Abstract
Objective:
If untreated, hepatitis C virus (HCV) leads to poor health outcomes, including liver disease and death, particularly among people with HIV (PWH). We describe trends over time in incidence rates of HCV diagnoses among PWH in the state of Georgia.
Methods:
We constructed a retrospective cohort of PWH in Georgia by using matched HIV and HCV case surveillance data from people diagnosed with HCV infection from January 1, 2014, through December 31, 2019. We calculated annual incidence rates per 1000 person-years and estimated trends over time in HCV diagnoses among the cohort of PWH by demographic characteristics and HIV care outcomes using Poisson regression analysis, with α = .05 considered significant.
Results:
From 2014 through 2019, among 49 530 PWH in Georgia, 1945 (3.9%) were diagnosed with HCV infection. During this period, overall incidence per 1000 person-years of newly diagnosed HCV infection among PWH decreased from 8.7 to 4.5 (P for trend < .001). However, from 2014 through 2019, the annual incidence rates of PWH who were newly diagnosed with HCV infection increased from 4.6 to 7.1 (P for trend = .003) among people born from 1980 through 1989 and from 3.3 to 12.8 (P for trend < .001) among people born in 1990 or later.
Conclusion:
Strategies are needed to increase prevention, diagnosis, and treatment of HIV/HCV coinfection, particularly among PWH born in 1980 and later. Routine linkage of state surveillance data can inform prioritization of PWH at highest risk of HCV infection.
In 2019, approximately 1.1 million adults in the United States were living with HIV, and approximately 67 000 people with HIV (PWH) resided in the state of Georgia. 1 PWH are at increased risk of comorbidities, including hepatitis C virus (HCV), given the similar transmission routes (ie, unsafe injection practices, condomless anal sex). The estimated prevalence of HIV/HCV coinfection among PWH in the United States is 20%, although this rate is likely an underestimate because of the low HCV testing rates among PWH who inject drugs and the underreporting of HCV infection. 2
People with HIV/HCV coinfection, as compared with people with neither infection nor monoinfection of HIV or HCV, are at increased risk of adverse health outcomes, including severe liver disease 3 and mortality.4,5 Direct-acting antiviral therapies can effectively treat HCV infection, preventing serious complications from liver disease and improving quality of life; however, direct-acting antiviral therapy is somewhat costly to the health care system. 6 Identifying characteristics of PWH who are at increased risk of HCV infection can help prioritize HCV prevention strategies, which is especially important because of shifts in the epidemiologic profile of people with HCV infection. Previously, HCV infections had mostly affected people in the baby boomer generation (ie, born during 1945-1965); however, HCV infections are now more prominent among younger people because of increasing injection drug use (IDU) associated with the opioid epidemic.7 -10 Additionally, PWH who are men who have sex with men (MSM), participate in condomless anal sex, or engage in condomless sex and drug use are at risk of HCV infection.11 -13
Because of geographic differences in HCV epidemics across the United States (varying rates of HCV infections by region and state), understanding geographic-specific changes over time in the characteristics of PWH who are diagnosed with HCV infection is important for informing local surveillance, prevention, and treatment strategies. Overall, only a few studies on HIV/HCV coinfection from the United States14 -17 have used state surveillance data to describe characteristics of coinfected populations; data are especially limited in the South. To our knowledge, no studies have examined trends over time in HCV diagnoses among PWH. We matched HIV and viral hepatitis surveillance data at the Georgia Department of Public Health (GDPH) to (1) determine the percentage of PWH who were subsequently diagnosed with HCV infection from 2014 through 2019 and (2) describe trends in demographic characteristics and HIV care outcomes among PWH diagnosed with HCV infection during this period.
Methods
Study Design
We matched HIV and HCV case surveillance data from GDPH to construct a retrospective cohort of PWH. Using this retrospective cohort, we described trends over time in the percentage and characteristics of PWH in Georgia who were subsequently diagnosed with HCV infection. The institutional review boards at GDPH and Georgia State University reviewed and approved this study.
