Abstract
The application of a care continuum model (CCM) can identify gaps in diagnosis, care, and treatment of populations with a common condition, but challenges are inherent in developing a CCM for chronic hepatitis B. In contrast with treatment for HIV or hepatitis C, treatment is not indicated for all people with chronic hepatitis B, clinical endpoints are not clear for those receiving treatment, and those for whom treatment is not indicated remain at risk for complications. This topical review examines the data elements necessary to develop and apply chronic hepatitis B CCMs at the jurisdictional health department level. We conducted a nonsystematic review of US-based publications in Ovid MEDLINE (1946–present), Ovid Embase (1974–present), and Scopus (not date limited) databases, which yielded 724 publications for review. Jurisdictional health departments, if properly supported, could develop locale-specific focused CCMs using person-level chronic hepatitis B registries, updated longitudinally using electronic laboratory reporting data and case reporting data. These CCMs could be applied to identify disparities and improve rates in testing and access to care and treatment, which are necessary to reduce liver disease and chronic hepatitis B mortality. Investments in public health surveillance infrastructure, including substantial enhancements in electronic laboratory reporting and case reporting and the use of supplementary data sources, could enable jurisdictional health departments to develop modified CCMs for chronic hepatitis B that focus, at least initially, on “early” CCM steps, which emphasize optimization of hepatitis B diagnosis, linkage to care, and ongoing clinical follow-up of diagnosed people, all of which can lead to improved outcomes.
Care cascades or care continuum models (CCMs) are used to evaluate patient retention through the successive stages of care required to achieve successful treatment outcomes. Such models have been applied to several infectious conditions, including HIV, hepatitis C, and tuberculosis, and have been used to identify gaps and disparities in diagnosis, access to treatment, and outcomes.1,2 The utility of such models hinges on several things: the capacity to diagnose the condition, the existence of treatment for all people with the condition, and measurable treatment outcomes indicating success. For conditions such as HIV or hepatitis C, diagnosis is typically straightforward, treatment is indicated for all, and an undetectable viral load indicates success; moreover, from a public health standpoint, this undetectability indicates prevention of further transmission.1,3
Conceptualizing a similarly structured CCM for chronic hepatitis B (CHB) is challenging. Although CHB diagnosis is straightforward, CHB has a variable and dynamic course. 4 Although many people with CHB are initially not eligible for antiviral treatment based on current guidelines, 5 long-term monitoring for active hepatitis and disease progression is recommended to determine whether an indication for treatment has developed. If treatment is initiated, however, no single indicator of ultimate treatment success exists.5,6 Although seroclearance of hepatitis B surface antigen (HBsAg; ie, functional cure) would be a useful clinical endpoint, it occurs infrequently among untreated people (about 1% annually), and frequency is even lower among people who are receiving continuous nucleos(t)ide analogue antiviral therapy (0.22%-0.31% annually).7-10 Furthermore, it is unclear how people diagnosed with CHB for whom antiviral therapy is not indicated would fit into such a model. Such people remain HBsAg positive, are at risk for disease reactivation and hepatocellular carcinoma (HCC), and continue to be a source for further transmission.5,6
Nonetheless, if the existing CCM in its entirety does not accommodate the natural history and comparative treatment shortcomings of CHB, it may be worthwhile to examine gaps and disparities in intermediate steps in the model (eg, frequency of linkage to hepatitis B–directed care). If high retention at an intermediate step is associated with improved outcomes (eg, lower rates of HCC), then such a metric might serve as a proxy measure for some degree of care success. Given the need to optimize the use of perennially limited resources, a focused model approach might be useful and feasible for US jurisdictional (ie, state, county, metropolitan area) health departments (JHDs).
