Abstract
Malakoplakia is a rare inflammatory condition characterized by the accumulation of granulomatous masses due to the incomplete destruction of infectious pathogens by monocytes and macrophages. While it commonly affects the genitourinary tract, pulmonary malakoplakia is infrequent and often associated with immunocompromised states. We present the case of a woman in her early 40s with rheumatoid arthritis who developed localized pulmonary malakoplakia in the airway. Initially presenting with constitutional symptoms and respiratory distress, she underwent a diagnostic evaluation revealing an endobronchial mass with neoplastic characteristics. Histopathological examination confirmed the presence of Michaelis–Gutmann bodies, pathognomonic for malakoplakia. Despite initial improvement postresection, the patient experienced disease recurrence 1 year later. Pulmonary malakoplakia, although rare, poses diagnostic challenges due to its resemblance to malignant lesions. Treatment typically involves antibiotic therapy, low-dose steroids, and localized resections. Differential diagnoses include primary or secondary malignancies, necessitating histopathological evaluation for accurate diagnosis. Malakoplakia should be considered in immunocompromised individuals presenting with pulmonary nodules or masses, especially without malignancy risk factors.
Introduction
Malakoplakia is a rare chronic inflammatory disorder characterized by defective intracellular killing of bacteria by macrophages, leading to accumulation of granulomatous masses that may mimic malignant tumors. 1 This phenomenon is attributed to inadequate phagocytosis related to a lysosomal defect. Due to this alteration, bacterial residues consisting of calcium and iron deposits, known as Michaelis–Gutmann bodies (M-G bodies), are generated, which are pathognomonic for this pathology.2,3
Due to the rarity of the disease and the similarity of this condition to malignant neoplasms, cytological or histopathological examinations are required to achieve the correct diagnosis. 4 This disease is most commonly observed in immunocompromised patients, especially those with human immunodeficiency virus infection, as well as in patients using immunosuppressants. The most common manifestations occur in the genitourinary, gastrointestinal, and cutaneous systems, with the former being the most affected, although it can also affect other systems. 5
Pulmonary malakoplakia is exceedingly rare, with most reported cases associated with Rhodococcus equi infections, especially in immunosuppressed hosts. 6 Therapeutic recommendations include prolonged antibiotic regimens with antibiotics like macrolides, fluoroquinolones, and rifampicin. Additionally, the possibility of surgical resection interventions is considered, if necessary, despite the absence of a standardized treatment guideline for this entity.1,7
Here, we present a rare case of endobronchial malakoplakia in a young woman with rheumatoid arthritis on immunosuppressive therapy. The lesion was localized to the right main bronchus and the right upper lobe bronchus. We describe her clinical presentation, diagnostic work-up, and disease course, including recurrence 1 year after surgical resection.
Case presentation
We present the case of a woman in her early 40s with a 1-year history of symptoms characterized by an unintentional weight loss of around 7 kg. Initially, she had a sporadic dry cough, for which she used loratadine and salbutamol, experiencing partial improvement in symptoms. However, a week before the medical evaluation, her condition worsened, manifesting frequent, intense, and irritating cough accompanied by chills, nocturnal diaphoresis, unquantified fever, pleuritic chest pain, and intermittent episodes of vomiting after coughing bouts.
The patient had a history of vitiligo and seropositive rheumatoid arthritis with a 6-year duration, with a positive rheumatoid factor of 279 IU/mL (normal range lower than 14) and negative antinuclear antibodies. For her underlying condition, the patient had been on chronic immunosuppressive therapy for the past 4 years, which included methotrexate at a dose of 20 mg weekly, folic acid, and sulfasalazine at a dose of 1.5g per day. She had no history of allergies, alcohol consumption, tobacco exposure, or transfusions. Initially, the patient went to another emergency service with a fever of 38.2°C, where she received treatment with nonsteroidal anti-inflammatory drugs, short-acting inhaled beta-agonists, and oral steroids. The specific drug names and doses were not documented in the medical record. However, due to persistent symptoms, she presented to Fundación Valle del Lili at the beginning of 2021 for medical evaluation.
The patient appeared fatigued, with respiratory distress marked by accessory muscle use and frequent coughing. Vital signs revealed tachycardia (145 bpm) and tachypnea (30/min), with normal oxygen saturation (98% on room air). Mucous membranes were normal, without cyanosis or jaundice. There were no masses in the neck. Cardiac exam showed a regular tachycardic rhythm without murmurs or rubs. Pulmonary auscultation revealed wheezes, rales at both bases, and decreased breath sounds. Abdominal, neurological, skin, and extremities examinations were normal.
