Abstract
The rate of angular correction (ROAC) is very unpredictable and may be affected by various factors in the treatment of genu valgum and varum by means of guided growth. The purpose of this study was to assess the ROAC in cases from our institution and to identify risk factors associated with the occurrence of lower ROAC.
We retrospectively reviewed the chart records of 68 patients undergoing guided growth with figure-eight plate for the correction of genu valgum and varum. Based on the data from these patients, the annual increment of physeal growth was calculated and compared with data from the Anderson chart. The associations between patient characteristics and ROAC were evaluated with the use of univariate logistic regression.
The mean rate of femoral angular correction was 10.29 degrees/year, while the mean rate of tibial angular correction was 7.92 degrees/year. In a univariate logistic regression analysis, the variables associated with a higher risk of lower ROAC included non-idiopathic coronal deformity of the knee (odds ratio = 13.58, p < 0.001) and body weight at or above the 95th percentile for children (odds ratio = 2.69, p = 0.020).
Obesity and non-idiopathic coronal deformity of the knee are risk factors for lower ROAC. It is still uncertain whether severity of deformity, race, and operative procedure have a substantial effect on the rate of correction.
Level III evidence.
Introduction
During skeletal growth, disturbance to the growth of the medial or lateral physis may lead to abnormal coronal alignment of the extremity, for example, genu valgum or varum. 1 The genu valgum or varum may be idiopathic or arise from diverse etiologies, such as trauma, dysplasia, metabolic disease, inflammatory, endocrine, tumor, neoplastic, or developmental pathologies.2–5 The presence of deformity may cause cosmetic problems, functional disabilities, activity-related pain, abnormal gait mechanics, and early-onset osteoarthritis in later life.6,7
Guided growth, which provides gradual deformity correction, is widely accepted for the management of limb angular deformity in skeletally immature patients by using various implant techniques (e.g. staple, figure-eight plate). Implant-mediated guided growth is reversible without permanently violating the physis. It is technically easy, minimally invasive, and associated with less morbidity and fewer complications with traditional osteotomy.8,9 The challenges include optimal timing of guided growth, implant removal, and unpredictable result. 10 Overestimating the rate of angular correction (ROAC) may lead to improper surgical management and follow-up strategy, even eventual surgical failure. 11 Therefore, accurately predicting the ROAC is useful for decision making and patient follow-up.
To date, the ROAC is very unpredictable and may be affected by various factors. Diverse values of ROAC were reported in various published studies.2,11–15 Paley’s multiplier method and Bowen’s chart,1,16 based on data from the work by Anderson et al.17,18 were introduced to calculate the timing of guided growth and to predict the ROAC. However, in clinical aspect, the error of prediction was still inevitable. 13 Other factors (e.g. surgical procedures applied, direction of the deformity, etiology, gender, body weight, and target physis) might also contribute to the error. 11
The purpose of this study was to assess the ROAC in cases from our institution and to identify the risk factors associated with lower ROAC after guided growth for the correction of genu valgum and varum.
Materials and methods
Patients
With the approval from Xinhua Hospital Ethics Committee (reference no. XHEC-D-2015-231), we reviewed medical charts and radiographical records to retrospectively evaluate the results of genu valgum and/or varum management. Written informed consent was obtained for the participant. All patients in the study underwent guided growth with figure-eight plate technique at our institution between 2010 and 2017. Inclusion criteria included coronal deformity of the knee of >15°, and a minimum of 1 year of remaining growth. Exclusion criteria included (1) patients with Blount’s disease; (2) history of knee surgery (e.g. osteotomy, external fixator); (3) patients with rickets undergoing non-standard metabolic therapy; and (4) no adequate preoperative or follow-up radiographs available for review. Patients were operated on by three fellowship-trained pediatric orthopedic surgeons at our institution during the study period.
