digestive tract mucosa. 6 And esophageal mucosal exfoliation may even occur in very few serious cases. Since 1892, Rosenberg coined the term “Esophagitis dissecans superficialis (EDS)” to describe the phenomenon of esophageal mucosal exfoliation. 7 It has been reported that more cases have been discovered with the widespread use of endoscopic, often accompanied by many serious diseases and certain medical treatment.8,9 However, in rare cases, tubular exfoliated esophageal mucosa may be spit out in the case of EDS. So far, it has been rarely reported about PQ poisoning causing EDS in medical literature. Therefore, retrospectively analysis of clinical characteristics, pathological features and clinical outcomes of patients with EDS caused by oral PQ poisoning can help people understand the pathogenesis of this disease and summarize relevant treatment and preventive measures.
Materials and methods
This is a retrospective observational study collecting patients with oral PQ poisoning combined by EDS in Qilu Hospital of Shandong University between January 1, 2011 and August 30, 2016.
This clinical study followed the Declaration of Helsinki and is compatible with the Medical Ethics Committee of Qilu Hospital of Shandong University. The trial was limited to patients with oral PQ poisoning. All patients had only a positive history of PQ intake, and the abnormal necrotic material vomited was pathologically confirmed as esophageal necrotic mucosa. Patients with mixed toxicity were excluded from this study. The diagnosis of PQ poisoning is based on the clinical history and the test of PQ concentration of blood and urine. After admission, clinical assessment and treatment of all patients were guided by the “Qilu scheme,” which was established in the Department of Poisoning and Occupational Disease at Qilu Hospital of Shandong University. 10 Severity of paraquat poisoning is assessed by urine paraquat concentrations and extent of organ injury. Gastrointestinal lavage was given immediately on admission, and other treatments included high doses of steroids, blood perfusion, protection of the liver, kidneys, and gastrointestinal mucosa. According to Qilu scheme, the steroid pulse therapy was given from the first day to the 21st day after admission or continued until the death of the patient. Activated carbon (30 g) and montmorillonite powder (30 g) with sufficient mannitol were administered after gastric lavage or after hospitalization. Patients are encouraged to autonomous eating in diet in our department unless there is gastrointestinal bleeding or severe gastrointestinal discomfort. A retrospective study was made on the patients with PQ poisoning combined with EDS. The patient’s case sheets and treatment charts were reviewed and medical data were recorded, including age, gender, toxic dose, treatment, clinical presentation, and complications. Additionally, we provide a simple review of related literature and summarize relevant treatment and preventive measures (Figure 1).

Flow chart of the study showing the design and the data analysis.
Statistical analysis
Continuous variables were presented as means ± standard deviation (SD). Independent sample t-test was used for comparison between groups. Data was analysed using SPSS software version 18.0 (SPSS, IL, USA), statistical significance was set at P<0.05.
Results
During the 5 years period, fifteen patients with oral PQ poisoning were complicated with EDS, which included six (40%) male and nine (60%) female patients, with a mean age of 32.9 years (32.9±11.4). The volumes of oral PQ ranges from 50 to 100 ml (a 20% wt/vol formulation), and the esophageal mucosa was stripped mainly in 3–8 days after poisoning through oral ingestion, with a mean time of 4.7 days (4.7 ± 1.8). The demographic and clinical variables of patients are shown in Tables 1 and 2. In death group, most patients demonstrate features of the multiple organ failure when the esophageal mucosal stripping happened, including lung injury, renal failure, and hepatic failure. nearly all patients have physical problems with swallowing before the intramural esophageal dissection occurred, other symptoms included sore throat or dysphagia (100%), nausea and vomiting (86.7%), heartburn or upper abdominal pain (73.3%), hematemesis (60%), abdominal distension (20%) and cough frequently (6.7%), two cases have mild gastrointestinal symptoms.
The demographic and laboratory parameters of EDS associated with PQ poisoning.

Continuous variables are presented as mean ± standard deviation.
Fisher exact probabilities test.
The demographic and clinical features of EDS associated with PQ poisoning.

PQ: paraquat.
The stripped of the esophagus were different shapes and sizes, and Among them, nine cases were tubular (60%) and six cases were irregular (40%) in shape (Figures 2 and 3). Pathological examination showed extensive injury of digestive tract epithelium, necrosis, hemorrhage and inflammatory reaction of epithelial tissue were obvious. Two typical histopathological examination of specimen in Figures 4 and 5. Most (80%) of patients died within few hours to days after ingested PQ and leading to EDS. A surviving case (No. 8) was followed up for 40 days without esophageal stricture whose esophageal barium swallow radiographs shown in Figure 6.