Among people identified with CHB, the goals of viral hepatitis programs in JHDs are to promote measures to prevent secondary hepatitis B virus (HBV) transmission; identify family members, household members, or other close contacts who are infected; and reduce CHB morbidity and mortality. According to the US Department of Health and Human Services Viral Hepatitis Elimination Plan for 2021-2025, mortality reduction refers to a decrease in the US hepatitis B mortality rate to 0.16 cases per 100 000 people by 2030. 11 As of 2020, the rate was 0.45 cases per 100 000 people, approximately 3 times higher than the 2030 target. 12
The objective of the current review was to examine some of the ways JHDs might develop focused CCMs to examine longitudinal follow-up and gaps in care among people living with CHB. To identify and address the challenges inherent in developing a CHB CCM and, more importantly, to examine metrics that might serve as proxy measures for interim care and management success, we provide a narrative (ie, nonsystematic) review of recent US-based publications. We selected publications that examined important elements of a typical CCM: screening and diagnosis of CHB, linkage to hepatitis B–directed care, eligibility for and receipt of antiviral therapy, and clinical outcomes among CHB patients in care. We review findings from these studies, discuss the relevance of these findings to JHDs, and explore potential practices, including the use of data from external health systems, that health departments might use to develop and conduct longitudinal, electronic laboratory reporting (ELR)–based follow-up of people with CHB identified through public health surveillance. To our knowledge, this is the first review to examine the data elements necessary for JHDs to develop and apply CCMs for people identified with CHB through public health surveillance.
Methods
For this review, we examined US-focused published English-language articles (primary data or commentaries) in Ovid MEDLINE (1946–present), Ovid Embase (1974–present), and Scopus (not date limited) databases. Our general search terms included “hepatitis B care cascades” or “cure cascades” or “care or cure continuum.” Additional terms included “hepatitis B diagnosis,” “hepatitis B access to care,” and “hepatitis B treatment,” each of which focused on specific elements of the CHB CCM. The base literature search was conducted on November 8, 2021; additional pertinent articles were added to this review subsequent to that date if they met the search criteria.
Discussion
We organized the findings of this review, which included 724 unique publications, according to specific elements of the CHB CCM: identification and enumeration of prevalent cases of CHB; linkage to and retention in hepatitis B–directed care; and antiviral treatment candidacy, initiation, continuation, and outcomes in the era of CHB antiviral therapy.
Identification and Enumeration of Prevalent Cases of CHB
While CHB is a nationally notifiable condition, it is not reportable by law in all US states. 13 Furthermore, JHDs, especially in regions where CHB prevalence is relatively high, may have limited resources available to develop and maintain longitudinal CHB registries. Surveillance reports published annually by the Centers for Disease Control and Prevention’s Division of Viral Hepatitis document newly identified CHB cases from a case-based reporting system; this system cannot currently provide person-based prevalence estimates or longitudinal assessments. 13 The National Health and Nutrition Examination Survey (NHANES), therefore, has been the basis for CHB prevalence estimates in the United States. 14
In an NHANES estimate for 2013-2018, 880 000 (95% CI, 580 000-1 170 000) or 0.3% (95% CI, 0.2%-0.4%) of the US noninstitutionalized population aged ≥6 years were living with CHB. 14 These estimates have been relatively consistent since 1999, with non–US-born people constituting approximately 70% of all cases and having a CHB prevalence roughly 10 times that of the general US population.15,16 Lim et al 17 supplemented CHB estimates in NHANES with additional prevalence data for groups underrepresented by NHANES (eg, people who are incarcerated, people who inject drugs, people experiencing homelessness, people born in HBV-endemic countries) and estimated overall US prevalence at 1.59 million people (range, 1.25-2.49 million) as of 2019. Wong et al 18 updated CHB prevalence among non–US-born people by using country-specific and US Census data, then added US-born prevalence figures from Lim et al 17 (0.42 million people) to the non–US-born total and estimated the prevalence of CHB in the United States in 2018 at 1.89 million people, with an upper limit of 2.4 million people.
