Abstract
Objective:
Although vaccination reduces the risk of severe COVID-19, fatal COVID-19 cases after vaccination can occur. We examined the characteristics of decedents with COVID-19–related mortality to help inform discussions about vaccination, boosters, and mitigation strategies.
Methods:
We examined COVID-19–related deaths in Kentucky resulting from infections occurring from July 1 through August 13, 2021. We used records from case investigations, medical records, the Kentucky Health Information Exchange, and the Kentucky Immunization Registry to determine demographic information, vaccination status, and underlying health conditions, including calculation of the Charlson Comorbidity Index (CCI). We calculated mortality incidence rates by vaccination status by using data for unvaccinated and fully vaccinated populations in Kentucky as of July 1, 2021.
Results:
In total, 777 COVID-19–related deaths occurred in Kentucky during the study period; 592 (76.2%) occurred among unvaccinated people. Compared with unvaccinated decedents, fully vaccinated decedents were older (median age, 77 vs 65 years; P < .001), had higher comorbidity levels (median CCI, 3 vs 1; P < .001), and were more likely to have immunocompromised health status (26.4% vs 16.0%; P = .003). Diabetes, hypertension, heart disease, and chronic lung disease were more common among vaccinated decedents than among unvaccinated decedents. Unvaccinated adults had a significantly higher risk of death than fully vaccinated adults (incidence rate ratio for age 20-49 years: 20.5 [95% CI, 6.5-64.8]; 50-64 years: 14.6 [95% CI, 9.4-22.7]; ≥65 years: 10.2 [95% CI, 8.3-12.4]).
Conclusions:
Immunocompromised health status, older age, and higher comorbidity were prevalent among fully vaccinated decedents, suggesting adults with these characteristics may benefit from additional protection strategies. Further understanding of the protection of additional and booster doses is needed.
COVID-19 vaccines are highly effective at preventing severe disease caused by SARS-CoV-2.1,2 However, breakthrough infections resulting in death do occur, suggesting that some vaccinated people may not mount a protective immune response or may not maintain that response over time.3-6 To better understand ongoing risk of fatal infections, detailed data, including vaccination history, are needed. We described characteristics of decedents with COVID-19–related mortality, by vaccination status, among Kentucky residents with SARS-CoV-2 infections occurring from July 1 through August 13, 2021, prior to the Centers for Disease Control and Prevention (CDC) recommendations for additional and booster doses of vaccine 7 and during a time when Delta was the dominant variant in circulation. 8
Methods
Cases of COVID-19 among Kentucky residents are investigated by local health departments (LHDs); confirmed and probable cases9,10 are subsequently reported to the Kentucky Department for Public Health (KDPH). Deaths with COVID-19 as a potential contributing factor are reported to KDPH by LHDs as they are detected. Death certificates from the Office of Vital Statistics are also queried for any indication of COVID-19 illness, either in the cause of death fields (“SARS,” “cov,” “coronav”) or with the International Classification of Diseases, 10th Revision (ICD-10) code of U07.1. 11 All potential COVID-19–related deaths are reviewed by a KDPH team member to determine whether COVID-19 contributed to the death. COVID-19–related deaths are initially counted when the decedent’s illness meets the COVID-19 case definition at the time of infection,9,10 death is within 6 weeks of the decedent’s positive SARS-CoV-2 test result, and investigative evidence collected by LHDs clearly corroborates COVID-19 as a contributing cause of death. Determination occasionally requires additional health records to be collected. Difficult-to-determine cases (eg, when COVID-19 is listed on the death certificate but the contribution to the death is unclear) are referred to a mortality review group. The mortality review group, comprising physicians, epidemiologists, and nurses, makes final determinations based on clinical judgment after reviewing all available records. If the contribution of COVID-19 is ultimately unclear, the final decision regarding whether to count the death as COVID-19–related is based upon information contained in the death certificate. The vaccination status of decedents is known to the mortality review group because it is typically noted in the medical documentation, but vaccination status is not considered in determining whether a death is COVID-19–related.