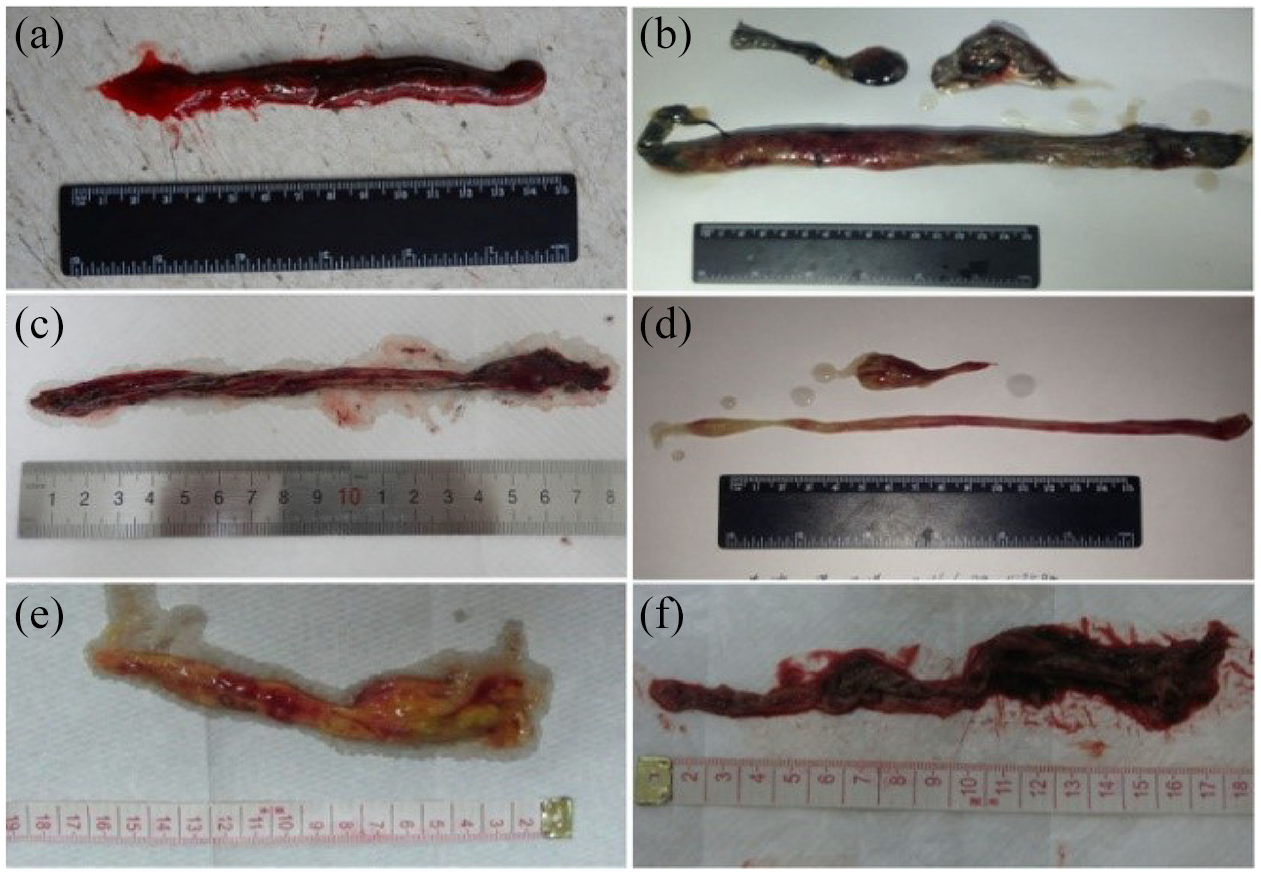
The vomited substance of some patients is almost tubular or strip-like with the size of 1 × 13 cm2 (a), 1.5 × 23 cm2 (b), 0.8 × 18 cm2 (c), 0.8 × 20 cm2 (d), 0.8 × 15 cm2 (e) and 1 × 17 cm2 (f).