Because people with CHB are generally asymptomatic, a valuable feature of NHANES is that participants, before serologic testing, are asked whether they have been previously diagnosed with hepatitis B. Kim et al 19 reviewed NHANES data from 2013-2016 and reported that only 33.9% of participants with CHB were aware of their infection status. In another recent analysis, using data from NHANES and the Truven Health MarketScan Database, investigators estimated that 18.6% of privately insured people living with CHB had been diagnosed. 20
Linkage to and Retention in Hepatitis B–Directed Care
Proper clinical management of people with CHB entails regular follow-up with clinicians who are knowledgeable about hepatitis B management and treatment guidelines. Despite differences in treatment thresholds in the guidelines issued by major liver disease societies worldwide, all recommend regular assessments of serum alanine aminotransferase (ALT) and HBV DNA levels, as well as hepatitis B e antigen (HBeAg) status.5,21,22 Study definitions for linkage to care vary to some degree among investigators; for example, it may be defined as people newly diagnosed with CHB who receive an initial CHB-focused clinical assessment within a defined period.23-26 Indicators of sustained care include documentation of ALT, HBV DNA, and HBeAg testing over time and of hepatic ultrasounds for HCC surveillance.23-26
In the United States, rates of linkage to sustained hepatitis B–related care among people diagnosed with CHB in general health care settings have been consistently low. In a study using administrative claims data (Truven Health MarketScan) for adults aged <65 years with private, employer-based health plans, Harris et al 25 examined linkage to care during 2008-2016 for 16 644 people with CHB. Defined as the presence of procedure codes for disease activity assessment >12 months after CHB diagnosis (ie, serum ALT with HBV DNA or HBeAg determination), linkage to care was 36%. Also using MarketScan data that included people aged ≥65 years, Tran et al 23 found that after 3 years of follow-up, only 33% of people with CHB had ALT, HBV DNA, or HBeAg assessments after CHB diagnosis. This detailed study, involving >55 000 US patients with CHB, also examined long-term disease monitoring and found that only 25% of people with CHB underwent ALT and HBV DNA assessments annually, and among those at higher risk of HCC, <40% underwent annual HCC surveillance. Among patients in the Veterans Health Administration (N = 12 450) during 1999-2013, Serper et al 27 found low frequencies of HBV DNA testing (44%) and HBeAg assessment (<50%) within 2 years of diagnosis; among those with cirrhosis, 31% never received an ultrasound for HCC surveillance.
More recent data suggest improvement in clinical monitoring among patients in care. Pham et al 28 analyzed Truven Clinformatics claims data from 2016-2019 and found that approximately half of patients with and without cirrhosis received ≥1 ALT and HBV DNA or HBeAg result, and approximately 50% of patients without cirrhosis (men aged >40 years and women aged >50 years) and 80% of patients with cirrhosis received 1 liver imaging study within 12 months of CHB diagnosis. Patients who were male, Asian, or privately insured or who had cirrhosis were more likely than those who were female, non-Asian, publicly insured, or without cirrhosis to receive ALT and HBV DNA or HBeAg tests and imaging studies within 12 months of diagnosis.
