Abstract
Amid present concerns over a potential scarcity of critical elements and raw materials that are essential for modern technology, including those for low-carbon energy production, a survey of the present situation, and how it may unfold both in the immediate and the longer term, appears warranted. For elements such as indium, current recycling rates are woefully low, and although a far more effective recycling programme is necessary for most materials, it is likely that a full-scale inauguration of a global renewable energy system will require substitution of many scarcer elements by more Earth-abundant material alternatives. Currently, however, it is fossil fuels that are needed to process them, and many putative Earth-abundant material technologies are insufficiently close to the level of commercial viability required to begin to supplant their fossil fuel equivalents “necessarily rapidly and at scale”. As part of a significant expansion of renewable energy production, it will be necessary to recycle elements from wind turbines and solar panels (especially thin-film cells). The interconnected nature of particular materials, for example, cadmium, gallium, germanium, indium and tellurium, all mainly being recovered from the production of zinc, aluminium and copper, and helium from natural gas, means that the availability of such ‘hitchhiker’ elements is a function of the reserve size and production rate of the primary (or ‘attractor’) material. Even for those elements that are relatively abundant on Earth, limitations in their production rates/supply may well be experienced on a timescale of decades, and so a more efficient (reduced) use of them, coupled with effective collection and recycling strategies, should be embarked upon urgently.
Keywords
Of all essential commodities, the fossil fuels may well run into production limits during the next few decades, and indeed, the underpinning determinant of how we extract resources, inducing those of energy, is the availability of energy itself, and those resources that provide it. As we, inevitably, use up high grade ores, and move on to poorer quality deposits, in which the desired element is increasingly diluted by other materials, the energy input to the whole extractive and processing mechanism increases: in terms of the production of energy resources, this is expressed as declining Energy Return on Investment (EROI). As the quality of mineral deposits declines, the volume of material that needs to be exhumed from the Earth, and processed, enlarges relentlessly, leading overall to increasing amounts of waste for each mass unit of metal, or other element recovered, and much more additional energy is needed. A coupling between the declining quality both of ores and energy sources can only compound the situation.
It has been indicated that there are insufficient, proven, reserves (and resources too, in certain cases) of several metals required to build a fully renewable energy system, to meet the global demand for energy that is expected by 2050. For scarce elements, recycling is indicated to be of limited value. It is possible that incorporating potentially less efficient technologies (but based on elements that are more widely available) might prove a viable strategy for reducing the risks of supply constraints. The future development of renewables may also rely on the recovery of materials from conflict zones and other politically unstable regions, which could pose problems for its large-scale expansion. Moreover, how such a fully renewable energy system might be maintained, beyond 2050, remains a serious open question. Meanwhile, major changes in our global demand for energy are necessary, and it may be wise to spend the fossil fuel equivalent of our remaining carbon budget on the extraction of metals required for low-carbon energy technologies.
Current consumption of resources means that the global materials base is unsustainable, and it is necessary to optimise our use of energy, to close material cycles, and to curb irreversible material losses, of all kinds. Mining of sand and gravel, used to furnish concrete, glass, asphalt and electronic devices, has risen to the point that their supply too is a matter of concern; levels of freshwater use are also now approaching newly defined planetary boundary limits. Natural resources are being consumed on an unprecedented scale, and currently, an annual 92 billion tonnes of raw materials are being extracted, which corresponds to around 12 tonnes for every person on the planet. The timescale of our intentions regarding the use of resources is critical, and the question of whether technology can solve our current problems, and meet future needs ‘sustainably’, has yet to be answered fully. Perhaps such considerations of what is sustainable only properly make sense, if societal viability over the duration of a civilisation (say, 500 years) is planned for; yet we have only the next few decades, at most, to undertake the appropriate actions to establish this. In regard to the sustainable use and regeneration of essential natural resources, it seems likely that the Earth Stewardship scenario, with the design system of permaculture as a creative response pathway towards achieving it, may be the most effective option.
The chemical elements
A chemical element is defined by its atomic number (i.e. by the number of protons in its atomic nucleus, as represented by the symbol, Z). 1 This definition allows for the existence of isotopes, for example, those of carbon, 12C, 13C, 14C, all of which have 6 protons in their atomic nucleus (Z = 6), but, respectively, 6, 7 and 8 neutrons, thus totalling 12, 13 and 14 nucleons, to achieve each particular mass number (atomic weight). A total of 118 elements has, so far, been characterised, and while the first 94 of the series have been identified to exist naturally on Earth, the later 24 were produced synthetically, by transmutation of the nuclei of naturally occurring elements. At least one stable isotope is known for 80 of these elements, but the isotopes of the other 38 are all radioactive (radionuclides), and decay into other elements, at rates set by their particular half-lives. 1 On the basis of mass, it is iron that dominates the elemental composition of the Earth, while, in the Earth’s crust, the most common element is oxygen. 1 Native (uncombined) forms of elements such as carbon, sulphur, copper and gold were known to the ancients, while those civilizations which followed them discovered that elemental copper, tin, lead and iron could be extracted from their combined form with other elements (ores), such as sulphur and oxygen, by heating (smelting) them with charcoal; the remainder of the naturally occurring elements had been identified by 1950. 1
Hydrogen and helium are the lightest of the chemical elements (Z = 1, Z = 2), and are thought to have been created by Big Bang nucleosynthesis (along with minor amounts of lithium and beryllium), during the first 20 min of the Universe’s existence,
2
in a mass ratio of around 3:1 (or an atomic ratio of 12:1).
3
Most of the other, heavier, elements were created by various other processes of nucleosynthesis,
4
mainly occurring in stars, as is indicated in Figure 1. The Periodic Table
5
was devised to group together elements that share similarities in their chemical properties, an explanation for which was later provided by electronic structure theory. A broad, first distinction of the chemical elements can be made between

Periodic table showing origin of elements in the Solar System. Elements from carbon up to sulphur may be made in small stars by the alpha process. Elements beyond iron are made in large stars with slow neutron capture (s-process). Elements heavier than iron may be made in neutron star mergers or supernovae after the r-process.
Chemical elements in the Universe
Less than 5% of the total mass of the Universe is made of atoms or ions (in other words, chemical elements as we understand them), which represents around 15% of its total matter, the remainder (85%) being dark matter, 7 whose nature is unknown, beyond that it contains no protons, neutrons or electrons. The remaining mass of the universe is attributed to dark energy (a source of anti-gravity, that is driving an accelerating expansion of the universe), 8 the constitution and qualities of which are more nebulous still; however, without its existence, the apparent near-flat geometry of space cannot readily be equated with the total amount of matter in the Universe. Thus, according to the Cosmic Microwave Background (CMB) spectrum, the total amount of matter in the Universe (including baryons and dark matter) accounts for only about 30% of the critical density, which implies that the remaining 70% must exist as an additional form of energy. 9 From measurements made, over 7 years, using the Wilkinson Microwave Anisotropy Probe (WMAP) spacecraft, it was estimated that the Universe comprises 72.8% dark energy, 22.7% dark matter and 4.6% ordinary matter (Figure 2) 10 ; however, on the basis of Planck spacecraft observations of the CMB, made in 2013, a more accurate estimate was obtained of 68.3% dark energy, 26.8% dark matter and 4.9% ordinary matter. 11
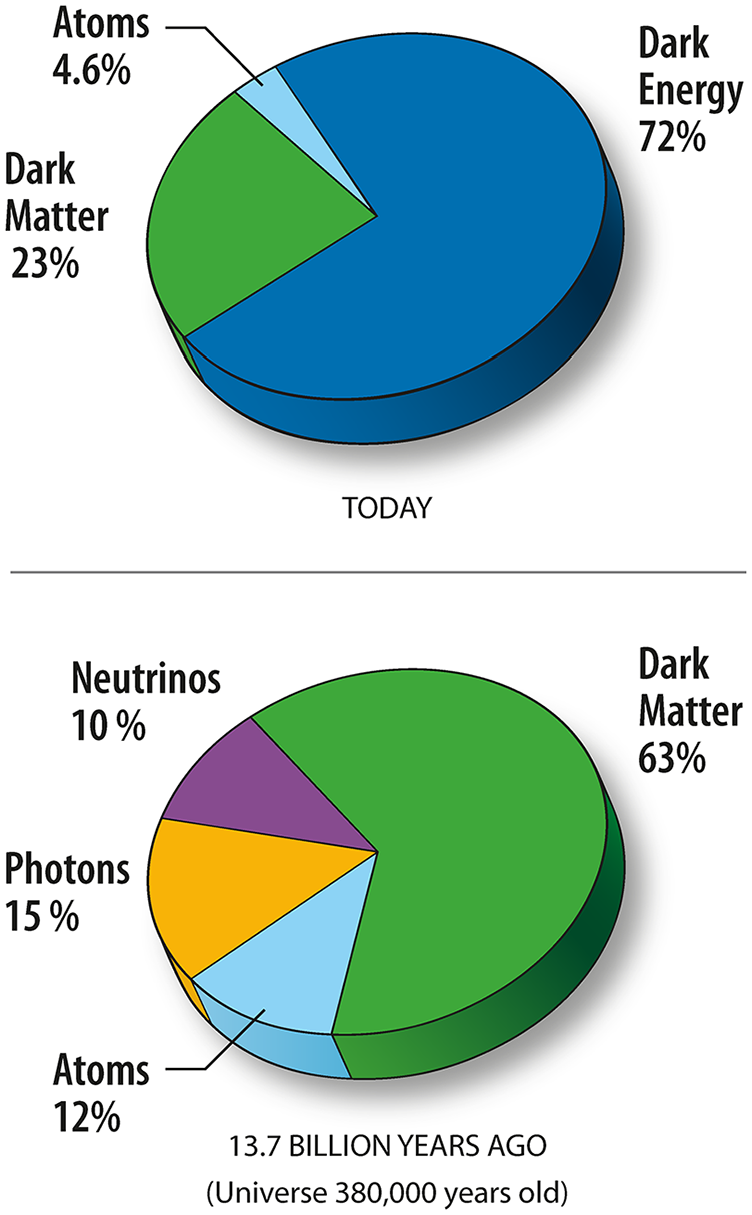
Estimated division of total energy in the Universe into matter, dark matter and dark energy based on 5 years of WMAP data.