Data collection and calculation
The following information was collected: sex, age at the time of surgery, age at time of implant removal, number of plates used, target physis (proximal tibia or distal femur), and etiology. We used the weight-for-age, which raised by Stettler N, to identify overweight children in our study because the height of a small number of patients was not available. 19 Standing full-length joint survey radiographs were reviewed and the mechanical lateral distal femur angle (mLDFA), anatomical medial proximal tibia angle (aMPTA) and the width of physis, were measured from the latest preoperative films, and the latest films before implant removal. The width of physis was defined the mediolateral radiographic extent of the physis (Figure 1). Radiographic images were retrieved using a picture archiving and communication system (PACS), and radiographic measurements were performed using PACS software (UniWed, 6.1, EBM Technologies, Shanghai, China). A consensus building session to define the radiographic measurements was held by two attending orthopedic surgeons. Evaluation of the outcome was conducted in a blinded fashion. The change of LDFA or MPTA after implant removal was calculated. If the value was less than 3° after more than 6 months of treatment, the rate was considered as 0. The width of physis was the mean value measured from the latest preoperative film and the immediate film before implant removal. The agreement between the two reviewers was evaluated with the Spearman correlation coefficient for interrater agreement and intraclass correlation coefficient (ICC). To calculate the longitudinal growth of the concave side of each operated physis, assumptions were raised as follows: (1) the concave side of the operated physis grows at the same speed as that of the equivalent one in the contralateral limb, and (2) the hinge point of angular correction is located at the perichondrium of the implanted physis. 18 The equation is obtained as the annual growth rate of increment = tan (α) × r/y. α = (the preoperative LDFA or MPTA) − (LDFA or MPTA after implant removal). r = the width of physis. y = duration of implant suspension (Figure 2). Based on the data from the Anderson chart,17,18 the standard annual increment of the proximal tibia and distal femur was calculated respectively. The equation is as follows. Annual growth rate of increment = (Lε+1 − Lε) × κ. ε+1 = 1 year older than the current chronologic age, ε = the current chronologic age, κ = the percentage of estimated contributions from the distal femur and proximal tibia to the longitudinal growth of their respective segments. The average growth percentile relative to entire bone until skeletal maturity: κ = 0.71(distal femur), 0.57(proximal tibia). 1
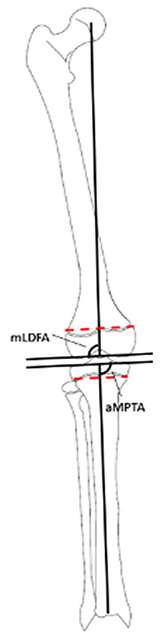
Sketch map of the knee (aMPTA: anatomical medial proximal tibial angle; mLDFA: mechanical lateral distal femoral angle; red dashed line: the width of physis).

Sketch map of proximal tibial physeal growth (black rectangle: normal physeal growth; black triangle: physeal growth with temporary hemiepiphysiodesis; r: the width of physis; α: the change of angle after temporary hemiepiphysiodesis).
