Abstract
Objectives
The New York City (NYC) Department of Health and Mental Hygiene (DOHMH) depends on reporting by health care facilities and laboratories for disease surveillance. Our objective was to evaluate the completeness of DOHMH surveillance to identify births to hepatitis B virus (HBV)–positive women to prevent perinatal transmission.
Methods
We identified infants born to HBV-positive women by matching mothers of all infants born in NYC during May 1, 2013–May 1, 2014, identified from the Citywide Immunization Registry (CIR) to persons with HBV-positive laboratory reports in the Electronic Laboratory Reporting (ELR) system. We then matched infants born to mothers identified in the CIR/ELR match to infants born to HBV-positive women from the DOHMH perinatal HBV surveillance database. We performed capture–recapture analysis to evaluate completeness of DOHMH case identification. We compared the proportion of infants born to HBV-positive mothers reported to DOHMH with the proportion of infants identified only through the CIR/ELR match for receipt of postexposure prophylaxis (PEP) and completion of the HBV vaccination series and post-vaccination serology testing.
Results
Of 1662 infants identified from the CIR/ELR match and 1554 infants in the DOHMH database, 1493 infants matched. Of 169 infants only in the CIR/ELR data set, 55 were born to HBV-positive women residing in NYC. Sixty-one infants were only in the DOHMH database. An estimated 2 infants were not identified by either method. The CIR/ELR match increased infant identification by 3.5%, from 1554 to 1609 infants. The proportion of infants who received PEP was significantly higher among infants whose mothers were reported to DOHMH (vs not reported to DOHMH).
Practical Implications
Use of the CIR/ELR match may further improve DOHMH identification of infants born to HBV-positive women and receipt of infant PEP.
Infants are at risk for acquiring hepatitis B virus (HBV) from infected women during childbirth. 1,2 Approximately 90% of infants who acquire HBV perinatally will become chronically infected, of whom an estimated 25% will die of HBV-related complications. 3,4 Timely administration of postexposure prophylaxis (PEP), consisting of hepatitis B immune globulin and the first dose of HBV vaccine at birth, followed by completion of the HBV vaccination series, is up to 95% effective at preventing transmission. 5,6
To identify HBV-positive pregnant women, New York State Public Health Law and New York City Health Code mandate prenatal serologic screening for hepatitis B surface antigen (HBsAg), an indicator of HBV infection, and the reporting of positive HBsAg results by health care facilities and laboratories to health departments. 7 -9 In 2016, approximately 1500 women infected with HBV delivered a live birth in New York City. 10 The New York City Department of Health and Mental Hygiene (DOHMH) Perinatal Hepatitis B Prevention Program conducts outreach to help prevent mother-to-child transmission of hepatitis B. Outreach involves educating HBV-positive pregnant women about HBV infection and the importance of infant PEP at birth and providing medical referrals if needed. DOHMH also works with prenatal care providers, delivery facilities, and pediatricians to ensure that infants born to HBV-positive pregnant women receive PEP at birth, complete the HBV vaccination series, and complete post-vaccination serology testing (PVST) to assess the infant’s immunity and infection status. 6 Identification of all HBV-positive pregnant women is critical to DOHMH prevention efforts.
Sources of reports of HBV infection in pregnancy to the DOHMH include laboratory reports indicating concurrent HBV infection and pregnancy; clinical reports from health care facilities, including prenatal care and delivery facilities; and Newborn Metabolic Screening Forms indicating maternal HBV infection. The DOHMH surveillance system depends on health care facilities to submit reports to the health department, but not all health care facilities are compliant.
We used a capture–recapture methodology to evaluate the completeness of the existing DOHMH surveillance system to identify infants born to HBV-positive pregnant women. This methodology was originally developed by ecologists to estimate animal populations, in which a portion of the population is captured, marked, and released. Another capture is performed at a later time, and the number of marked animals in the second portion is used to estimate the total population size, using the assumption that the proportion of marked animals in the second capture is proportional to the number of marked animals in the total population. 11 The capture–recapture methodology is used to evaluate disease surveillance systems, including a perinatal HBV surveillance system, by comparing 2 independent data sources to determine the number of records captured by each source and by both sources. 12 -19 A model is then applied to estimate the total population size and derive the number of records not captured in either data source. 20 -22 We evaluated the completeness of DOHMH surveillance to identify births to HBV-positive women and compared rates for receipt of PEP, completion of the HBV vaccination series, and completion of PVST among infants born to women who were reported to DOHMH with rates among infants born to women who were not reported to DOHMH.