Management for bronchospasm was initiated with salbutamol and ipratropium bromide (salbutamol, four puffs every 20 minutes for 1 hour, then every hour for 2 hours, then every 2 hours, and ipratropium bromide, 40 mcg inhalation every 8 hours), along with intravenous hydrocortisone 50 mg every 6 hours. Additionally, oxygen was administered via nasal cannula at a rate of 1.5 L/min. Four hours after initial management, the patient experienced an improvement in dyspnea and cough, with no chest pain. Follow-up vitals showed improvement: respiratory rate 22, heart rate 108, blood pressure 133/70, SpO₂ 100%.
Blood tests showed mild respiratory alkalosis on arterial gases, with elevated inflammatory markers, including increased C-reactive protein (14.13 mg/dL) and leukocytosis of 15.170 µL, predominantly due to neutrophilia. Polymerase chain reaction testing for COVID-19 was negative. Cardiac enzymes, renal function, and electrolytes were all within normal limits. D-dimer was mildly elevated. Chest X-ray showed normal cardiomediastinal contours, clear lungs and pleura, and no osseous or soft-tissue abnormalities.
Due to the history of pleuritic chest pain with dyspnea, with no significant changes on chest X-ray and a slight elevation of D-dimer, a thromboembolic event was suspected. Therefore, a computed tomography (CT) angiography was performed, which ruled out acute pulmonary embolism. Additionally, following the institutional protocol in place at the time, a sequential venography scan of the pelvis and thighs was conducted simultaneously, ruling out deep vein thrombosis in the thighs. However, the imaging revealed a right mediastinal and hilar mass radiologically suspicious for neoplasm, accompanied by endobronchial involvement of the right main bronchus. The mass showed heterogeneous enhancement with irregular and lobulated contours, enveloping the right main bronchus and the bronchus leading to the ipsilateral upper lobe (Figure 1).

Chest CT scan. (A) and (B) Mediastinal window; (C) and (D) lung window, where a mass with heterogeneous enhancement, irregular lobulated contours, is identified. It is in the subcarinal region, surrounding the right main bronchus and the bronchus leading to the right upper lobe, measuring 45 × 20 × 35 mm. It presents endoluminal representation in the right main bronchus (red arrows). CT: computed tomography.
Due to the history of autoimmune disease, antinuclear antibodies and antineutrophil cytoplasmic antibodies were requested and came back negative. Angiotensin-converting enzyme was normal. The results for HIV antibodies using enzyme-linked immunosorbent assay, cryptococcal antigen immunoassay, urinary histoplasma antigen immunoassay, hepatitis C antibodies, and hepatitis B surface antibody tests were negative.
A bronchoscopy revealed a mass with tumor-like appearance on the anterior wall of the right main bronchus, accompanied by small smooth nodular lesions (Figure 2). Bronchoalveolar lavage cytology showed cells with atypical changes, and some cocci were observed in the Papanicolaou staining (Figure 3). The cytological findings suggested the diagnosis, which was later confirmed by histopathological examination. The endobronchial lesion was resected using an electrocautery loop. Additionally, endobronchial ultrasound with fine needle aspiration was carried out for sampling at right stations 4, 7 subcarinal, and 11, which were negative for malignancy.