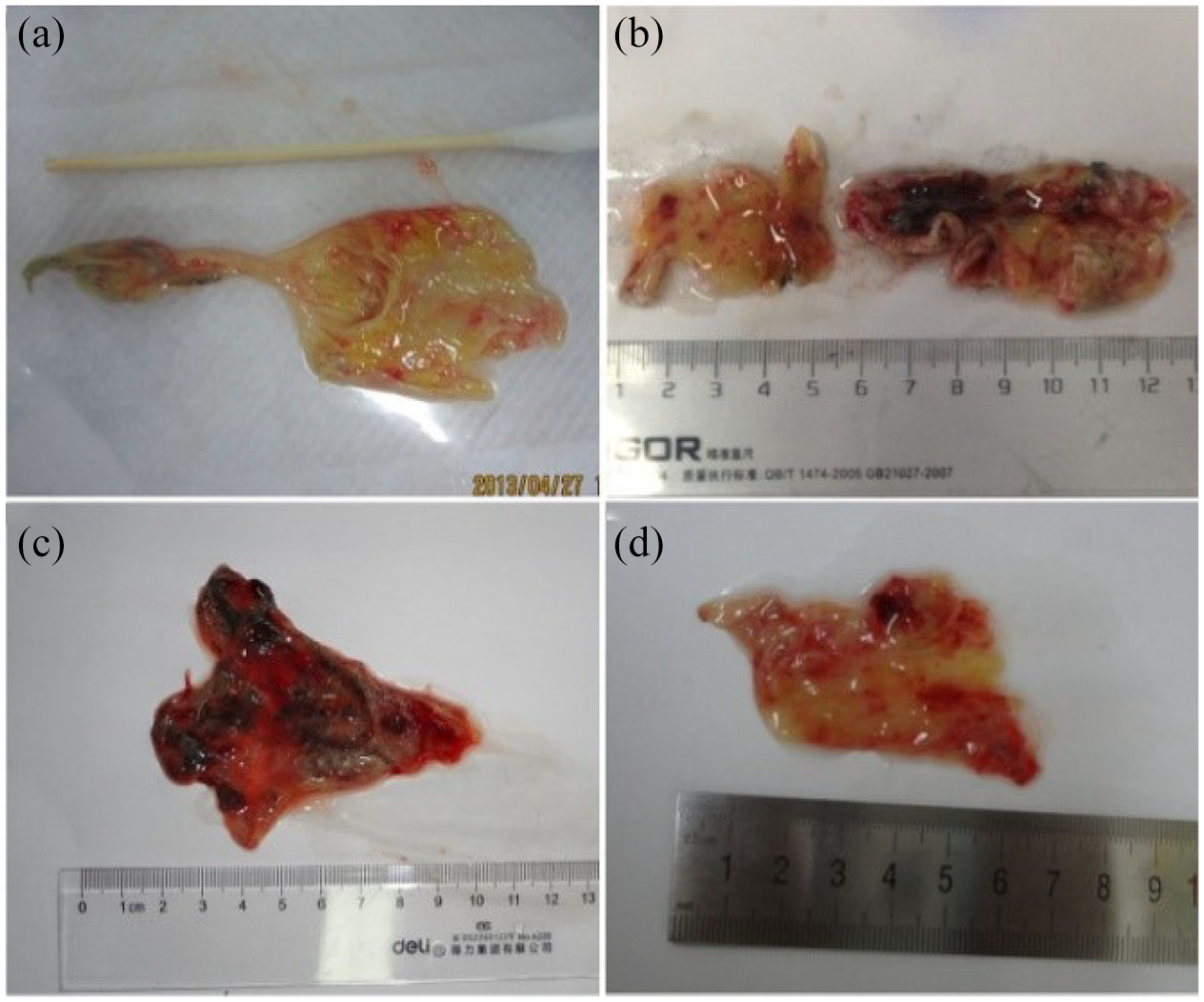
Some patients vomited large fragments of esophageal mucosa in irregular shapes (a–d).

The histopathological lesion to capture the laceration between the esophageal mucosa and submucosa, as in this view. Many nuclei have become undergone karorrhexis (fragmentation) and karyolysis (dissolution), the cytoplasm and cell borders are not recognizable: (a–c) H&E ×100, (d) H&E ×400, and (b) histopathological examination of the esophageal mucosa, showing separation at the suprabasal level of epithelium.

Pathological mechanism showed that the area of lesions is very wide, from mucosa to muscularis: (a) H&E ×40 and (b–d) H&E ×400.

