Abstract
Neonatal mitochondrial disease is occasionally observed in patients with intraventricular cysts in the brain. Atypical morphology is rarely seen in these cysts. Here, we report a case of neonatal lethal mitochondrial disease with IBA57 gene mutation. We have, for the first time, described a subependymal pseudocyst (SEPC) with a fluctuating membrane. Our findings suggest that SEPCs with fluctuating membranes can be a potential diagnostic indicator of neonatal mitochondrial disease.
Introduction
Neonatal mitochondrial disease occasionally presents as intraventricular cysts in the brain.1,2 The size and site of the cysts are often evaluated in clinical management. 3 Further, atypical morphology is rarely seen in these cysts. We here report a case of neonatal lethal mitochondrial disease with IBA57 gene mutation. For the first time, we demonstrate the presence of a fluctuating membrane in a subependymal pseudocyst (SEPC) detected by brain sonography.
Case report
The mother was 36 years old (gravida 4, para 3). Fetal growth restriction was detected at 24 weeks of gestation (estimated weight, 560 g, −1.5 standard deviation [SD]). At 28 weeks of gestation, dilated ventricles were observed on fetal brain sonography. Fetal magnetic resonance imaging (MRI) at 34 weeks of gestation showed a few brain cysts with dilated lateral ventricles (Figure 1). The mother’s serum test did not indicate TORCH infection. The female newborn was delivered by cesarean section at 37 weeks and 3 days of gestation. The birth weight was 2142 g (−1.72 SD), and the Apgar scores were 3 and 6 at 1 and 5 min after birth, respectively. She was admitted to our neonatal intensive care unit owing to hypotonia. Brain sonography on admission detected enlargement of lateral ventricles and a few SEPCs (Figure 2), which were located posterior to the caudothalamic notch, adjacent to the temporal horns. The largest cyst measuring 8 × 18 mm was located in the left ventricle. Its membrane was not tense and showed fluctuating movements, which was an atypical finding. Because of the frequent apnea attacks, starting on day one after birth, the baby was put on a ventilator. On day 18, her blood test revealed metabolic acidosis (pH 7.049, PCO2 54.3 mmHg, HCO3− 14.3 mmol/L, BE −16.6 mmol/L) with a high lactate to pyruvate ratio of 41.3. Mitochondrial disease was suspected, and the baby was given vitamin cocktail therapy 20 days after birth. At 28 days of age, her general condition suddenly deteriorated due to infection, and she died 30 days after birth, despite intensive treatments.

Fetal brain magnetic resonance image (T2-weighted image). Shown are the (a) axial, (b) coronal, (c) sagittal images of multiple SEPCs (arrows).
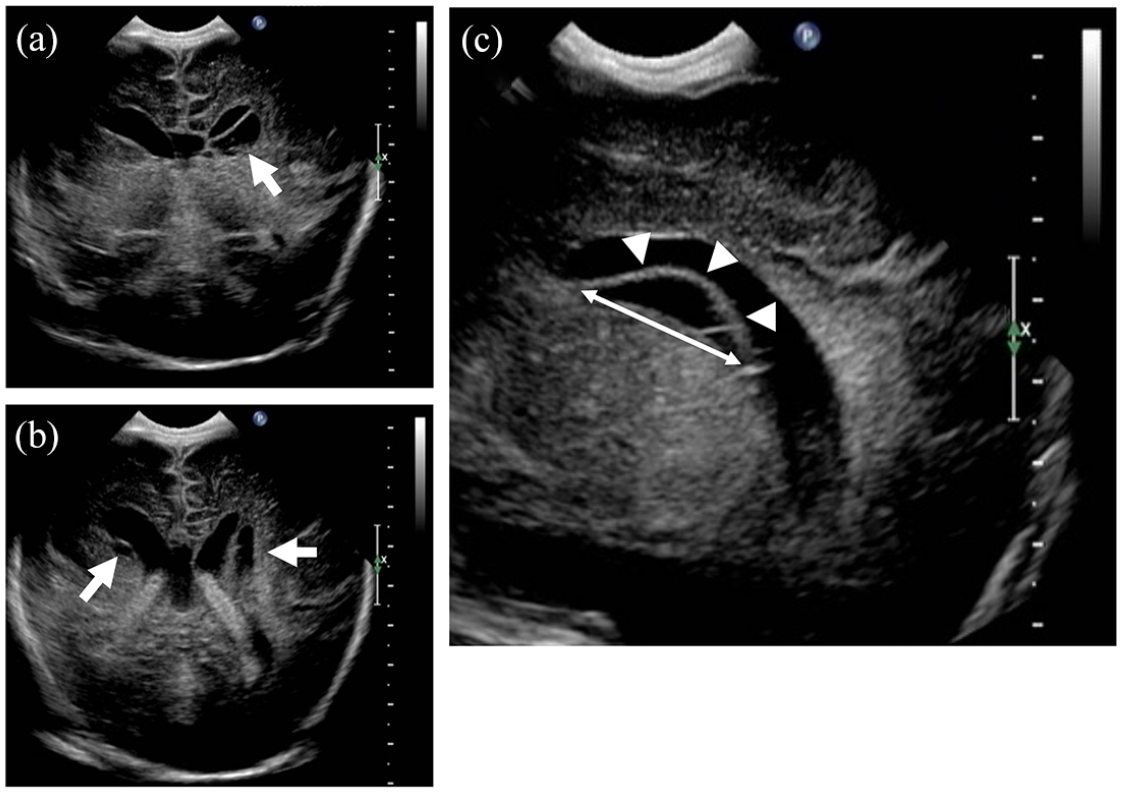
Brain sonography. Shown here are the coronal images of multiple SEPCs (arrows) at the (a) lateral body level and (b) choroid plexus level. (c) A sagittal image of the largest SEPC detected (arrowheads) at the left lateral ventricle level, with a maximum diameter of 18 mm (double arrow).
After the baby’s death, blood and various tissue samples by biopsy were obtained for investigation of the etiology with the consent of her parents. An enzyme activity test for the mitochondrial respiratory chain complexes revealed extremely low complex I and II activities in her liver, heart, and kidney and I, II, II+III, and IV in muscle (Table 1). Analysis of genomic DNA from the blood of the baby and her parents revealed compound heterozygous mutations in the IBA57 gene (c.49_67dup and c.310G>T, Figure 3). Based on these findings, she was diagnosed with mitochondrial disease. Permission to publish these findings was obtained from the patient’s parents.
Enzyme activity tests for the mitochondrial respiratory chain complexes.