Study Population
For our cohort, we included confirmed HIV cases 18 among people aged ≥13 years at the time of diagnosis who were diagnosed by December 31, 2013, and living in Georgia. We excluded cases with reported perinatal, hemophilia, or blood transfusion exposure to HIV. We included in our analysis all HCV cases that were diagnosed from January 1, 2014, through December 31, 2019, as confirmed by positive RNA test results.19,20
Data Linkage
We matched HIV case records that met inclusion criteria to HCV case records to identify people who were diagnosed with HIV and HCV infections. GDPH staff used Registry Link Plus 21 to conduct a probabilistic data linkage using an algorithm that included first name, last name, and date of birth. GDPH staff manually reviewed cases to determine final matches.
Measures
We defined a case of HIV/HCV coinfection by the following criteria: diagnosed with HIV on or before December 31, 2013; residing in Georgia (updated yearly for 2014 through 2019); and newly diagnosed with HCV infection between January 1, 2014, and December 31, 2019.
We obtained data on characteristics of people in the cohort from HIV surveillance records because the key variables were more complete than in the HCV surveillance records. We defined retention in HIV care as PWH who had ≥2 CD4 tests or viral load measures at least 3 months apart and had these tests or measures annually during the study period. We defined viral suppression as viral load <200 copies/mL at most recent viral load measure and was measured yearly. We also categorized people according to the following cohorts: birth year (before 1960, 1960-1969, 1970-1979, 1980-1989, 1990 or later), year of HIV diagnosis (before 2000, 2000-2010, after 2010), race and ethnicity (Hispanic/Latino, non-Hispanic Black, non-Hispanic White, other/unknown [American Indian or Alaska Native, Asian, Native Hawaiian or other Pacific Islander, multiple race and ethnicity]), transmission (sexual contact in MSM, IDU, IDU in MSM, heterosexual contact, other/unknown), and sex at birth (female, male).
Statistical Analysis
We summarized demographic characteristics using frequencies and medians. For each year from 2014 through 2019, we determined newly diagnosed HCV infections among PWH matched to the HCV case surveillance registry who were previously undiagnosed with HCV infection, alive, and living in Georgia at the beginning of each year. We calculated median time between HIV diagnosis and HCV diagnosis. We compared characteristics of people diagnosed with HIV and HCV infection with people diagnosed with only HIV infection by using percentage distributions and Pearson χ 2 tests. We calculated annual crude incidence rates of HCV diagnoses via Poisson regression, with person-years in cohort as the offset, and computed crude incidence rates of HCV per 1000 person-years overall and by demographic characteristics of people in the cohort. We also used Poisson regression to assess the trend over time in annual incidence rates of HCV. We performed additional post hoc descriptive analysis to calculate the frequency of risk factors for HIV (ie, transmission category) stratified by year of HIV diagnosis and birth year cohorts separately to characterize the risk of HCV among PWH with IDU and MSM risk factors. We used the Pearson χ 2 test to define significance and trends at α = .05. We conducted all analyses using SAS version 9.4 (SAS Institute, Inc).
Results
Demographic Characteristics of PWH and PWH Diagnosed With HCV Infection
As of December 31, 2013, 49 530 people aged ≥13 years at the time of diagnosis were diagnosed with HIV in Georgia, living in Georgia at some time during the study period (2014-2019), and undiagnosed with HCV infection as of December 31, 2013 (Table 1). Among 49 530 people, 1945 (3.9%) were subsequently diagnosed with HCV infection from 2014 through 2019. The median age at diagnosis of HCV infection was 50 years (IQR, 39-58 y), and the median time from HIV diagnosis to HCV diagnosis was 12 years (IQR, 8-19 y). We observed significant differences in characteristics between PWH who were newly diagnosed with HCV infection and people diagnosed with HIV only: PWH who were diagnosed with HCV infection were, on average, older and more likely to have IDU risk factors (14.6% vs 4.2%, P < .001) than people diagnosed with HIV only.
