Abstract
Data from 3 US public health surveillance systems indicate that an estimated 67 400 people acquired hepatitis C virus infections in 2022, 2.2 million adults had hepatitis C during January 2017–March 2020, and 12 717 people died in 2022 from hepatitis C–related causes, despite the availability of curative treatment since 2013. The 3 surveillance systems that provided these data—the National Notifiable Diseases Surveillance System, the National Health and Nutrition Examination Survey, and the National Vital Statistics System—were selected to monitor progress toward elimination of hepatitis C as a public health threat in the United States by 2030. However, some limitations of these surveillance systems compel the use of additional data sources with more timely information for the general population and for populations with higher incidence, prevalence, or mortality of hepatitis C, such as those experiencing homelessness, incarceration, or injection drug use. Commercial laboratories, health systems, and programs serving these populations could provide such data. This topical review of hepatitis C surveillance describes the history, long-term trends, and recent investments in public health surveillance for hepatitis C. Strengthening and modernizing the hepatitis C surveillance workforce and systems, improving data system interoperability, linking complementary data sources, and leveraging multiple data systems can aid in the measurement of public health efforts to meet hepatitis C elimination goals.
Data from 3 US public health surveillance systems indicate that an estimated 67 400 people acquired hepatitis C virus (HCV) infections in 2022, 2.2 million adults had hepatitis C during January 2017–March 2020, and 12 717 people died in 2022 from hepatitis C–related causes, despite availability of curative treatment since 2013.1,2 These surveillance systems—the National Notifiable Diseases Surveillance System (NNDSS), the National Health and Nutrition Examination Survey (NHANES), and the National Vital Statistics System (NVSS)—were selected to monitor progress toward elimination of hepatitis C as a public health threat in the United States by 2030.3-8
NNDSS, NHANES, and NVSS have provided data for decades.3-5 Changes in methods and lags in data availability, as well as incomplete information on race and ethnicity, health insurance, and health risks, limit their use for a data-directed response to hepatitis C, such as linkage to care and treatment and the monitoring of long-term trends.4,6,9-15 More timely and complete data can help link populations with risk of infection to testing, care, and treatment, such as people who are experiencing homelessness or incarceration or have substance use disorder.16-19
In this topical review, we describe trends in hepatitis C prevalence in the United States using data from these 3 sources. We discuss the strengths, limitations, and recent investments in public health surveillance for hepatitis C and the complementary data sources to support the achievement of hepatitis C elimination goals in the United States. This project was reviewed by the Centers for Disease Control and Prevention (CDC), was exempted from institutional review board oversight because it did not involve human subjects, and was conducted consistent with applicable federal law and CDC policy (eg, 45 CFR 46.102[e]).
Epidemiology, Surveillance, Testing, Treatment, and Elimination Goals in the United States, 1982-2024