Numerous studies during the past few decades have aimed to identify specific (and potentially modifiable) barriers to hepatitis B–directed care. Those with CHB, especially non–US-born people, can face stigmatization and other discriminatory practices, creating disincentives for screening and engagement in medical care. 29 In a review of US-based studies published during 2007-2018, Ispas et al 30 classified obstacles to CHB care in the United States. Patient barriers included cultural stigma, lack of knowledge of CHB risks, a perception of limited treatment options, and financial concerns. Health care provider barriers centered on lack of disease knowledge and familiarity with CHB guidelines, which were more problematic among primary care providers than among specialists.
Antiviral Treatment Candidacy, Initiation, Continuation, and Outcomes in the Era of CHB Antiviral Therapy
Antiviral drugs for CHB, although not curative, suppress viremia, prevent progression to cirrhosis, lower the risk of HCC, and reduce all-cause and liver-related mortality.31-33 The 2 CHB antiviral agents most commonly in use, tenofovir and entecavir, are relatively well-tolerated and have a high threshold for resistance despite administration as monotherapy. People with CHB and evidence of active liver disease (ie, elevated ALT and HBV DNA levels) or cirrhosis are eligible for treatment according to major guidelines.5,21,22 Among those without cirrhosis, eligibility criteria for antiviral treatment are complex. In contrast with HIV and hepatitis C virus infection—for which treatment is typically indicated at diagnosis—determination of treatment eligibility for CHB initially necessitates determination of fibrosis status,34,35 accompanied by longitudinal assessments of ALT and HBV DNA levels and HBeAg status,5,21,22 although appeals have been made to simplify and expand treatment eligibility criteria. 36
In US cohorts, the estimated frequencies of treatment eligibility and initiation, cirrhosis prevalence, and the distribution of CHB phases can be observed from studies that examined some of these issues. Among Alaska Native people with HBeAg-negative CHB, 25% met criteria for immune-active CHB (a major determinant of treatment eligibility). 37 Among CHB patients attending community-based clinics in California, investigators found that 29% of treatment-naïve CHB patients became eligible for treatment after 3 years of follow-up. 38 Evidence suggests, however, that many eligible CHB patients do not receive antiviral treatment. In a large analysis using NHANES and MarketScan data collected during 2007-2014, only one-third of privately insured CHB patients with cirrhosis and one-half with HCC had received antiviral therapy. 20 Pham et al 28 reported that only 29% of patients with cirrhosis during 2016-2019 received antiviral treatment within 12 months of CHB diagnosis. 28 Data suggest that many patients discontinue, or have interruptions in, CHB treatment after initiating it.38,39 Despite evidence of suboptimal antiviral coverage, other data from North American cohorts suggest a decline in the frequency of severe hepatic outcomes and mortality among closely monitored and treated patients.27,31-33,37,40-42
Public Health Practice Implications
