Abstract
Compression ignition engines are one of the world’s largest consumers of fossil oil but have energy extraction efficiency limited to 35%. Addition of hydrogen alongside diesel fuel has been found to improve engine performance and efficiency; however, after a certain limit, hydrogen begins to show adverse effects, mainly because the ratio of oxygen to fuel decreases. This can be overcome by using oxyhydrogen, which is a mixture of hydrogen and oxygen gas.
In this study, effects of addition of oxyhydrogen generated by electrolysis, with varying flows at the intake manifold, on a 315 cc compression ignition engine alongside diesel were analyzed.
The engine was mounted on a Thepra test bed and torque measurements were taken at predetermined test points for diesel and 6 and 10 standard cubic feet per hour flowrates of oxyhydrogen.
H10 showed the maximum improvement in engine performance equating to a 22.4% increase in both torque and power at 3000 r/min, and a 19.4% increase in efficiency at 2600 r/min was recorded. The large increase in engine performance as compared to previous results is because of high oxyhydrogen flowrate to displacement volume ratio.
The oxyhydrogen flowrate to displacement ratio is the most important factor as it is directly impacts engine performance. The difference in engine performance because of oxyhydrogen becomes prominent at higher engine speed due to high suction pressure. No experimental flowrates of oxyhydrogen showed any adverse effect on the engine performance.
Introduction
Use of fossil fuels is increasing globally to fulfill energy demands of the modern world. The reserves of fossil fuels are depleting at an exponential rate. 1 Oil is expected to last 34 years at current trend of consumption; coal, about 107 years; and natural gas, about 37 years. 2 Energy is extracted from fossil fuels by combustion. Much of this energy is lost during conversion to useful work due to inefficiencies in the conversion system.
Compression ignition (CI) engines, which power the heavy machinery of the world, are also used extensively for automobile and marine transportation due to their high torque and efficiency and their ability to run on lean air fuel mixtures as compared to spark ignition (SI) engines, which mainly use diesel or heavy fuel oil (HFO) as the source of energy. The efficiency of CI Engines varies from 20% to 35% with maximum efficiency recorded with diesel only is about 54.4%. 3 The basic problem for CI engines is improper mixing of air and fuel because fuel is injected near the end of the compression stroke. Also, combustion occurs spontaneously at any point in the air–fuel mixture. The spontaneous combustion occurs at multiple points throughout the power stroke. These points act as centers of flame propagation. 4 Adding a fuel with high flame travel speed would result in complete combustion of the diesel fuel immediately after the first spontaneous combustion giving high torque and power.
Hydrogen, a highly combustible fuel, has a flammability range of 4%–75% by volume in air which is very high as compared to diesel’s 0.7%–5% by volume. It also has a high diffusion coefficient, 0.61 cm2 s−1, thus it can produce a homogeneous air–fuel mixture inside the combustion chamber. 5 Hydrogen also has a high laminar flame velocity, the speed at which flame propagates, and thus, rapid combustion occurs. 6 Hydrogen air mixtures have a minimum ignition energy (MIE) of 0.065 MJ, significantly less than the hydrocarbon air mixture MIE, 0.2–0.3 MJ. 7 Although low MIE is preferable for combustion, it may result in ignition of the air–fuel mixture before the engine completes its compression stroke. Hydrogen alone cannot be used in CI engines because it would require a very high compression ratio due to hydrogen’s high auto ignition temperature, 858 K. 8 It can be used as a supporting fuel to diesel in a CI engine. It has shown promising results, increasing efficiency and torque output of a CI engine.9–13 However, induction of hydrogen with diesel affects the air–fuel ratio of the diesel engine, which limits the amount of hydrogen that can be added for favorable results. This is because hydrogen uses up some of the oxygen present in the air and thus results in poor diesel combustion and decreased efficiency.14,15
As a solution to the low oxygen problem, oxyhydrogen (HHO) was introduced. HHO is a mixture of hydrogen and oxygen. When H2 and O2 are in the ratio 2:1 by volume, the mixture is called Brown’s Gas. 16 When HHO is injected into a CI engine, all the hydrogen is accompanied by the stoichiometric amount of oxygen required by hydrogen for combustion. Thus, diesel will have proper supply of oxygen even at high flowrates of HHO. HHO can be expected to improve engine’s combustion characteristics greatly and in turn improve the engine performance. This study analyses the use of HHO alongside diesel in CI engine using an engine which has a swept volume of 315 cc, far less than any of previous research. Such small engine, if made more efficient by the use of HHO, could be used to in small automobiles and motorbikes which contribute to a large fraction of the fossil fuel used world over.