Materials and Methods
The first independent data source was the DOHMH perinatal HBV surveillance database, which includes reports of infants born to HBV-positive women reported to DOHMH, hereinafter referred to as the DOHMH database. 23 The DOHMH database contains maternal and infant identifiers and HBV laboratory results for the women. 23 We created a data set from this source, which included all maternal–infant pairs with an infant birth date from May 1, 2013, through May 1, 2014, for which the woman was a New York City resident and had a positive HBsAg result collected during the prenatal period.
The second independent data source was the resulting data set from a match between the New York City Citywide Immunization Registry (CIR) and the New York City Electronic Laboratory Reporting (ELR) system, referred to hereinafter as the CIR/ELR data set. 24,25 The CIR is the DOHMH’s immunization information system and contains vital records data for all infants born in New York City, regardless of vaccination status, and maternal identifiers. 24 The CIR data set functioned to identify women who gave birth in New York City during the study period. The ELR database contains HBV laboratory results for New York City residents and was used to identify all persons with a positive HBV laboratory result during the study period. 25 We matched the CIR and ELR databases by using a deterministic matching algorithm that incorporated mother’s name, date of birth, telephone number, and address. To maximize the number of matching records returned, we conducted multiple matches of decreasing likelihood categories consecutively, and the results were de-duplicated to remove matches that were identified in multiple matching categories (Table 1). The CIR/ELR data set included only infants born from May 1, 2013, through May 1, 2014, to women who were residing in New York City and had at least 1 positive maternal HBV laboratory result, including HBsAg, hepatitis B e-antigen, or HBV DNA results, collected from August 1, 2012, through July 31, 2014. We chose the collection dates to account for the earliest possible prenatal period corresponding to the earliest delivery date and for the latest possible prenatal period corresponding to the latest delivery date plus 3 additional months to account for women who may have been tested soon after delivery (ie, prenatal testing was not performed).
Algorithms and results for matches between the Citywide Immunization Registry (CIR) and the Electronic Laboratory Reporting (ELR) system and between the CIR/ELR matched data set and the Department of Health and Mental Hygiene (DOHMH) Perinatal Hepatitis B Virus Surveillance Database among infants born from May 1, 2013, through May 1, 2014, New York City a

Abbreviation: DOB, date of birth.
aData sources: Electronic Clinical Laboratory Reporting System, Citywide Immunization Registry, Perinatal Hepatitis B Virus Surveillance Database. 23 -25
bLikelihood refers to the probability that the match of the selected variables is a true match. High indicates a definite match that did not require manual review, medium indicates a probable match that required manual review, and low indicates a possible match that required manual review.
cVariables pertain to the mother unless otherwise indicated.
We conducted a match between the 2 independent data sources (DOHMH database and CIR/ELR data set) to determine the number of infants born to HBV-positive pregnant women identified by both sources and by each source. This match was a deterministic match, which incorporated a CIR key (ie, a unique identifier assigned to each person in the CIR) and mother and infant demographic characteristics from both data sources. We conducted multiple matches of decreasing likelihood categories (Table 1) consecutively.
We further investigated infants identified only in the CIR/ELR data set but not in the DOHMH database through medical record review to confirm that the infant was truly born to an HBV-positive woman who resided in New York City at the time of delivery. We included only infants who were verified as born to an HBV-positive woman residing in New York City at delivery in subsequent analyses; we excluded all other infants. We investigated reasons for reporting failures. We collected data on receipt of PEP, completion of the HBV vaccination series, and completion of PVST for infants with reporting failures.