Statistical analysis
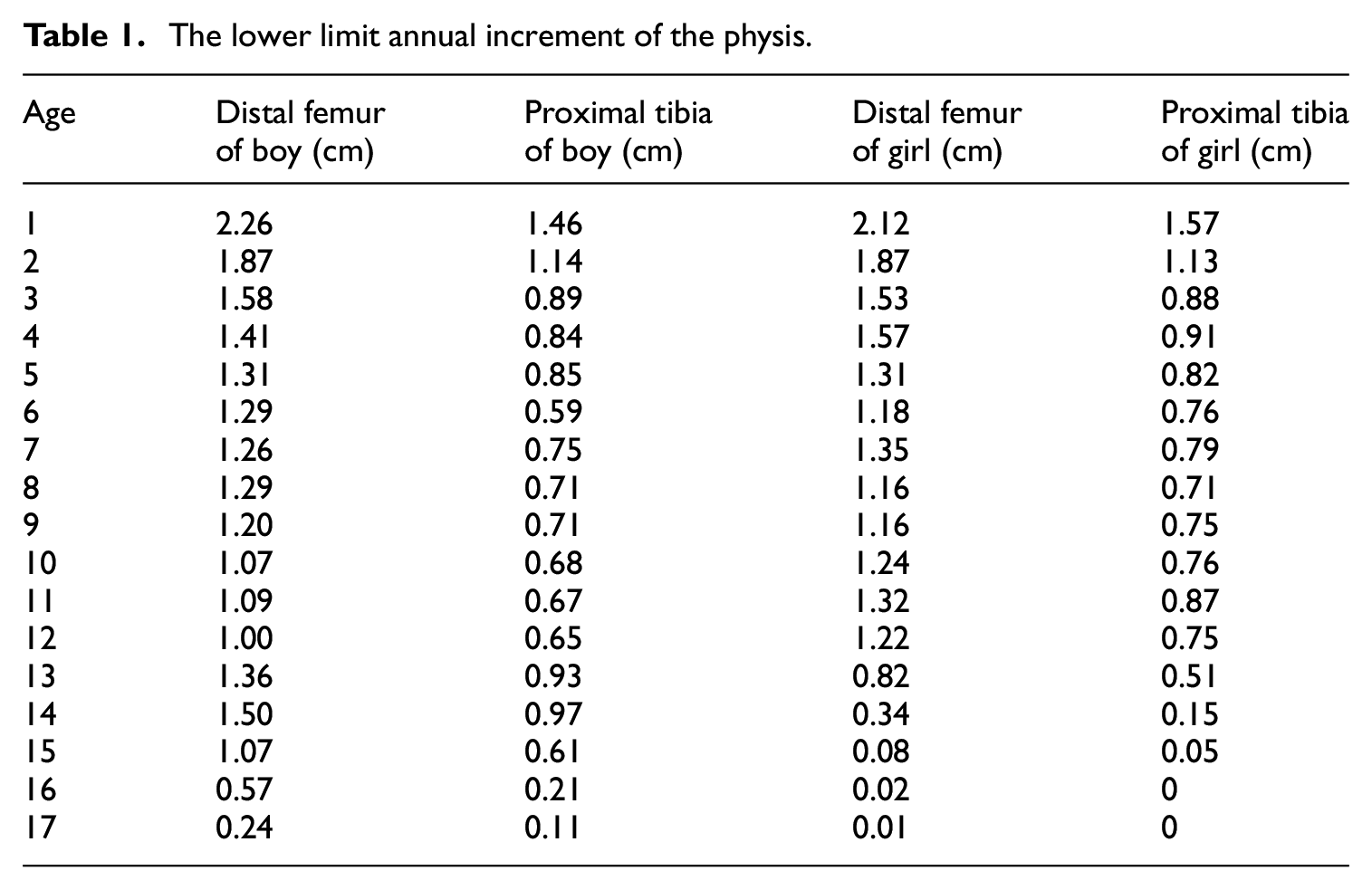
We defined the value of mean-2 standard deviation (SD) of the Anderson chart as the lower limit value of annual increment of physis (Table 1). A comparison was made between the annual increment from the Anderson chart and our data. If the value from our patients was lower than the lower limit value of annual increment according to chronological age, it was considered as a lower ROAC (Figure 3). After that, the associations of patient characteristics with lower ROAC were assessed with the use of univariate logistic regression. The number of plates used, direction of the deformity, etiology, gender, body weight, and the physis treated were assessed as binary variables. The etiology included idiopathic coronal deformity of the knee (ICDK) and non-idiopathic coronal deformity of the knee (non-ICDK). Idiopathic coronal deformity of the knee would be diagnosed only if all the other etiologies were excluded. Obesity in children and adolescents was defined as body weight at or above the 95th percentile for children of the same age and sex in the Centers for Disease Control and Prevention growth charts. 20 Statistical significance was set at p < 0.05.
The lower limit annual increment of the physis.

Graphs showing the annual increment of each physis from clinical patients (red circles) compared with the lower limit from Anderson’s data (blue line). If the value was lower than the limit line, it was defined as a lower rate of correction.