Literature survey
Hydrogen and oxygen can be generated separately by many chemical reactions. Oxygen can also be extracted from air. But the most effective and efficient method of producing hydrogen and oxygen simultaneously with a near stoichiometric condition is by electrolysis of water. Water itself is a poor conductor; however, due to the presence of an ionizing agent such as metal hydroxides and halides, water becomes a conductor. It forms
Aydin et al. found an increase of 19.1% in maximum torque, 27% in maximum power, and a 14% decrease in specific fuel consumption (SFC) when HHO was injected through the intake manifold of a four-cylinder, 3657 cc engine. Hydrogen Electronic Control Unit (HECU) was present to inject HHO which regulated the supply as per engine need. 8 Uludamar found an increase of 1.3% in efficiency of a 3907 cc engine with HHO induction along with a small increase in the maximum power and torque output of the engine. 24 Baltacioglu et al. also reported an increase in engine torque and power output when HHO was injected with 10% and 15% biodiesel blends. The brake-specific fuel consumption (BSFC) decreased and the maximum efficiency of the engine increased by 0.7%. 25 Matienzo performed similar experimentation on a 930 cc diesel engine and reported an average increase of 2.79% in the efficiency. 26 Yilmaz et al. also reported an increase in the thermal efficiency and maximum torque output of a 3567 cc engine with HHO flowrate of 5 L/min (LPM). A control unit had been introduced to improve fuel economy that would reduce the HHO production below the engine speeds of 1750 r/min. It was observed that HHO had adverse impact on the engine performance without the use of a control unit below the speed of 1750 r/min. This may have been due to pre-ignition of the fuel or the inability of the engine to draw in hydrogen due to low suction at this speed. 27 Baltacioglu et al. 28 posted similar results indicating increased efficiency and torque of a 3600 cc diesel engine. In another study, Arat et al. 29 found that the efficiency of the engine increased with induction of HHO compressed natural gas (CNG) Mixture through the air inlet. Many other studies have been conducted using HHO as an additive fuel, through the intake manifold, with diesel or biodiesel and found results complementing each other.29–31 Another study analyzed the vibration response and found that the amplitude of vibration of a diesel engine reduced with the addition of HHO. 32 However, one of the most recent studies by Rimkus et al. 33 observed a 2.6% decrease in brake torque and a 2% increase in SFC on a 1.9 L CI Engine. The adverse effect of HHO addition was due to pre-ignition of fuel caused by the presence of small fractions of monoatomic hydrogen and oxygen which reacted before the fuel mixture reached the autoignition temperature.
Methodology
HHO generator
HHO was produced by an Ironside Group HHO Generator (Figure 1). The generator comprised an electrical unit which plugged into the alternating current (AC) mains. The electrical unit outputs 35 V 0–60 A DC power. This DC power is supplied to square plate reactor, which produced HHO gas. HHO gas flowed through the bubbler and then a flow meter before entering the engine intake manifold. The generator was capable of producing up to 10 standard cubic feet per hour (SCFH) or 4.72 LPM. The reactor specifications are given in Table 1.

HHO generator.
General specifications of HHO reactor.

HHO: oxyhydrogen; SCFH: standard cubic feet per hour.
Concentrated potassium hydroxide solution is used because KOH has higher solubility and affinity for water as compared to sodium hydroxide and thus produces a highly ionized solution containing hydrogen and hydroxide ions. When a potential is applied across this solution, the hydroxide ions move toward the anode and give off oxygen, while the hydrogen ions move toward the cathode producing hydrogen gas. Figure 2 shows the components for the production of HHO gas.
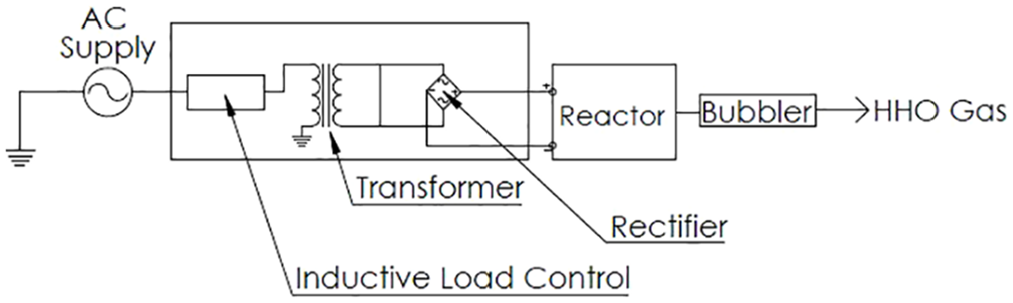
Schematic for HHO generation.