Epidemiology and Surveillance
Identification of HCV in 1989 led to testing of the blood supply in 1992. 20 Although now rare in the United States, exposure through medical procedures and transfusions before 1992 and injection drug use contributed to a high prevalence of chronic hepatitis C among people born during 1945-1965.21-24 The burden of newly reported chronic hepatitis C cases shifted as the incidence of acute hepatitis C increased among younger people who inject drugs (PWID).1,20 Massachusetts reported a bimodal distribution of its newly diagnosed hepatitis C cases, with the highest proportions among people aged 24 and 49 years in 2007, shifting by 2022 to people aged 31 years. 25 National data revealed a bimodal distribution of newly diagnosed chronic hepatitis C cases by age and sex during 2018-2022.1,26,27 Acute cases among people aged 20 to 29 years decreased after 2018 following 8 years of increases.1,26,27 Studies have reported high rates of hepatitis C among people experiencing homelessness, incarceration, or substance use disorder.16,28-32 The increase in hepatitis C among young adults has placed more neonates at risk for perinatally acquired HCV infection, a third wave of the hepatitis C epidemic.1,33
Acute HCV infection rarely results in death; however, untreated chronic hepatitis C can lead to liver damage and premature death. 27 Disparities in hepatitis C–related death rates by geographic area and age group emerged during the past 2 decades. 34 Long-standing disparities persisted, with higher death rates among non-Hispanic American Indian and Alaska Native people and non-Hispanic Black people than among non-Hispanic White and non-Hispanic Asian people. 1
Testing and Treatment
Hepatitis C testing recommendations and guidance evolved during 1991-2023, influencing trend data from NNDSS and commercial laboratories. 35 Hepatitis C antibody testing has been recommended for donors of blood, organs, tissue, and semen since 1991; people at risk of infection or severe disease outcomes since 1998; and, since 2012, people born during 1945-1965.36-40 The 2003 guidelines added HCV RNA reflex testing following a confirmed reactive antibody test to distinguish between current and past infection; the 2013 guidelines indicated HCV RNA testing following a reactive screening antibody test, then an optional confirmatory antibody test following a nondetected HCV RNA result.38,40 Recommendations published during 2018-2023 added screening during every pregnancy and screening of adults aged 18 to 79 years at least once, updating guidance for testing of perinatally exposed infants and children in 2023.24,33,41-45 Each screening recommendation followed developments in the HCV epidemic, treatment, and care. Treatment with direct-acting antivirals results in cure in >95% of treated people and reduces hepatitis C–related morbidity and mortality. 24 Among people with ongoing risks, direct-acting antiviral treatment, substance use disorder treatment, and harm reduction can prevent new infections. 46
Elimination Goals and Other Key Indicators