These recent publications outlined many of the challenges in developing CCMs for CHB. Nationally, CHB prevalence estimates range widely, many people remain undiagnosed, and many diagnosed people are not linked to care. Treatment is recommended only for some people and, if initiated, is often interrupted, and achievable endpoints are undefined. People with CHB not indicated for treatment remain at risk for reactivation, HCC, and HBV transmission.
Despite the challenges, some aspects of these publications revealed potential interim care steps for which high retention appeared favorable to the reduction of severe CHB outcomes. In the United States, people with CHB in regular care who have access to antiviral treatment demonstrated low frequencies of severe hepatic outcomes and liver-related mortality. JHDs with sufficient resources and access to pertinent ELR data (Table 1) could develop focused CHB CCMs specific to their own jurisdictions (Table 2). These publications also illustrated CCM elements that would be unreliable and resource intensive to pursue (eg, determinations of treatment eligibility, initiation, and interruption). Given the variability in resources available, JHDs might restrict CCM development to certain core elements and reserve other enhanced elements for later, as resources become available. For example, JHDs with established CHB registries might focus on identifying gaps in CHB diagnosis and linkage to care (Table 3).
Strategic components a for development of a chronic hepatitis B (CHB) care continuum model in jurisdictional health departments
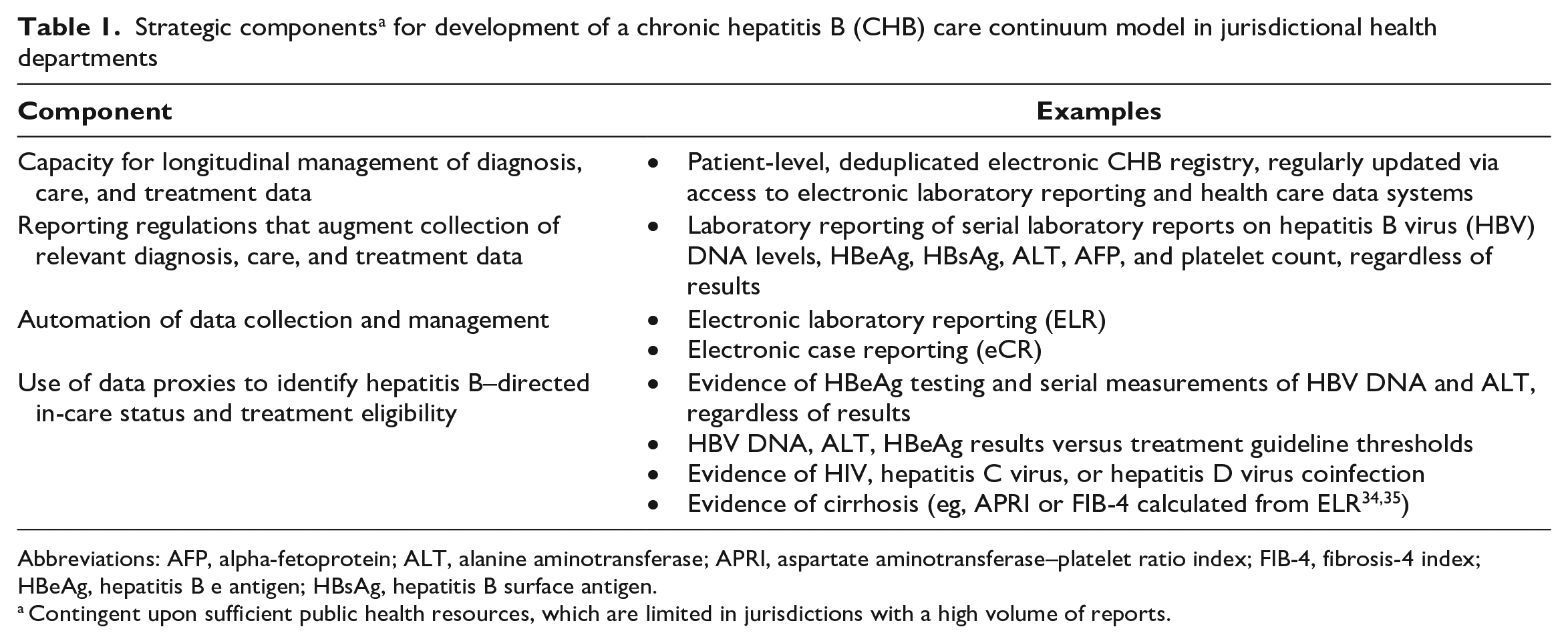
Abbreviations: AFP, alpha-fetoprotein; ALT, alanine aminotransferase; APRI, aspartate aminotransferase–platelet ratio index; FIB-4, fibrosis-4 index; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen.
Contingent upon sufficient public health resources, which are limited in jurisdictions with a high volume of reports.
Strategic data elements for monitoring chronic hepatitis B (CHB) care continuum models in jurisdictional health departments
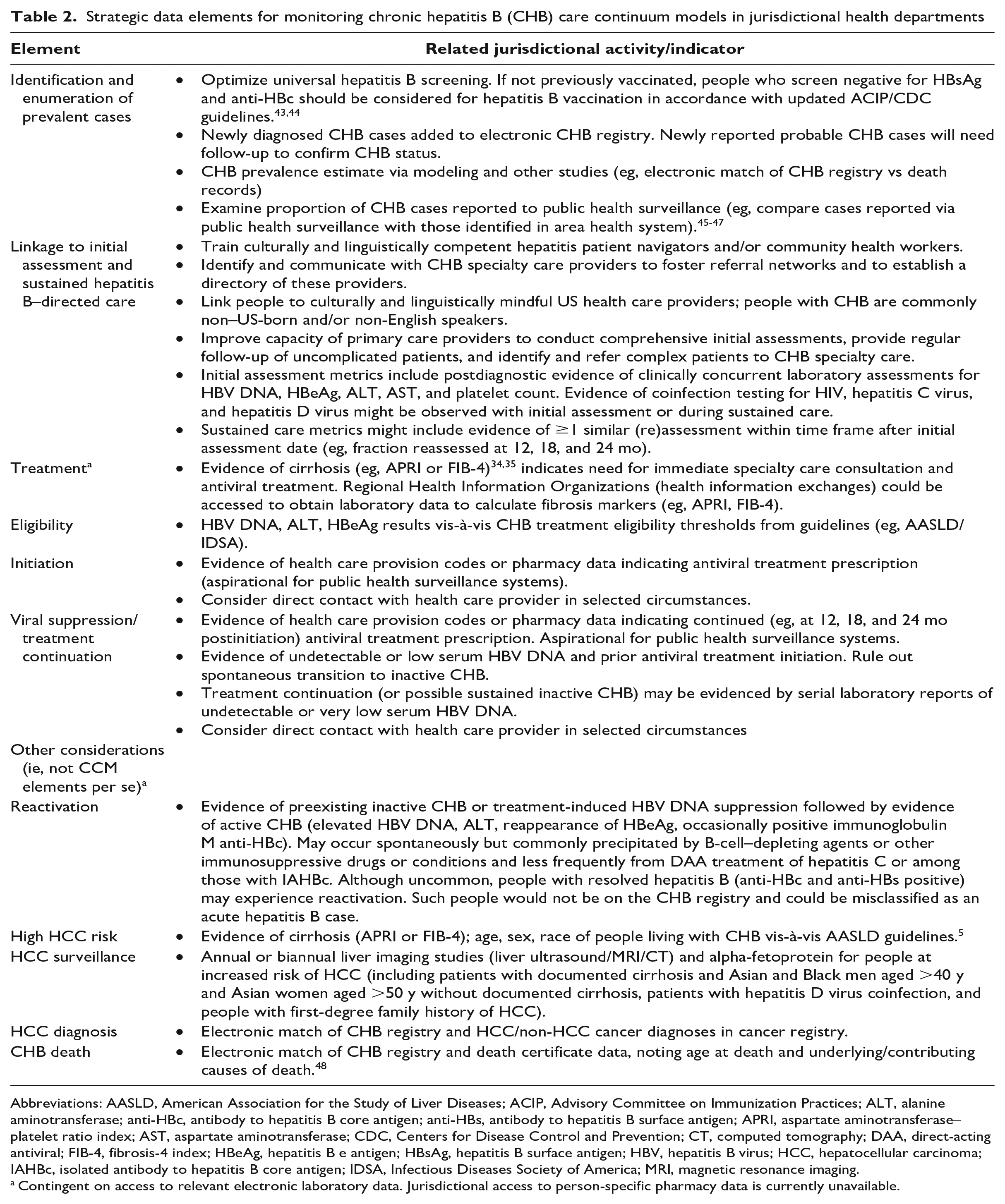
Abbreviations: AASLD, American Association for the Study of Liver Diseases; ACIP, Advisory Committee on Immunization Practices; ALT, alanine aminotransferase; anti-HBc, antibody to hepatitis B core antigen; anti-HBs, antibody to hepatitis B surface antigen; APRI, aspartate aminotransferase–platelet ratio index; AST, aspartate aminotransferase; CDC, Centers for Disease Control and Prevention; CT, computed tomography; DAA, direct-acting antiviral; FIB-4, fibrosis-4 index; HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; IAHBc, isolated antibody to hepatitis B core antigen; IDSA, Infectious Diseases Society of America; MRI, magnetic resonance imaging.
Contingent on access to relevant electronic laboratory data. Jurisdictional access to person-specific pharmacy data is currently unavailable.
Public health data-to-care indicators and potential actions a for jurisdictional health departments to optimize hepatitis B–directed care

Abbreviations: HBeAg, hepatitis B e antigen; HBsAg, hepatitis B surface antigen.
Contingent upon sufficient public health resources, which are limited in jurisdictions with a high volume of reports.