We used the Fisher exact test to compare the proportions of infants who received PEP, completed the HBV vaccination series, and completed PVST who were identified via the CIR/ELR match only with the proportion of those in the DOHMH database. We assessed PVST completion regardless of HBV vaccination status. Using the total number of infants identified by both data sources and each data source individually, we applied the unbiased version of the Lincoln–Petersen formula (Figure 1) to estimate the total number of infants and derive the estimated number of infants who were not identified by either data source. 26 We used the standard error formula to calculate 95% confidence intervals (CIs) for the total population estimate. 27 We used SAS version 9.3 to conduct matches between data sets and analyses. 28 We conducted this evaluation as part of routine surveillance to identify infants born to HBV-infected women; as such, we did not pursue institutional review board review.

Results
We compared 182 003 HBV records in the ELR system for persons not reported as male with positive HBV test results with 117 273 birth records in the CIR based on matching with maternal identifiers. After de-duplication of the CIR/ELR data set and removal of match results with non–New York City addresses, we found 1662 possible records for infants born to HBV-positive women residing in New York City. The comparison of 1662 records in the CIR/ELR data set with 1554 records in the DOHMH database resulted in 1493 matched infants. Records of 169 infants were in the CIR/ELR data set but not in the DOHMH database, of whom 55 were verified to be born to HBV-positive women residing in New York City after investigation; the remaining 114 were excluded. An additional 61 infants were not identified by the CIR/ELR data set but were in the DOHMH database (Figure 2).
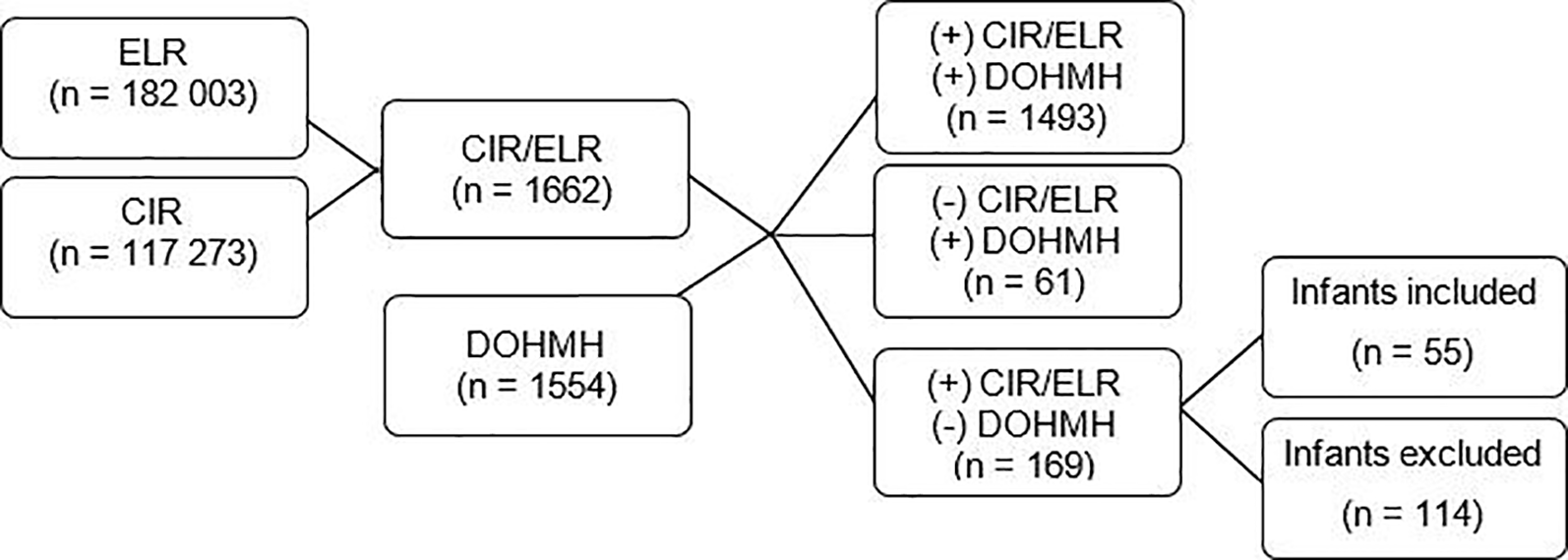
Results of the Citywide Immunization Registry (CIR) and Electronic Laboratory Reporting (ELR) system match and comparison with the New York City Department of Health and Mental Hygiene (DOHMH) Perinatal Hepatitis B Virus (HBV) Surveillance Database for infants born to HBV-positive women in New York City, May 1, 2013, through May 1, 2014. The presence (+) and absence (–) of infants identified in