A 2017 Institute of Medicine assessment determined that elimination of hepatitis C as a public health threat in the United States by 2030 was feasible. 47 In 2020 and 2021, CDC and the US Department of Health and Human Services published strategic plans for 2025 that established goals to eliminate hepatitis C as a public health threat in the United States by 2030.6,7 The strategic plans built on earlier strategic and action plans and contributed to international goals for viral hepatitis elimination.48-50 The 2025 strategic plans and Healthy People 2030 use NNDSS, NHANES, and NVSS to track indicators of new infections, viral clearance, mortality, and racial and ethnic disparities.6-8 NNDSS and NVSS data also are used to monitor indicators in elimination plans developed by health departments and partner organizations. 51 The prevalence of hepatitis C is estimated by using NHANES alone or augmented with data from populations experiencing homelessness, incarceration, or drug use who are not well represented in NHANES.2,16,52
Surveillance Strategies and Long-term Trends
Case Surveillance Data From NNDSS
NNDSS was launched in 1951 with case definitions developed independently by jurisdictions and then standardized nationally in 1990 (Figure 1, eTable 1 in Supplement).53-55
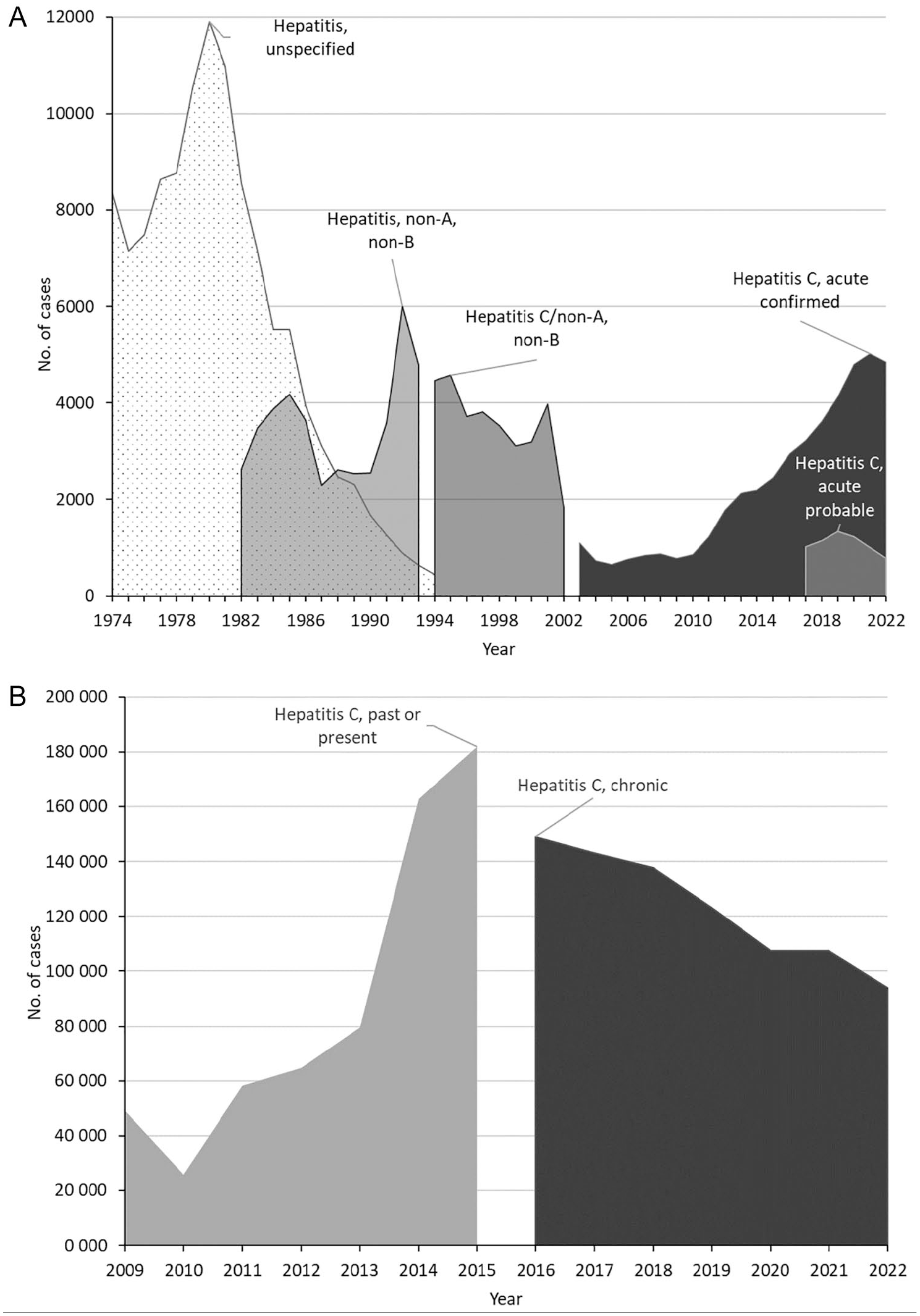
Trends in reported cases of unspecified hepatitis, non-A/non-B hepatitis, confirmed and probable acute hepatitis C, past or present hepatitis C, and chronic hepatitis C from the National Notifiable Diseases Surveillance System, United States, 1974-2021. (A) Reported cases of unspecified hepatitis, non-A/non-B hepatitis, and acute hepatitis C, 1974-2022. The non-A/non-B hepatitis case definition was established in 1990 as one of many newly standardized case definitions for notifiable diseases. Acute hepatitis C case definitions were published for 1995 and 1996. Case definitions for acute hepatitis C were revised for 2000, 2004, 2007, 2011, 2012, 2016, and 2020 (eTable 1 in Supplement). Surveillance reports published combined hepatitis C and non-A/non-B hepatitis case counts from 1994 through 2002 because jurisdictions implemented the acute hepatitis C case definitions at different times (eTable 2 in Supplement). (B) Newly reported cases of past or present hepatitis C and chronic hepatitis C, 2009-2022. Case definitions for hepatitis C, past or present, were published for 2003, 2005, 2011, and 2012. Case definitions for chronic hepatitis C were published for 2010, 2016, and 2020 (eTable 1 in Supplement). Surveillance reports published annual case counts for hepatitis C, past or present, from 2009 through 2015 and for chronic hepatitis C since 2016 (eTable 2 in Supplement). Data source: National Notifiable Diseases Surveillance System. 3
NNDSS receives notifications of new hepatitis C cases reported to jurisdiction health departments; provisional data are published weekly and final data are published annually. 56 Viral hepatitis has been a nationally notifiable disease since 1951; hepatitis C cases were among the unspecified hepatitis cases before 1982 and non-A/non-B hepatitis cases during 1982-1993 because the etiologic agent was not discovered until 1989 (Figure 1, eTables 1 and 2 in Supplement). 36 The CDC case definition for acute hepatitis C was first implemented in 1995 and then revised 8 times during 1996-2020.57-65 Seven chronic hepatitis C case definitions were published between 2003 and 2020: 4 for past or present hepatitis C and 3 for chronic hepatitis C (eTable 1 in Supplement).66-72