Abundance of chemical elements in the Universe
On the basis of spectroscopic measurements, estimates of the mass fractions of the 10 most abundant elements in the Milky Way Galaxy have been made, as are listed in Table 1 12 :
List of the 10 most abundant elements in the Milky Way Galaxy. 12
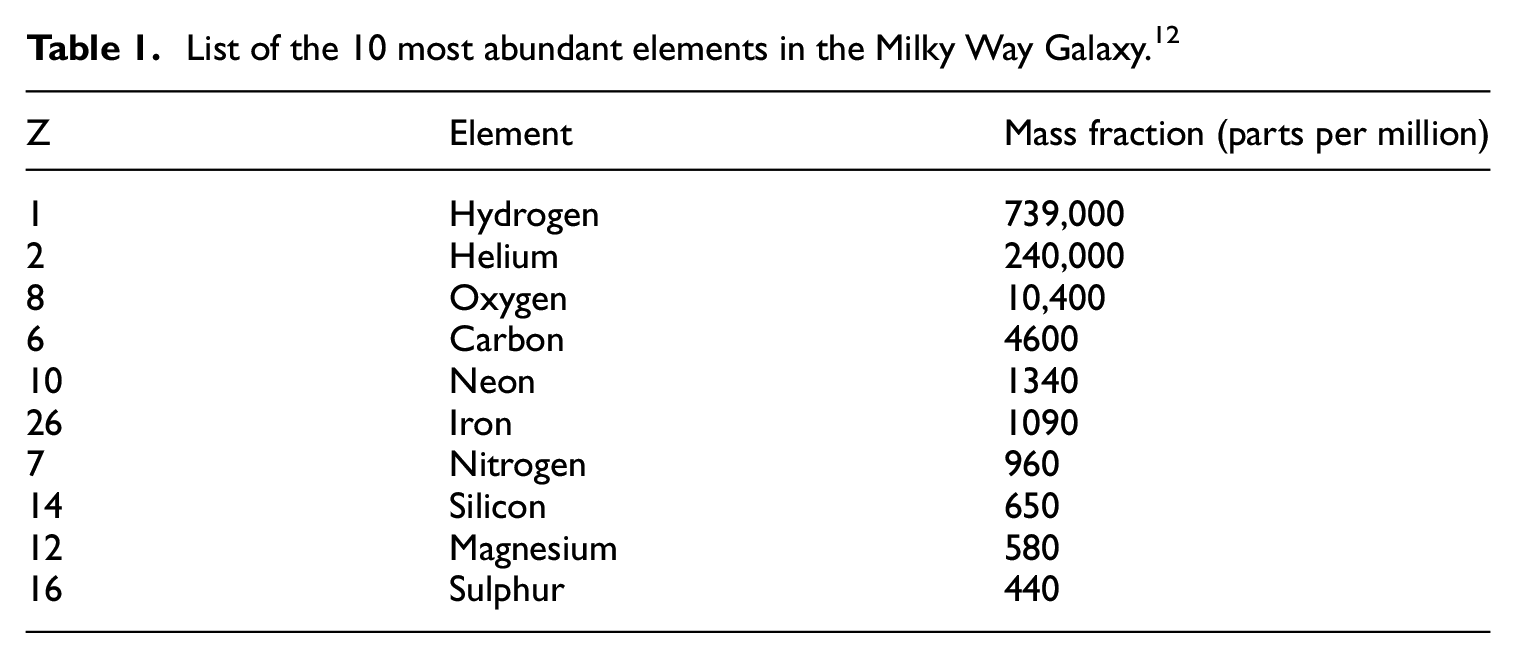
The standard cosmological model accounts for the abundance of the lightest elements, since, as we have noted, they were mostly produced within the first minutes of the universe’s existence, by Big Bang nucleosynthesis, while heavier elements were mainly produced within stars (Figure 1), and at much later times. Although roughly 74% and 24% of all baryonic matter in the universe is in the form of hydrogen and helium, 3 respectively, the far less abundant ‘heavy elements’ nonetheless have an appreciable influence on different kinds of astronomical phenomena. 13 For its mass, 56Fe appears surprisingly abundant (Table 1), but this is because it is the most stable element that can be synthesised from α-particles ( 4 He nuclei: each nucleus being a bound state of 2 neutrons plus 2 protons): 56Ni arises from the ultimate combination of 14 4He nuclei but is radioactive and decays to form 56Fe. Elements of still greater atomic number are created by energy-absorbing processes in large stars, and, in general, as their atomic number increases, their abundance in the Universe (and on Earth) decreases. 13
Abundance of chemical elements on Earth
The mass of the Earth is approximately 5.98 × 1021 tonnes, and the planet, overall, has an elemental composition (by mass) of 32.1% iron, 30.1% oxygen, 15.1% silicon, 13.9% magnesium, 2.9% sulphur, 1.8% nickel, 1.5% calcium, 1.4% aluminium, with the remaining 1.2% being the sum of trace quantities of other elements. 14 Hence, in comparison with the bulk composition of the solar system, the Earth appears to have lost a substantial fraction of its volatile elements, that is, H, He, Ne, N and also C (in the form of volatile hydrocarbons). However, in terms of the heavier elements, the distribution is more or less typical of the ‘rocky’ inner planets, 14 which formed in the thermal zone where heat from the Sun drove volatile materials into space. It is significant that oxygen is the second-largest elemental component of the Earth’s mass (and largest fraction in terms of number of atoms), and this is primarily due to the presence of silicate minerals which have high melting and boiling points, and very low vapour pressures. 15 Although estimates have been made of the elemental composition 13 of the Earth’s atmosphere, oceans, mantle and core, it is the availability of those elements (and other raw materials) in the upper continental crust that sets limits to the creation and expansion of particular technologies that require them.
Thus, by mass, the Earth’s crust consists of 13 46% oxygen, 28% silicon, 8.2% aluminium, 5.6% iron, 4.2% calcium, 2.5% sodium, 2.4% magnesium, 2.0% potassium and 0.61% titanium, with the abundance of other elements being less than 0.15% each. Figure 3 illustrates the relative atomic-abundance of the chemical elements in Earth’s upper continental crust, in which many of the elements are classified into different (and partially overlapping) categories:
rock-forming elements (major elements are identified in the dark green field, and minor elements in the light green field);
rare earth elements (REE; lanthanides, La-Lu and Y; are marked by blue chemical symbols);
major industrial metals (with a global production of greater than ∼30,000 tonnes/year) are identified by red chemical symbols;
precious metals (marked with purple chemical symbols);
the nine rarest ‘metals’– the six platinum group elements, plus Au, Re and Te (a metalloid) – are shown in the yellow field; due to their solubility in iron, they are concentrated in the Earth’s core, and hence only present in the crust in relatively low abundances. In relation to its cosmic abundance, tellurium (Te) is the single most greatly depleted element in the bulk silicate Earth, and this is because not only is it concentrated in the planetary core in the form of dense chalcogenides, but the element had already undergone severe depletion by pre-accretional sorting in the nebula, in the form of highly volatile H2Te. 15
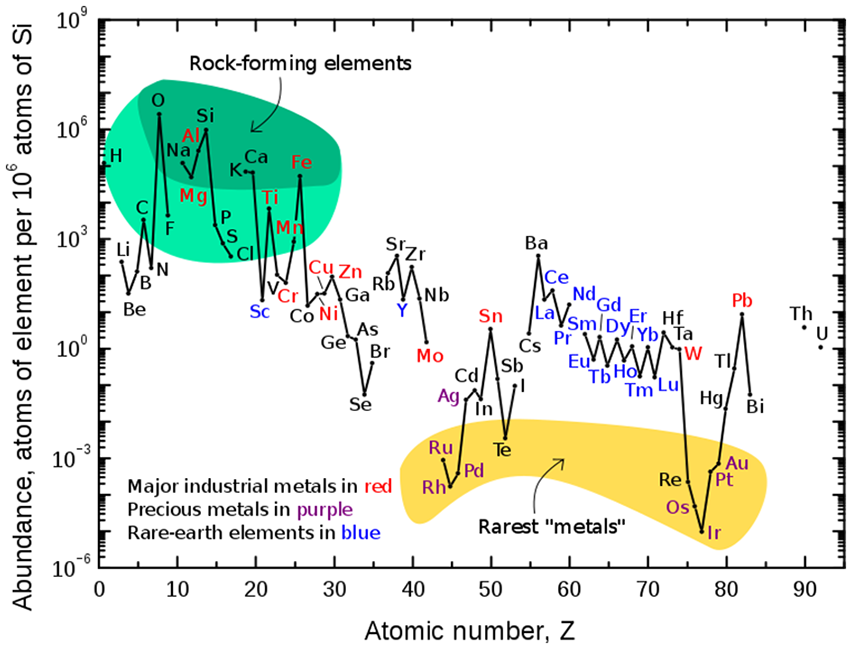
Abundance (atom fraction, on a logarithmic scale) of the chemical elements in Earth’s upper continental crust as a function of atomic number. The rarest elements in the crust (shown in yellow) are rare due to a combination of factors: all but one are the densest siderophiles (iron-loving) elements in the Goldschmidt classification, meaning they have a tendency to mix well with metallic iron, depleting them by being relocated deeper into the Earth’s core. Their abundance in meteoroids is higher. In addition, tellurium has been depleted by pre-accretional sorting in the nebula via formation of volatile H2Te. 15
Rare earth elements
Despite the term ‘rare’, a number of REE have abundances in the Earth’s crust that are similar to chromium, nickel, copper, zinc, molybdenum, tin, tungsten and lead, and even thulium and lutetium (which are the least common of the group) are almost 200 times more abundant than gold. 13 Nonetheless, in contrast to the more usual base metals and also precious metals, REE tend not to become concentrated in exploitable deposits (ores), with the result that the majority of their global supply originates from only few sources, mainly China (Figure 4). 16 As a further complication to the production of a particular rare earth metal in a state of purity, for a given application, the fact that all members of the group have rather similar chemical properties makes it quite difficult to separate them from one another, and they tend to occur in deposits that contain mixtures of them. There are two factors that contribute to the different abundances of particular REE in the upper continental crust of the Earth: first, those with even atomic numbers (e.g. 58Ce, 60Nd) are present in greater cosmic and terrestrial abundances than those with odd atomic numbers (e.g. 57La, 59Pr), and second, the larger ionic radii of the lighter rare earths tends to make them less compatible, than their heavier counterparts, with rock-forming minerals, and so they tend to partition more strongly into a melt phase, while the heavier elements may prefer to remain in the crystalline residue, particularly if it contains minerals like garnet that can accommodate them well. 17 Hence, all magma formed from partial melting has greater concentrations of the lighter than the heavier rare earths, while individual minerals may be dominated by either heavier or lighter elements, depending on which range of ionic radii best fits into the crystal lattice. 17 In most rare earth ores, the first four elements of the series, lanthanum, cerium, praseodymium and neodymium, account for somewhere in the range of 80%–99% of the total amount of rare earth metals present. 17
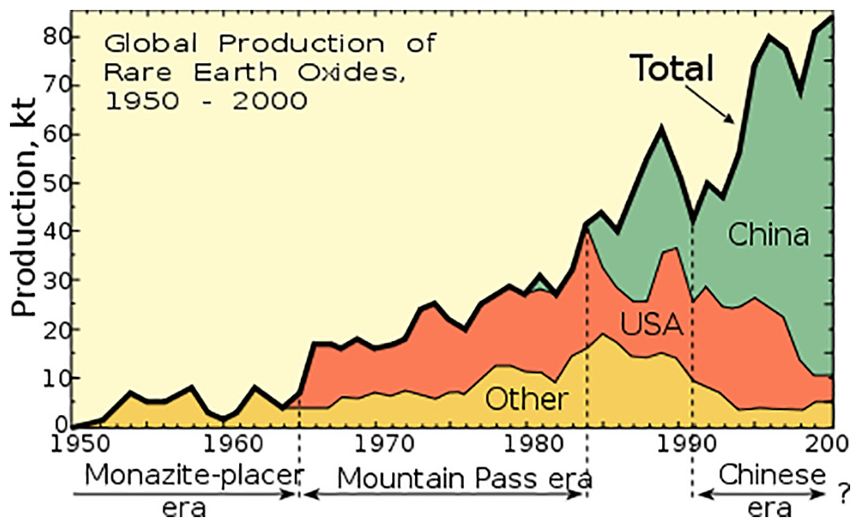
Global production of rare earth metal oxides, 1950–2000.