each data set are specified. ELR refers to electronic laboratory reports for all positive hepatitis B results. CIR refers to infant births from the Citywide Immunization Registry. CIR/ELR refers to a data set of matched records indicating potential births to HBV-positive women. DOHMH refers to reports received by DOHMH surveillance database of births to HBV-positive women. Infants included were those who, after investigation, were confirmed as born to an HBV-positive woman residing in New York City at delivery; all other infants were excluded. Data sources: Perinatal Hepatitis B Virus Surveillance Database, Citywide Immunization Registry, Electronic Clinical Laboratory Reporting System. 23 -25
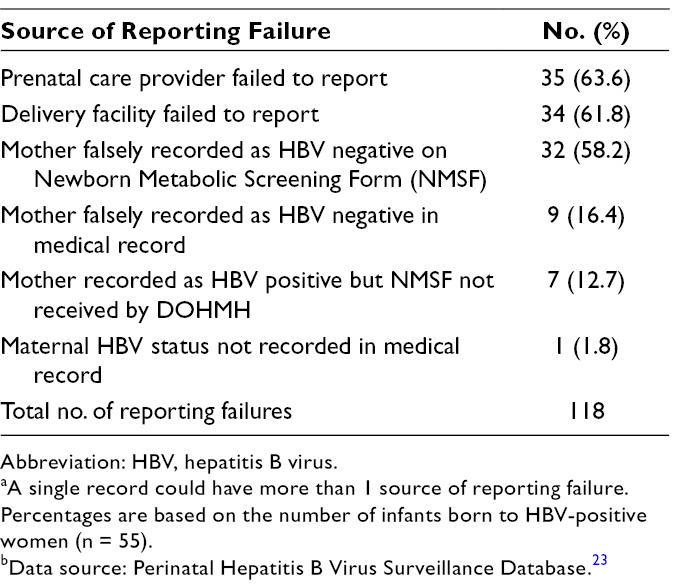
The most common sources of reporting failure in the records of the 55 infants who were identified by the CIR/ELR match but not in the DOHMH database were (1) the prenatal care facility failed to report the mother (n = 35 records), (2) the delivery facility failed to report the mother (n = 34 records), and (3) the mother was falsely recorded as HBV negative on the Newborn Metabolic Screening Form (n = 32 records) (Table 2). These 55 unreported infants were born at 19 facilities, 4 of which accounted for 29 (53%) infants. The proportion of infants who received PEP was significantly higher among infants of women who were reported to DOHMH (99.3% [1543 of 1554]) than among infants of women who were not reported to DOHMH (87.3% [48 of 55]) (P < .001) (Table 3). Compared with infants of women who were not reported to DOHMH, infants of women who were reported to DOHMH were also more likely to have completed the HBV vaccination series (90.6% [1408 of 1554] vs 87.3% [48 of 55]) and to have completed PVST (86.6% [1346 of 1554] vs 81.8% [45 of 55]), but these differences were not significant (P = .53 and P = .40, respectively). Among infants who completed PVST, none of the 55 infants whose mothers were not reported to DOHMH and 6 of the 1346 (0.4%) infants whose mothers were reported to DOHMH were infected; this difference was not significant (P > .99).
Abbreviation: HBV, hepatitis B virus.
aA single record could have more than 1 source of reporting failure. Percentages are based on the number of infants born to HBV-positive women (n = 55).
