Abstract
Introduction
β-eudesmol and atractylodin (Figure 1a and b, respectively) are the major volatile components of the rhizome of

Structure of (a) β-eudesmol and (b) atractylodin.
As
Methods
Compounds
β-Eudesmol and atractylodin, in their pure form (99.99%), were bought from Wako Pure Chemical Industries Ltd (Osaka, Japan). Solvents used for dissolving β-eudesmol and atractylodin were ethanol (EtOH) and dimethyl sulfoxide (DMSO), respectively. Preparation of different concentrations of the test compounds was done by diluting the stock solutions in embryo medium (E3). The highest concentrations of EtOH and DMSO in the final test solutions were 1.11% and 0.6%, respectively, not exceeding the lowest permitted concentrations that cause phenotypic and molecular level defects in the zebrafish embryos.9–11
Zebrafish embryo collection
Ethical approval to report this case was obtained from the Ethics Committee for Animal Research of the National Science and Technology Development Agency, Thailand (Number 005/2561). Zebrafish were maintained and handled according to the recommendations provided in OECD Test No. 236: FET. 12 To get the eggs, adult male and female fish were made to mate in spawning tanks an hour before the collection of the eggs. The eggs were then collected and raised in E3 water. Naturally fertilized eggs were selected for the tests using a stereomicroscope (Olympus SZX7, Olympus Corporation Shinjuku Monolith, Tokyo, Japan).
Treatment of embryos with test compounds
Appropriate concentrations of the test compounds were selected based on the preliminary experiment. The concentrations selected for β-eudesmol were 12.5, 50, and 100 µM and for atractylodin were 12.5, 25, and 50 µM. The control consisted of only the solvent in the vehicle. Naturally fertilized eggs aged 0.75–1.25 h post-fertilization (PF) were selected and transferred to a 12-well plate (30 embryos/well). The test solutions, either β-eudesmol (12.5, 50, and 100 µM) or atractylodin (12.5, 25, and 50 µM) and the control were added to respective wells. The plate was then incubated at 28.5°C for 72 h. An equal number of embryos from each concentration was transferred to a centrifuge tube, and the exposure media was completely removed. The embryos were washed three times with type II deionized water for use in further experiments.
RT-qPCR analysis
Total mRNA of the embryos was extracted using the RNeasy Plus Kit (Qiagen, Hilden, Germany) following the company's instructions. One microgram of mRNA was reverse transcribed into cDNA using iScript™ Reverse Transcription Supermix for RT-qPCR (Bio-rad, CA, USA). cDNA (500 ng) was used for RT-qPCR using Bio-Rad CFX96 thermal cycler and iTaq™ Universal SYBR® Green Supermix (Bio-rad).
13
The primer sequences used are presented in Table 1. The reaction process included incubation at 50°C for 2 min, denaturation for 10 min at 95°C, and extension of 40 cycles of 15 s at 95°C, 30 s at 60°C, and 30 s at 72°C. Melt curve analysis was immediately performed, starting at 65°C and increasing to 0.5°C
Nucleotide sequences of primers used in the R-qPCR.

ELISA assay of whole-body cortisol
Whole-body cortisol of the embryos was extracted as previously described. 15 In order to obtain a sufficient amount of cortisol, 70 embryos/concentration were used. Cortisol was extracted by adding diethyl ether in embryos homogenized in 1xPBS, and the concentration was determined using a commercial ELISA kit (Fish cortisol ELISA kit, MyBioSource, CA, USA). Samples were prepared in threefold dilutions following optimization tests; each assay was performed in duplicates. Fifty microliters each of the standard and the sample were added to each well, followed by an antibody (1x, 50 μl). The plate was incubated at 37°C for 40 min, and the content in each well was washed with 200 μl of wash buffer. After that, HRP conjugate (1x, 100 μl) was added. The plate was again incubated at 37°C for 30 min. The plate was rewashed, and 90 μl of TMB substrate was added and further incubated at 37°C for 20 min. At last, 50 μl of stop solution was added, and the absorbance was measured at 450 nm in a Spectramax M5 microplate reader (Molecular Devices, California, USA).
Statistical analysis
Statistical analysis was performed using SPSS version 23 (SPSS Statistics 23.0, International Business Machines Corp., Armonk, NY, USA). Data are presented as mean ± SD values of three independent experiments (duplicates each). Comparison of the differences in more than two and two groups of quantitative data was performed using one-way ANOVA followed by Fisher's LSD test The statistical significance level was set at
Results
Na/k-ATPase genes
The selected nonlethal (12.5 μM) and lethal (50 μM) concentrations of β-eudesmol suppressed the expression of

Relative expressions of Na/K-ATPase genes in zebrafish embryos exposed to different concentrations (μM) of the test compounds (a) β-eudesmol and (b) atractylodin for 72 h. Expressions were compared to β-actin as the internal control gene.