Demographic characteristics of people diagnosed with HIV and HCV infection versus people diagnosed with HIV infection only, Georgia, United States, 2014-2019 a
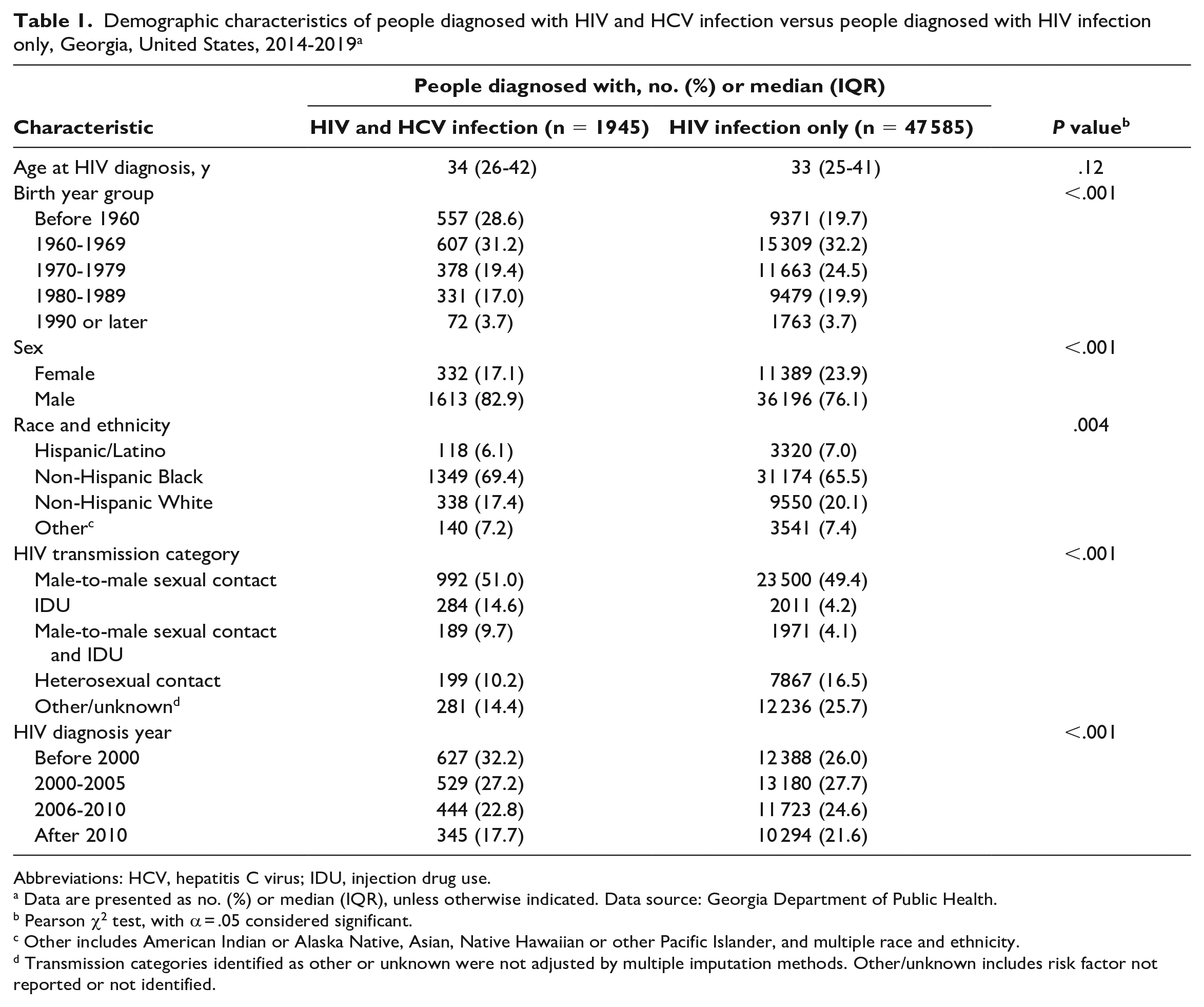
Abbreviations: HCV, hepatitis C virus; IDU, injection drug use.
Data are presented as no. (%) or median (IQR), unless otherwise indicated. Data source: Georgia Department of Public Health.
Pearson χ 2 test, with α = .05 considered significant.
Other includes American Indian or Alaska Native, Asian, Native Hawaiian or other Pacific Islander, and multiple race and ethnicity.
Transmission categories identified as other or unknown were not adjusted by multiple imputation methods. Other/unknown includes risk factor not reported or not identified.
Trends in Selected Characteristics
Overall, the annual incidence rate per 1000 person-years of newly diagnosed HCV infection among PWH decreased from 8.7 in 2014 to 4.5 in 2019 (P for trend < .001) (Table 2). The annual incidence rate of newly diagnosed HCV infection decreased from 12.9 in 2014 to 7.5 in 2019 (P for trend < .001) among PWH who were retained in HIV care during that year and decreased from 12.4 in 2014 to 6.7 in 2019 (P for trend < .001) among PWH who were virally suppressed during that year. Conversely, the annual incidence rate of newly diagnosed HCV infection increased from 4.6 in 2014 to 7.1 in 2019 among PWH who were born in 1980 through 1989 (P for trend = .003) and increased from 3.3 in 2014 to 12.8 in 2019 among those who were born in 1990 or later (P for trend < .001) (Figure). The annual incidence rate of newly diagnosed HCV infection among PWH who were non-Hispanic Black decreased from 9.0 in 2014 to 6.3 in 2019 (P for trend < .001).