CS: citrate synthase; I: complex I; II: complex II; III: complex III; IV: complex IV.

Genetic analyses using genomic DNA. Genetic analysis reveals that c.49_67dup and c.310G>T were the origins in the mother’s and father’s DNA, respectively.
Discussion
We have presented a case of neonatal lethal mitochondrial disease with a unique and rare SEPC characterized by a fluctuating membrane. SEPCs are of two types. While one is an acquired, posthemorrhagic cyst, the other is congenital and related to germinolysis. 4 The latter has been reported in association with congenital viral infections (cytomegalovirus and rubella),3,5,6 metabolic disorders (Zellweger syndrome), 7 chromosomal abnormalities,3,5 and maternal cocaine consumption. 8 However, an SEPC is commonly not an isolated finding in otherwise healthy newborns. Heibel et al. 9 showed that the incidence of SEPCs in full-term newborns was 0.35%. Of the 34 newborns with SEPC, 30 were healthy, while the remaining four had symptoms such as hemiparesis and infantile spasm. 9 Rohrbach et al. 10 reported a case of SEPC with mitochondrial disease for the first time using fetal brain sonography and MRI. Egloff et al. 1 reported that 14 (5%) out of 296 cases with pyruvate dehydrogenase deficiency and other mitochondrial diseases had cystic cerebral lesions based on fetal brain sonography, and seven (2.5%) of them had cysts including SEPCs in the ventricles. García-Cazorla et al. 11 reported four cases with pyruvate carboxylase deficiency had periventricular cysts based on postnatal brain MRI.
Abnormal SEPCs present as infections, metabolic disorders, or mitochondrial disease. They are diagnosed based on cyst size and location in fetal brain sonography using the following criteria: (1) maximum diameter ≥9 mm, (2) adjacent to the occipital and temporal horns, (3) located posterior to the caudothalamic notch, and (4) atypical morphology (ill-defined margins, square shape or height greater than the anteroposterior diameter). 3 The SEPC, in our case, clearly satisfied the first three of the above-mentioned features. As for atypical morphology, we for the first time, detected fluctuating movements of the SEPC membrane. The image of the SEPC membrane (sum of 283 images after subtracting the average background) captured as a movie by brain sonography, 12 was analyzed using ImageJ (https://imagej.nih.gov/ij/). The fluctuating movements of the SEPC membrane showed more dense contractions than the lateral ventricle wall (Figure 4).

SEPC image and background subtraction.
This present case also revealed compound heterozygous mutations in the IBA57 gene. The IBA57 gene encodes a protein involved in the iron-sulfur cluster (4Fe-4S) assembly. 13 Defects in IBA57 have been reported to cause a general deficiency of mitochondrial proteins with 4Fe-4S clusters, such as those in the respiratory complexes I and II.14,15 We detected extremely low complex I and II activities in this case. Cavitating leukoencephalopathy has been reported in a large number of cases of infantile mitochondrial leukoencephalopathy, particularly in those with multiple mitochondrial dysfunction syndrome related to ISCA2 and IBA57 mutations.16–18 Based on these findings, we speculate that fluctuating movements in the SEPC membrane may be indicative of neonatal mitochondrial leukoencephalopathy.
Conclusion
SEPCs are seen in cases of neonatal mitochondrial disease. The fluctuating movement of the SEPC membrane on brain sonography could be a diagnostic marker of neonatal mitochondrial disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Grants-in-Aid for Young Scientists (no. 19K20194) of JSPS KAKENHI and the Practical Research Project for Rare/Intractable Diseases from the Japan Agency for Medical Research and Development, AMED (JP19ek0109273, JP20ek0109468).