Results
Of the 103 patients treated during the period of this study, 68 patients (26 females, 42 males), with 150 involved physes, met the inclusion criteria. The mean age of patients was 6.3 ± 0.4 years (2.1–13.0), the mean time of implant remaining in the body was 1.0 ± 0.1 years (0.7–2.4), the mean rate of femoral angular correction was 10.29 degrees/year, and the mean rate of tibial angular correction was 7.92 degrees/year. The mean width of the physis at the distal femur was 6.24 ± 0.12 cm, while that of tibial physis was 5.20 ± 0.12 cm. We found the mean rate of angular correction was 6.07 degrees/year in the age 8–10 years old group, and the mean rate of angular correction was 5.22 degrees/year in the age 10–14 years old group. We didn’t compare these two groups by t-test due to limit sample size. The Spearman’s correlation coefficients for both the interrater agreement and intraclass correlation coefficient (ICC) were 0.82 and 0.87, respectively. We compared the rate of angular correction in our study with this in other literature (Table 2). There were 28 cases of idiopathic coronal deformity of the knee (Figure 4), 10 cases of benign tumor, 11 cases of posttraumatic coronal deformity of the knee, 5 cases of rickets, and 14 cases of skeletal dysplasia (Figure 5). There were 26 obese patients. Based on the rate of correction and the width of physis, with an illustration of the trigonometry, the growth rate at the contralateral side was calculated. In terms of the operated sites, the incidence of lower ROAC was 74/150. In the femur group, the incidence of lower ROAC was 39/82. In the tibia group, the incidence of lower ROAC was 35/68. Two significant risk factors were identified for the group with lower ROAC: obesity and non-idiopathic coronal deformity of the knee (Table 3). The risk of lower ROAC had a 13.58-fold increase (95% confidence interval = 5.02–36.76, p < 0.001) in the patients with non-ICDK compared to those with ICDK. The risk of lower ROAC had a 2.69-fold increase (95% confidence interval = 1.17–6.20, p = 0.02) in obese patients compared to non-obese patients. There was no statistically significant difference in the ratio of boys and girls, the number of the plates used, the operated sites (either proximal tibia or distal femur), and the direction of deformity (genu valgum or varum) as to the risk of lower ROAC.
The comparison of angular correction rate between other literature and our results.

ICDK: idiopathic coronal deformity of the knee; non-ICDK: non-idiopathic coronal deformity of the knee.
Age at the time of surgery (years).
Degrees/year.