Annual incidence rates per 1000 person-years of HCV diagnoses and HIV care outcomes among people diagnosed with HIV, by demographic characteristics, Georgia, United States, 2014-2019 a
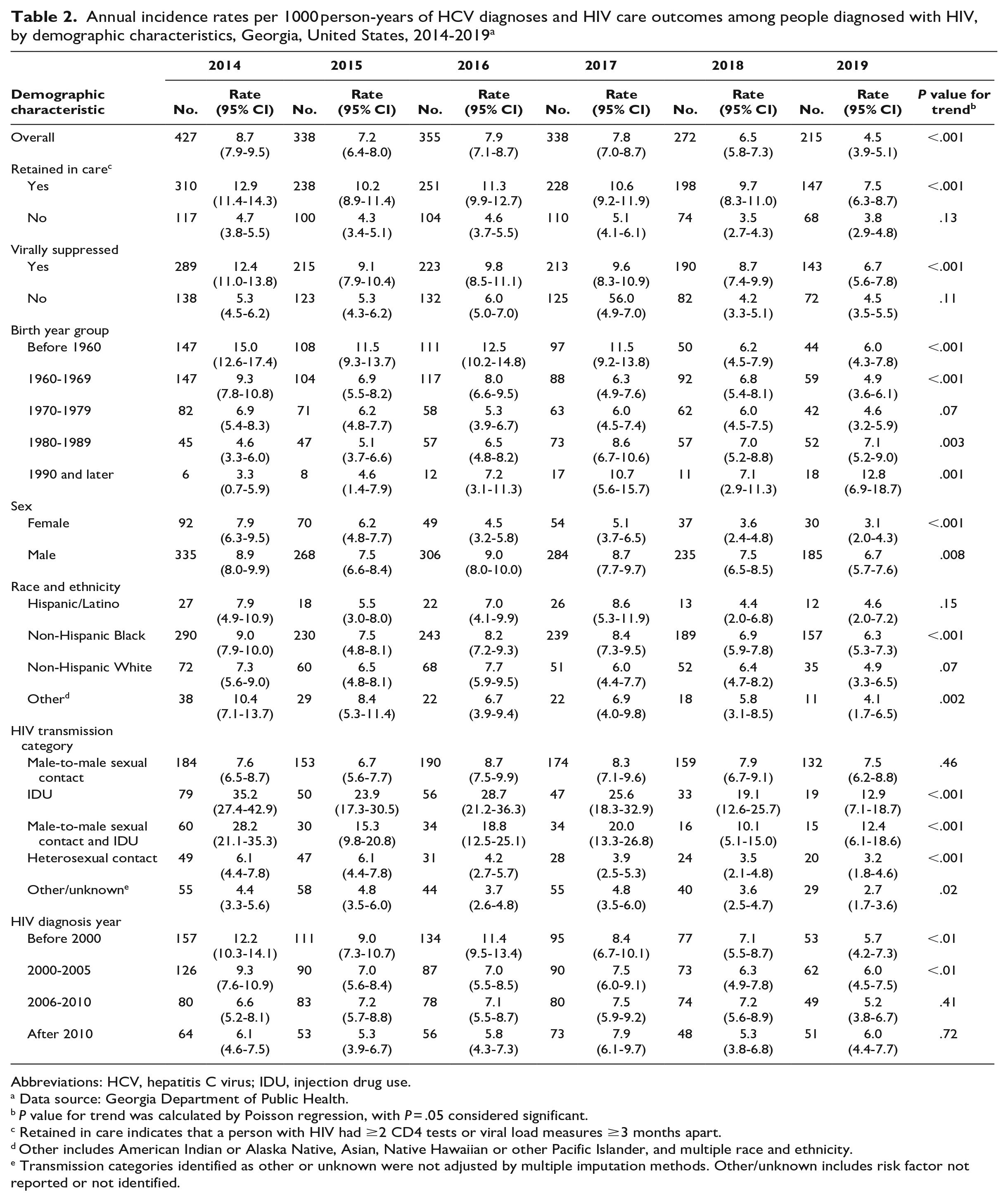
Abbreviations: HCV, hepatitis C virus; IDU, injection drug use.