If universal screening 43 leads to increases in CHB diagnoses, then newly identified cases should, in most jurisdictions, be reported to their respective surveillance programs. Consequently, these newly diagnosed cases could be incorporated continually into the CHB registry. A robust CHB registry provides the foundation for CCM development and focused application of public health actions to reduce CHB morbidity and mortality. As a core exercise, programs could compare characteristics of people with CHB in their registries with the demographic composition of their jurisdiction, potentially illustrating gaps and disparities in screening and diagnosis. Furthermore, identification of case contacts for testing and vaccination is necessary to prevent further transmission, and people of reproductive age with CHB should be monitored to prevent perinatal HBV transmission should they become pregnant. Jurisdictional CHB surveillance programs should work closely with perinatal hepatitis B prevention programs to identify pregnant people with CHB, so that preventive measures are instituted if indicated.5,49 People in the CHB registry should be tested for HIV and hepatitis C virus and considered for hepatitis D virus testing, as recommended by the American Association for the Study of Liver Diseases. 5 Among CHB cases with HIV coinfection, public health viral hepatitis programs might benefit from collaboration with affiliated public health HIV programs to augment data collection and case follow-up efforts. The American Association for the Study of Liver Diseases recommends that antiretroviral treatment among people with HBV/HIV coinfection include 2 drugs with activity against HBV (ie, tenofovir with emtricitabine or lamivudine). 5 As for HIV preexposure prophylaxis among people with CHB, recent data suggest that tenofovir/emtricitabine-based preexposure prophylaxis can reduce the transmission of HBV among HIV-negative men who have sex with men. 50 Always, programs should promote hepatitis B screening of all adults and vaccination for all people aged 19-59 years and hepatitis A vaccination for people with CHB and for high-risk unvaccinated people regardless of HBV infection status.43,44,51-53
To examine linkage to care, programs with CHB registries could determine the frequency of completed initial hepatitis B assessments within a designated period after diagnosis as a primary data point and the frequency of sustained follow-up as a secondary measurement. To do so, however, these programs would need longitudinal access to patient-level data, including HBV DNA and ALT levels. For JHDs with adequate resources, access to other laboratory data, such as platelet counts, could enable surveillance programs to identify people with cirrhosis,34,35 who should receive treatment and undergo HCC surveillance every 6 to 12 months.5,21,22
Using ELR data to determine antiviral treatment initiations and interruptions is unreliable using ELR data, because untreated people with inactive disease might be indistinguishable from people with formerly active CHB who have responded to treatment. JHDs should delay efforts to determine treatment eligibility and delivery unless they have access to pharmacy or electronic case reporting (eCR) data, which are aspirational at present. 54 We outline proxy indicators of treatment in the absence of pharmacy or eCR data (Table 2), but as mentioned, these indicators are unreliable and resource intensive to examine.
