Abstract
No consistent results have been reached on the relationship of interleukin-1 (IL-1) rs1800587 polymorphism with the susceptibility to rheumatoid arthritis (RA) so far. Therefore, the current meta-analysis was designed to thoroughly review relevant studies, in order to examine the relationship of IL-1A rs1800587 polymorphism with RA risk. Electronic databases were retrieved for literature regarding the relationship between IL-1A rs1800587 polymorphism and RA vulnerability according to the inclusion and exclusion criteria. Stata 12.0 software was adopted to examine the enrolled literature. Meanwhile, odds ratio (OR) and corresponding 95% confidence intervals (95% CI) were used to evaluate the association. A total of seven case-control researches (3267 patients and 2960 healthy controls) were eventually enrolled into the current meta-analysis. Our data indicated no correlation of IL-1A rs1800587 polymorphism with RA risk (TT vs CC: OR = 0.90, 95% CI = 0.73–1.11; TC vs CC: OR = 1.02, 95% CI = 0.78–1.34; Dominant model: OR = 1.04, 95% CI = 0.80–1.35; Recessive model: OR = 1.91, 95% CI = 0.74–1.12). Similarly, no association was found in subgroup analysis stratified by ethnicity. Our findings indicated no relationship between IL-1A rs1800587 polymorphism and RA vulnerability.
Introduction
Rheumatoid arthritis (RA), a common chronic systemic inflammatory disorder, can cause destruction of joints. 1 However, the etiology of RA remains largely unclear at present. Multiple studies have identified proinflammatory cytokines to be involved in bone destruction and joint inflammation pathophysiology in RA. 2 In addition, the imbalanced activities between anti- and pro-inflammatory cytokines have also been reported to trigger and aggravate tissue destruction, chronic inflammation and autoimmune responses in joints during RA.
All interleukin-1 (IL-1) family members exert vital roles in regulating both adaptive and innate immunity, which are involved in the pathogenesis of autoinflammatory, autoimmune, and infectious disorders. In addition, they systemically mediate inflammatory responses, including leukocyte activation, acute-phase proteins in liver and fever. 3 Moreover, IL-1 has been identified as an important mediator for tissue destruction and inflammation during RA, with important significance in the pathogenesis of RA. 4 Knockdown of IL-1 mitigates arthritis in several mouse models, and consistently, clinical trials have suggested that IL-1 knockdown can ameliorate RA in human. 5 The IL-1 family plays a role as the crucial inflammatory response mediator, which is constituted by one IL-1 receptor antagonist (IL-1Ra) as well as two agonists (IL-1a/IL-1b).
The IL-1 gene cluster is localized within 430 kb region on chromosome 2 (2q13-21), consisting of IL-1RN, IL-1A, and IL-1B and (encoding IL-1Ra, IL-1a, and IL-1b, respectively) genes. 6 The IL-1A gene is localized on the long arm of chromosome 2 (2q14), harbors a common polymorphism (rs1800587) on the 5′-regulatory region: C to T transition at position −889, which is within a transcriptional regulatory region. Additionally, previous researches have shown the TT genotype was reported to increase promoter activity, producing increased levels of IL-1A mRNA and IL-1A, compared with those of CC genotype.7,8 Several meta-analyses have reported an association between rs1800587 polymorphism and the presence of various autoimmune diseases, including systemic lupus erythematosus, multiple sclerosis, and Graves’ disease.9–11
Recent researches has been conducted to assess the relationship of IL-1A rs1800587 polymorphism with RA risk. Nonetheless, no consistent findings have been obtained among existing studies. Meta-analysis is an efficient approach to detect the relationship that may be masked among studies with small sample size, particularly in those that evaluate the unusual allele frequency polymorphism. Herein, in this current meta-analysis, we aimed at examining the relationship of rs1800587 polymorphism with RA risk based on relevant case-control studies.
Methods
Retrieval protocol
The meta-analysis was conducted independently per the PRISMA (preferred reporting items for systematic reviews and meta-analyses) guidelines. 12 We systematically searched the electronic databases, including China National Knowledge Infrastructure (CNKI), Embase and PubMed for identification of the potentially eligible studies using the following key words, “RA,”“IL-1,”“rheumatoid arthritis,” and “interleukin-1.” The study language was restricted to Chinese and English (up to February 2021). Additionally, we manually retrieved the related references in those articles so as not to omit any eligible studies. The protocol has been registered in PROSPERO (ID: CRD42021235813).