bData source: Perinatal Hepatitis B Virus Surveillance Database. 23
Differences in management and outcome for infants born to HBV-positive women in New York City among pregnant HBV-infected women reported and not reported to the New York City Department of Health and Mental Hygiene, May 1, 2013, through May 1, 2014 a
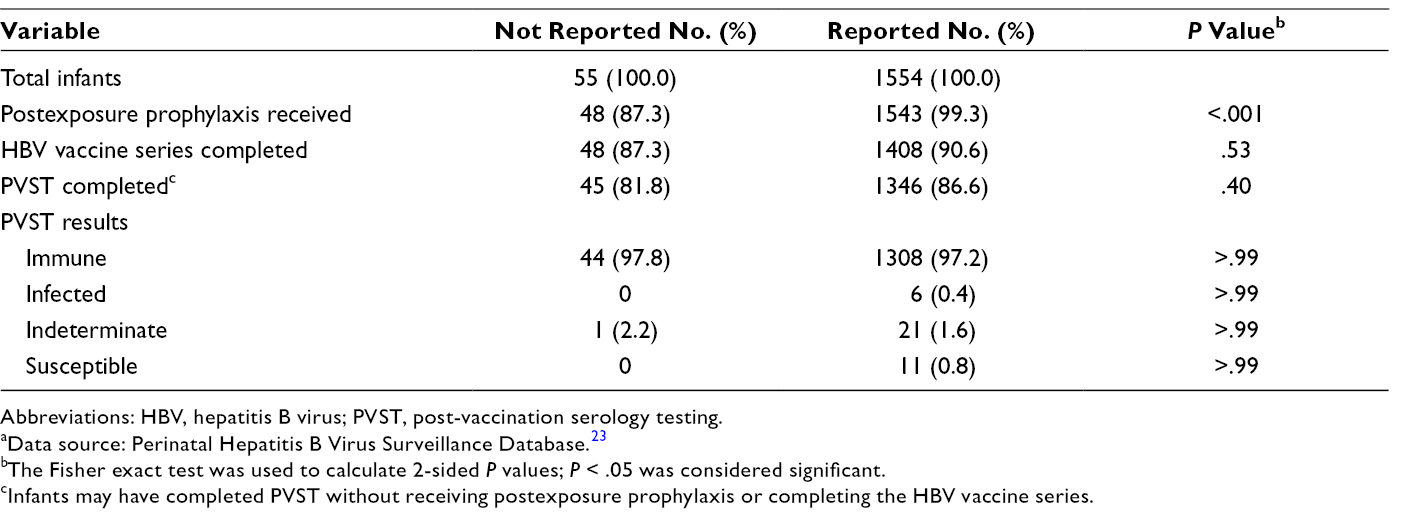
Abbreviations: HBV, hepatitis B virus; PVST, post-vaccination serology testing.
aData source: Perinatal Hepatitis B Virus Surveillance Database. 23
bThe Fisher exact test was used to calculate 2-sided P values; P < .05 was considered significant.
cInfants may have completed PVST without receiving postexposure prophylaxis or completing the HBV vaccine series.
The unbiased Lincoln–Petersen formula estimated that 1611 infants were born to HBV-positive women in New York City during the study period. Based on this estimate, the mothers of 57 (3.5%) infants were not reported to DOHMH, including 2 (0.1%) who were not identified by either the DOHMH database or the CIR/ELR data set. The CIR/ELR match resulted in a 3.5% (n = 55; from 1544 to 1609 infants) increase in the number of infants identified by DOHMH.
Discussion
This capture–recapture analysis demonstrated that most (96.5%) infants born to HBV-positive women in New York City were identified by DOHMH through routine surveillance. However, the proportion of infants reported fell short of the DOHMH Perinatal Hepatitis B Prevention Unit’s goal of identifying 100% of infants born to HBV-positive women. 10 Infants born to women who were not reported to DOHMH were significantly less likely to have received PEP and, although not a significant result, were also less likely to have completed the HBV vaccination series than infants born to women who were reported to DOHMH. Given that PEP and completion of the HBV vaccination series are up to 95% effective at preventing perinatal HBV transmission, this difference supports the importance of timely prenatal reporting to DOHMH and the effectiveness of the DOHMH infant case management protocol. 5 Our findings further supported this point because, although the result was not significant, infants born to women who were not reported to DOHMH were less likely than infants born to women who were reported to DOHMH to have completed PVST, which is necessary to assess the infant’s HBV status and to determine the success of PEP and HBV vaccination in preventing transmission. 29 The Philadelphia Department of Public Health conducted a similar analysis and identified a larger proportion of infants born to HBV-positive women who were not reported to the Philadelphia Department of Public Health (45.6% vs 3.5% not reported in New York City). 19 These findings suggest that the use of a similar capture–recapture methodology could have an even greater benefit to perinatal HBV prevention programs at other health departments, where a smaller proportion of HBV-infected pregnant women might be reported to the department of health, as compared with those reported to the New York City DOHMH.