Data source: Georgia Department of Public Health.
P value for trend was calculated by Poisson regression, with P = .05 considered significant.
Retained in care indicates that a person with HIV had ≥2 CD4 tests or viral load measures ≥3 months apart.
Other includes American Indian or Alaska Native, Asian, Native Hawaiian or other Pacific Islander, and multiple race and ethnicity.
Transmission categories identified as other or unknown were not adjusted by multiple imputation methods. Other/unknown includes risk factor not reported or not identified.
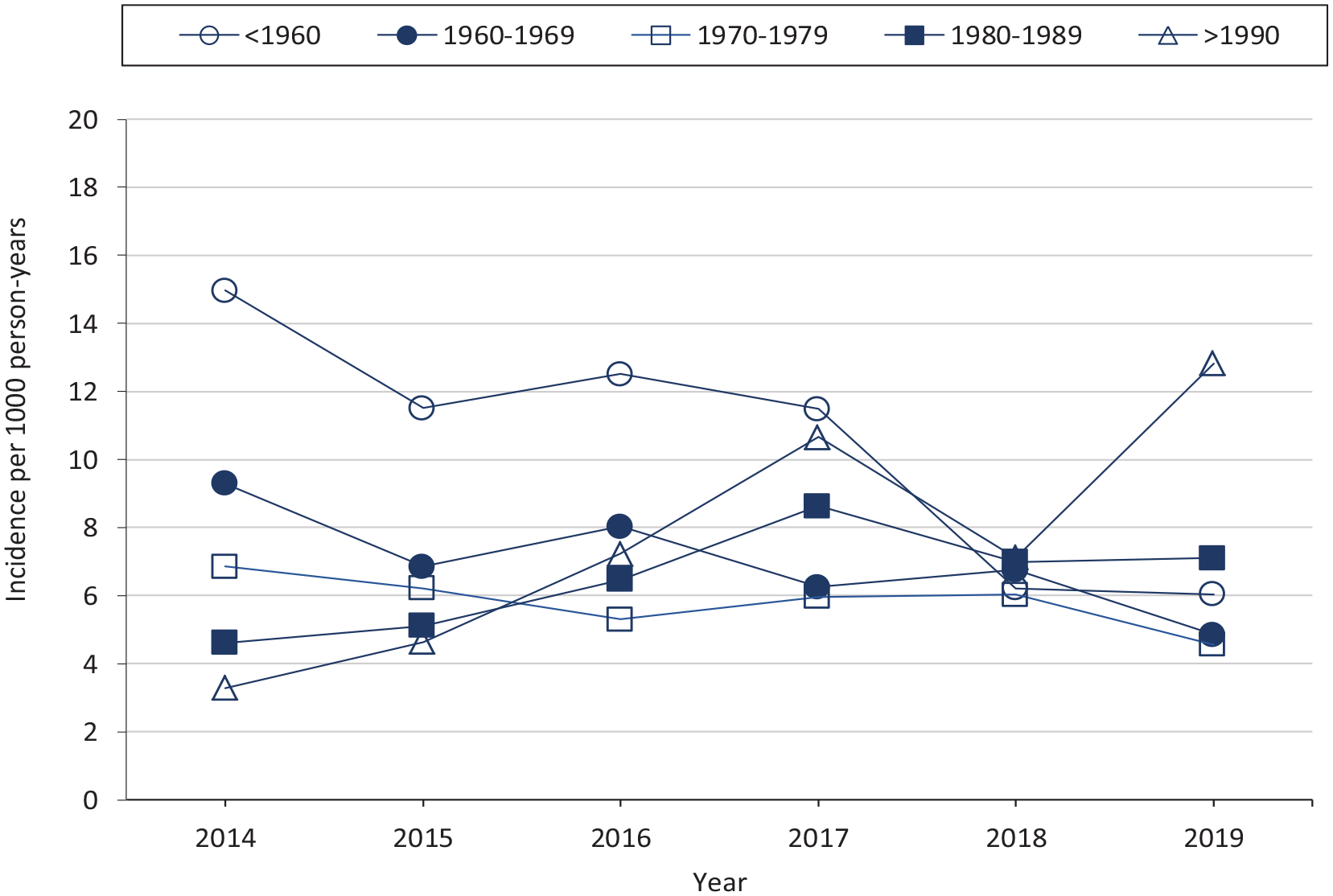
Annual incidence rates per 1000 person-years of hepatitis C virus diagnoses among people diagnosed with HIV, by birth year cohort, Georgia, United States, 2014-2019. Data source: Georgia Department of Public Health.