Criteria for inclusion and exclusion
The inclusion criteria of enrolling articles in current study were shown as follows: (1) relevant case controlled researches of RA cases and normal controls; (2) articles regarding the relationships of IL-1A rs1800587 polymorphism with RA susceptibility; and (3) researches including enough genotype information for extraction. The exclusion criteria were as follow: (1) not case-controlled researches; (2) case reports, reviews, or meta-analyses; and (3) studies with insufficient original data. Abstracts and unpublished reports were not considered. Investigations in subjects with family history or cancer-prone disposition were also excluded.
Information extraction
Two investigators independently evaluate the possibly eligible articles in accordance with the above exclusion and inclusion criteria. Moreover, these data were retrieved from every research: first author, country of origin, ethnicity, year of publication, genotyping approaches, number of cases, and normal controls and polymorphisms. Conflicting data were discussed with all authors, and missing data were requested by e-mail. We also used the Newcastle-Ottawa Scale (NOS) system to assess the study quality and generate an NOS score. An NOS score <5 means the study was poor quality, and such studies were excluded.
Quality evaluation
The authors used the methodological quality of each included article using the Newcastle-Ottawa quality assessment scale (NOS). An ultimate score of 6 stars or more was regarded as high-quality study.
Statistical analysis
The deviation from HWE for each genotype distribution in the control group was assessed by Fisher exact test. Odds ratio (OR) along with 95% confidence interval (95% CI) was utilized to evaluate the strength of correlation of IL-1A rs1800587 polymorphism with RA risk under a homozygote comparison (TT vs CC), a heterozygote comparison (TC vs CC), a dominant model (TT + TC vs CC) and a recessive mode (TT vs CC + TC). Additionally, I2 test was employed to assess the heterogeneity in the enrolled researches. Random effects model was adopted in the case of I2 > 50% (suggesting heterogeneity), otherwise the fixed effects model was performed. We also performed the subgroup analyses according to the characteristics of ethnicity. Afterwards, sensitivity analysis was performed by sequentially eliminating one single study, showing suspect on one study of excessive sensitivity because the omission of this specific study yielded to the estimation beyond the 95% CI of the pooled analysis. Finally, possible publication bias was evaluated by visualizing the Begg’s funnel plot. STATA version 12.0 was utilized for statistical analysis. Power analysis was performed using the statistical program G*Power (http://www.gpower.hhu.de/).
Results
Characteristics of included studies
After systematic retrieval from database and manual review of reference lists in all eligible articles, altogether 117 publications were preliminarily included. Finally, seven articles were enrolled in our meta-analysis, including 3267 RA cases and 2960 control subjects.13–19 All studies were published from 1995 to 2017. According to the NOS for case-control studies, the results of the quality assessment are shown in Table 1. And all studies were defined as high-quality. The detailed information of the seven included papers was summarized in Figure 1 and Table 1. Of these, four studies focused on Caucasians, one on Asians and two on Mestizos. The genotype distributions in healthy controls from each research were consistent to the HWE. The statistical powers of these seven studies ranged from 29.3% to 58%. None of the studies had a statistical power that exceeded 80%.
Characteristics of the included studies for meta-analysis.

HWE: Hardy–Weinberg equilibrium; PB: population-based.

The flowchart of the included studies in the meta-analysis.
Overall and subgroups analyses
The major findings in this study and heterogeneity were listed in Table 2. Overall, IL-1A rs1800587 polymorphism was not significantly correlated to RA risk under all genetic models (Figure 2: TT vs CC: OR = 0.90, 95% CI = 0.73–1.11; TC vs CC: OR = 1.02, 95% CI = 0.78–1.34; Dominant model: OR = 1.04, 95% CI = 0.80–1.35; Recessive model: OR = 1.91, 95% CI = 0.74–1.12).
Summary ORs and 95% CI of IL-1A rs1800587 polymorphism with RA risk.