Case definition changes to incorporate evolving testing and treatment guidance and jurisdiction surveillance practices affected trends in published data during 1974-2022 (Figure 1, eTables 1 and 2 in Supplement).73-82 Cases of unspecified hepatitis peaked in 1980 (Figure 1A) and then declined steeply as case reports of non-A/non-B hepatitis rose, peaking in 1992.74,75 While jurisdictions adopted a new hepatitis C case definition in different years during 1994-2002, combined case counts were published for hepatitis C and non-A/non-B hepatitis. The emergence of infections arising from injection drug use related to the opioid crisis increased the number of acute and chronic hepatitis C cases from 2011 to 2021.28-32 The large increase in the number of newly reported chronic hepatitis C cases from 2013 to 2014 reflects, in part, increased screening and testing during the early days of direct-acting antiviral availability and implementation of the 2013 recommendations for testing the 1945-1965 birth cohort.21,35,39 A discontinuity in trends in chronic hepatitis C cases from 2015 to 2016 reflects a change in case definitions (Figure 1B, eTable 1 in Supplement).83,84
Hepatitis C surveillance data have had limitations since surveillance for non-A/non-B hepatitis began in 1982.74,85,86 Lack of completeness of information for race and ethnicity and risks was evident in 2005-2007 and persisted through 2022.1,87 Other considerations include changes in case definitions, differences in timing of case definition implementation by jurisdictions, and constraints on health department resources for detection and investigation of the large volume of hepatitis C cases. 88
Data modernization efforts, such as electronic laboratory reporting from commercial laboratories and electronic case reporting from health systems, have the potential to increase the timeliness and completeness of case reports through improvements in interoperability of health care and public health information systems. 89 Automation of time-consuming data management, case classification, and case prioritization tasks could free jurisdiction health department staff time for case investigation aspects that cannot be automated. 90 Data linkages can improve the completeness of surveillance data and utility for analyses to support elimination efforts.
Hepatitis C Prevalence Data From NHANES
Launched in 1959, NHANES is the only national survey that conducts a questionnaire, a clinical examination, and laboratory assessments for many risks and health conditions, including hepatitis C, with consenting participants.4,91-94 NHANES collects blood and information from a randomly selected sample of the civilian, noninstitutionalized US population, with about 5000 people from 15 areas participating annually. 4 NHANES data have been released in 2-year cycles since 1999, with survey weight and design variables needed to produce nationally representative estimates (Figure 2, eTable 3 in Supplement). NHANES conducted viral hepatitis tests for participants starting in 1974 and hepatitis C tests starting in 1988 (eTable 3 in Supplement).
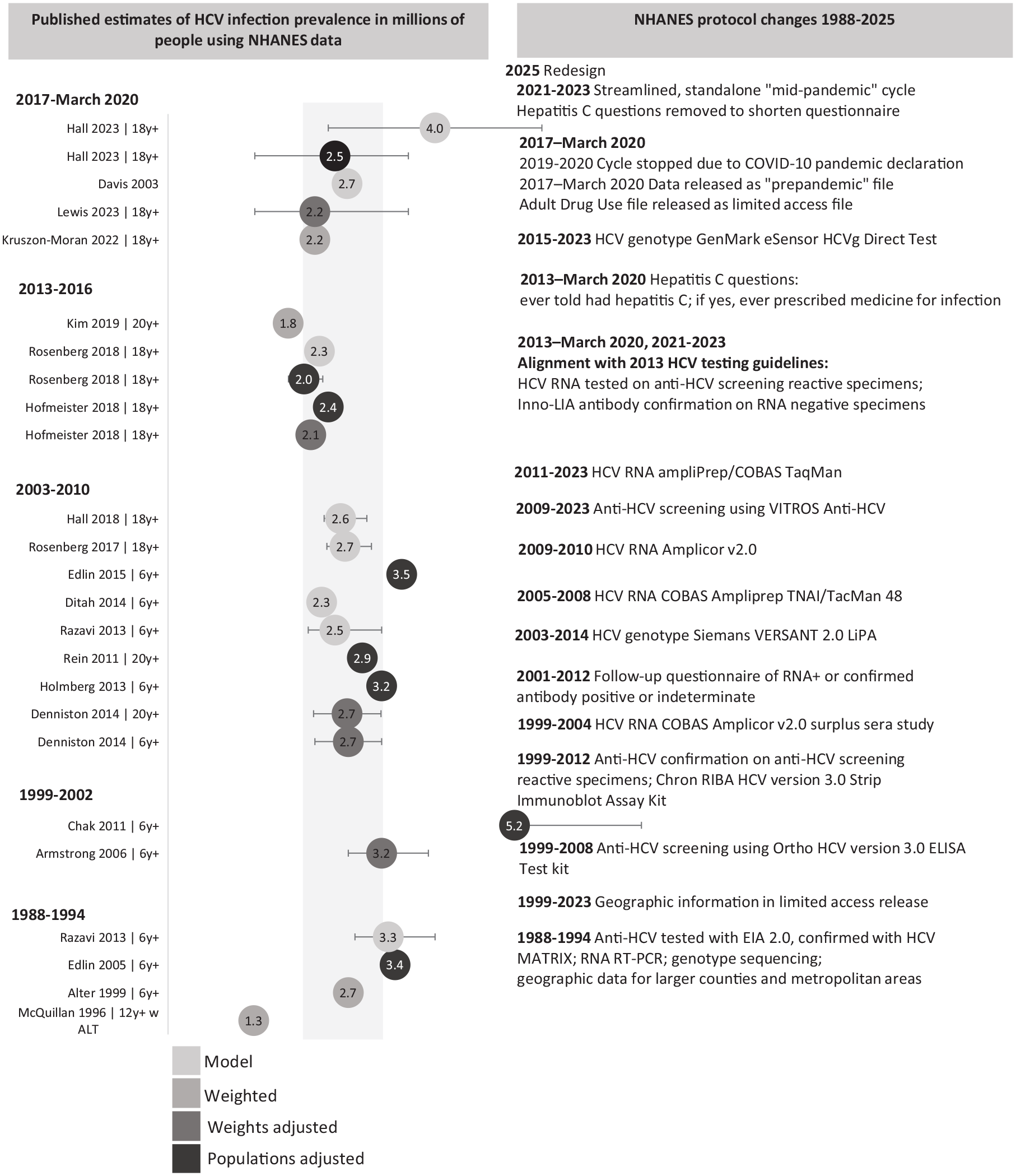
Published estimates in millions of people with detected hepatitis C virus (HCV) RNA and selected protocol changes for the National Health and Nutrition Examination Survey (NHANES), United States, October 1988–August 2023. Data source: NHANES. 