Although our review focused on the application of CHB CCMs in JHD settings, the same CCM framework could be used in other settings and populations. For example, large private-sector health systems operate by using longitudinally based interconnected electronic information systems with patient-level data on laboratory and diagnostic test results, clinical encounters, and drug prescriptions—all of which are requisite components for CCM development (Table 1). Given the expansive catchment areas of many of these health systems,45,46,55 JHDs could collaborate with such systems, leveraging health care data to monitor clinical follow-up of people in the surveillance CHB registry. Completeness of case reporting to JHDs could be evaluated by comparing CHB patients diagnosed in area health systems with CHB cases reported to public health.45,46
Future Directions
Because limited resources are available to conduct comprehensive CHB surveillance, jurisdictions might leverage other tools to improve CCM component estimates. For example, as health care facilities throughout the United States transition from traditional reporting to eCR, 56 CDC has launched the Data Modernization Initiative (DMI), which includes a nationwide effort to improve case reporting for all reportable conditions. 57 A major objective of DMI is to modernize public health surveillance systems so that core public health data are ready for use in cloud-enabled public health systems. Response to the COVID-19 pandemic has accelerated the implementation of eCR, improved completeness of health care provider–based reporting, and enabled the collection of patient demographic and clinical information that were inaccessible or incomplete via traditional reporting. 58
Activities to increase reporting of people newly diagnosed with CHB could include matching a list of area clinical providers and facilities with those currently reporting hepatitis B cases to the JHD, potentially identifying where outreach is needed to implement (or improve) eCR. Because case detection and investigation require a laboratory report indicating infection, similar outreach to clinical laboratories might identify gaps in laboratory reporting. Additional hepatitis surveillance staff may be needed to adequately classify cases (ie, acute vs chronic, confirmed vs probable) and then conduct the necessary follow-up if nonengagement in care or a need for immediate case management referral (eg, pregnant people) is indicated. To bolster case completeness, data sources such as electronic health records and health insurance claims data could be incorporated into a jurisdictional CHB registry. 59 Data proxies from laboratory data could indicate a person is not in care or needs immediate case management referral (Table 3).
In the future, annual US viral hepatitis surveillance reports might include data analyzed by JHDs from their CHB registry. Enhanced jurisdictional investigations to augment the CHB CCM with outcome data might include matching cases in the CHB registry with death certificate and/or cancer registry data. Such matching could provide valuable clinical outcome data on registry cases, including underlying and contributing causes of death, age at death, and incident hepatic and nonhepatic malignancies. 48
Despite the challenges in developing CHB CCMs akin to those of HIV and hepatitis C virus, JHDs could nonetheless apply focused CCMs with improvements to electronic infrastructure, including access to supplementary data sources such as private health care networks. Initial, core CCM activities should focus on the development of CHB registries. JHDs with registries might use longitudinal ELR data to gauge linkage to and retention in hepatitis B–directed care, which can lead to the reduction of severe outcomes and mortality. As resources increase and data sources improve, JHDs might expand CCM activities to include assessments of treatment eligibility and delivery, HCC surveillance delivery and case detection, and causes of death among people in the CHB registry.
Footnotes
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily reflect the official position of the Centers for Disease Control and Prevention or the authors’ affiliated institutions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received funding from the United States government.