Endangered elements
The growth in the population of humans on Earth (1.1%/year) 18 has been accompanied – and partly driven – by an accelerating rate of extraction of materials from the Earth (Figure 5), which are consumed to provide energy and for other purposes. This has prompted various different authors to express their reservations (e.g. in previous studies19–22 and articles cited therein) as to how much longer this can be maintained, before the planetary boundaries of the Earth are transgressed. 23 Although many studies regarding this matter have been viewed as being more of academic than practical significance, 22 it is a truth that the implementation of new technologies, and their long-term viability, is dependent on the availability of essential, and mostly finite, resources. Deposits of minerals and fossil fuels are a one-off bestowal to us from the Earth’s geological history, and any rates of their renewal are insignificant, in comparison with the timescale of human, technological progress. It is not merely that our rate of resource consumption has risen in step with population growth, but has relentlessly accelerated beyond it, and for example, consumption of energy trebled 24 (40,589–144,424 TWh during the same 50 year period (1960–2011)) when the population ‘merely’ doubled 25 (3 billion–7 billion), meaning a per capita increase of 50%: this may be taken as a proxy for humankind’s consumption of materials, more generally. In 2018, global energy use increased by 2.3% (its fastest pace in a decade), 70% of which was provided by an increased burning of fossil fuels. 26 Given their finite nature, whatever of these resources we use now leaves a deficiency of them – tonne by tonne – for future generations. In order to fabricate cars, flat-screen TVs, DVD players, mobile phones and related devices, compact batteries, miniaturised machinery and cameras, 50 different metals and other elements are required; increasing technological complexity is also an issue, and for example, some 60 different elements are now employed in the manufacture of computer chips, while only around 10 were necessary in 1980. 27 Indeed, the sufficiency of various elements that are used to make essential components of various items in common use, including mobile phones and other handheld devices, and as might be required to vastly expand renewable energy production, as is necessary to reduce carbon emissions and meet climate change targets, is a matter of particular concern (see section on ‘Shortage of elements for renewable energy?’).

Mir diamond mine in open pit diamond mine located in Mirny, Sakha Republic, in the Siberian region of eastern Russia. The mine is more than 525 m (1722 ft) deep, has a diameter of 1200 m (3900 ft), and is one of the largest excavated holes in the world. The mine is now inactive, but is so deep that the surrounding airspace was closed to helicopter traffic because several were pulled downward by the airflow. 29
The viability of the supply of a particular material is often estimated, in a handy, but crude, ‘back of an envelope’ way, on the basis of the reserves-to-production (R/P) ratio,19–22 which is the ratio of the size of the reserve base to the annual rate of production: thus, for example, a 100,000 tonne reserve produced from 1000 tonnes per year should last for 100 years. This is, of course, a naive piece of arithmetic, since no material can be produced at a constant rate, right up to the bitter end, and instead, a production peak is to be expected, as the quality of particular deposits/ores declines, and the energy input, per tonne of a material recovered, increases relentlessly – eventually to the point that further extraction from a particular source is no longer worthwhile. (In terms of the extraction of resources to provide energy, such as oil, gas or coal production, the term Energy Return on Investment (EROI), is used, to describe a ratio that declines as more energy must be put into the system to recover energy from material of declining quality; see Appendix 1). Hence, production is maintained for longer that the R/P value indicates, but with an inexorably declining annual output. However, a simple economic prediction is that, as the lifetime of a reserve shortens (on an R/P basis), its price will increase, with the consequence that other sources, once thought too expensive, may enter the market. 19 This tends to make the original reserve last longer, in addition to the volume from the new reserves (this can, however, result in an oversupply to the market, as we have seen recently with oil, which causes the price to crash 28 ). Price is a critical determinant of the size of the reserve, compared to the resource, as might be anticipated from the definitions for both: the reserve is what can be both technically and economically (and legally) recovered, while the resource is the reserve plus everything else that is estimated to be in place, that could potentially be produced. Thus, as the price of a material increases, more passes from the resource to the reserve category and vice versa. 29
Various methods exist for estimating the rate and production lifetime for a given material, including those based on fitting various (e.g. logistic or Gaussian) functions to existing production data, or using system dynamics models.20,21 Thus, production peaks were predicted to occur by 2050 for gold, silver, copper, zinc, lead, indium, iron, chromium, molybdenum, nickel, platinum group metals, phosphorus, all fossil fuels (oil, gas, coal), and also global fish catches and the global area of tilled soils.20,21 Peaking, as in ‘peak oil’, does not mean the point of running out of something, but where the production rate reaches its maximum level, and thenceforth, that the output of a given material (from the particular source being evaluated) can be expected to decline relentlessly, the material eventually becoming scarce.
30
The concept of the ‘burn-off time’ has been introduced,20–22 which has the same formal definition as the R/P ratio; however, it has been emphasised that, while this is applicable in a stagnant economy (constant production rate), in a growing economy, it overestimates the production lifetime.20,21 From a pragmatic perspective, a low R/P value can be taken to draw attention to an element or raw material which might become scarce in the not too distant future, or whose production might prove difficult to enlarge sufficiently to meet rising demand from the expansion of new technologies. Sverdrup and colleagues20–22 have examined the implications of ‘peaking’ of the supply of metals, minerals and also of energy, wealth, food and population, in the context of devising appropriate policy for a sustainable society.20–22 Burn-off times were estimated using either a Hubbert curve, or a system dynamics model, under different scenarios for recycling, materials use and population, and the ‘time to scarcity’ (defined as being where the production rate has fallen to 10% of the maximum –‘peak’– value) is expressed as being
21
‘the double of this estimate’, which is broadly in accord with the values shown against burn-off times in Table 2 of Ragnarsdóttir et al.
22
A time to scarcity is indicated for gold at about 2070; and while iron is (as noted in section ‘Abundance of chemical elements on Earth’) highly abundant on Earth, there are supply limitations for extraction of the metal at a reasonable cost, with indications of a first production peak at 2030, probably followed by a second one at around 2060, due to increased prices, recycling and the consequences of a global recession.20,21 Nickel, copper, zinc, manganese, tin, molybdenum, lead, tungsten (wolfram), niobium, helium, arsenic, lithium and antimony were all indicated as elements whose supply might also become insufficient within the present century, but, in all cases, the results indicate that any supply shortages could be obviated by increasing recycling to a rate of 95%.20–22 Furthermore, if the global population were to be reduced to 3 billion, along with an additional
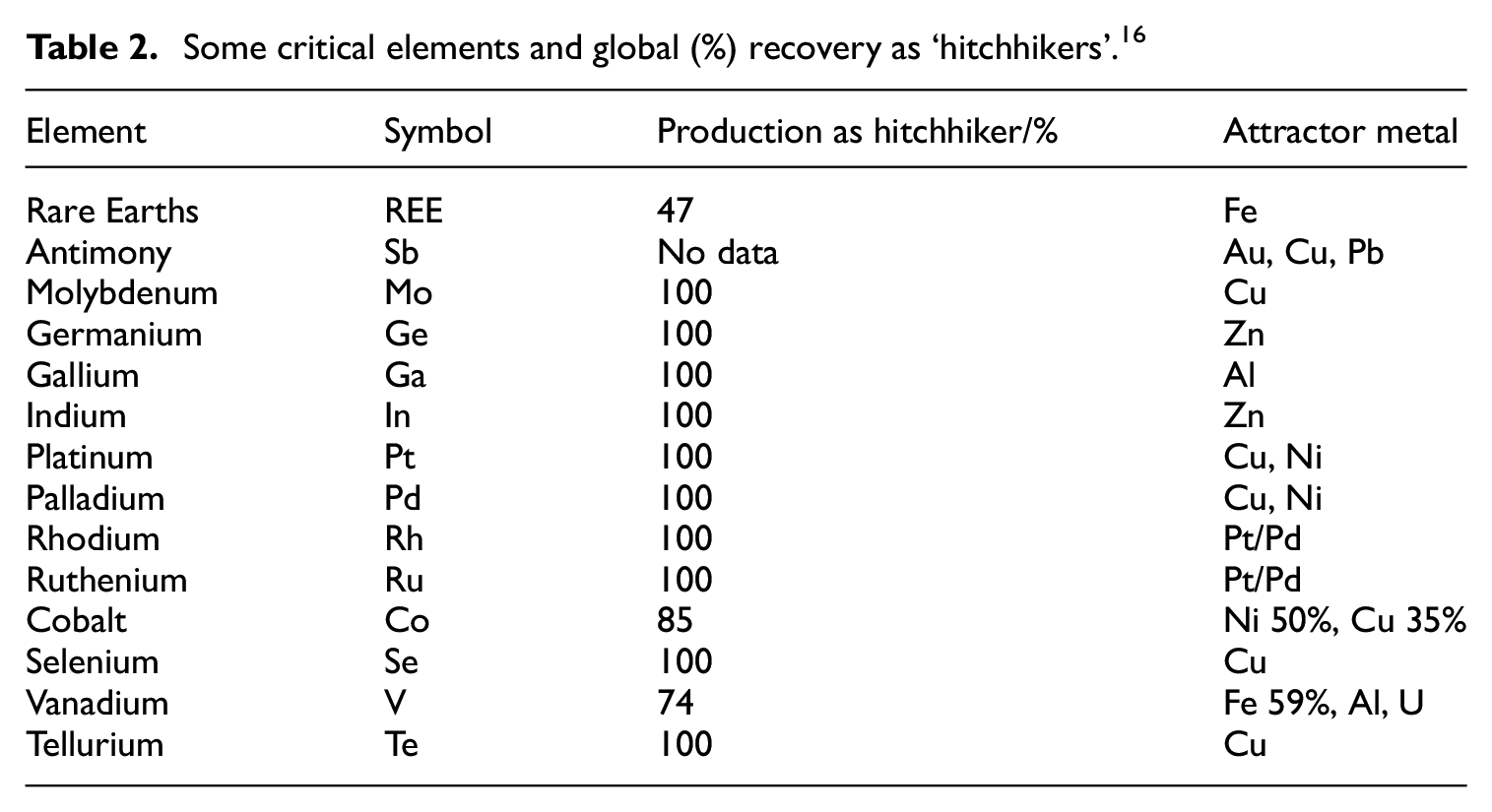
Some critical elements and global (%) recovery as ‘hitchhikers’. 16
The supply of phosphorus is a much debated and complex topic, but a production peak at some point during the present century is possible, 30 with potentially very serious consequences for industrialised agriculture. 30 It has also been determined that the global area of tilled soils peaked in 2005, while global fish production peaked in 2002–2003, with the alarming forecast that, by 2060, the fish catch will have decreased to just 10% of its maximum level.20,21 Such factors as limited provisions of phosphorus and soil to grow food, along with falling fish catches, are likely to restrict global food supplies and therefore set limits to world population. In some regions, at least, the population is projected to peak at some point during the present century (although probably not globally, mainly due to robust growth prevailing in Africa). 25 Peaking of global conventional crude oil production is indicated to have occurred 21 in 2008 (which is probably accurate, since the global oil supply has been enlarged over the past decade only by light tight oil production from shale, using hydraulic fracturing (‘fracking’), along with production of extra-heavy oil from oil sands31,32), while ‘peak coal’ is indicated 21 in 2018 and global ‘peak wealth’ in 2035. 21 In summary, it is necessary to close all cycles of finite materials, and restrain production from renewable resources to significantly below their critical extraction rates, if sustainability is to be achieved. Further details of the techniques involved and their application to various different resources are available in a book-length article, by Sverdrup and Ragnarsdóttir. 33 Thus, one model predicts 34 that, for copper, nickel and zinc, scarcities may emerge at some point during the next 40 years, while, within the next 30 years, the main sources of iron, aluminium and copper will be scrap metal. However, it is more probable that we will run out of energy to produce – and money to buy – iron, aluminium or copper, before we run out of the metals themselves. 34 A production peak in around 2040 was predicted 35 for stainless steel of the type containing Mn–Cr–Ni, because of nickel supply limitations, followed by a production decline around 2045; however, it was concluded that the price of stainless steel will thus be forced to rise, and stimulate more recycling, thus partially offsetting its scarcity. However, if the rate of recycling is increased to above 80%, it is predicted that there is sufficient nickel, chromium and manganese to last for several centuries. 35