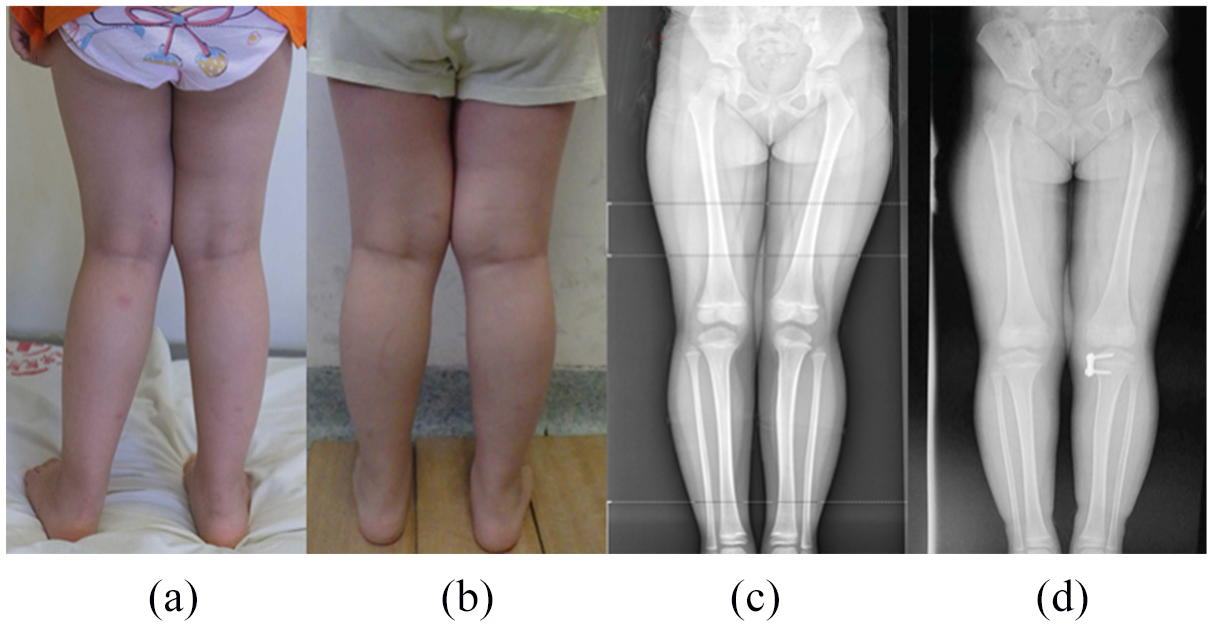
Case 1: a 4.9-year-old, 18.6 kg girl with idiopathic genu valgum. After 1 year of treatment, the valgum was corrected. The rate of angular correction was higher than the predicted lower limit value from the Anderson chart: (a, c) before operation and (b, d) 1 year after operation.

Case 2: a 2.7-year-old, 17.1 kg obese boy with skeletal dysplasia. After 1.2 years of treatment, for peer review the varum was partially corrected. However, the rate of angular correction was lower than the predicted lower limit value from the Anderson chart: (a, c) before operation and (b, d) 1.2 years after operation).
Results of univariate logistic regression analysis for the lower rate of correction.

ICDK: idiopathic coronal deformity of the knee; p > |z|: p values; z: statistical magnitude.
Discussion
Guided growth allows gradual correction of the deformity. In pediatric patients with an open physis, angular deformity can be corrected by asymmetrically suppressing the physis and taking advantage of the physeal growth. In view of the close relationship between the growth potential and rate of correction,1,16 a predicted rate of angular correction is necessary to help surgeons decide the timing for surgery and make a suitable follow-up strategy. In previous studies, the values of ROAC were reported to be inconsistent. The mean angular correction per year in terms of mLDFA and mMPTA ranged from 2.63° to 12° and from 2.11° to 12°, respectively.2,4,5,11–15,21,22 Some studies also presented that the rate of angular change varied from the first year after the guided growth through to the subsequent years.13,23 Our results showed that the mean value of ROAC at the distal femur and proximal tibia was 10.29 and 7.92 degrees/year, respectively.
Why do ROAC values have such great diversity? Many prognostic factors and baseline characteristics have been proposed to play roles in producing such diversity. The variation in etiology, gender, operative technique, and the condition of the involved physis is believed to contribute to the occurrence of lower ROAC. In our study, based on the width of the physis and the rate of correction, the longitudinal growth rate at the concave side of the operated physis can be approximated by applying a mathematical formula and comparing the result with the data from Anderson. The incidence of lower ROAC was very high (49.3%). Factors were analyzed, such as gender, etiology, body weight, direction of the deformity, and number of plates. Two significant risk factors were identified for lower ROAC: non-ICDK and obesity.