Hepatotoxicity genes
β-eudesmol at nonlethal (12.5 μM) and lethal (50 μM) concentrations caused significant downregulation of the
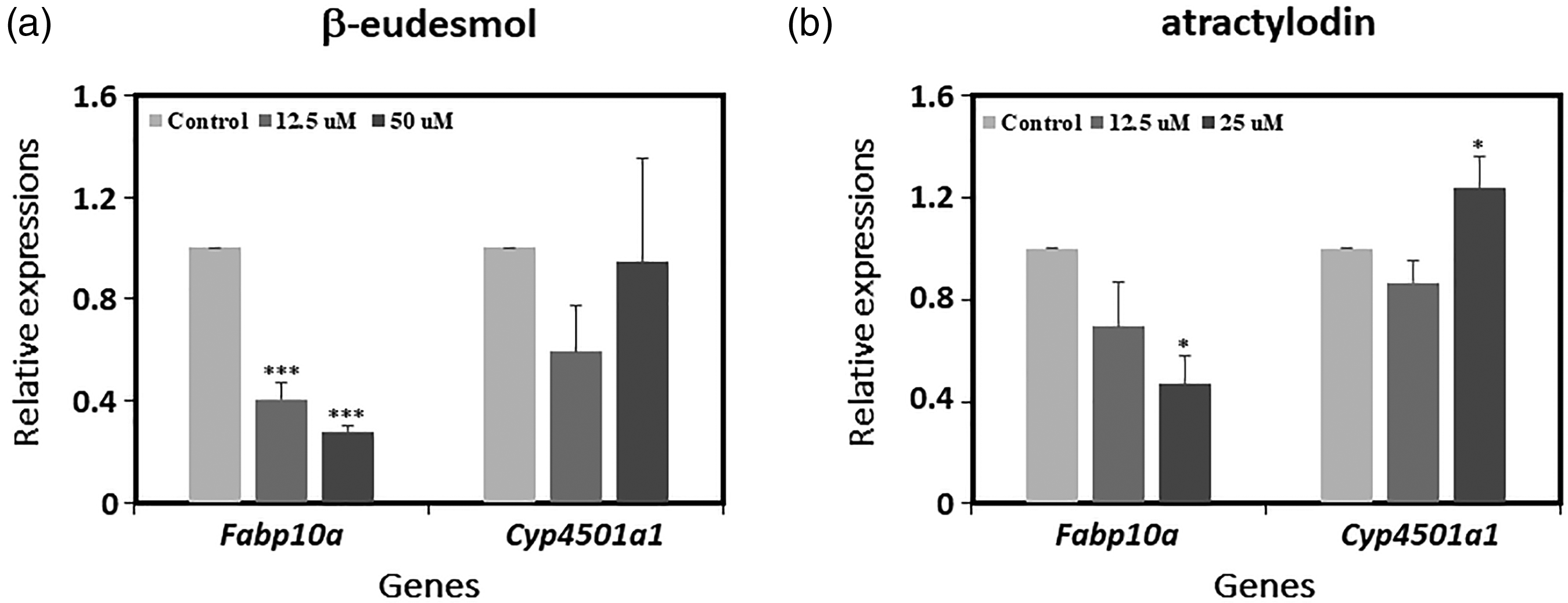
Relative expressions of genes involved in hepatotoxicity in zebrafish embryos exposed to different concentrations (μM) of the test compounds (a) β-eudesmol and (b) atractylodin for 72 h. Expressions were compared to GAPDH as the internal control gene.
Whole-body cortisol levels
Using selected concentrations of β-eudesmol (50 and 100 μM), it was found that only the highest concentration (100 μM) caused significant suppression of whole-body cortisol concentration (8.08 ng/ml) in embryos when compared with control (12.06 ng/ml) (

Cortisol levels (ng/ml) obtained from zebrafish embryos exposed to different concentrations of (a) β-eudesmol and (b) atractylodin compared to the control group.
Discussion
Apart from the pharmacological properties, toxicological effects also significantly determine the successful development of a candidate compound for the targeted disease. 16 Even after the drug has been launched for clinical use, some serious safety issues may also lead to the withdrawal of the drug from the markets like in the case of thalidomide.7,8 Therefore, the ideal approach would be to investigate the possible toxic effects of the test compounds/candidate drugs on critical organs and organ systems during the preclinical studies to avoid the occurrence of serious drug reactions in humans.