The analytic population for this study included deaths deemed to be COVID-19–related among Kentucky residents aged ≥20 years and that were reported as of December 15, 2021. The study population was limited to cases with COVID-19 onset from July 1 through August 13, 2021. We used the specimen collection date for onset date because of completeness of this variable; we used the case investigation date when the specimen collection date was missing. We selected the time period for 2 reasons: (1) the circulating virus in Kentucky was almost exclusively the Delta variant, 8 and (2) the period was prior to recommendations for additional or booster doses of vaccine. 7
We collected data on demographic characteristics and health information for all cases using a data abstraction form. We used 3 sources to gather information on cases and linked them using first and last name and date of birth: (1) case investigation records, (2) hospitalization records, and (3) Kentucky Health Information Exchange (KHIE) data. Case investigation records included self-reported information from patient or proxy interviews, case report forms submitted by medical providers, and medical records, when available. LHDs entered case investigation information into Kentucky’s National Electronic Disease Surveillance System, which was queried for this analysis. We reviewed records for inpatient COVID-19–related deaths, which were requested from hospitals by LHD or KDPH investigators, to obtain information on diagnosed underlying health conditions and clinical course of illness. The third data source, KHIE, allows sharing of health and medical information between health care providers and facilities in Kentucky. KHIE contains medical diagnoses and ICD-10 codes for health care encounters, as well as social history information from routine intake questions.
We used data from the Kentucky Immunization Registry (KYIR) to determine vaccination status. We defined “fully vaccinated” as completion of a US Food and Drug Administration–authorized COVID-19 vaccine series (1 dose of Janssen or 2 doses of mRNA vaccine, Pfizer-BioNTech, or Moderna) at least 14 days prior to infection onset. We defined “partially vaccinated” as receipt of ≥1 vaccine dose prior to infection onset but primary vaccination series was not complete as of 14 days prior to infection onset. If patient/proxy interview or medical records documented vaccination status but immunization records were not in KYIR, we used the reported vaccination status.
During the study period, KDPH had requested that Kentucky hospitals submit to the public health laboratory all available respiratory specimens that tested positive for SARS-CoV-2 for vaccinated, hospitalized patients. We obtained sequencing results from the National Electronic Disease Surveillance System for fatal SARS-CoV-2 cases among vaccinated residents, when available. CDC reviewed this activity consistent with applicable federal law and CDC policy (eg, 45 CFR part 46.102(l)(2), 21 CFR part 56; 42 USC §241(d); 5 USC §552a; 44 USC §3501 et seq). The Kentucky Cabinet for Health and Family Services Institutional Review Board additionally reviewed and approved this project.
Demographic and Health Characteristics
We determined age of resident by date of specimen collection relative to date of birth. We used case interview information obtained during the initial case investigation to categorize long-term care facility residence (yes/no) and health care worker status (yes/no or unknown).
Medical record abstraction focused on identifying the presence of underlying conditions that are associated with an increased risk of severe COVID-19.12,13 These conditions included chronic kidney disease, chronic lung disease, hypertension, heart disease, dementia, diabetes, Down syndrome, liver disease, immunocompromised health status (cancer, HIV/AIDS or primary immune deficiency diseases, rheumatoid arthritis/lupus/psoriasis, stem cell or organ transplant recipient, receipt of immunosuppressive therapy), stroke or other cerebrovascular disease, pregnancy, sickle cell anemia, former or current smoking status, former or current substance abuse, and obesity. For our analyses, heart disease included congestive heart failure, history of myocardial infarction, coronary artery disease, arrhythmias, history of pacemaker placement, and conditions deemed to require lifelong cardiology follow-up. Because of limitations in identifying the use and timing of immunosuppressive medication or therapies through available records, people with autoimmune conditions associated with use of immunosuppressive therapies (rheumatoid arthritis/lupus/psoriasis) were considered as having immunocompromised health status.
We categorized smoking status (current, former, never, unknown) and substance abuse or illegal drug use (current or former/never or unknown) using case investigations and social history questions on hospitalization records or in KHIE. We defined current smoking status as reports of current smoking or tobacco use during case interviews, at time of hospital admission, or in medical records (including ICD-10 code 17.x) within the year prior to infection date. We classified former smoking status as report of a history of, but not current, smoking or tobacco use during case interviews, at time of hospital admission, or in medical records (including ICD-10 code Z87.891) within the year prior to infection date. We categorized substance abuse or report of illegal drug use (including marijuana) as a single group because of differences in social history questions among Kentucky hospitals. Current or former refers to reports of current alcohol or substance abuse or illegal drug use, or history of such abuse, at the time of case investigation, hospitalization, or medical encounters captured in KHIE within the year prior to infection date. In the event of conflicting information between data sources, we used a hierarchy of current over former over never to determine categorization for smoking status and substance abuse/illegal drug use.