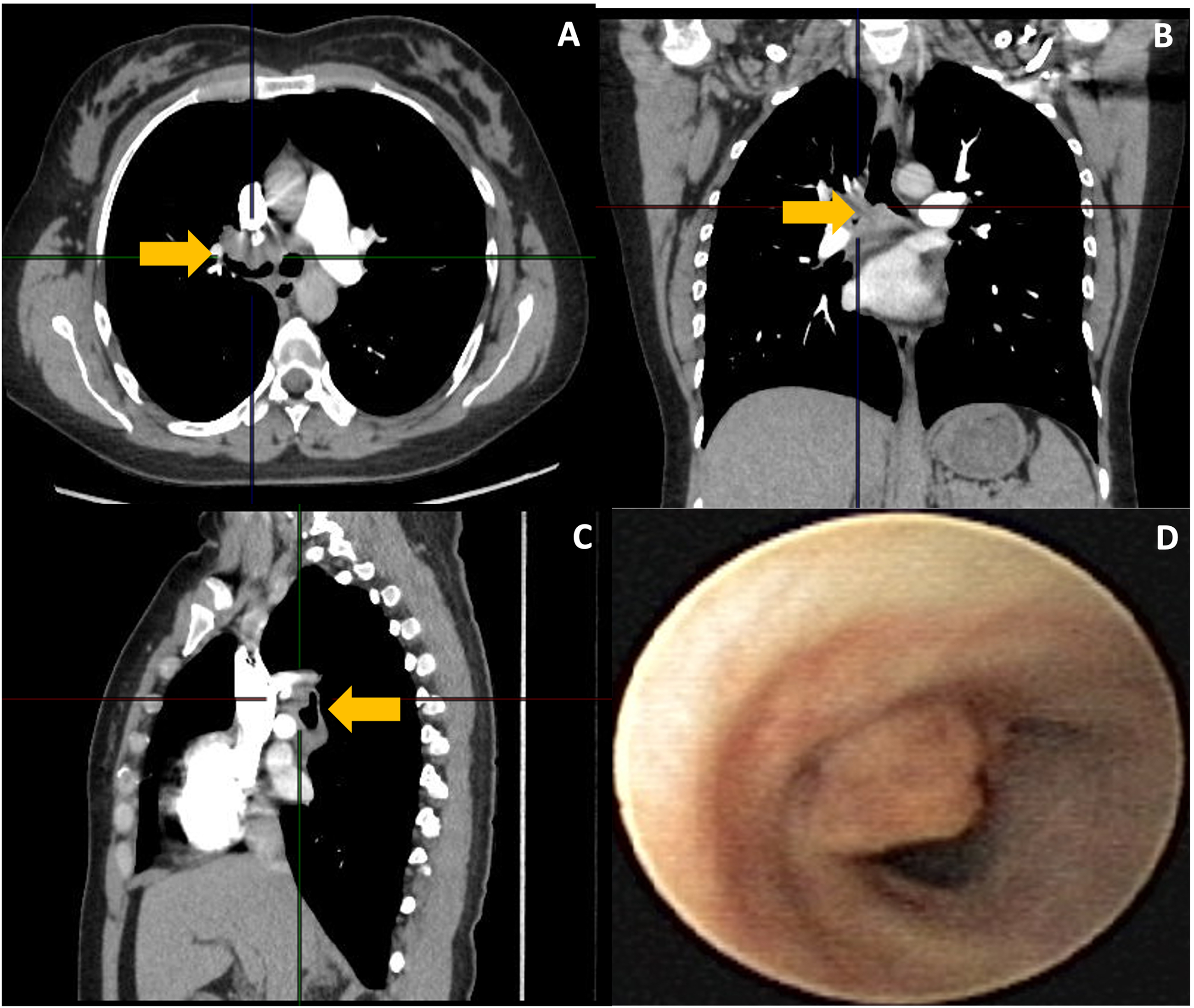
Chest CT scan. (A) Coronal section; (B) coronal section; (C) lateral section showing the mediastinal, subcarinal lesion and its endobronchial representation (yellow arrows); and (D) endoscopic appearance of the irregular nipple-like lesion partially obstructing the lumen of the right main bronchus prior to resection. CT: computed tomography.

Bronchoalveolar lavage, Giemsa staining 40×. Black arrows indicate cell clusters with atypical changes.
Histopathological examination with hematoxylin and eosin (HE) staining revealed aggregates of histiocytes with abundant, granular, eosinophilic cytoplasm (von Hansemann cells), together with M-G bodies (Figure 4). Based on these findings and the suspicion of malakoplakia, periodic acid-Schiff (PAS) and Von Kossa stains were performed. PAS staining demonstrated microgranules within histiocytes, while Von Kossa staining highlighted calcium deposits. The identification of M-G bodies, pathognomonic for malakoplakia, together with the other histological findings, confirmed the diagnosis. Additionally, extensive chronic inflammation with pseudogranuloma formation, but without necrosis, was observed (Figure 5).

Hematoxylin and eosin stain, 40×. Clusters of histiocytes with abundant granular eosinophilic cytoplasm (von Hansemann cells), along with Michaelis–Gutmann bodies (black arrow).

(A) Xanthogranulomatous stroma is observed, with multinucleated giant cells (black arrow) accompanied by lymphocytes, plasma cells, and neutrophil polymorphonuclear cells, with the presence of phagosomes in histiocytes (hematoxylin and eosin staining, 40×). (B) Abundant foamy histiocytes with microgranular cytoplasm (black arrow). (hematoxylin and eosin staining, 40×). (C) Periodic acid-Schiff (PAS) staining, showing microgranules in foamy histiocytes (black arrow) (40×). (D) Von Kossa staining positive for calcium, consistent with Michaelis–Gutmann bodies (black arrow) (40×).