Cardiotoxicity and hepatotoxicity are the most common causes of withdrawal of candidate drugs during preclinical and clinical development.
16
In this study, the cardiotoxic and hepatotoxic effects, and in addition, the effect on the endocrine system of β-eudesmol and atractylodin were investigated in the zebrafish model. The effects of both compounds on the expression of the
Cyp1a1 is a member of the cyp450 heme monooxygenases enzyme family.
32
Cyp enzymes are responsible for the detoxification of xenobiotics as well as endogenous substrates.33,34 Zebrafish cyp1a1 has been reported to be prominently expressed in the liver,
35
while other members like cyp1b1, cyp1c1, cyp1c2, and cyp1d1 are found in small quantities in the liver, gill, and kidney.35,36 Exposure to external substances can have varying effects on the production of cyp enzymes. Some xenobiotics tend to increase the production of enzymes (enzyme inducers), while others might suppress enzyme production (enzyme inhibitors). Enzyme inducers can accelerate the metabolism of other compounds, while enzyme inhibitors can delay the metabolism.
37
β-Eudesmol showed a nonsignificant increase in the expression of the
The stress hormone cortisol is the major corticosteroid in most mammals and fishes, which is produced in the hypothalamic–pituitary–interrenal (HPI) axis. Its production is mostly triggered by various stressful conditions such as confinement, handling, the sight of predators, and exposure to extremes of temperatures and toxicants.
38
Zebrafish's response to stress by the increase in the production of cortisol has been confirmed in several studies. Zebrafish exposed to a sublethal concentration of ammonia (equivalent to the amount of ammonia found in the environment) for 24 h led to more than a threefold increase in the whole-body cortisol levels when compared with the control group. The response to stress through an increase in cortisol production was maintained as long as the inducer was present, whereby the cortisol levels were about twofold higher than the control group even after 14 days of exposure.
39
Cortisol levels can also be reduced when the fish are exposed to certain compounds with stress-reducing properties. For instance, zebrafish treated with essential oil from
There are earlier studies that reported the toxicity profile of the extract of
Conclusions
This study demonstrated the effects of β-eudesmol and atractylodin on some of the critical organ systems in developing zebrafish embryos. Results revealed some adverse effects on the functions of the
Limitations of the study
This study focused only on a few target genes that were earlier reported to be markers of toxicity of the particular organ system. As such, it may not be representative of the overall toxicity study of the systems discussed here. Other targets at different levels might have to be further studied. However, our study indeed confirms the effects of the test compounds on the selected targets in various organ systems as
Footnotes
Acknowledgements
The authors would like to thank Nano Safety and Risk Assessment Laboratory, National Nanotechnology Center, and National Science and Technology Development Agency (NSTDA), Pathum Thani, for their kind support of laboratory facilities.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This study was supported by Chulabhorn International College of Medicine (CICM) of Thammasat University, Center of Excellence in Pharmacology and Molecular Biology of Malaria and Cholangiocarcinoma of Thammasat University, National Research Council of Thailand (NRCT), National Research University Project of Thailand (NRU), Office of Higher Education Commission of Thailand, and National Nanotechnology Center (NANOTEC). Kesara Na-Bangchang is supported by the National Research Council of Thailand under the Research Team Promotion grant (grant number NRCT 820/2563). Mr Gyem Tshering is supported by Thammasat University for PhD program and also received financial support from the research fund of Chulabhorn International College of Medicine contract No. T1/2562.
Ethical approval
Ethical approval to report this case was obtained from the Ethics Committee for Animal Research of the National Science and Technology Development Agency, Thailand (Number 005/2561).
Author biographies
Gyem Tshering is a PhD graduate in Bioclinical Sciences in the field of Pharmacology and Toxicology. He has a background in Pharm D. He has three international publications published as the first author.
Wittaya Pimtong is a senior researcher at the Nano Environmental and Health Safety Research, National Nanotechnology Center, National Science and Technology Development Agency of Thailand. He is an expert in Developmental Biology, Molecular Biology, Biochemistry and Toxicology. He has around 20 international publications.
Tullayakorn Plengsuriyakarn is an assistant professor at the Graduate Studies at Chulabhorn International College of Medicine, Thammasat University. He is an expert in Pharmacology (Animal Research). He has more than 30 international publications.
Kesara Na-Bangchang is a professor in pharmacology and an expert in pharmacology and toxicology, especially pharmacokinetics. She has more than 200 international publications. Recently, she won the 2021 National Outstanding Researcher in Chemical and Pharmaceutical Science of Thailand. Currently, she serves as the director of Graduate Studies at Chulabhorn International College of Medicine, Thammasat University.