Engine and test bed
A Lombardini 15LD 315 Diesel Engine was used on Thepra Hydraulic Brake Engine Test Bed. The Lombardini 15LD series is specifically designed for this Thepra Test Bed. The engine specifications are listed in Table 2. Thepra Test Bed used during experimentation has a digital torque meter and a tachometer with least counts of 0.1 N m and 1 r/min. The test bed employs a highly viscous oil which the engine has to pump through a valve. Changing the valve opening changes the load that is applied onto the engine. The engine and test bed arrangement are shown in Figure 3.
Engine specifications.


Engine mounted on Thepra test bed.
The engine used in this experimentation has a swept volume of 315 cc far less than any of previous research. Such small engine if made efficient could be used to in small automobile and motorbikes which contribute to large fraction of the fossil fuel used world-wide.
Fuel
Services of the Fuel Laboratory, Department of Chemical Engineering, University of Punjab, Lahore were employed for the testing of the diesel that was used in the experimentation. ASTM D4737 standard was employed for the calculation of the cetane index of the fuel. The fuel sample was distilled and every 10 mL recovery temperatures were noted. The results of the experimental analysis of the fuel are given in Table 3.
Properties of diesel.

CCI: Calculated Cetane Index.
Experimental procedure
Before each experimentation, the engine was warmed to its operating oil temperature of 80°C at medium-load, full throttle. Then, the engine load was reduced to zero. After operating the engine at zero load for a few minutes, the load was increased gradually and adjusted for specific predefined engine speed. A video camera was installed to record any minor changes in the engine rotations per minute and torque at experimental point, while the fuel consumption was measured manually, four times, when the engine was in steady state. Once the minimum speed point was reached, that is, 1000 r/min, the load was gradually reduced to zero while taking measurements at the predefined engine speed, again. For each measurement, a total of 200 data points obtained from the video were averaged to attain a single-plot point. This was done for diesel and 6 SCFH (H6) and 10 SCFH (H10) flowrates of HHO.
Results and discussion
Effect on engine torque
The introduction of HHO into the engine cylinder along with air resulted in an increase in torque (Figure 4).

Variation of torque with engine speed.
This corresponds to our initial hypothesis that HHO induction would improve combustion and cements previous researches.8,18,25–28 No limiting engine speed nor any experimental flowrate was observed to have an adverse impact on engine performance contradictory to the findings of Rimkus et al. 33 This negates the possibility of monoatomic hydrogen and monoatomic oxygen entering the combustion chamber and causing premature combustion as suggested by Rimkus. Furthermore, we observe that the curves tend to converge at low engine speed indicating that there may be a point 1000 r/min where the suction pressure of the engine is so low that it is unable to suck hydrogen in as suggested by Yilmaz et al. 27 However, this engines lowest operable speed was 1000 r/min.
Comparing this to other studies,8,18,25–28 we see that none reported such drastic increase in torque to a maximum of 22.4%. This is due to the fact that the engine used is of significantly low capacity while the flowrate is high. This informs us of another factor at play, the ratio of flowrate to engine capacity.
Effect on engine power
Figure 5 shows the power curve for the engine. The power curves are found to be almost linear. This is because the engine has a relatively flat torque curve making torque a constant in the empirical relationship of power, torque, and engine speed. Thus, power becomes directly dependant on engine speed. As there is a slight dip in power for diesel after 2900 r/min, it can be said that the maximum power point of the power curve has shifted to the right for HHO. At H10, for such a small engine, the power output is higher than diesel at all points with a difference of 0.527 kW at the peak corresponding to a 22.4% change. Moreover, a 12% increase is seen for H6.