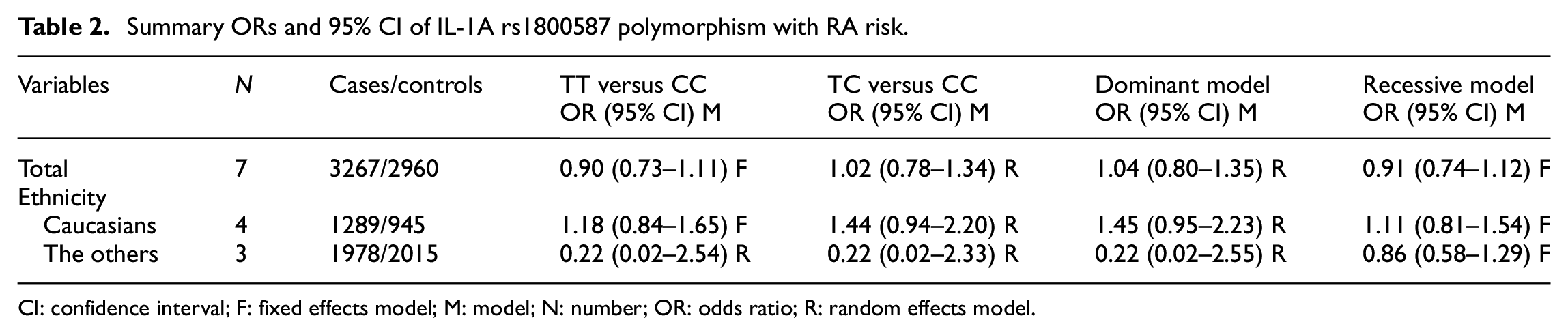
CI: confidence interval; F: fixed effects model; M: model; N: number; OR: odds ratio; R: random effects model.
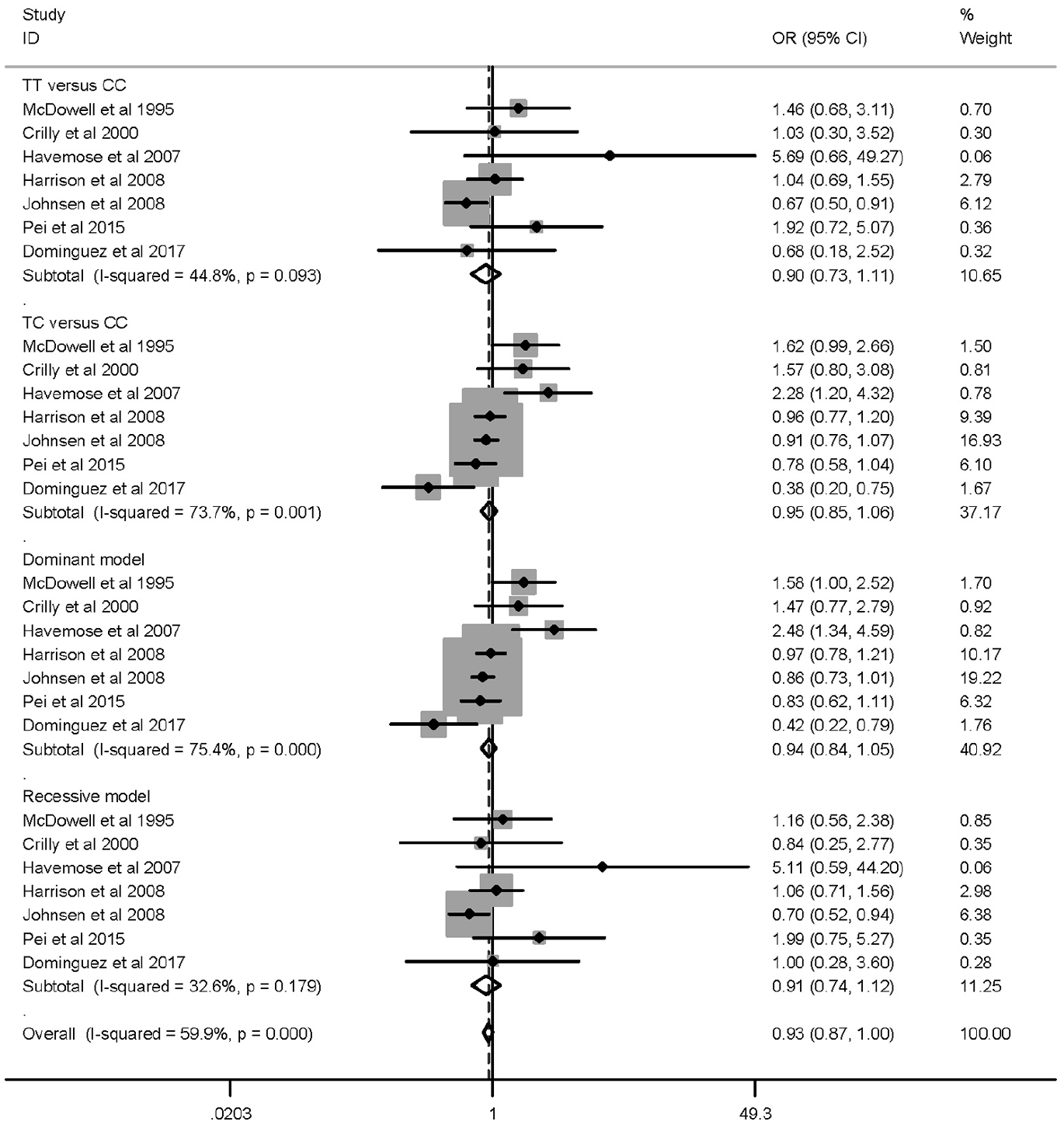
Forest plot for meta-analysis of the association between the IL-1A rs1800587 and RA risk.