Another analysis
36
indicates that peak lithium may be reached in 2060, followed by a slow production decline; however, this scenario might be ameliorated through the introduction of large-scale recycling, and, at least initially, it should be possible to meet rising demand from the expected number of new electric vehicles. After 2050, however, and if the replacement of
A theoretical assessment was made 37 of those minerals and metals commonly used in the construction industry, and how much of each might be needed up to 2050 (based on growth trends from 1950) in order to develop infrastructure for the mass production of metal-intensive items such as cars, and agricultural machinery, given projected demand for goods and food, by then. The model predicts that demand for aluminium may rise by a factor of 8 from current levels, while the production of cement would need to increase sevenfold, and requirements for lead would double by 2050. 37 The implications of the necessary expansion of mining for different elements, across the nations of the world, to expand technologies as required to meet climate change targets, as set-out both in the 2015 Paris Agreement and in the United Nations UN Agenda 2030, have been surveyed by the Earth Science Community. 38
Hitchhiker elements
There are a number of critical elements that are mainly not produced in their own right, but are instead by-products of, or ‘hitchhikers’ on, extraction from primary, or ‘attractor’ metal deposits (Table 2). 16 Thus, once the supply of the main attractor is exhausted, so are those of its hitchhikers: hence, the lifetimes of Ga, Ge, In and Te, which are hitchhikers on the production of Zn, Al and Cu, are limited by the production timescales of the main (base) metals. Furthermore, to meet a rising demand for a given hitchhiker, it would be necessary to expand the rate of production of the major ‘attractor’ metal; improve the extraction efficiency of the minor by-product from the base metal ore; find alternative, and independent, sources of it, or substitute for the material with something else. The first option is not likely to prove feasible, or at least not economically worthwhile, unless demand for the base metal were to increase dramatically, while the other options require technological innovations and increased initial capital investment. 16 A better strategy is through improved resource recovery technologies, better recycling and by demand reduction and substitution with alloys or more accessible elements. 16
There is a further, geopolitical, dimension 16 attendant to the above set of critical elements, since the REE, W, Sb, Mo, Ge, Ga and In, all mainly come from China; most of the world’s Pt, Pd, Rh, Ru and V are produced in South Africa (which provides 89% of the world’s platinum group metals); while 60% 39 of the global supply of Co is mined in the Democratic Republic of the Congo (DRC), with its further implication as a ‘conflict mineral’ (see section on ‘Conflict minerals’). When a single nation has a monopoly on the supply of a particular material, questions naturally arise over how secure this might prove in the longer term, and those nations whose manufacturing/technology base and economy are underpinned by imports of particular critical elements begin to search elsewhere for alternative sources. In the case of REE, various companies and countries that depend on them are making assiduous efforts to gain control of mineral rights 16 in South Africa, Greenland and Australia, while, China, though plentiful in REE, is securing access to other minerals across the world. It is speculated that President Trump’s reported recent interest 40 in ‘buying’ Greenland may have been motivated by its substantial mineral wealth, particularly REE, although the chances of this acquisition actually transpiring seem fairly low. 41
Recycling and substituting
Given their likely availability against projected demand, it is clear that metals such as copper, zinc, the platinum group metals and tantalum, along with other potentially limited resources, must be used sustainably. 22 At the outset, the use of these materials can be reduced through better design of products containing them, which can be made more robust and durable, in antithesis to the concept of ‘planned obsolesce’. In terms of recycling, however, certain limitations are imposed by how a particular material has been used: thus, zinc, for example, is difficult to recycle once it has been dispersed across large area, metallic surfaces, where it is typically applied in relatively low concentrations for galvanising purposes. 22 A general principle has also been demonstrated that the rate of recycling of different metals increases in proportion with their price. 42 Therefore, increasing scarcity (and price) may well encourage more effort towards achieving higher recycling rates; however, anywhere near full recycling is only likely to be approached once the various metals and other elements have become relatively expensive.
According to the United Nations Environment Programme (UNEP), 43 the recycling rates of various metals are >50% for platinum, palladium, rhodium, niobium, cobalt and chromium; 25%–50% for magnesium and iridium; 10%–25% for ruthenium and tungsten; 1%–10% for antimony; and <1% for rare earths (both light and heavy), osmium, beryllium, gallium, germanium and indium. 43 Reasons for the very low recycling rates, especially of metals and semi-metals such as indium, germanium, gallium and the REE, as are used in electronics and energy applications, are discussed elsewhere in the context of feedback control of mineral supply. 44 The specific case of indium has been addressed in another study, 45 in which the importance of end-of-life collection of the metal (which is currently almost nonexistent) to improve its recycling rate is stressed. 45 This point was also highlighted in a recent European Commission report. 46
In order to obviate such potential scarcities of critical materials, means to replace scarcer elements by more abundant and readily obtainable alternatives are being investigated (see section on Technologies based on Earth-abundant elements). Given that global industrial production has expanded by a factor of 40 during the past century, 22 demand for a supply of materials to support this growth has increased accordingly. However, for a number of metals, the available virgin stocks appear too small to provide for all people on Earth, at a level of material consumption equal to that in the industrialised nations, at least using contemporary technology. 47 It is, in principle, possible to extract from lower grade ores, to mine them from greater depths within the Earth, or to recover metals from seawater, but the associated energy costs may prevent such approaches being actualised. Energy requirements may also prove prohibitively high for the practical recovery of elements and metals that were initially recovered from concentrated deposits, but then ‘diluted’ by their subsequent use and dissipation into the environment, at least on the very large scale.22,29
By way of a substitution strategy, 22 it is for example, possible to make pin-head capacitors in mobile phones from aluminium, to replace the yttrium, tantalum or hafnium, that they are currently made from; while silicon, silicon carbide or carbon nanotubes might be used to make transistors that are currently made from germanium and indium; and it has been found that silicon/polyvinyl chloride can be employed to make solar cells that have twice the energy conversion efficiency of thin-film cells based on indium and gallium. 22 Nothing, however, can replace phosphorus, which is required by all living organisms, 30 and the proposed recovery of phosphorus from seawater would most likely fail due to the necessarily very large energy inputs required to produce it on the scale that it is consumed globally. 29 It appears that production peaks will occur on the longer timescale, first in mining and producing from high grade reserves, followed by that from low grade, and finally from the ultralow grade reserves.30,48 Hence, if we wish to maintain its overall supply, it may be necessary to recover phosphorus from animal and indeed human waste (Figure 6). 30 There are complex issues over what the demand will be for phosphorus in the future, as measured against a rising population 25 (from 7 billion to over 9 billion in 2050), and a greater per capita demand for fertiliser to grow more grain, in part to feed animals and meet a rising demand for meat by a human species that is not merely more populous but more affluent. 30 As a counterweight to this, we may expect that greater efficiencies in the use of phosphorus – including recycling from farms and of human and animal waste – will reduce the per capita demand for phosphate rock. 30 A further game changer may be a declining future global oil supply (‘peak oil’), since phosphate is mined and recovered using machinery powered by liquid fuels refined from crude oil. 30 Hence, there is no unequivocal case that we can afford to ignore the likelihood of a supply–demand gap for phosphorus occurring sometime this century, and it would be perilous to do so. 30
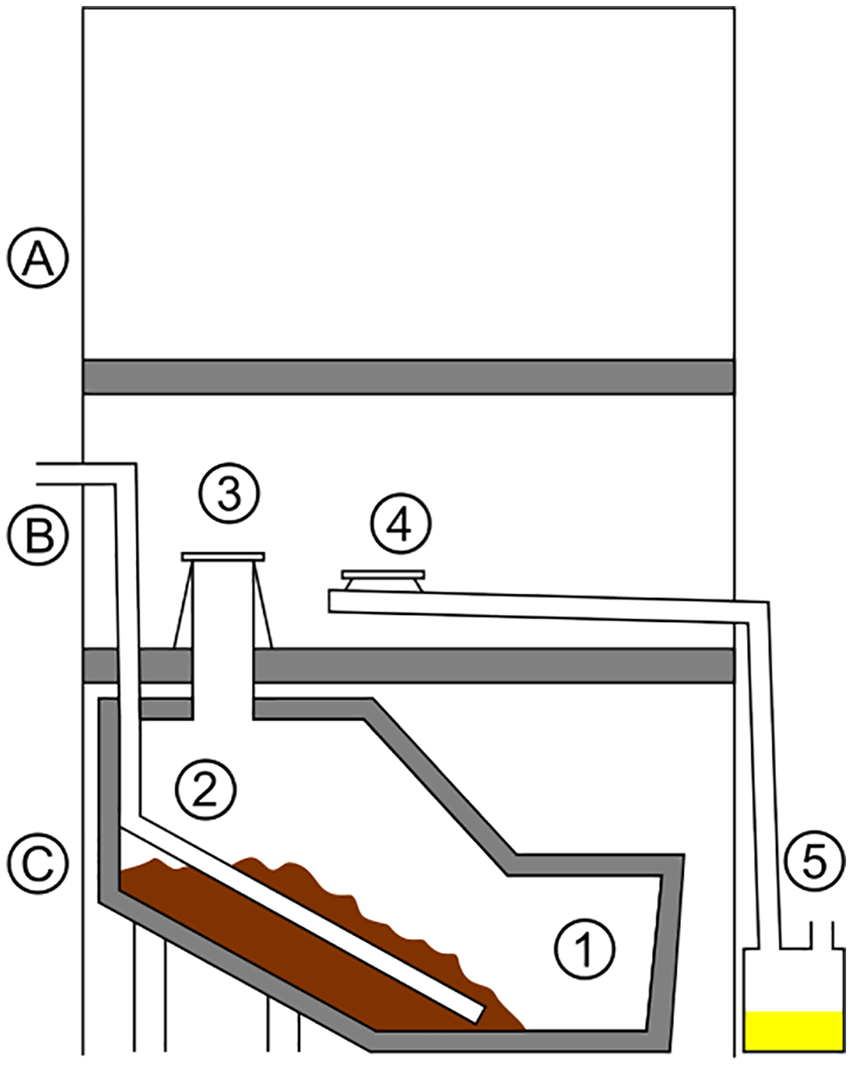
A schematic of a composting toilet with urine diversion and dehydration (UDD). A. second floor; B. first floor; C. ground floor; 1. humus compartment; 2. ventilation pipe; 3. water closet; 4. urinal; 5. urine collection and dehydration.