The X-ray barium meal signs of the terminal injury without esophagitis stenosis (a and b).
Discussion
Superficial exfoliative esophagitis (EDS) is a rare but benign disease characterized by the formation of esophageal casts and the shedding of large segments of the inner layer of the esophageal mucosa found by endoscopy. 11 Although EDS has been reported to be associated with serious diseases and certain drugs, there are few reports about of EDS caused by transoral PQ poisoning.
The causes of EDS include medications, candidiasis, skin conditions, chemical irritants, physical trauma, and esophageal structure.12,13 In addition, EDS may also be caused by ingestion of hot drinks, alcohol or spicy foods, exposure to corrosive substances, and mechanical or iatrogenic trauma such as strong vomiting, esophageal sclerotherapy, and nasogastric tube intubation. 14 Common symptoms are dysphagia, swallowing pain, chest pain, heartburn, bloating, and nausea or vomiting. The lesions are mainly found by endoscopy, but hematemesis or vomiting esophageal casts occurs rarely.8,15
PQ poisoning through oral ingestion is common in China. Severe poisoning can lead to multiple organ failure, including pulmonary edema or fibrosis, liver failure, renal failure, cardiotoxicity, leading to high mortality. Chaudhuri et al. 16 reported a fatal case of paraquat poisoning complicated with bilateral spontaneous pneumothorax secondary to lung injury, also called the “Daisley Barton Syndrome”. Adejumo et al. 17 reported a case of paraquat poisoning complicated with delayed AKI, which indicating of the importance of monitoring renal function. Somu et al. 18 reported a 41-year-old male accidentally consumed paraquat, subsequently developed an esophageal injury, and finally succumbed to lung fibrosis. Yano and Kinjo 19 reported a case of paraquat poisoning complicated with swelling of the epiglottis and corrosive esophageal injury. A recent study revealed that among paraquat patients, those with respiratory distress had the highest mortality, and dose of poison, blood sugar level, and serum aspartate transaminase are significant predictors for mortality. 20
In our hospital, all patients are usually treated using a method known as the “Qilu scheme” (called the Qilu Plan). 10 Some patients can survive by comprehensive treatment. Also, we have noticed that some transoral PQ poisoning can cause severe digestive tract damage. However, it is a rare phenomenon that patients with acute transoral PQ poisoning spit out esophageal mucosal casts.
Fifteen cases of EDS in patients with transoral PQ poisoning have similar signs and symptoms, including nausea and vomiting, swallowing difficulties, ulcer sores and sore throat, non-cardiac chest pain. Rarely, it may be accompanied by snoring, bloating, and one case complicated by persistent cough. Usually after acute vomiting, the patients with oral PQ poisoning vomit esophageal mucosal cast, and two patients vomit esophageal mucosal cast suddenly after mild gastrointestinal symptoms. As reported in the literature, patients can develop esophageal mucosal casts, which can eliminate the esophageal lumen and cause obstructive symptoms. 2 Our clinical observation found that gastric lavage was not carried out in a timely and completely manner in almost transoral PQ poisoning patients with EDS, and in addition to different sizes and shapes of esophageal mucosal debris, esophageal mucosal desquamation mainly occurred within 3–8 days in patients with oral PQ poisoning, accompanied by a certain degree of bleeding. EDS can occur at any segment of upper gastrointestinal, such as pharyngeal, esophagus and even in fundus of stomach, more in esophagus, which means they come in many different shapes and length.
The histological features of necrotic tissue in patients with vomiting have important reference value in this study. The vomited necrotic tissue in poisoned patients was examined pathologically to determine the condition of the esophageal mucosa. The esophageal mucosal epithelium is extensively destroyed according to the general and histopathological examination of the vomit specimen. Epithelial tissue necrosis, bleeding and inflammatory response are obvious and are considered to be the pathological basis of EDS. The necrotic tissue has been confirmed by pathological examination and has different clinical and pathological characteristics. Red blood cells and inflammatory cells can be observed. It has been reported that esophageal biopsy revealed a “two-toned” appearance in intact squamous mucosal debris, which manifested as superficial epithelial necrosis with parakeratosis.21,22 Our observations are not completely consistent with the above literature. The pathological changes of esophageal mucosa are mainly showed the presence of cell necrosis and severe inflammatory reaction (Figure 3), which is relatively common in patients with EDS, indicating the presence of cell necrosis and a severe inflammatory response, suggesting that the esophageal mucosa is separated from the lamina propria. The peeling layer is located in the lamina propria below the epithelium. In some patients, the esophagus is severely damaged by PQ, and pathological examination shows that the lesion extends from the mucosa to the muscular layer. Figure 4 shows the presence of a hematoma form and a severe inflammatory response in the esophageal muscle layers, and the lesions extend from the muscle layer to the submucosal site, stripping the esophageal mucosa.