4 Based on data for HCV RNA from NHANES collected during 1988-1994 and 1999–March 2020, 1 or more estimates of the prevalence of current, likely chronic, HCV infection were abstracted from each of 21 studies. The number in millions of people with current HCV infection (HCV RNA detected) is shown, with 95% CIs if available from the study. Studies used different methods: modeling the prevalence by using NHANES with other data sources, such as mortality from overdoses; weighting the estimates by using the survey weights available in the datasets; adjusting the survey weights to account for missing HCV antibody or HCV RNA results; or augmenting the estimates with information from studies of populations that were excluded or underrepresented in NHANES. Studies used different age groups (eg, ≥6, ≥12, ≥18, or ≥20 y). One study did not specify the age group used, and another limited the analysis to NHANES participants with elevated alanine transaminase levels. The hepatitis C laboratory and interview protocols changed during this time, as did the NHANES sampling design and data selected for public release. A major redesign of NHANES launched in 2025 (eTable 3 in Supplement). Abbreviations: ALT, alanine aminotransferase; anti-HCV, antibody to HCV infection; INNO-LIA, INNO line immunoassay; ELISA, enzyme-linked immunosorbent assay; RT-PCR, reverse-transcription polymerase chain reaction.
During 2017–March 2020, the prevalence of chronic hepatitis C in the United States was estimated at 2.2 million people when using NHANES data and higher when accounting for populations excluded from or underrepresented in NHANES, such as people living unsheltered or incarcerated or PWID.2,16,52 The lack of change in hepatitis C prevalence during 1988–March 2020 is likely due to evolving injection drug use and the past decade’s opioid epidemic offsetting reductions in infections from blood supply screening, deaths among older adults with hepatitis C, and expanding treatment (Figure 2, eTable 3 in Supplement).2,16,22,52,95-110 Studies have refined NHANES hepatitis C prevalence estimates by pooling multiple cycles to improve the statistical reliability of estimates,2,96,97,102,107,109 adjusting weights for the few missing laboratory results,2,16,22,107 augmenting estimates to account for populations excluded from the NHANES sample design,16,52,98,101,104-106,108 and incorporating other data sources into models or forecasts52,95,98-100,102,103 (Figure 2, eTable 3 in Supplement). Most of these studies estimated that 2 to 3 million people had chronic HCV infection.2,16,22,52,95,96,98-100,102-104,109 Studies that incorporated other data to account for populations omitted from or underrepresented in NHANES estimated that 3.2 to 5.2 million people had chronic HCV infection (Figure 2, eTable 3 in Supplement).52,101,103,105-108
Strengths of NHANES include completeness of sociodemographic information and availability of data to monitor decades-long trends in hepatitis C prevalence; however, several aspects of NHANES limit its use for supporting elimination efforts. NHANES is not designed to produce state or local area estimates and has encountered declining participation rates. 111 NHANES omits or underrepresents people who are living unsheltered, are in carceral facilities, or inject drugs. 16 Declining participation rates could worsen representation of PWID. Few estimates among groups categorized by race and ethnicity, age, type of health insurance, history of drug use, and awareness or treatment of HCV infection meet statistical reliability standards.2,22,27,112,113 Changes in the survey limit the number of years of data that can be pooled to meet these standards (Figure 2, eTable 3 in Supplement).