Post Hoc Descriptive Results
Among our cohort, most PWH with an IDU transmission risk factor at HIV diagnosis were born before 1960 (44.3%) or from 1960 through 1969 (37.8%), whereas most PWH with an MSM risk factor were born during 1960 through 1969 (30.1%), 1970 through 1979 (24.1%), or 1980 through 1989 (26.0%) (P < .001). Most PWH with an IDU risk factor were diagnosed with HIV before 2000 (55.3%) or from 2000 through 2005 (26.6%). PWH with an MSM risk factor were diagnosed with HIV at a similar rate across those years: 27.0% were diagnosed before 2000, 25.7% from 2000 through 2005, 24.4% from 2006 through 2010, and 22.9% after 2010 (P < .001). Among PWH who were diagnosed with HCV infection from 2014 through 2019, the percentage with an MSM risk factor for HIV ranged, by year of HCV diagnosis, from 78.7% to 86.7% among those born from 1980 through 1989 and from 83.3% to 100.0% among those born in 1990 or later.
Discussion
From 2014 through 2019, 1945 PWH were newly diagnosed with HCV infection in Georgia. We observed increasing trends over time in the annual incidence rates of HCV diagnoses among PWH born from 1980 through 1989 and born in 1990 or later. This increase in the incidence rates of HCV diagnoses among young people supports previous findings.9,10,22,23 In 2019, 2.9 and 3.2 acute HCV cases per 100 000 people aged 20-29 and 30-39 years, respectively, were reported to national surveillance, 23 as compared with 2.2 and 1.7 acute HCV cases per 100 000 people from the same age groups in 2014. 24 The opioid crisis and associated increasing IDU have contributed to increases in HCV infections, especially in the Midwest and Appalachia and among young people. 8 Although the most notable increases in newly diagnosed HCV infections in our study were among young people, people born before 1960 and from 1960 through 1969 accounted for the largest percentages of new HCV diagnoses among PWH, although these cases were likely chronic infections rather than acute infections. To our knowledge, our study is the first in the South to present age-specific information on new HCV diagnoses among PWH and the second in the South 15 to evaluate the characteristics of people diagnosed with HIV/HCV coinfection and how these characteristics have changed over time. Combining public health HIV and HCV surveillance data with data on opioid and drug overdose may help to further identify areas where PWH at risk of HCV infection reside for implementation of age-specific harm reduction, prevention, and treatment strategies.
To our knowledge, our study is the first to include HIV care outcomes (eg, retention in care, HIV viral suppression) as potential predictors of HCV diagnoses among PWH. We found significant decreasing trends in the annual incidence rates of newly diagnosed HCV infection among PWH who were retained in HIV care and virally suppressed. The incidence rates of newly diagnosed HCV infection among PWH who were retained in care and among PWH who were virally suppressed decreased significantly over time but remained consistently higher than the rate among PWH who were not retained in care or not virally suppressed. A reason for the decreasing trend may be that people diagnosed with HIV/HCV coinfection are more engaged with the health care system than people with monoinfection (HIV or HCV only) because of comorbidities or because PWH who are engaged in care are more likely than PWH not engaged in care to have their HCV infection diagnosed. Either possibility underscores the importance of retention in care among PWH, which improves HIV-related health outcomes and increases the detection and management of comorbidities such as HCV infection.25 -27
Across all years (2014 through 2019), the rate of newly diagnosed HCV infection among PWH with an IDU risk factor and among PWH with MSM and IDU risk factors was higher than the rate of newly diagnosed HCV infection among PWH with other risk factors. However, the rate of newly diagnosed HCV infection declined significantly during some years among PWH with an IDU risk factor for HIV and increased during some years among PWH with an MSM risk factor for HIV, although the increase was not significant. Although most new HCV infections in the United States are attributable to IDU,7,23,28 condomless anal sex also increases the risk of HCV infection among MSM,11,29 -32 making MSM another often overlooked priority population for routine HCV testing and other prevention interventions. Most young people with incident HCV diagnoses during 2014 through 2019 had MSM risk factors for HIV. These young men may have acquired HCV through either MSM sexual behavior or IDU initiated after diagnosis of HIV, which is concerning from the perspective of a risk network crossover (ie interchanging networks of sex and IDU). MSM with HIV infection may also engage in risky sexual behaviors and concurrent drug use, further increasing the risk of HCV infection.12,13 The Centers for Disease and Control and Prevention recommends HCV testing at least once for all PWH and routinely for people who inject drugs. 33