Studies for fungal and acid-fast bacilli detection and slow-growing bacteria were negative both in direct examination and final cultures, and there were no suggestive inclusions of cytomegalovirus in the lavage cytology. After resection, improvement in cough was observed, becoming sporadic. Low-dose steroids were initiated, and baseline treatment for rheumatoid arthritis was continued, as indicated by the rheumatology service, which determined that the patient met criteria to maintain therapy at the time of discharge. The patient was discharged and had periodic outpatient follow ups with rheumatology and pulmonology, with the first follow up conducted 1 month after surgery by the pulmonology service.
One year later, the patient was consulted again due to the recurrence of constitutional symptoms, such as weight loss and general discomfort associated with cough, hoarseness, dyspnea, and posterior chest pain. At the time of consultation, she was still on methotrexate once a week and low-dose steroids, had received two doses of the Sinovac vaccine against COVID-19, and had SARS-CoV-2 infection in December 2021 without requiring hospitalization.
Chest CT revealed a smaller recurrent endobronchial lesion with reduced peribronchial tissue involvement. Bronchoscopy identified a nodular lesion at the distal end of the right intermediate bronchus, raising suspicion of recurrence 1 year after the previous resection. Consequently, a new bronchoscopic resection was performed. The lesion was successfully removed using an electrocautery loop without complications (Figure 6). Histopathological findings were consistent with those of the prior procedure.

Bronchoscopy images. (A) View at the level of the carina showing a smooth vascularized single lesion in the right main bronchus. (B) and (D) Close-up of the lesion in the right main bronchus. (C) Appearance of the distal airway beyond the lesion showing normal bronchi of the right middle and lower lobes. (E) Electrocoagulation process with electrocautery loop. (F) Image showing the forceps extracting the resected lesion.
For a summarized overview of the patient's clinical course, diagnostic work-up, and management across both episodes, please refer to Supplemental Figure 1. For the preparation and publication of this case report, we have de-identified all the patient's details. The patient provided written informed consent to undergo all diagnostic and therapeutic procedures described in this report, as well as signed informed consent for publication of the case, including clinical details and accompanying images. Furthermore, the reporting of this study conforms to CARE guidelines. 8
Discussion
Malakoplakia is an inflammatory disease more commonly observed in immunocompromised individuals than in immunocompetent ones. It typically affects the genitourinary tract, although its presence has been reported in other organ systems, including pulmonary, gastrointestinal, musculoskeletal, lymphatic, integumentary, and nervous systems.1,4
Fewer than 500 cases of malakoplakia have been reported in the literature to date 9 ; it is a rare, noninfectious granulomatous disease characterized by the accumulation of histiocytes containing layered basophilic inclusions, secondary to compromised phagocytic function. These inclusions, known as M-G bodies, are pathognomonic of the disease.10,11 It has been suggested that this condition occurs secondary to a bactericidal defect in macrophages. 1
Risk factors for developing malakoplakia include immunosuppressive therapy, chemotherapy lymphomas, myelodysplastic syndrome, postradiotherapy, diabetes mellitus, alcoholism, autoimmune diseases such as rheumatoid arthritis, and other conditions that have in common the presence of immunosuppression. 12 Malakoplakia typically occurs in the sixth or seventh decade of life. 13
The genitourinary system is the most common site where malakoplakia has been described, accounting for up to 80% of cases, mainly associated with Escherichia coli. However, other opportunistic pathogens are involved due to the immunosuppressive state of these patients.1,9 Other microorganisms that have been described include Klebsiella sp., R. equi, Proteus mirabilis, Pseudomonas aeruginosa, Yersinia sp., Staphylococcus aureus, and fungal infections.14,15
Pulmonary malakoplakia is an uncommon diagnosis, with only a limited number of cases described in the literature worldwide, 4 resulting in fewer than 30 reported cases after the search for related articles. Pulmonary lesions may include nodules, masses, and cavitations, usually associated with R. equi infections.4,16 However, it is rare to identify the germ or achieve its growth in special media. Its location is generally in the parenchyma of the upper lobes. It is even rarer to find malakoplakia located in the airway in the tracheobronchial tree. 16
When presenting in the lung, radiologically, it is characterized by well-defined round solid lesions, single or multiple, without calcifications, easily confused with malignant lesions, abscesses, or infections such as tuberculosis; hence, the importance of evaluation to confirm the etiology and define prognosis and treatment. Treatment is generally conservative, consisting of targeted antibiotic therapy and low-dose steroids; extensive resections are not recommended unless performed to identify the diagnosis and rule out malignancy. In the case of endobronchial presentation, local resections are recommended to ensure airway patency and close monitoring of symptoms, imaging, and bronchoscopy when necessary.1,14