A redesigned NHANES launched in January 2025. Planned changes could support statistically reliable hepatitis C prevalence and viral clearance estimates for more population groups than in previous cycles.114,115 An ongoing NCHS program that links NHANES data with national administrative data, geographic characteristics, and social determinants of health offers the potential for new insights into trends in hepatitis C prevalence. 116
Mortality Data From NVSS
NVSS, through the Vital Statistics Cooperative Program, receives data from death certificates filed in jurisdictions’ vital records offices; provisional data are published monthly and final data are published annually. 5 Summaries of NVSS mortality data have been published since 1950. 117 Estimates of deaths with hepatitis C as the underlying cause published beginning in 1996 showed that hepatitis C accounted for much of the increase in overall viral hepatitis deaths during 1996-1998 (Figure 3A, eTable 4 in Supplement).118,119 Since 1999, hepatitis C deaths have been summarized with hepatitis C listed as one of multiple causes of death because hepatitis C is often recorded as a contributing cause but not the primary or underlying cause; also beginning in 1999, causes of death have been coded by using the International Classification of Diseases, Tenth Revision instead of the International Classification of Diseases, Ninth Revision (Figure 3B, eTable 4 in Supplement). 120 Hepatitis C accounted for most deaths with any viral hepatitis listed among the multiple causes during 1999-2022.121,122 Hepatitis C–related deaths peaked in 2014, accounting for >90% of the 21 000 deaths with any viral hepatitis listed in that year (Figure 3B, eTable 4 in Supplement). 121
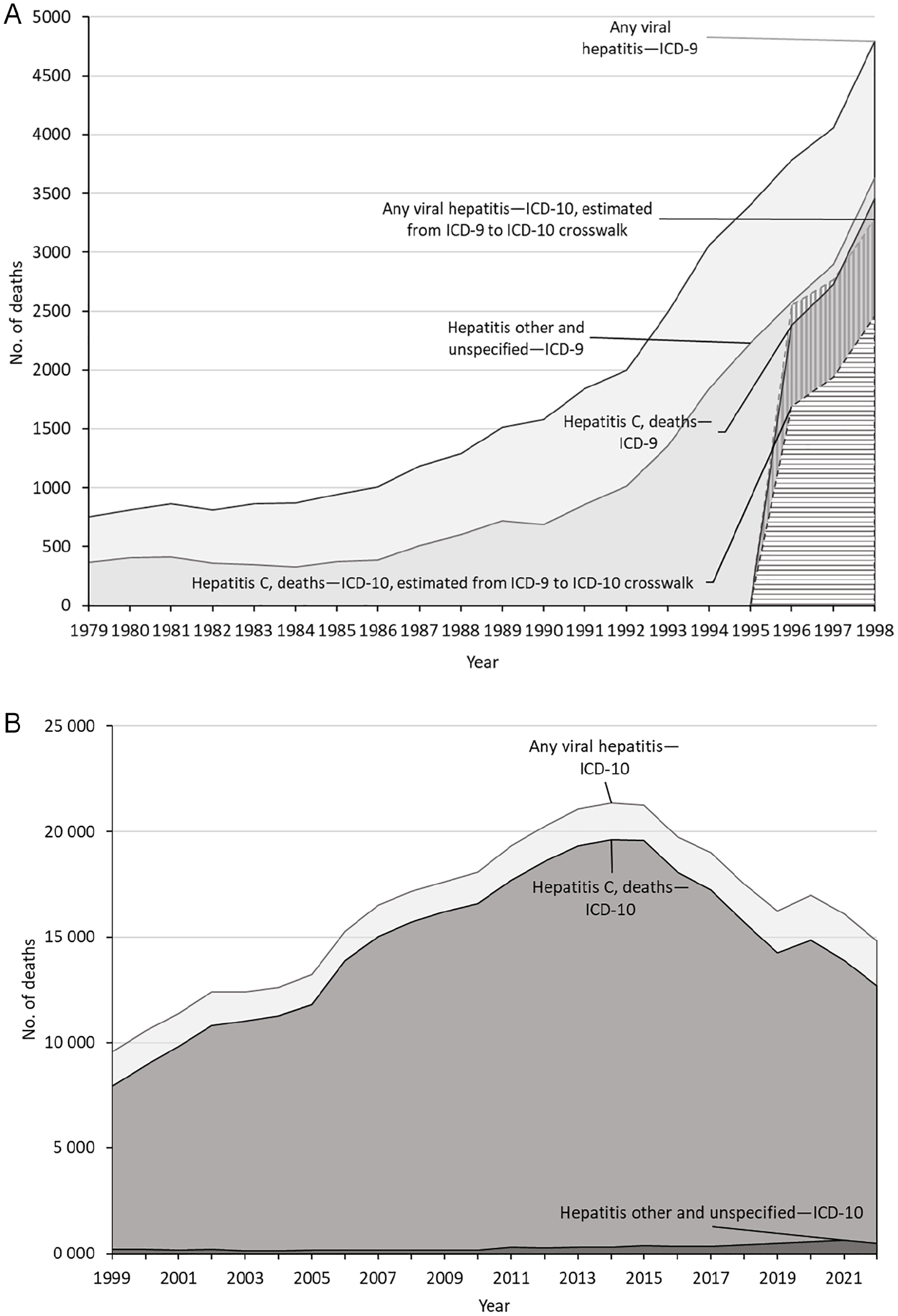
Trends in reported deaths with any viral hepatitis, hepatitis C, and other and unspecified hepatitis from the National Vital Statistics System, United States, 1979-2022. Data source: National Vital Statistics System. 5 (A) Deaths with underlying causes of any viral hepatitis and other and unspecified hepatitis during 1979-1998 and hepatitis C during 1996-1998 were coded by using the International Classification of Diseases, Ninth Revision (ICD-9). In preparation for the transition to the International Classification of Diseases, Tenth Revision (ICD-10) in 1999, deaths during 1996-1998 were recoded by using a crosswalk from ICD-9 to ICD-10 coding. (B) Deaths with any viral hepatitis and hepatitis C as 1 of multiple causes of death, 1999-2022. Beginning in 1999, deaths with hepatitis C as 1 of multiple causes of death were published instead of deaths with hepatitis C as the underlying cause of death (eTable 4 in Supplement).