We determined obesity from body mass index (BMI), defined as weight in kilograms divided by height in meters squared, during hospitalization or nearest date prior to infection and classified as the following: not obese (BMI < 30 kg/m2), Class I obesity (30 to <35 kg/m2), Class II obesity (35 to <40 kg/m2), Class III obesity (≥40 kg/m2), or obesity, unspecified BMI. We noted obesity, unspecified BMI when obesity was reported during case interviews or included in hospital records but BMI was not available. Self-report of nonobese was used unless BMI measurements were available and conflicted with self-report.
We calculated the Charlson Comorbidity Index (CCI) (eTable in Supplemental Material)14,15 according to the presence of 17 health conditions. We reviewed case investigation and hospital records, as well as ICD-10 codes and diagnoses in KHIE, 14 to ascertain underlying conditions prior to SARS-CoV-2 infection date. We used indications of the presence of underlying health conditions in any of the 3 data sources to classify the condition as yes versus no indication of condition.
We calculated age-stratified incidence of death among fully vaccinated and unvaccinated populations. Incidence of death for fully vaccinated people included number of deaths of fully vaccinated people divided by the fully vaccinated Kentucky population as of July 1, 2021. We included vaccine doses from US Food and Drug Administration–authorized vaccines (Janssen, Moderna, Pfizer-BioNTech) administered prior to July 1, 2021, and available in KYIR. We used first name, last name, and date of birth to identify unique people vaccinated. The fully vaccinated denominator included those who were fully vaccinated by July 1, 2021. The partially vaccinated population included those with only a single dose of mRNA vaccine by July 1, 2021. The unvaccinated population was the remainder of the 2019 US Census population estimate for Kentucky after removing the fully and partially vaccinated population as of July 1, 2021.
Statistical Analyses
We used Pearson χ2 and Wilcoxon tests to compare categorical and non-normally distributed continuous data among fully vaccinated, partially vaccinated, and unvaccinated decedents, respectively, with P < .05 considered significant. We used the Fisher exact test for categorical data when expected cell counts were <5. We used the Cochran–Armitage test for trend for categorical age (20-29, 30-39, 40-49, 50-59, 60-69, 70-79, ≥80 years). We limited between-group comparisons to unvaccinated and fully vaccinated decedents because of small numbers of deaths among partially vaccinated decedents. We estimated incidence rate ratios (IRRs) using Poisson regression. We used SAS version 9.4 (SAS Institute Inc) for all analyses.