In the ICDK group, the physis was considered as normal, while in the non-ICDK group, the physis was influenced by various etiologies. The non-ICDK in our study included benign tumor, posttraumatic coronal deformity of the knee, rickets, and skeletal dysplasia. The results showed that patients with non-ICDK had a significantly higher incidence of lower ROAC. Boero et al. 24 reported that the correction was faster in idiopathic deformity than that in pathological deformity. Castañeda et al. 25 reported that the corrective speed of an “abnormal physis” was lower than that of a normal physis. Cho et al. 26 speculated that the physeal growth rate could be retarded in the patients with multiple epiphyseal dysplasia. Our findings were similar to these studies. The annual increment of an abnormal physis was significantly lower compared with that of a normal physis. This lower increment at abnormal physis may explain why some authors suggested the treatment be started as soon as possible and the efficacy of correction was unpredictable.2,26
Obesity was another prognostic factor for lower ROAC. It is known that greater body weight increases compressive loads. High loads retard the growth of physis. Patients with Blount’s disease were excluded from this study due to the strong relationship with obesity, which could lead to risk of bias in the analysis. The conventional BMI-for-age method was not applied to identify obesity for three reasons: (1) The cut-off point of BMI-for-age percentile was diverse in different ethnic groups in the population; (2) the stature of patients with genu deformity might be abnormal in terms of different etiologies; and (3) the height of a small number of patients was not recorded in our hospital. The weight-for-age method, introduced by Stettler et al. 19 had reasonable value of positive prediction. Although body weight, as a constant variable in Bushnell et al.’s 27 study, was not found to be associated with correction, obesity was found to be associated with higher likelihood of lower ROAC in our study. The result was similar to the findings by McIntosh et al. 10 In both our paper and McIntosh’s, obesity was taken as a binary variable. We speculate there is a threshold load specified at each physis regulating the skeletal growth. If the actual load exceeds the threshold, lower ROAC might occur. Conversely, if the load is under the threshold, the ROAC is normal. Congdon et al. 20 also supported this opinion.
Considering the variation of load contribution across the medial and lateral compartments of the knee, 28 we tried to see whether the direction of deformity is related with lower ROAC. Castañeda et al. 25 found that the rate of valgum correction was higher than that of varum correction. However, their data included patients with Blount’s disease. Sung et al. 11 also suggested that direction of deformity might influence the ROAC. However, our results did not support the role of these factors in the occurrence of lower ROAC.
The number of plates was taken into the analysis in our study. It was the surgeon’s preference whether to apply one or two plates at the individual physis. Guzman et al. 29 suggested that two plates provided a faster rate of deformity correction. Stevens 9 speculated that two plates would offer no biomechanical advantage. In our study, we found no significant difference in outcome measure between the use of either one or two plates. Although some surgeons have speculated that two plates can reduce the rate of implant failure, little evidence exists to support this conclusion. The cost for patients is also a concern.
Bone age was not assessed in this study. As a more objective criterion to determine the endpoint of growth, it is theoretically preferable in planning asymmetric growth arrest. Clinically, it is difficult and expensive to obtain the bone age for all patients because of patient transportation problems and fear about radiation exposure. For patients with skeletal dysplasia or rickets, it is difficult to estimate bone age and predict remaining growth. 26 The recordings of chronological age are more convenient for doctors and patients. Although some authors have suggested it is unreliable to use chronological age in predicting the growth potential,30–32 Sander et al. 33 suggested that it might be more reliable before the growth spurt. The mean age of our patients was younger than 10, therefore, chronological age was employed as one of the parameters in our research.
The limitations of our study include its retrospective design, a relatively small sample size, the fact that power analysis for sample size calculation was not done, and the fact that all the data is from a single center. The young patients would receive the guided growth procedure only in this condition that children with pathological coronal deformity of the knee, and it maybe lead to the select bias. Preliminary medial proximal tibial angle was not analyzed in our study because the deformity was moderate or even mild in most of our cases (the angle <25°). The power of this study was not great enough to assess the relationship between the rate of angular correction and severity.
Conclusions
The mean rate of angular correction in the distal femur and proximal tibia was 10.29 and 7.92 degrees/year, respectively. Obesity and non-idiopathic coronal deformity of the knee are the risk factors for lower ROAC. It is still uncertain whether severity of deformity, race, and operative procedure make any substantial contribution to the rate of correction.
Footnotes
Acknowledgements
The authors acknowledge Mr. Daniel Edward Porter and the writing assistant center in Children’s mercy hospital of Kansas City (64108 MO.USA) as writing assistant.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China grant number 81501930.
Ethics approval
Ethical approval for this study was obtained from Xinhua Hospital Ethics Committee (NO. XHEC-D-2015-231).
Informed consent
Written informed consent was obtained from legally authorized representatives of the minor patients.