A revised approach to the issue of substitution of critical elements and other raw materials, by more abundant and readily available versions, has been presented in a report
49
by the European Commission, which is summarised there as: In the revised methodology for criticality assessment, the availability of substitutes is considered as a reducing element in both the economic importance (SIEI) and the supply risk (SISR) dimensions. The assessment only takes into account the proven substitutes that are readily available today and able to reduce the consequences of a disruption and/or influence the risk of a disruption. Commercial information and published patents are only used to identify proven substitute alternatives readily available and applicable at the market today. Neither ‘substitutability’ nor ‘potential future substitution’ is considered in this methodology.
The method involves the calculation of a Substitution Index, 49 in which the following factors and influences are taken into account:
Technical performance (extent to which the substitute can replace the functionality of a candidate raw material in an application, for example, it is very unlikely that a tantalum capacitor would be substituted with an aluminium capacitor because a mobile phone would weigh 1 kg).
Cost performance (costs often drive decisions in business).
Substitute production (availability of substitutes in sufficient quantities needs to be considered).
Substitute criticality (substituting one critical material by another is unlikely to decrease the RM supply risk in a given application).
Substitute by-/co-production (if the proposed substitute is mainly obtained as a by- or co-product, its supply is dependent on a demand for another raw material).
Full details of the procedure are given and illustrated for three materials – Indium, lithium and tungsten – in the Annex of the report. 49
A ‘criticality index’ has been established for 62 metals and metalloids within a three-dimensional ‘criticality space’, which factors-in supply risk, environmental implications and vulnerability to supply restriction. 50 The results show that extreme values of the index are driven by high primary production from few sources, where suitable substitutes are not readily available, and where the source is in a politically unstable region. It is concluded that, for many metals (such as gallium and selenium) that are important in emerging electronics technologies, supply risk is a major factor; environmental factors pose significant risks for the platinum group metals, gold and mercury; while supply restriction affects significantly metals used both in steel alloys (e.g. chromium and niobium) and in high-temperature alloys (such as tungsten and molybdenum). As noted in the section on ‘Hitchhiker elements’, metals that are largely, or entirely, produced as by-products from the production of other metals are especially vulnerable, as are those used in small quantities for highly specialised applications, with no effective substitutes. 50 A further study 51 was made by the same group, paying particular attention to the principal uses of 62 elements that underpin the materials basis of modern society; for 12 of these, the potential for substitution was found to be either inadequate or apparently nonexistent, and for none of them could a substitute be found for all its major uses. 27
Recycling from low-carbon energy sources: photovoltaic modules and wind turbines
If an electrical generation system is to be established, based on a wholesale expansion of low-carbon energy (renewables), it is clear that it will be necessary to recycle those elements that are critical for the fabrication of the devices employed to construct it, particularly thin-film solar cells and wind turbines. For silicon solar panels, landfill is currently the cheapest option for end-of-life treatment, 51 because the cost of recycling is probably greater than the value of the embodied materials that might be recovered; however, a lifecycle analysis has indicated that thermal recycling could become financially viable, so long as various cost criteria are met and appropriate technological improvements are achieved. 51 One proposed scenario involves the introduction of a delamination process that can recover silicon wafers intact and in high yield, but a manufacturing method is also required whereby these can be fabricated into high performance solar modules. 51 The International Energy Agency (IEA) has published a report 52 on the end of life management of photovoltaic (PV) panels, which categorises, separately, crystalline silicon cells, and thin-film cells made from cadmium–telluride (CdTe) and copper–indium–gallium–selenium (CIGS), due to the different materials and module structures attendant to each. While noting that some commercialisation of PV module-recycling is already underway, the report considers 52 trends in technological developments from the perspectives of patents and national R&D projects. One of the more challenging tasks in module-recycling is to remove the encapsulant from the laminated structure, for which various methods exist: thermal, mechanical and chemical. The recovery of metals (e.g. silver) from crystalline silicon (c-Si) cells may be accomplished by chemical means. 52 Another study 53 suggests that, due to the rapid growth in PV technology, in 2050, there will be some 5.5–6 million tonnes of modules coming to the end of their lives; currently, however, the volumes recycled are small, with only the European Union (EU) having a sufficiently strong regulatory framework to support the necessary expansion of the process. 53 Thus, while it is currently unprofitable to recycle c-Si cells, 53 it is likely that the practice will become mandatory, both on environmental grounds (reducing pollution and decarbonising the electricity supply) and to recover critical and valuable metals. The most widely employed thin-film technology is currently based on CdTe, with a market share of 65%, followed by CIGS at 25% and amorphous silicon (a-Si) at around 10%. 53 A survey was made of different methods for recycling thin-film cells, to recover elements such as Cd, Se, Ga, In and Te, and their relative merits were compared. 53
The wind energy industry body, Wind Europe, 54 has formed a partnership with the European Chemical Industry Council (Cefic) and the European Composites Industry Association (EUCIA), with the aim to improve recycling options for wind turbine blades (Figure 7), which are made from composite materials. Currently, they are recycled using cement co-processing: this involves the separation of the composite components, so that the glass fibres and fillers can be used to replace some of the raw materials normally used for manufacturing cement, while the organic fraction is burned, partially in place of coal as a fuel, thus lowering overall CO2 emissions by 16%. 54 However, it is proposed to investigate alternative recycling methods, such as mechanical recycling and pyrolysis, which potentially have a lower environmental impact. Presently, the wind energy sector uses around 2.5 million tonnes of composite material, although a considerable expansion of this energy source is anticipated in the coming years. 54 It is expected that, within the next 5 years, some 12,000 of the EU’s 130,000 wind turbines (from which 14% of the EU’s electricity was generated in 2018) will be decommissioned. 55 The issues attendant to decommissioning offshore wind farms are complex,56,57 but the rapid growth in wind energy, in recent years, is likely to become matched by an equivalent rate of dismantlement, and for the technology to be considered as a ‘clean’ energy source, a significant degree of repurposing and recycling, with minimal final disposal of the wind turbine components, is necessary. 58 While existing experience is limited, it seems clear that there are considerable knowledge gaps regarding the various steps involved, and due to the complexity of the offshore environment, and the considerable variation among different wind energy projects, it is vital to have clear procedural plans in place, and to make appropriate risk assessments, in advance of any specific decommissioning programme being undertaken. 58 There is also, currently, a lack of a specific regulatory framework for decommissioning wind farms, which complicates such issues as liabilities, and the drawing up of necessary plans both for the process itself, and in dealing with any consequent environmental impacts. 58

Wind turbines, Thanet wind farm. Thanet Wind Farm viewed from Joss Bay. The wind farm is 12 km off Foreness Point and covers an area of 13.5 square miles (35 km2), which includes numerous OS grid squares. The turbines are spaced at 500 m (1600 ft) with 800 m (2600 ft) between the rows. The wind farm has operated since 2010 and consists of 100 Vestas wind turbines. When it was built, it was the largest offshore wind farm in the world. https://www.geograph.org.uk/photo/5092483
In order to address the overall viability of renewable energy systems, a comprehensive study has been published
59
in the journal
Stockpiling
In order to address the potential problem of extreme price volatility, and interrupted supplies, caused by the depletion of cheap, ‘high grade’ mineral ores, and the necessary exploitation of lower grade ores, with attendantly higher extractive costs and diminishing economic returns, it has been proposed to create a ‘buffer’ by stockpiling critical materials. 63 Instability arises from the reaction of the market, which either pushes up prices, thus tending to reduce demand, or brings down prices, so that the producer suffers a loss or elimination of profits, and is forced to cut production. It is vital to ensure a sufficient supply of materials to maintain the economy, and to avoid price shocks, which may be a result of market idiosyncrasies, or conflicts, be they social or strategic. 63 The critical and rare metals are particularly vulnerable, as are needed, for example, to perform vital tasks such as the conversion and transmission of electrical energy, such as that produced by PV power plants, and as is required by the electronics industry. It has been proposed that by keeping stockpiles of mineral resources above ground, a buffer is created against both price and supply shocks. The notion of a ‘metal bank’ is proposed, to manage the supply of rare and sensitive minerals. 63 Other authors have reinforced 64 the importance of maintaining a supply of critical metals, from the viewpoints of politics, science and society, given that the quantities of mineral resources on Earth are limited. However, they question 64 whether the observed declining quality of mineral ores should be interpreted as a sign of resource exhaustion, and argue instead that this has more to do with increasing demand and the implementation of technological advances that have made mining them profitable, which they take as a positive sign. 64 Since this view is based mostly on the situation for copper mining in Australia, it is not compelling to extrapolate a case that technology will come to the rescue for the wider mineral base, and across the world. ‘Peak discovery’ was in 1996 for copper, and for of all kinds of materials, this is typically followed by a production peak 40 years later, 20 thus suggesting 2036 for ‘peak copper’. 20 Other economists have argued 65 that there is no certainty that higher-grade ores no longer exist in the environment and that humans have found themselves able to access ever-increasing amounts of material from often increasingly lower-grade sources; hence scarcity cannot be averred definitely. As the authors conclude, ‘we remain critical optimists’. 65 However, such optimism is based on a kind of faith that human technological ingenuity and innovation will ever prevail.