Results
In total, 777 COVID-19–related deaths were included: 592 (76.2%) were unvaccinated, 26 (3.3%) were partially vaccinated, and 159 (20.5%) were fully vaccinated. No decedents received >1 dose of Janssen or 2 doses of mRNA vaccine. Three decedents had medical report documentation stating that they had previously received vaccine (2 fully vaccinated and 1 partially vaccinated), but vaccination records were not in KYIR. These decedents were considered fully or partially vaccinated, but time from vaccination was unable to be determined.
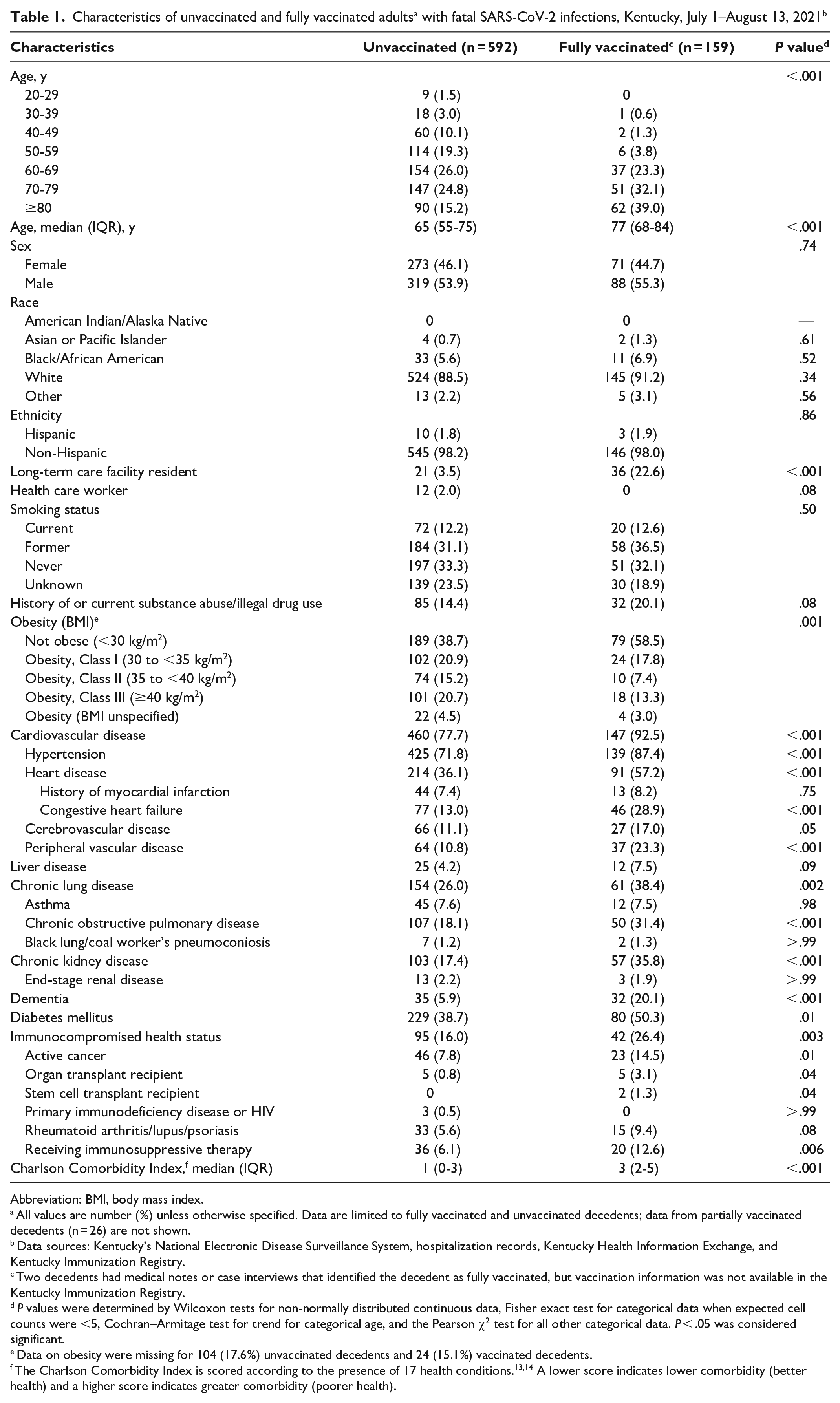
Vaccinated decedents were significantly older than unvaccinated decedents (median age, 77 vs 65 years; P < .001; Table 1) and had significantly more comorbidities, as measured by the CCI (median CCI, 3 vs 1; P < .001). Hypertension, heart disease, diabetes, and chronic lung disease were prevalent underlying conditions among both vaccinated and unvaccinated decedents, with higher prevalence noted among vaccinated decedents. We found immunocompromised health status among 26.4% of vaccinated decedents compared with 16.0% of unvaccinated decedents. A higher percentage of vaccinated decedents (22.6%) than unvaccinated decedents (3.6%) were long-term care facility residents. Twelve deaths occurred among health care workers, all unvaccinated; 10 (83.3%) were aged <65 years.
Abbreviation: BMI, body mass index.
All values are number (%) unless otherwise specified. Data are limited to fully vaccinated and unvaccinated decedents; data from partially vaccinated decedents (n = 26) are not shown.
Data sources: Kentucky’s National Electronic Disease Surveillance System, hospitalization records, Kentucky Health Information Exchange, and Kentucky Immunization Registry.
Two decedents had medical notes or case interviews that identified the decedent as fully vaccinated, but vaccination information was not available in the Kentucky Immunization Registry.
P values were determined by Wilcoxon tests for non-normally distributed continuous data, Fisher exact test for categorical data when expected cell counts were <5, Cochran–Armitage test for trend for categorical age, and the Pearson χ2 test for all other categorical data. P < .05 was considered significant.
Data on obesity were missing for 104 (17.6%) unvaccinated decedents and 24 (15.1%) vaccinated decedents.
Among 91 decedents aged 20 to 49 years, 3 were fully vaccinated and all 3 had immunocompromised health status (Table 2). Among unvaccinated decedents in this age group, 41.4% had a CCI score of 0. Obesity (86.6%), hypertension (56.3%), and current or former smoking status (29.9%) were prevalent health conditions among unvaccinated decedents aged 20 to 49 years.