Subgroup analysis stratified by ethnicity also revealed no significant relationship of IL-1A rs1800587 polymorphism with RA risk in either Caucasian population or other populations. Sensitivity analysis was additionally performed to determine whether the outcomes were stable via the sequential deletion of a single research, which demonstrated that the pooled ORs were not significantly influenced by any individual study (Figure 3).

Sensitivity analysis of the IL-1A rs1800587 polymorphism.
Publication bias
The possible publication bias was evaluated by funnel plot and Begg’s test, which showed no evidence of publication bias (Figure 4), indicating low publication bias in our meta-analysis.

Begg’s funnel plot analysis of the IL-1A rs1800587 polymorphism.
Discussion
Similar to the majority of autoimmune disorders, both genetic and environmental factors contribute to the pathogenesis and severity of RA. Many studies have been carried out to examine the relationship of gene polymorphisms with the risk of RA. IL-1, one of the multi-factorial cytokines, is suggested to exert a vital role in the pathogenesis of RA. 20 Genetic variant is possibly and vitally involved in the susceptibility of RA, but the underlying mechanism is quite complicated. As indicated in several studies, IL-1A rs1800587 polymorphism is related to RA susceptibility; however, without consistent results. Thus, to provide the reliable and comprehensive conclusions, the current study was carried out to enhance statistical power to acquire more creditable results.
According to our meta-analysis, the data indicated that IL-1A rs1800587 polymorphism had no relationship with RA risk. In consideration of the different environmental factors, ethnicity-specific subgroup analysis was further conducted, similarly revealing no such association in either Caucasian population or other population. Due to the contribution of various genetic products and environmental factors to RA risk, we should concentrate on gene-gene and gene-environment interactions. 21 To be specific, the association of between IL-1A rs1800587 polymorphism with other candidate genes have investigated. Dominguez-Perez et al. 18 has demonstrated that IL-1A rs1800587 polymorphism is not related to RA risk, however, the IL-1A rs1800587(C)/IL-1b511A gene-gene polymorphism combination has an association with RA predisposition. Besides, a number of environmental factors, like age, are found to increase the RA susceptibility. Therefore, these variables must be examined in subsequent articles, regardless of the difficulties in assessing environmental exposures and study design.22,23
Some limitations exist in our meta-analysis. To begin with, we only enrolled researches in Chinese and English, thus, some potentially eligible researches published in other languages might be omitted. Secondly, every eligible case-control study assessing IL-1A rs1800587 polymorphism was enrolled and related data were extracted. However, the relatively small sample size in this study still limited its statistical power for attaining the definitive conclusion. Thirdly, similar to meta-analysis on SNPs, our outcomes were mainly based on unadjusted estimates, which were also restricted by the absence of original information from eligible researches. Hence, the evaluation of effects of gene-gene or gene–environment interactions was restricted. Fourthly, it is worth analyzing the combined influence of different SNPs or cytokine genes, when more casecontrol studies become available.
To sum up, the current meta-analysis indicates IL-1A rs1800587 polymorphism is not related to RA vulnerability. Considering that IL-1 is not currently considered one of the central cytokines in RA’s pathogenesis or in the biological therapies for RA treatment. Further studies on this polymorphism are needed. Due to the advantages and drawbacks in our study, large-scale, delicately-designed studies are required to validate these outcomes by considering both gene-gene and gene-environment interactions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