The decrease in HCV diagnoses that we observed among people with an IDU risk factor for HIV was likely due to the age distribution of this group, in which 82% were born before 1970. In the absence of new risk factors for HCV, PWH who were born before 1970 would have had diminishing risk for new HCV diagnoses during our study period given the likelihood of a previous diagnosis among PWH receiving clinical care. 34 Many people in this age group with a history of IDU would have been diagnosed with HCV infection before 2014 and, thus, excluded from our cohort. Importantly, having an MSM risk factor at HIV diagnosis does not preclude having an IDU risk factor for HCV infection. Although young MSM may have initiated IDU after diagnosis of HIV, HIV risk factors among young MSM may have been misclassified at the time of diagnosis.
The use of surveillance data can help guide prioritization efforts for HCV prevention, screening, and treatment interventions. According to our findings, PWH born in 1980 and later should be prioritized for HCV screening and prevention in Georgia and in states with similar HCV epidemics. Although we observed no increases in HCV diagnoses among those born from 1970 through 1979, 1960 through 1969, and before 1960, people born during these years still accounted for 79% of HCV diagnoses, demonstrating an ongoing need for screening, diagnosis, and treatment among these age groups. Although annual incidence rates of HCV diagnoses decreased significantly among people who were non-Hispanic Black, this population still accounted for most of the new HCV diagnoses and continued to have disproportionate rates of HCV 35 and HIV 1 infections nationally; in addition, both infections may be less likely to be treated among non-Hispanic Black people than among non-Hispanic White people.36,37 Addressing the social and structural barriers (eg, cost, access to HCV testing and direct-acting antiviral therapy, transportation) that challenge uptake of HIV and HCV prevention and treatment strategies can help eliminate the racial and ethnic disparities that currently exist.
Limitations
Our study had several limitations. First, our findings relied on the last known reported address in Georgia, which may have underestimated retention and HIV viral suppression measures for people whom the health department was unaware had moved. Second, our data represented only diagnosed HIV and HCV cases reported to GDPH. Many cases of HIV and HCV are undiagnosed and were not represented in our study. Therefore, our findings are an underestimate of the number of PWH coinfected with HCV in Georgia during 2014 through 2019. Third, because we were unable to differentiate between acute and chronic infections in these data, many HCV infections may have occurred earlier than the year of HCV diagnosis. Finally, after direct-acting antiviral therapies became available in the 2000s, HCV testing increased. 38 Some of the increases in HCV diagnoses observed during our study period may reflect increased HCV testing rather than incident infections.
Conclusions
Strategies are needed to prevent, diagnose, and treat HCV infection among PWH. Our study showed increasing annual incidence rates of HCV diagnoses among PWH born in 1980 and later. These findings may reflect increased HCV testing or incident HCV infections related to the US opioid epidemic and associated risk factors, such as IDU and sexual behavior associated with HCV transmission. The need to prevent new HCV infections and diagnose and treat existing infections is urgent, especially among younger people born in 1980 and later. In addition, increasing routine HCV testing among PWH, particularly those with risk factors for HCV (eg, MSM with HIV, people who inject drugs), can lead to early diagnosis and treatment, both necessary to prevent ongoing transmission. Treatment of HCV among PWH can also reduce poor health outcomes, including severe liver disease and death. Strategies to accomplish these efforts may vary by geography and will rely on routinely linking state surveillance data to inform prioritization of PWH at highest risk of HCV infection. Our study provides a methodological framework for states with HCV epidemics similar to Georgia and a model for using surveillance data to target prevention and treatment strategies.
Footnotes
Acknowledgements
The authors thank Rodriques Lambert, PhD, MPH, with the Georgia Department of Public Health for conducting the data linkage.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