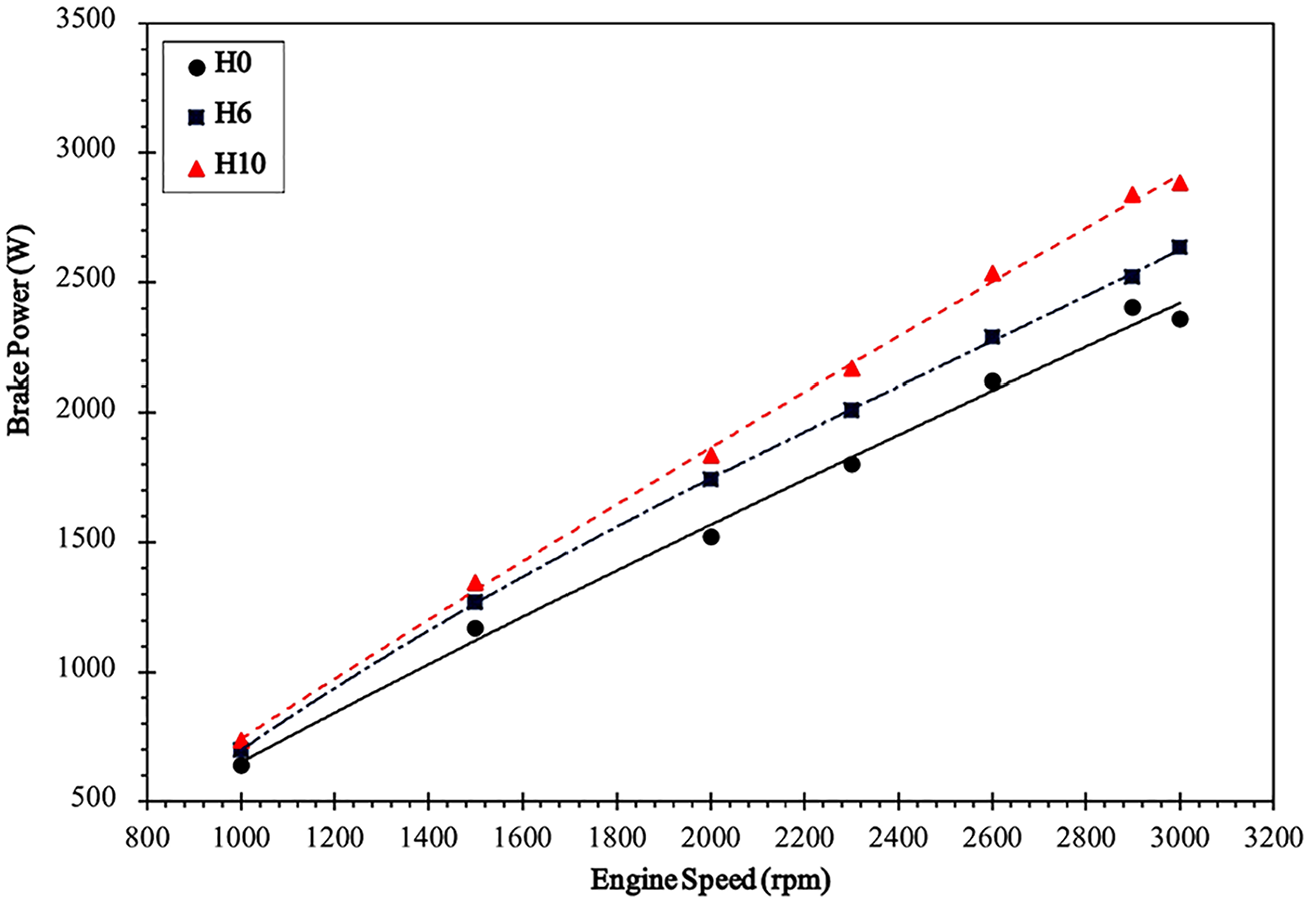
Variation of power with engine speed.
Effect on engine efficiency
The efficiency plotted against engine speed is shown in Figure 6.
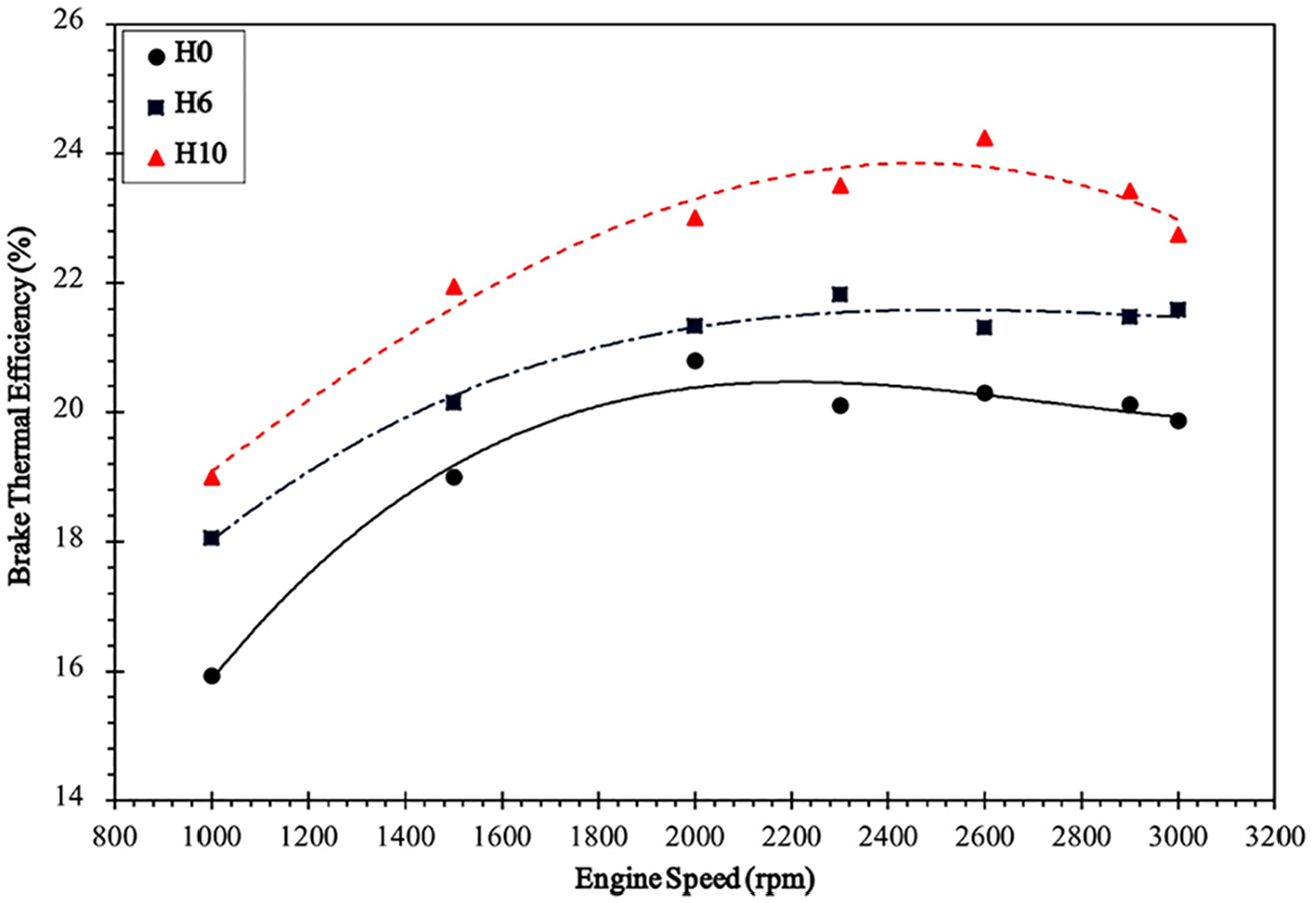
Variation of efficiency with engine speed.
It is observed that the efficiency of the engine is greatly improved with a 19.4% increase for H10 and a 8.6% increase for H6. This again cements our initial hypothesis that the combustion of diesel is improved with HHO induction and more energy is released by diesel than before. This can also be related to Carnot’s law. HHO induction into the combustion chamber increases the combustion temperature. 34 Higher flame velocity also enhances the combustion properties.6,7 Carnot’s efficiency of the cycle is improved as the temperature difference between the source and the sink is increased. The optimum flowrate of HHO as found in this study is 10 SCFH. However, we must take the flowrate to cc ratio into account. Higher the value of this ratio, higher would be the peak efficiency. The maximum efficiency observed for the engine running on HHO–diesel mixture is 24.24%. The maximum efficiency observed for diesel is 20.8%. Hence, HHO has increased the maximum efficiency by 3.4%.
Conclusion
From the results, we conclude that addition of HHO along with diesel in a small-capacity diesel engine greatly improved all performance factors of the engine. The minimum percentage increase in efficiency recorded is 2.5% and 10.5% for H6 and H10, while the minimum increase in torque is 8% and 15%. These parameters are significantly larger than any previous research. The simple explanation is that after the first spontaneous combustion in the power stroke of the diesel engine, the hydrogen ignites instantly causing the entirety of diesel to burn at a single moment. As hydrogen is accompanied with its required molar oxygen, we do not observe any adverse effect. Furthermore, such small engine is provided a much larger amount of HHO. If such an on-deck production system is developed, these small-capacity engine could power much of the smaller automobiles as they would be providing larger torque at a greater efficiency. We conclude that such systems would be much better than existing systems. We see that the engine performance is improved throughout the range of engine speed although the change is much significant at speed over 2400 r/min. The most important conclusion that we draw from this study is that no adverse effects of HHO addition were observed, neither any limiting speed nor critical point for all test flow rates.
Footnotes
Acknowledgements
We are thankful to the administration of the Department of Mechanical Engineering, University of Engineering and Technology, Lahore, Pakistan for providing us the equipment and work space necessary for performing this study. We are also thankful to the Department of Chemical Engineering, University of Punjab, Lahore, Pakistan for providing us access to their Fuel Testing Laboratory for analysis of diesel fuel used in this experimental work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