Health characteristics of unvaccinated and fully vaccinated adults with fatal SARS-CoV-2 infections, by age, Kentucky, July 1–August 13, 2021 a
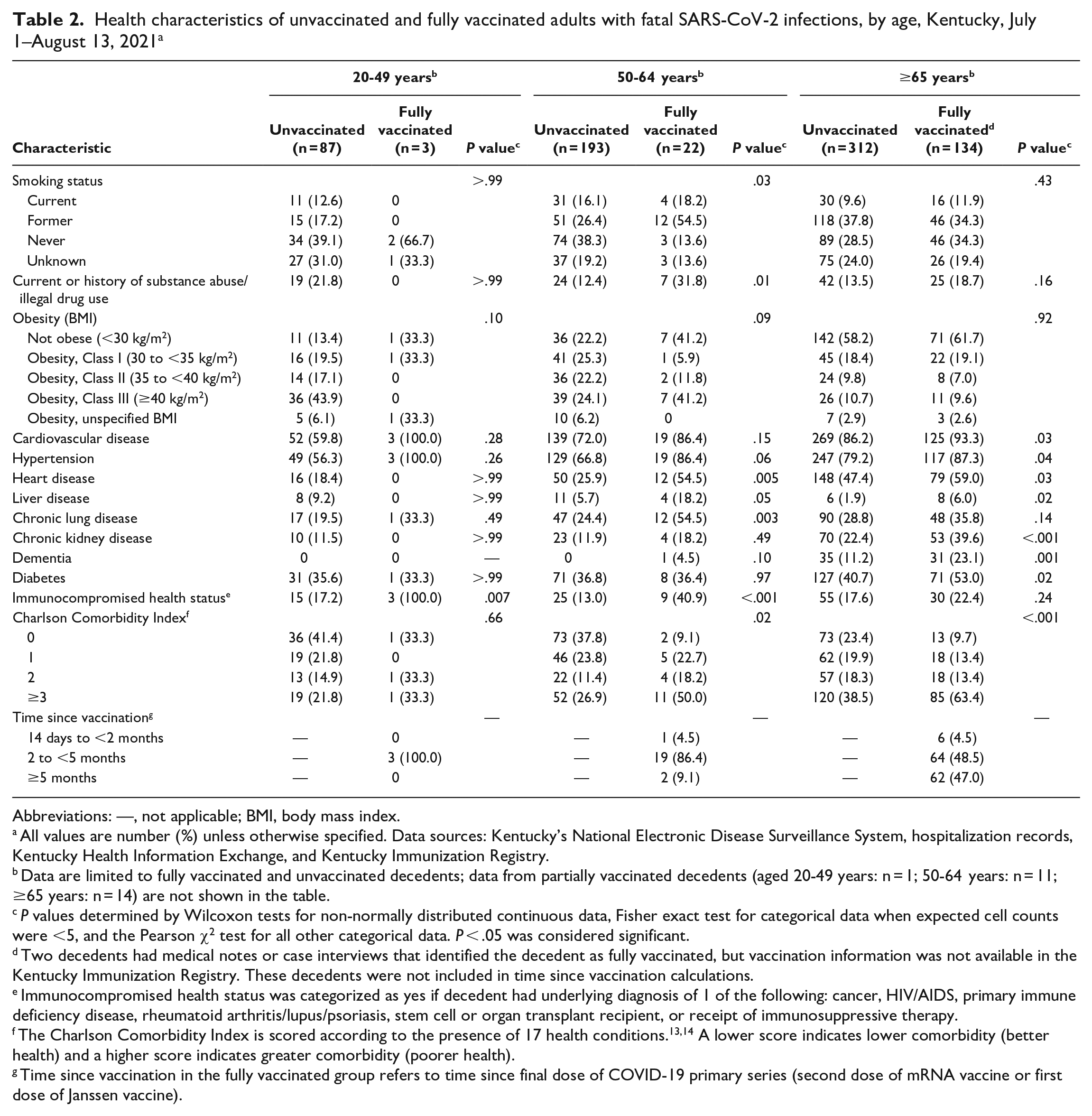
Abbreviations: —, not applicable; BMI, body mass index.
All values are number (%) unless otherwise specified. Data sources: Kentucky’s National Electronic Disease Surveillance System, hospitalization records, Kentucky Health Information Exchange, and Kentucky Immunization Registry.
Data are limited to fully vaccinated and unvaccinated decedents; data from partially vaccinated decedents (aged 20-49 years: n = 1; 50-64 years: n = 11; ≥65 years: n = 14) are not shown in the table.