Modified periodic tables
As noted in the section ‘The chemical elements’, the periodic table 5 was devised as a revolutionary means to group together elements that share similarities in their chemical properties, an explanation for which was later provided by electronic structure theory. Initially, its discoverer, Dimitri Mendeleev, accommodated the chemical elements that were known in 1869, but confidently left ‘gaps’, into which were later slotted new elements as they were discovered. The essential form (Figure 1) of the more familiar periodic table was later devised by Glenn Seaborg, in 1945, showing the actinides as a second, f-block series. 5 There have been various different formats devised since then, and in 2011, the United Kingdom’s Chemistry Innovation Knowledge Transfer Network published a new, colour-coded, periodic table, 66 which highlighted as ‘endangered’ 44 of the known 118 elements whose supply will either be limited, or it will no longer be economically viable to extract or use them, whereupon they will need to be replaced by alternatives. In part, this situation is exacerbated by the inauguration and expansion of new technologies, meaning that demand for some elements has increased manyfold within just the last two decades. 66 These include neodymium (a rare earth element), zinc, gallium, germanium, helium, silver and phosphorus. A version of this periodic table was also published by the Royal Society of Chemistry. 67
To mark 2019 as the international year of the periodic table, in January 2019, the European Chemical Society
68
released a modified format for it, which indicates not only which elements are potentially ‘endangered’ but also their relative abundances by means of a logarithmic areal scale (Figure 8). The infographic in Figure 8 also indicates up to 30 of the different elements that may go into a smartphone, for over half of which we may struggle to meet demand, in the coming years, since 10 million phones
68
are replaced or discarded in the EU alone,
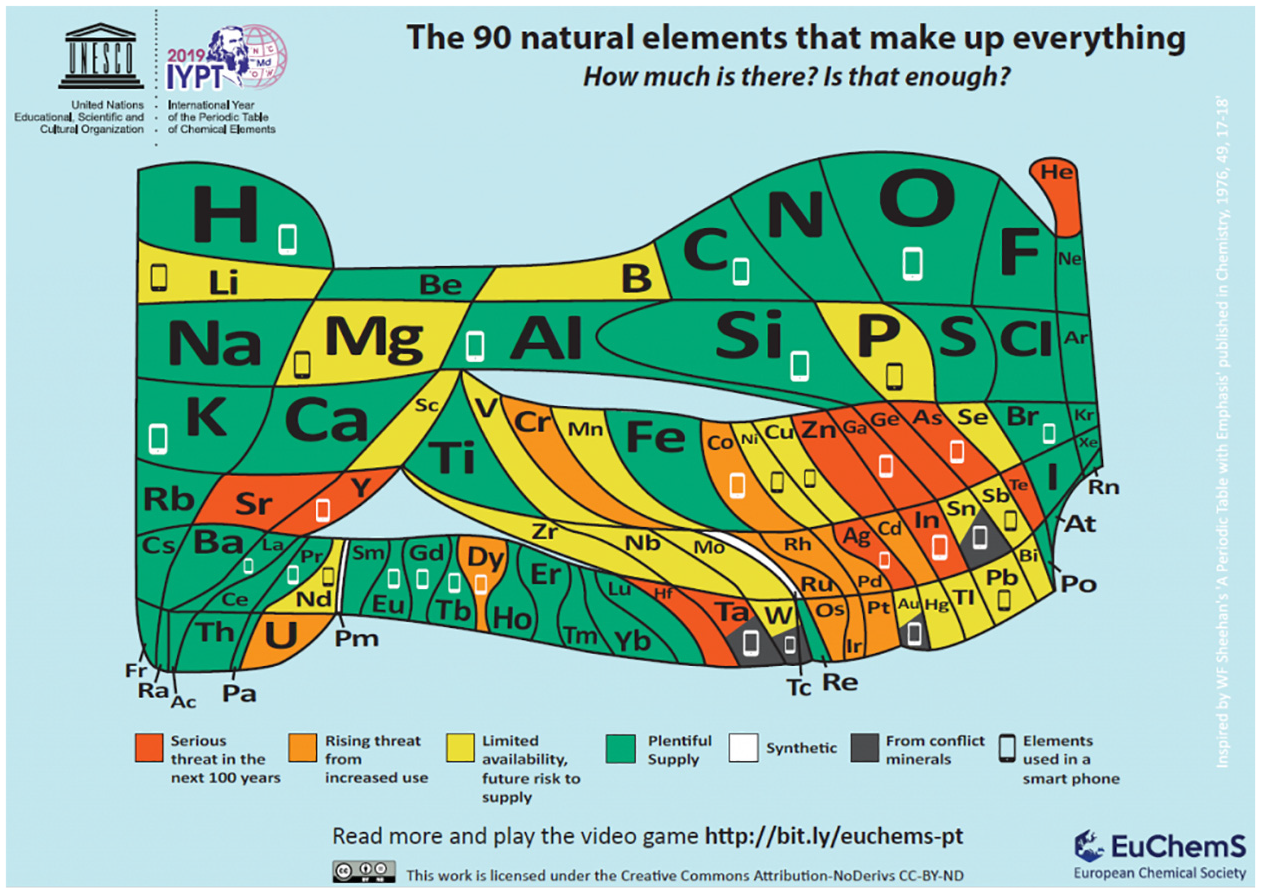
The 90 natural elements that make up everything. How much is there? Is that enough?
Shortage of elements for renewable energy?
The question of whether a fully renewable energy system, based on wind, water and solar, to provide for electricity, transportation, heating/cooling and industry, across the continental United States, can be installed 70 by 2050–2055 has been the subject of intense debate,71–73 and which became sufficiently acrimonious to result in a legal skirmish, 74 although the case was later dropped. 75 On the basis 76 of a mass balance of 29 metals and five different energy scenarios, it was concluded that the reserves of 8 metals (Cd, Co, Au, Pb, Ni, Ag, Sn, Zn) would probably have been depleted in advance of a large-scale, fully renewable energy system being in place by 2050. 76 Surprisingly, this seems to be the case irrespective of different levels of energy demand that were considered. It is thought that competition might occur over Cd, Co, Li and Ni, which already are widely deployed in traditional industries, with the expanding new renewable energy industry. 76 It seems likely that it will be necessary to make changes in global energy demand, along with the development of technologies that do not require speciality metals. It is also possible that incorporating potentially less efficient technologies (but based on elements that are more widely available) might prove a viable strategy for reducing the risks of supply constraints. 76 For scarce elements, recycling is indicated to be of limited value. 76 While choosing between either high-tech or low-tech technologies for future energy production is expected to have a considerable effect on resource depletion, it appears sensible to allocate some of our remaining carbon budget towards the extraction of metals, as is necessary to build a renewable energy system. 76
Although there are concerns over its impending scarcity, the element indium was not included explicitly in the above analysis, due to an insufficiency of available data 76 ; however, other workers did investigate 77 the likely availability of indium and also that of both gallium and germanium. They concluded that, due to the different potential sources of these elements (all being ‘hitchhikers’– see section ‘Hitchhiker elements’), their supply potential exceeds current production by a factor of about 10 for Ga and Ge, but only a factor of 3 for In, which suggests that, of the three, the latter is the most likely to reach its production maximum in the near future. A new metric was introduced, 77 ‘time-to-maximum extraction as a by-product (TMEB)’, which could efficiently be used to assess the different supply scenarios for the three by-product elements: Ga, 58 (42–74) years; Ge, 85 (67–110) years; and In, 20 (20–47) years. 77 The authors stress that this does not necessarily mean that these elements will become scarce in 58, 85 or 20 years, respectively, but that increasing their supply after those times may be difficult. (It also does not rule out other sources of each element being found.) They conclude that there is no cause for concern over the provision of any of these three elements for at least the next decade, but it cannot be assumed that the same is true for other hitchhiker elements that are used in high-tech applications (Re, Te, Se, Sc, Zr, Eu, Gd, Tb, Dy, Er, Yb, Hf, etc.), each of which should be assessed, separately, using the same methodology. 77
Critical raw materials
The European Commission 78 has compiled a list of ‘critical raw materials’ (CRMs), to which ‘unhindered access’ is required for the essential running of society. This list is subject to regular review and update, and the most recent version features 27 different materials: antimony, beryllium, borates, cobalt, coking coal, fluorspar, gallium, germanium, indium, magnesium, graphite, niobium, phosphate rock, silicon, tungsten, platinum group metals, light rare earths and heavy rare earths, baryte, bismuth, hafnium, helium, rubber, phosphorus, scandium, tantalum and vanadium. In this context, the word ‘critical’ does not mean, necessarily, that the CRMs are thought to be scarce, rather that 79
they have a significant economic importance for key sectors in the European economy, such as consumer electronics, environmental technologies, automotive, aerospace, defence, health and steel;
they have a high-supply risk due to the very-high import dependence and high level of concentration of set CRMs in particular countries;
there is a lack of (viable) substitutes, due to the unique and reliable properties of these materials for existing, as well as future applications.
Hence, we see that the CRMs are irreplaceable for a range of applications, 79 such as in solar panels, wind turbines, electric vehicles and energy-efficient lighting, along with new and growing technologies: for example, a smartphone might contain up to 50 different kinds of metals, all of which contribute to its small size, low weight and functionality. To determine ‘criticality’ is a complex matter, and the different methods used around the world to evaluate it are quite diverse. 79
Technologies based on Earth-abundant materials
Due to limitations in the supply of critical metals and other elements, it is likely that a sufficient enlargement of the low-carbon energy base, in order to meet climate change targets, can only be achieved through the development of technologies that are based on Earth-abundant materials (EAMs). That noted, even for those elements that are relatively abundant on Earth (see section ‘Abundance of chemical elements on Earth’), limitations in their production rate, from technically and economically viable sources, may well be experienced on a scale of decades, and so a more efficient (reduced) use of them, coupled with effective collection and recycling strategies, is still necessary.20–22 Indeed, future limitations in the supply of energy, currently mainly furnished from the fossil fuels,20–22 may prove to be the restricting factor in the mining and production of even ‘abundant’ elements and also for their recycling. The majority of the EAM-based technologies, so far investigated, concern harvesting solar energy, for the purpose of PV applications, and to make solar fuels (primarily hydrogen), and for environmental decontamination strategies, including the conversion of CO2 to useful products. Electrocatalytic materials provide a parallel topic of research. The following is a very brief summary of some of the more recent and salient developments.
The ‘trash to treasure’ approach
80
has been used to create a three-dimensional (3D) form of porous carbon, containing both micropores and mesopores, from abundant biomass, which is intended to be used for a variety of energy-related and environmental applications.
80
Although thin-film solar cells based on Cu2ZnSnS4 (CZTS) and Cu2ZnSn(S, Se)4 (CZTSSe) were investigated in detail, during the past decade, more recently, a new class of chalcogenide materials has entered the field:
An effective, bifunctional Ni-Co-S electrocatalyst has been reported,
85
which produced the necessary voltage of 1.58 V (
A low-cost, high-efficiency system has been demonstrated 91 for the conversion of CO2 into hydrocarbons, which employs a PV cell coupled to an electrochemical cell (EC) for CO2 reduction. Using Cu-based catalysts at both electrodes of the electrolyzer, and by minimising all possible energy losses associated with the device, the reduction of CO2 to ethylene and ethane was demonstrated, with a 21% energy efficiency. 91 As coupled with a state-of-the-art, low-cost, PV mini-module, based on a perovskite, a 2.3% solar-to-hydrocarbon efficiency was achieved, which is considered to set a benchmark for an inexpensive, all-Earth-abundant PV–EC system. 91 It has been proposed 92 to instal floating ‘solar methanol islands’ with the facility to produce H2 from the PV electrolysis of seawater, from which CO2 is also extracted. The reaction between these materials using a selective catalyst (e.g. Cu/ZnO/Al2O3) might be used to produce methanol as a fuel, or possibly, and alternatively, higher (C5+) liquid hydrocarbons, using a direct CO2 Fischer–Tropsch hydrogenation process, with a CuFeO2 catalyst. Naturally, there are many outstanding questions regarding the feasibility of the overall approach, 91 and the scale on which it might be deployed. In order to make enough methanol to substitute for all the fossil-derived fuels that are used globally by long-haul transportation, it was reckoned that probably 170,000 such islands would be needed, and which would have to be located in ocean regions where the waves are less than 2 m high, and where sufficient sunlight is received, annually, to meet the energy requirements of the process. 92
Conflict resources (conflict minerals)
Conflict resources (conflict minerals) are natural resources that are extracted in a conflict zone and sold to perpetuate the fighting there; armed groups often use forced labour to mine minerals, which are then sold to fund their activities, such as buying weapons.