Strengths of NVSS data include availability of data during decades, availability to jurisdictions through their vital statistics offices, and relatively complete coverage of geographic areas and populations. Limitations include underreporting of hepatitis C on death certificates and transitions in coding standards. 123
Modernization of NVSS might improve the timeliness, completeness, and quality of death data, overall and for hepatitis C–related deaths. 124 Analyses of NVSS data by geography integrated with social determinants of health data could help direct treatment resources to medically underserved areas. 34
Achieving the 2030 Elimination Goals With Improved Surveillance Data
NNDSS, NHANES, and NVSS have strengths for tracking progress toward hepatitis C elimination goals but also have limitations in their use to support hepatitis C elimination efforts. Recent investments in public health aim to strengthen hepatitis C surveillance, address the limitations of NHANES, and support the development and use of additional data sources.
In May 2021, CDC funded 59 jurisdictions to improve the timeliness of case reporting and the completeness of demographic, outbreak, and risk information. 125 The journey from new infection to cure can be traced through a continuum, based on laboratory data alone, or on person-level registries built from multiple data sources that support efforts to link patients to care and treatment and monitor the proportion of patients who are cured and reinfected.17,18 Implementation of electronic laboratory reporting and electronic case reporting can increase the timeliness of case reports.126-128 Implementation of electronic case reporting might support the inclusion of prescription and laboratory results from health system records into case reports; however, features of the electronic health record will influence the completeness of electronic case reporting case reports.128,129 Receipt of negative and positive hepatitis C antibody and RNA results aids in the detection of new infections and viral clearance.87,89,130,131 Matching case report data to vital records, immunizations, Medicaid, or other disease surveillance systems can improve the completeness of demographic information and identify neonates requiring hepatitis C testing. 132 Implementation of self-testing, which is not yet available in the United States, and point-of-care testing could link more people with hepatitis C to care and timely treatment, which could lead to an increase in case notifications via NNDSS. Yet, implementation of these testing strategies presents challenges, such as ensuring the accuracy of self-reported results and effectively integrating the data into surveillance systems.