P values determined by Wilcoxon tests for non-normally distributed continuous data, Fisher exact test for categorical data when expected cell counts were <5, and the Pearson χ2 test for all other categorical data. P < .05 was considered significant.
Two decedents had medical notes or case interviews that identified the decedent as fully vaccinated, but vaccination information was not available in the Kentucky Immunization Registry. These decedents were not included in time since vaccination calculations.
Immunocompromised health status was categorized as yes if decedent had underlying diagnosis of 1 of the following: cancer, HIV/AIDS, primary immune deficiency disease, rheumatoid arthritis/lupus/psoriasis, stem cell or organ transplant recipient, or receipt of immunosuppressive therapy.
The Charlson Comorbidity Index is scored according to the presence of 17 health conditions.13,14 A lower score indicates lower comorbidity (better health) and a higher score indicates greater comorbidity (poorer health).
Time since vaccination in the fully vaccinated group refers to time since final dose of COVID-19 primary series (second dose of mRNA vaccine or first dose of Janssen vaccine).
Five deaths occurred among people who had a previous COVID-19 case report at least 90 days prior to the positive test result in July–August 2021; these were considered reinfection cases. Of these, 2 were unvaccinated, 2 were partially vaccinated, and 1 was fully vaccinated prior to reinfection; ages ranged from 38 to 85 years. Specimens for 1 fully vaccinated and 1 partially vaccinated decedent had been available at the testing laboratory and were sent to the state public health laboratory; whole-genome sequencing was possible for 1 specimen with cycle threshold values low enough for sequencing (case with partial vaccination) and identified the Delta (AY.4) variant. There were no indications of immunocompromising health conditions among the 5 cases with prior infections.
Whole-genome sequencing results were available for specimens from 19 vaccinated decedents (2 partially vaccinated and 17 fully vaccinated); 18 (94.7%) corresponded with the Delta variant (13 B.1.617.2; 2 AY.3; 1 AY.3.1; 2 AY.4). One result corresponded to a non-Delta lineage (B.1.628).
Overall, 51.0% of Kentucky residents aged ≥20 years were fully vaccinated by July 1, 2021; vaccination coverage was lowest among those aged 20-49 years and increased with age (Table 3). The incidence of death per 100 000 population among vaccinated adults was lower among younger adult age groups and comparatively higher among older adults (20-49 years: 0.5; 50-64 years: 4.4; ≥65 years: 24.2). The incidence of death per 100 000 population among unvaccinated adults compared with vaccinated adults was higher in all age groups (IRR for age 20-49 years: 20.5 [95% CI, 6.5-64.8]; 50-64 years: 14.6 [95% CI, 9.4-22.7]; ≥65 years: 10.2 [95% CI, 8.3-12.4]).
Incidence per 100 000 population of fatal SARS-CoV-2 infections among vaccinated and unvaccinated populations by age, Kentucky, July 1–August 13, 2021 a
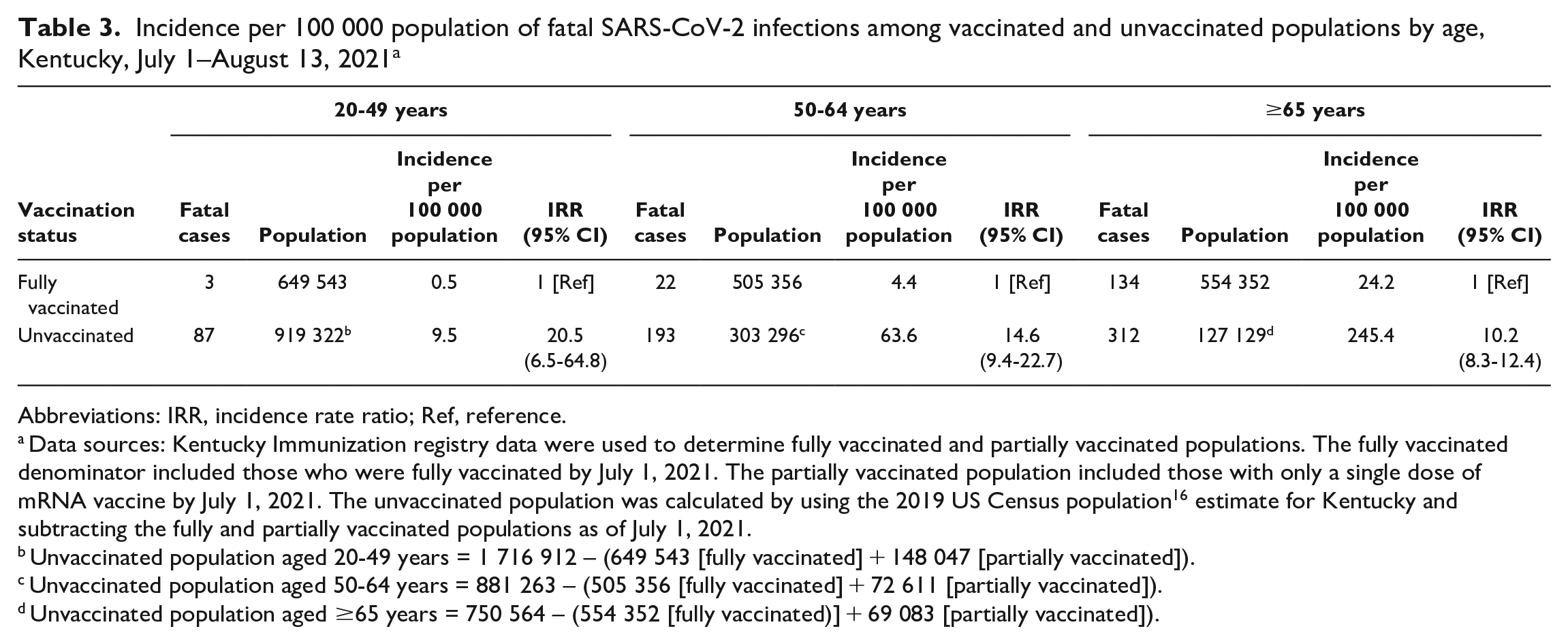
Abbreviations: IRR, incidence rate ratio; Ref, reference.