93
During wars in the eastern provinces of the DRC, various armies, rebel groups and external players have profited from the extraction of minerals, while perpetuating further violence and exploitation there.
94
The term ‘conflict minerals’ (Figure 9) currently refers to cassiterite (for tin), wolframite (for tungsten), coltan (for tantalum) and gold ore (these are sometimes referred to as ‘ Ensure that EU importers of 3TG (tin, tungsten, tantalum and gold) meet international responsible sourcing standards, set by the Organisation for Economic Co-operation and Development (OECD) •Ensure that global and EU smelters and refiners of 3TG source responsibly •Help break the link between conflict and the illegal exploitation of minerals •Help put an end to the exploitation and abuse of local communities, including mine workers, and support local development The regulation covers minerals and metals of: •gold •tin •tungsten •tantalum The regulation requires EU companies in the supply chain to ensure they import these minerals and metals from responsible and conflict-free sources only.
98
In some cases, it is possible that new sources of these metals will need to be found, if current parts of their supply chain are found wanting, which might introduce supply issues.

Conflict minerals: clockwise from top left: coltan, cassiterite, gold ore and wolframite.
Although it is not on the current official list of conflict minerals, the extraction of cobalt, of which 60%
39
of the world’s supply comes from the DRC, is also a matter for concern, in terms of the human cost of its production.
99
Cobalt
100
is an important material in renewable energy applications, for example, solar PV: although it is not employed directly in the panels themselves, it plays a crucial role in the cathode of rechargeable lithium-ion batteries, where the energy produced can be stored. The following statement appears in the UK government’s guidance document on conflict minerals
95
: The DRC’s mineral wealth is enormous. It is estimated that the country contains between 65-80% of the world’s columbite-tantalite (coltan) reserves, 49% of its cobalt reserves, and 3% of its copper reserves. Gold and diamond deposits remain under explored. Industrial Diamond reserves are estimated at 25% of world reserves.
95
A report has been published 101 by the International Institute for Sustainable Development with an interactive map showing countries assessed according to a ‘Fragile States Index’ and a ‘Corruption Perceptions Index’, along with particular locations where major deposits of materials, that are critical to green energy technologies, are to be found. Thus, global, political ‘hotspots’ are identified, which may be significant for the production of bauxite and alumina, chromium, copper, graphite, iron ore, lead, lithium, manganese, molybdenum, nickel, rare earths, selenium, silver, tellurium, tin, titanium metal concentrates and zinc. 101 These data make clear that the future development of renewable energy may rely on the recovery of materials from conflict zones and other politically unstable regions, and consequently pose problems for the large-scale expansion of the technology.
Other potentially constrained raw materials
Sand and gravel
Although normally thought of as being practically unlimited, the amounts of sand and gravel currently being consumed are now so large as to exceed their rate of natural renewal, and by the mid-21st century, production may no longer be able to meet demand. 102 Although the exact figures are sketchy, it is estimated that somewhere in the region of 32–50 billion tonnes are used globally each year, 102 primarily to make not only concrete, glass and asphalt but also electronic devices. Thus, sand and gravel are the most greatly extracted group of materials: by mass, exceeding our use of fossil fuels and biomass, 103 and by volume, are second only to our use of water. 104 An explosion in the use of these materials is being driven by urbanisation and global population growth, especially in China, India and Africa, and according to one estimate, by 2060, annual demand will have risen to 82 billion tonnes. 104 In many parts of the world, sand mining is not regulated (Figure 10), and is the province of ‘sand mafias’; sand has also been described as a ‘conflict mineral’. 105

Loading illegally dredged sand. Illegal sand mining is extensive around cities like Mumbai where it is required for construction.
The extraction of sand from ‘active’ regions, including rivers and deltas, can cause social, economic and environmental problems, as compared with ‘passive’ regions, such as geological deposits, from where its recovery is less damaging. In a recent report from the United Nations, 106 it is recommended that ways of reducing demand for ‘new’ sand should be encouraged, while policies that aim to discourage the harmful environmental impacts of sand mining must be reinforced. The report also proposes that the sand supply chain should be made more traceable, through better monitoring and the sharing of information internationally. 106 Even though it is believed that that there are trillions of tonnes of sand present on Earth, much of this is not useful, such as for construction purposes, 107 where the grains may be too small, or too smooth/round to bind well into cement, whereas crushed sand and gravel contain particles with rough/sharp edges that serve this purpose well. According to a system dynamics model, the production of sand and gravel will show plateau behaviour, with a maximum being reached in 2060–2070, partly due to an expected human population decline, along with rising prices for the materials, which will limit their demand. 107
In a kind of ‘every cloud has a silver lining’ way, it has been suggested 108 that the increased rate of melting of the Greenland ice sheet will increase the supply of sediment to the coastal zone, and therefore furnish the nation with an ample supply of sand and gravel. Accordingly, the changing Arctic conditions, as a result of climate change, may give Greenland an opportunity to become a global exporter of aggregates and assist in its goal of achieving economic independence. However, a caveat was added that a careful assessment of the resulting environmental impact should be made, and any such developments must be undertaken only with the approval of the wider Greenlandic society. 108
Water
Although water is a self-renewing resource, being circulated through the Earth’s hydrological cycle, there are, nonetheless, considerable challenges attendant to providing sufficient supplies of freshwater to meet the growing needs of an enlarging population. The term ‘peak water’ has been coined, 109 in analogy with ‘peak oil’, to describe a situation where water is being withdrawn from particular sources, such as aquifers, faster than they can recharge naturally, leading to an eventual maximum in production rate. Peak ‘ecological’ water is defined as ‘the point beyond which the total costs of ecological disruptions and damages exceed the total value provided by human use of that water’. 109 Although the global use of freshwater was previously reckoned to lie within its planetary boundary, 23 a call has been made that this should be reassessed, particularly in regard to environmental flow requirements 110 and the influence of water on critical Earth system functions such as regional climate and biodiversity. 111 Thus viewed, current freshwater use is found to lie within a much less comfortable margin of the newly evaluated boundary limits. 111
The use of ‘unconventional water resources’ (UWRs) is a global initiative 112 of the United Nations University’s Institute for Water, Environment and Health (UNU-INWEH), which aims to inform future policy and investment decisions on innovative water resources management. Various types of UWRs were considered, of which the first five can be classified as ‘new water’ and the remaining two as ‘used water’ 112 :
Desalinated seawater and highly brackish groundwater;
Groundwater in regions with no culture or experience in its large-scale use and groundwater confined in deep geological formations or in offshore aquifers;
Physical transportation of water through tankers and icebergs;
Micro-scale capture of rainwater where it otherwise evaporates;
Atmospheric moisture harvesting such as cloud seeding, fog water collection;
Collection and treatment of wastewater, grey water and storm water; and
Collection and use of agricultural drainage water.
Possible ways to adapt towards drought and climate change using unconventional water, with a focus on water stressed regions of Southern Spain, have been highlighted.
113
It has been determined
114
that there are 15,906 desalinisation plants (Figure 11) operating worldwide, which deliver a combined 95 million m3 of water/day for human use, of which 48% is produced in North Africa and the Middle East. Desalination is far from being a panacea for water shortages, due to the high capital costs of building desalination plants, the expense of the water produced and its transportation, the appreciable

Multi Stage Flash Desalination Plant at Jebel Ali G Station, UAE.
Fossil fuels
Although the future supply of crude oil remains a complex and contentious issue, especially in respect of future production rates of unconventional oil, which is necessary to compensate for the declining supply of conventional oil, all evidence is that the challenges involved in making such a substitution are considerable. 115 Some analyses predict that demand for oil will be mitigated through the use of gas and natural gas liquids (NGLs) (the non-methane fraction 115 of natural gas (mostly ethane to pentane) that is either liquid at normal temperatures and pressures, or can be relatively easily turned into a liquid with the application of moderate pressure 115 ), the availability of which has grown in hand with an increased production of shale gas by means of hydraulic fracturing (‘fracking’). 115 However, per unit volume, NGLs contain around one-third less energy than crude oil, and only about one-third of this volume can be blended into transportation fuels. 115 It is possible that NGLs may partially counterbalance an enlarging contribution of heavy oil to the refinery input mix, but it seems clear that the relentlessly declining production of conventional crude oil must finally reach a stage where it can no longer be made up for with NGLs. 115 Other analyses indicate that production of light tight oil (light crude oil 115 contained in shale or carbonate rocks with very low permeabilities that can be produced using horizontal wells with multi-stage hydraulic fracturing 115 ), also mainly produced by fracking shale (Figure 12), will provide the compensating unconventional oil; however, while it is true that in the past decade or so, it is only this source, along with extra heavy oil from ‘oil sands’, that has allowed the global production of ‘oil’ to grow – conventional production having flat-lined over the past decade – the prospects that a worldwide massive production might be achieved, similar to that in the United States, are highly debatable. 31 Indeed there is much speculation over the longer term viability of the entire fracking industry,31,116 and if it were to stall, the implications for maintaining the global supply of oil, gas and NGLs would likely be severe.

In areas where shale-drilling/hydraulic fracturing is heavy, a dense web of roads, pipelines and well pads turn continuous forests and grasslands into fragmented islands.