We identified several sources of reporting failure among health care facilities. The most common reason for reporting failure was that the health care facility did not report the woman, despite having a record of the woman’s maternal HBV-positive status. Nonreporting also occurred when the maternal HBV status was mistranscribed from the medical record of an infected patient and mistakenly recorded as hepatitis B negative on the Newborn Metabolic Screening Form, as was observed for 32 infants. Four delivery facilities accounted for more than half of the unreported infants, indicating systematic reporting issues at these hospitals to target for follow-up. In contrast with findings from this New York City evaluation, only 11 (3.1%) of the unreported infants in Philadelphia were the result of failure among health care facilities to report the case despite knowing the maternal HBV-positive status. 19 These findings suggest that reasons for reporting failure may vary across jurisdictions.
Limitations
This evaluation had several limitations. First, a number of births captured by routine DOHMH surveillance were not identified by the CIR/ELR match, indicating a need to improve the matching algorithm between the CIR and ELR databases. Second, however, regardless of the strength of the matching algorithm, the CIR/ELR match may still miss births to HBV-positive women; infants born in delivery facilities outside New York City to women who are residing in New York City are not reported to New York City vital records, which populate the CIR database. In addition, despite reporting requirements, women who were residing in New York City at the time of delivery but were tested for HBV in a non–New York City facility may not have had an HBV result reported to the New York City ELR. Likewise, laboratory reports for women who moved to New York City after prenatal HBsAg testing was performed would not have been reported to the New York City ELR prenatally. Third, because the Lincoln–Petersen model assumes that each member of a population has an equal chance of being captured for a given group, the matching limitations may have resulted in a slight overestimation in the number of births to HBV-positive women. However, if we assume that all 61 infants who were identified by DOHMH and not by the CIR/ELR match had no chance of being captured by the CIR/ELR match (for the aforementioned reasons), the estimated percentage of births captured by DOHMH would be reduced by <0.1 percentage point; as such, the effect of this bias on the estimate would be small. Lastly, we did not assess maternal HBV DNA levels and the use of antiviral treatment during pregnancy as part of this evaluation, which could have affected PVST results.
Practical Implications
Efforts by DOHMH to improve case reporting and identification of HBV-positive women are needed given incomplete and delayed reports from health care facilities. This evaluation demonstrates that capture–recapture methodology is an effective tool for enhancing perinatal hepatitis B surveillance for public health programs. Our future work includes improving the CIR/ELR matching algorithm and implementing the match routinely, to improve case identification efforts and to routinely evaluate our surveillance system. Because our capture–recapture evaluation relied on birth records and, as such, women are not identified until the postnatal period, we also intend to consistently follow up with health care facilities that fail to report infants to identify the causes and to prevent future occurrences of reporting failure. In 2014, DOHMH implemented an amendment to Article 13 of the New York City Health Code for pregnancy status to be recorded on all electronic HBV laboratory results for both probable and confirmed pregnancies to increase case ascertainment and potentially identify cases not reported due to transcription errors. 9 However, laboratory reporting of HBV-positive pregnant women to DOHMH is conditional on health care facilities either indicating pregnancy status on laboratory requisitions or ordering prenatal panels containing the hepatitis B test request. Automated electronic reporting of notifiable diseases from electronic medical records to health departments is also promising; efforts to implement such systems are underway in New York City and nationally, but implementation is challenging and resource intensive. The development of a modern electronic reporting infrastructure should continue to be supported by resources at the national and local level.
This evaluation supports continued efforts to educate health care providers on the importance of prenatal HBV testing and reporting of HBV-positive pregnant women to health departments. Effective case management by health departments is crucial to ensure prevention of perinatal hepatitis B transmission.
Footnotes
Authors’ Note
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the New York City Department of Health and Mental Hygiene or the Centers for Disease Control and Prevention.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support with respect to the research, authorship, and/or publication of this article.