Data sources: Kentucky Immunization registry data were used to determine fully vaccinated and partially vaccinated populations. The fully vaccinated denominator included those who were fully vaccinated by July 1, 2021. The partially vaccinated population included those with only a single dose of mRNA vaccine by July 1, 2021. The unvaccinated population was calculated by using the 2019 US Census population 16 estimate for Kentucky and subtracting the fully and partially vaccinated populations as of July 1, 2021.
Unvaccinated population aged 20-49 years = 1 716 912 – (649 543 [fully vaccinated] + 148 047 [partially vaccinated]).
Unvaccinated population aged 50-64 years = 881 263 – (505 356 [fully vaccinated] + 72 611 [partially vaccinated]).
Unvaccinated population aged ≥65 years = 750 564 – (554 352 [fully vaccinated)] + 69 083 [partially vaccinated]).
Discussion
During the first weeks of the Delta surge in Kentucky, at least 777 residents died from SARS-CoV-2 infections, most of whom (76%) were unvaccinated. Vaccinated decedents were older and had a higher comorbidity score, as measured by CCI, than unvaccinated decedents. Hypertension, heart disease, diabetes, and chronic lung disease were prominent underlying health conditions among vaccinated people with fatal SARS-CoV-2 infections. While deaths among vaccinated people aged <50 years were rare, immunocompromised health status was noted in all fatal infections in this age group.
While most deaths were among unvaccinated people, >20% of deaths occurred among fully vaccinated people. Understanding the characteristics of fully vaccinated people with fatal SARS-CoV-2 infections can help to guide discussions on additional and booster doses of vaccine. Evidence suggests that immunosuppressed people may have a limited immune response after vaccination, thus potentially limiting the protection provided by vaccines.5,6,17 Immunocompromised health status was prevalent among vaccinated decedents and particularly notable among nonelderly adults in our study, where all 3 deaths among adults aged 20 to 49 years and 40.9% of decedents aged 50 to 64 years had immunocompromised health. Evidence also suggests that vaccine effectiveness wanes over time, and booster doses were recommended by CDC after the study period.3,4 More than 40% of fully vaccinated decedents from the study period had completed their primary series at least 5 months prior to infection. Future studies should address the improved protection afforded by additional and booster doses.
While a focus on the need for additional or booster doses in the vaccinated population has increased, most transmission, as well as fatal disease, during the Delta variant circulation occurred among unvaccinated populations. 1 In our study, the incidence of death among unvaccinated people was higher in all age groups but most notably among adults aged 20 to 49 years, in which unvaccinated people had more than a 20-fold increased risk of death relative to vaccinated people. These findings highlight the protection against fatal COVID-19 infection that is afforded by vaccines and the need to continue vaccination efforts among the unvaccinated population.