There are many reasons for the pathogenesis of EDS caused by PQ poisoning, and local corrosion is one of the main reasons. In addition, treatments that induce vomiting, lavage or the use of neutralizers have the potential to further damage the mucosa. 23
Frequent nausea, vomiting, intractable hiccups, as well as bloating, difficulty swallowing and eating problems can aggravate damage to the digestive tract and cause mucosal damage. Reactive oxygen species (ROS) produced by PQ poisoning has an important influence on the immune system and may lead to apoptosis, 24 which may be one of the reasons for the deterioration of secondary esophageal injury. High-dose corticosteroids in the treatment of PQ poisoning may be another important risk factor for esophageal mucosal injury. For example, methylprednisolone is widely used as a treatment for PQ poisoning, and it also increases the risk of gastrointestinal mucosal damage. A study has shown that corticosteroids are associated with non-ulcer, non-variceal gastrointestinal bleeding. 24 Finally, under the action of external force, the esophageal mucosa is stripped. Unfortunately, this study was retrospective, and all patients did not undergo endoscopy. The diagnosis of EDS mostly depends on endoscopy, but the final diagnosis depends on esophageal biopsy. 25
It seems that if routine upper gastrointestinal endoscopy is performed on patients with PQ poisoning, it may indicate that esophageal involvement is more common than current findings, and the actual incidence of EDS in PQ poisoning patients may be higher.
In fact, endoscopy is generally not considered as a part of the routine assessment of PQ poisoning patients, and there are some worried by physicians to perform endoscopy on paraquat patients due to erosion of the oral and digestive tract mucosa in most patients. Regarding these risks, endoscopy may not be used in all patients with oral paraquat poisoning, so some patients with EDS may be missed.
In general, EDS seems to be a benign accidental finding with no persistent esophageal lesions. 11 However, the long-term prognosis of most patients with transoral PQ poisoning is not very satisfied, most patients die of multiple organ failures, mainly a consequence of respiratory failure from either pulmonary edema in the first few days or extensive alveolar damage and fibrotic changes in later stages. 26 Patients with transoral PQ poisoning have different survival times and are mainly associated with vital organs damage. In addition, some patients have varying degrees of improvement in gastrointestinal symptoms as the disease progresses, and some patients may survive more than half a month after exfoliation of the esophageal mucosa. In surviving transoral PQ poisoning patients, vital organs damage, such as lung, liver, and kidney function damage, is mild during the course of the disease. Therefore, intramural esophageal dissection may not be the main cause of death.
Currently, there is no published information to guide the treatment of esophageal mucosal detachment. For the treatment of EDS associated with paraquat poisoning, in addition to a reasonable diet, timely and complete gastric lavage, mucosal protective agents and antacids may be beneficial. Proton pump inhibitors may be considered when using steroids and acute exfoliation and erosion of the patient’s esophageal mucosa. Natural product extracts have been proved to ameliorate paraquat-induced lung and other organ injuries. 27 In vivo study, Xuebijing, an intravenous traditional Chinese medicine, is protective against paraquat-induced lung injury by attenuating oxidative stress responses and organ inflammation. 28 Nanostructured lipid carriers had a significant effect of preventing PQ-induced toxicity. 29 However, research associated with treatment for paraquat-induced EDS was still rarely reported.
Conclusion
For PQ poisoning complicated with EDS, the etiology of EDS is not completely clear, and the standardized treatment protocol is not available. By summarizing the features of the patients, this study increased awareness of EDS associated with PQ poisoning, indicating that it is significant to do basic research and explore the targeted treatment of EDS associated with PQ poisoning.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Shandong University Qilu Hospital of Ethics Committee (KYLL-2018-166).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National key clinical speciality (no.2012650) and the Taishan Scholar Program of Shandong Province (no. ts20130911).
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
China Clinical Trial Registry: CHICTR1800015288.