The diagnostic approach to malakoplakia is histopathological and becomes crucial to differentiate this entity from its main differential diagnoses, the most important being primary or secondary malignant lesions. Malakoplakia can present in various forms, such as a polyp, a mass, an ulceration, or may even involve lymph nodes. Its rapid growth can manifest as single or multiple nodules. 17
Microscopic characteristics pose a broad differential diagnosis. There are numerous conditions with histiocytic proliferation, such as xanthoma, Crohn's disease, granular cell tumor, sarcoidosis, tuberculosis, leprosy, leishmaniasis, granulomas due to reactions to drugs or fungal organisms, infection with Mycobacterium avium, melanosis coli, Langerhans cell histiocytosis, Chediak–Higashi syndrome, and metabolic storage diseases.1,17 However, the presence of M-G bodies is definite for malakoplakia. These bodies stain positive with PAS reagent, Von Kossa reaction for calcium, and Perl reaction with ferricyanide for ferric iron, these stains are essential to differentiate malakoplakia from other granulomatous or histiocytic disorders.10,18
In our patient, the finding of pulmonary malakoplakia may be related to her underlying condition, rheumatoid arthritis, a chronic autoimmune disorder associated with immunodeficiency, together with her long-term use of immunosuppressive therapy. 19 Malakoplakia has been commonly reported in immunosuppressed individuals, including those with lung transplantation, HIV infection, tuberculosis, and other immunosuppressive conditions.2,4–6,11–13,20,21 However, only a few cases have been described in patients with seropositive rheumatoid arthritis, with presentations ranging from esophageal malakoplakia 22 to soft tissue malakoplakia of the neck. 23 To our knowledge, endobronchial involvement has not previously been reported, making this the first documented case of endobronchial malakoplakia in a patient with rheumatoid arthritis, notable for its recurrence during clinical follow up.
Regarding treatment, despite the lack of standardized treatment guideline for this entity, therapeutic recommendations include prolonged antibiotic regimens, low-dose steroids, and, in some cases, surgical resection.1,7 Furthermore, given the clinical outcome of our patient, in whom recurrence of the disease was observed during follow up, we emphasize the vital importance of strict clinical and imaging surveillance in patients with this condition.
Despite the rarity of this diagnosis, it is essential to consider it, particularly in young, immunosuppressed individuals without conventional risk factors for malignancy. 4 Given its close clinical and radiological resemblance to neoplasia, careful assessment of risk factors is crucial when establishing the differential diagnosis. In the context of our patient, bronchoscopy and biopsy played a fundamental role confirming the diagnosis. Although our study has limitations such as relying on medical records for retrospective analysis, it contributes to a better understanding of the behavior of this entity.
Conclusion
Malakoplakia is a rare inflammatory disease most frequently observed in immunocompromised individuals, classically affecting the genitourinary tract but occasionally involving the lungs. Its diagnosis is challenging and requires histopathological confirmation to differentiate it from malignant lesions. Management typically combines antibiotic therapy, low-dose steroids, and localized resections. Our case underscores the importance of considering malakoplakia in the differential diagnosis of endobronchial lesions in immunosuppressed patients, particularly when histological or radiological findings are atypical for malignancy. Greater awareness of this entity may facilitate timely recognition and appropriate treatment.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251411205 - Supplemental material for Endobronchial malakoplakia in a young woman with rheumatoid arthritis: A case report
Supplemental material, sj-docx-1-sci-10.1177_00368504251411205 for Endobronchial malakoplakia in a young woman with rheumatoid arthritis: A case report by Liliana Fernández-Trujillo, Luz F Sua, Ana Isabel Castrillón and Ciro D Ibarra in Science Progress
Footnotes
Abbreviations
Acknowledgements
We acknowledge the use of ChatGPT (OpenAI, San Francisco, CA, USA) exclusively for English language improvement. The tool was not used for idea generation, data analysis, interpretation, or original drafting of any part of the manuscript.
Ethical considerations
This report was prepared in accordance with the ethical standards of the Institutional Ethics Committee and the 1964 Helsinki Declaration. Ethical approval for publication was granted by the Ethics Committee in Biomedical Research of Fundación Valle del Lili, Cali, Colombia (approval no. 146-2024; 23 February 2024).
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review upon request.
Author contributions
All authors have read and approved the manuscript and significantly contributed to this article. LFT, LFA, and AIC were involved in conception and design, literature review, manuscript writing correction, and final approval of the manuscript; CDI in literature review, manuscript writing correction, and final approval of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data related to our study has not been deposited in a publicly available repository. Nevertheless, all data and materials will be available upon request by contacting the correspondence author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