There are predictions of a peak in the production of coal, due to supply limitations, 117 and it is significant that demand for coal, despite having fallen briefly, is now once again on the increase, especially for electricity generation. 118 Overall, it appears likely that the provision of energy from fossil fuels will decrease during the latter half of this century,20,21 and in any case, their use must be curbed in order to reduce CO2 emissions. 119 However, to maintain the current overall use of energy, by replacing fossil fuels with renewables, will require challenging rates of expansion of the latter 120 ; moreover, as we have seen in some of the previous sections of this article, there may be limits to the elements needed to fabricate such a system at the necessary scale. It is possible, therefore, that even for those minerals whose burn-off times/times to scarcity are centuries long, it may be limits to energy that curb their production rates and availability.20–22,34,76
Resources and energy
Fossil fuels are finite resources, and once burned can neither be renewed (or only on geological timescales), nor recycled; thus, even if no attempt were made to curb their use, in order to limit CO2 emissions and meet global warming targets, their supply may become compromised within the next few decades.20–22 Currently, more than 80% of the total energy used by humans on Earth is furnished by the fossil fuels,
119
which necessarily underpin both the extraction and recycling of other resources. Thus, fossil fuels will be needed to drive the production of materials to establish renewable energy sources, until sufficient capacity can be installed to feed back into the system, overall, and replace the fossil energy that is being used to power such an energy transformation. Even if the holy grail of fusion power could be achieved,
29
which is often hailed as ‘creating a star on Earth’, and that it produces its own fuel (tritium), in fact the process requires a supply of 6Li (which constitutes just 7.5% of naturally occurring lithium) that must be mined from the Earth. Due to the increasing demand for batteries, supplies of lithium are already a matter of concern, although this could be improved by wholesale recycling
36
(as seems to be true for all metals).20–22 In all cases, not only does the aphorism hold true that ‘it takes energy to produce energy’, but other, underpinning, resources are required too. It should be stressed that the majority of the lithium required by a fusion power plant is not consumed by its conversion into tritium (0.5 g of tritium s−1 for a 1 GWe (gigawatt of electricity and around one-third of the thermal power of the reactor) output
121
) but it is needed for other purposes, including as a neutron moderator and heat transfer medium. Thus, according to different estimates of lithium
As the quality of mineral ores becomes relentlessly impoverished, 42 more energy is necessary 34 to recover each mass (or energy) unit of their useful component: for energy sources, this is expressed by falling EROI (see Appendix 1). The extraction of metals from increasingly low grade ores also means that there is more waste produced, that is, a 5% metal content (1:20) of an ore means that every kilogram of metal produced is accompanied by 19 kg of ‘waste’, while for a 0.5% content (1:200), the waste amounts to 199 kg. The huge amount of additional energy needed 34 is inversely proportional to the declining ore quality, and since this, for the immediate future, will almost all come from the fossil fuels (for the actual mining, mainly from oil), it also means more CO2 being produced, and most likely, more water will be needed for the mining and processing, as an increasing further input to the system. It can also be expected that extraction from resources of diminishing quality translates into lower production rates, and, for energy sources, falling EROI (and therefore, net energy). A coupling between the falling quality of both ores and energy sources can only compound the situation. 29
Conclusion
The UK Government’s Chief Scientific Adviser at the Department of Environment, Food and Rural Affairs from September 2012 to August 2019, Professor Sir Ian Boyd, has recently exhorted that, 124 in order to meet carbon emissions targets, we need to travel less, eat less red meat, buy fewer clothes and generally reduce demand for consumer goods: all of which is underpinned by resources extracted from the Earth and underpinned by energy, more than 80% of which is currently drawn from the fossil fuels. 119 Indeed, natural resources are being consumed on an unprecedented scale, and currently, an annual 92 billion tonnes of raw materials are being extracted, which corresponds to around 12 tonnes for every person on the planet. 125 As part of a significant expansion of renewable energy production, it will be necessary to recycle elements from wind turbines and solar panels (especially thin-film cells). The interconnected nature of particular materials, for example, cadmium, gallium, germanium, indium, tellurium, all mainly being recovered from the production of zinc, aluminium and copper, and helium being obtained from natural gas, means that the availability of such ‘hitchhiker’ elements, is a function of the reserve size and production rate of the primary (or ‘attractor’) material. Given the potential limitations in the supply of various critical elements, it is likely that the necessary huge expansion of renewable energy sources will be best achieved using technologies based on ‘Earth Abundant Materials’, but currently, it is fossil fuels that are needed to process them, and most EAM technologies are insufficiently close to the level of commercial viability required to begin to supplant their fossil fuel equivalents necessarily rapidly and at scale. Moreover, even for those elements that are, indeed, relatively abundant on Earth, limitations in their production rates/supply may well be experienced on a timescale of decades, and so a more efficient (reduced) use of them, coupled with effective collection and recycling strategies should be embarked upon urgently.20–22
Of all essential commodities, the fossil fuels may well run into production limits during the next few decades, and indeed, the underpinning determinant of how we extract resources, inducing those of energy, is the availability of energy itself and those resources that provide it. As we, inevitably, use-up high grade ores, and move on to poorer quality deposits, in which the desired material is increasingly diluted by other materials, the energy input to the whole extractive and processing mechanism increases: in terms of the production of energy resources, this is expressed as declining EROI. As the quality of mineral deposits declines, the volume of material that needs to be exhumed from the Earth, and processed, enlarges relentlessly, leading overall to increasing amounts of waste for each mass unit of metal, or other element recovered, and much more additional energy is needed. 34 This is sometimes described as a “Red Queen’s race”, after the character in Lewis Carol’s fantasy novel, “Through the Looking Glass”, where everyone in her kingdom needed to run as fast as they possibly could, just to stay in the same place. 29 Using more fossil fuel–derived energy also means more CO2 being produced, and most likely, more water will be needed for mining and processing, as an increasing further input to the system. It can also be expected that extraction from resources of diminishing quality will translate into falling production rates, and, for energy sources, falling EROI (and therefore, net energy). A coupling between the declining quality both of ores and energy sources can only compound the situation.
Some points are worth highlighting regarding the overall provision of global energy:
Various different analyses predict a production peak for fossil fuels sources within the next few decades, but, in any case, it is necessary to curb current levels of fossil fuel use, in order to reduce CO2 emissions and meet global warming targets;
Hence, for both the latter reasons, low-carbon energy (i.e. renewables and possibly nuclear energy) must be introduced, rapidly and at scale, to substitute for fossil fuel energy;
If low-carbon energy cannot be introduced sufficiently rapidly to meet the growing total global demand for energy, fossil fuel energy use will increase (and accordingly CO2 emissions will also increase), until their production peaks, whereupon overall available energy will begin to decline, and CO2 emissions then fall;
It is the fossil fuels that will provide the initial energy necessary to furnish low-carbon energy sources, which will eventually pay back energy into the overall system;
If low carbon energy cannot be introduced sufficiently rapidly and at scale, it will be necessary to reduce overall energy demand, but this will happen anyway following the fossil fuel production peak, if low-carbon energy cannot keep up with the resulting decline;
The EROI for renewables being >1 means that energy recovered overall is greater than that originally contained in the fossil fuel; however, the extra energy is ‘bought’ by the mining, processing and de-concentration of mineral ores (the energy costs of decommissioning/recycling various materials should also be factored into the overall denominator);
In terms of low-carbon ‘nuclear energy’, the ‘holy grail’ of nuclear fusion power (D + T) depends on finite supplies of lithium (to generate T), and unless the extraction of Li from seawater, for example, can be made energetically favourable, the most likely type of ‘nuclear’ energy that could be viable for centuries or millennia is that generated using ‘breeder’ reactors, based on uranium (or thorium), which would extract the full complement of the primary energy from the fuel, rather than from fissioning a minor isotope - 235U -”; although, there are many and various unattractive issues attendant to this;
A voluntary reduction in our overall use of energy, for example, through energy efficiency (such as insulating/draught-proofing buildings) and localisation measures (reducing transportation and adopting less energy intensive – more locally scaled – food production and distribution), would mitigate the challenges of dealing with the peaking, followed by declining production rates of fossil fuels, curbing CO2 emissions, and mitigate the level of low-carbon energy that must eventually be installed.
However, it has been determined that there are insufficient, proven, reserves (and resources too, in certain cases) of several metals required to build a fully renewable energy system, to meet the global demand for energy that is expected by 2050. For scarce elements, recycling is indicated to be of limited value, as judged against the scale of their use required. It is possible that incorporating potentially less efficient technologies (but based on elements that are more widely available) might prove a viable strategy for reducing the risks of supply constraints. The requirements of demand-side technologies such as electric vehicles, fuel cells and energy efficiency measures are a critical additional factor, and it is also necessary to make an accurate evaluation of the demand for speciality metals by non-energy industries. (The future development of low-carbon energy may also rely on the recovery of materials from conflict zones and other politically unstable regions, 101 which could pose further problems for its large-scale expansion). Moreover, how such a fully renewable energy system might be maintained, beyond 2050, remains a serious open question. 76 Meanwhile, major changes in our global demand for energy are necessary, and it may be wise to spend some of the fossil fuel equivalent of our remaining carbon budget on the extraction of metals required for renewable energy technologies. 76
The present manner of our resource use means that the global materials base is unsustainable, and it is necessary to optimise our use of energy, to close material cycles, and to curb irreversible material losses, of all kinds.20–22 Such is our consumption of sand and gravel, to furnish concrete, glass, asphalt and electronic devices, that their supply too is now a matter of concern; levels of freshwater use are also now approaching newly defined planetary boundary limits. It is probable that a sustainable human population on Earth is less than 3 billion,20,21,30 but who have significantly attenuated their consumption of resources, of all kinds, from present per capita levels. 22 To undertake the necessary transformation, profound social restructuring is likely to be necessary, in terms of equity, education, employment, goals, values, expectations and measures of ‘success’ and ‘worth’. It is thought that the conversion of resources and human labour to ‘money’, as well as the current growth of debt (extensive borrowing from the future), may mean that peak oil and peak materials will lead to ‘peak wealth’ and hence to the end of the golden age that is currently experienced by those of us in the industrialised nations.20,21 As some economists have pointed out,64,65 there is no absolute guarantee that no new sources of materials will be found, nor better technologies be discovered that make recovery from poorer quality reserves viable. However, there are severe limitations to the economic ‘linear model’, 126 and, as EF Schumacher 127 pointed out almost half a century ago, using ‘natural capital’ as income is ultimately unsustainable and destructive to the environment.
The timescale of our intentions regarding the use of resources is critical, and the question of whether technology can solve our current problems, and meet future needs ‘sustainably’, is yet to be answered fully. Perhaps such considerations of what is sustainable only properly make sense, if societal viability over the duration of a civilisation (say, 500 years) 128 is planned for; yet we have only the next few decades, at most, to undertake the appropriate actions to establish this. Indeed, one is reminded of the possible four ‘future scenarios’ 129 proposed by David Holmgren, one of the originators of permaculture, which is a design system, based on three core ethics: Earth Care, People Care and Fair Shares (return of surplus). Holmgren looks towards the future on the timescale of development of an old growth forest (say, 500 years), and concludes as ‘technofantasy’ the idea that technological advances will be able to not only meet all energy and other needs but also absorb and repair the damage already done, and which we may continue to do, over that same time period, and, presumably, beyond it. Holmgren sees permaculture as a pathway (creative response) to achieving a state of harmony between humans and Nature, ‘Earth Stewardship’, where resources are used not only sustainably but also regeneratively, and that a more apposite icon for a solar economy is a tree, rather than a solar panel, since, arguably, all solar panels (and wind turbines too) will have become waste (possibly toxic waste) long before then, unless better initial design and the cycling back of materials and solar/wind-generated energy into the system, overall, can be achieved. Critical drivers will be resources, especially of energy, but ultimately it seems likely that Earth Stewardship may prove the only truly sustainable scenario, and indeed the only one that is regenerative of essential natural resources. 130
Footnotes
Appendix 1
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Author biography
![]() ), which cover radiation chemistry, catalysis, zeolites, radioisotopes, free radicals and electron paramagnetic resonance spectroscopy, and more recently have developed into aspects of environmental decontamination and the production of sustainable fuels. Chris has given numerous radio and televised interviews concerning environmental issues, both in Europe and in the United States – including on BBC Radio 4’s Material World. Latest invitations include a series of international lectures regarding the impending depletion of world oil supply, and the need to develop oil-independent, sustainable societies. He has published more than 250 peer-reviewed scientific articles and six books. He is also a published novelist, journalist and poet.
), which cover radiation chemistry, catalysis, zeolites, radioisotopes, free radicals and electron paramagnetic resonance spectroscopy, and more recently have developed into aspects of environmental decontamination and the production of sustainable fuels. Chris has given numerous radio and televised interviews concerning environmental issues, both in Europe and in the United States – including on BBC Radio 4’s Material World. Latest invitations include a series of international lectures regarding the impending depletion of world oil supply, and the need to develop oil-independent, sustainable societies. He has published more than 250 peer-reviewed scientific articles and six books. He is also a published novelist, journalist and poet.