The 2025 redesign of NHANES presents an opportunity to improve operational efficiency, participation rates, and stability of estimates and to extend data linkage activities.114-116 Additional approaches to supplement surveillance data could focus on leveraging other systems and surveys and developing nationally representative sampling studies for groups disproportionately affected by hepatitis C but not well represented in NHANES. For example, CDC’s HIV program has conducted the National HIV Behavioral Surveillance (NHBS) since 2003. 133 Every 3 years, NHBS is implemented among people who have injected drugs in the past 12 months (NHBS-PWID cycle). 133 PWID are recruited via respondent-driven sampling in 15 to 24 US cities with high HIV prevalence, and participants are interviewed and offered HIV testing.133,134 NHBS asks questions about hepatitis C testing and diagnosis in all participating cities and has funded a subset of cities to offer hepatitis C testing in 2012 (antibody only) and in 2018, 2022, and 2024 (antibody and RNA).133-135 Expanding the offer of hepatitis C testing to all participants and coverage of the project beyond urban areas with high HIV prevalence could increase understanding of the prevalence of hepatitis C among people with current or recent injection drug use. 136 Additionally, CDC is working with commercial laboratories to estimate the national prevalence of HCV infection using randomly selected, anonymized remnant sera from people who received any laboratory testing. 137 The much larger sample sizes could support stable estimates among sex and age groups, although race and ethnicity data are not available from this study. For NVSS, the transition to codes from the International Classification of Diseases, 11th Revision has begun in other countries; the timeline for adoption and impact on hepatitis C mortality rates in the United States is unknown, but the National Committee on Vital and Health Statistics has called for swift action to assess the impact of adoption on US health care systems, including public health and mortality statistics.15,138
Health system, pharmacy claims, and commercial laboratory data could provide timely indicators of prescribing, treatment uptake, and viral clearance.17,18 Data on testing and treatment from health care providers and health programs in settings that serve populations with ongoing risk for HCV transmission, such as syringe service programs, drug treatment programs, and carceral facilities, could ensure that resources are placed where they are needed most to cure hepatitis C and prevent new infections. CDC funded 18 jurisdictions to undertake special projects in these settings during May 2021–April 2026. 125 CDC also funded 8 jurisdictions to increase hepatitis C testing in these settings that serve populations with ongoing risk for HCV transmission and to build public health laboratory capacity for hepatitis C testing. CDC also funded 2 jurisdictions to explore the utility of integrating genomic and epidemiologic surveillance for outbreak detection and response. 125 In 2022, CDC funded a 5-year project to develop and survey a national network of syringe service programs. 139 These efforts could increase the availability of data to address hepatitis C elimination among PWID.
Public Health Implications
Achievement of hepatitis C elimination goals for the United States requires timely, accurate, and complete data, integrated with social determinants of health data. Funding from CDC supports efforts to strengthen the workforce and modernize systems for hepatitis C surveillance. Comprehensive national surveillance supports public health action to implement prevention and treatment efforts to eliminate hepatitis C.
Supplemental Material
sj-docx-1-phr-10.1177_00333549251351880 – Supplemental material for Hepatitis C Surveillance in the United States: Past, Present, and Future
Supplemental material, sj-docx-1-phr-10.1177_00333549251351880 for Hepatitis C Surveillance in the United States: Past, Present, and Future by Laurie K. Barker, Mona Doshani, Ruth B. Jiles and Eyasu Teshale in Public Health Reports®
Footnotes
Acknowledgements
The authors acknowledge Allison Ertl, PhD, and Neil Gupta, MD, of the Centers for Disease Control and Prevention Division of Viral Hepatitis for their helpful discussions, editorial comments, and guidance on the content of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily reflect the official position of the Centers for Disease Control and Prevention.
Supplemental Material
Supplemental material for this article is available online. The authors have provided these supplemental materials to give readers additional information about their work. These materials have not been edited or formatted by Public Health Reports’s scientific editors and, thus, may not conform to the guidelines of the AMA Manual of Style, 11th Edition.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