Complacency or a lack of perception of an urgent need to be vaccinated may contribute to lower vaccination uptake, particularly among younger adults. 18 In our study, more than 40% of unvaccinated decedents aged 20 to 49 years had a CCI score of 0. Designed to predict 1-year mortality risk for hospitalized patients, the CCI is one of the most widely used predictors for all-cause mortality. 19 It is possible that some of these adults with a “healthy” CCI score had a self-perceived low risk for severe COVID-19, thus contributing to a delay or refusal to receive COVID-19 vaccination. Yet, while many unvaccinated decedents aged <50 years had low mortality risk according to CCI scores, the prevalence of obesity, including Class III obesity, hypertension, and smoking history was high. Obesity, hypertension, and smoking are associated with COVID-19 mortality20-22 but are not accounted for in the CCI. Obesity, hypertension, and smoking history, even among those with a CCI of 0, are health conditions that would place individuals at risk for severe COVID-19. It is possible that health care–seeking behaviors or access to health care among unvaccinated decedents was lower than among vaccinated decedents, resulting in artificially lower CCI scores. Nonetheless, the finding that a substantial proportion of decedents aged <50 years had no or few underlying documented medical diagnoses prior to SARS-CoV-2 infection may be important in vaccine outreach strategies. This finding stresses the importance of discussing vaccination at every health care interaction, particularly for populations that may have limited access, seek health services infrequently, or do not have a primary care provider. Taking vaccines to the client in community-facing events may be another strategy to reach unvaccinated people with limited health care utilization. Discussions with unvaccinated people about the risk of severe disease for those with obesity, hypertension, or smoking history could be beneficial, as these characteristics may not be self-perceived as risk factors for severe disease.
Limitations
This study had several limitations. First, it is likely that substantial differences in the baseline health status of vaccinated and unvaccinated populations exist. While we have data sources to investigate health conditions among decedents, we do not have information on population denominators to adjust for these factors for risk comparisons of COVID-19–related death. Second, while the use of 3 data sources helps to ensure more complete data, some underlying health conditions may not have been captured for some decedents. For example, Kentucky residents who typically receive their medical care out of state or at federal sites would have limited data available in KHIE. Hospitalization records or case interviews may help to ensure ascertainment of underlying medical conditions, but variability in data quality exists. Third, we used conditions that are frequently treated with immunosuppressive therapies as a proxy to estimate immunosuppressed health status, which may overestimate prevalence. Fourth, immunizations administered out of state or at federal sites are typically not entered in KYIR. While vaccination status misclassification is possible, case investigation interviews and hospital records typically identify vaccination status of patients. Hospital records were concordant with KYIR in all but 3 cases in which vaccination was reported but records were not in KYIR. The denominator of the fully vaccinated population may be slightly underestimated because of the uncaptured vaccinations received out of state or at federal sites. Therefore, the protective effect of vaccination is likely underestimated. Finally, additional deaths from July through mid-August COVID-19 cases may not have been included in these analyses because of delays in death reporting. However, we do not expect that there is a bias toward vaccination status in the cases currently reported.
Conclusion
Most fatal SARS-CoV-2 infections occurring in Kentucky during the Delta surge and prior to CDC’s recommendations for additional or booster doses of vaccines were among unvaccinated people. Fully vaccinated decedents were more likely to be adults aged >65 years and have more comorbidities or immunocompromised health status than unvaccinated decedents. Future studies should detail the characteristics and underlying health conditions among decedents with fatal SARS-CoV-2 infections following the availability of additional and booster doses to better understand the additional protection provided.
Supplemental Material
sj-pdf-1-phr-10.1177_00333549231155867 – Supplemental material for Characteristics of Decedents With COVID-19–Related Mortality in Kentucky, July 1–August 13, 2021
Supplemental material, sj-pdf-1-phr-10.1177_00333549231155867 for Characteristics of Decedents With COVID-19–Related Mortality in Kentucky, July 1–August 13, 2021 by Alyson M. Cavanaugh, Likhita Raparti, Mishaal Bhurgri, Amy Herrington, Claire Holladay, Vaneet Arora, Kevin B. Spicer, Douglas A. Thoroughman and Kathleen Winter in Public Health Reports
Footnotes
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention or the Kentucky Department for Public Health.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online. The authors have provided these supplemental materials to give readers additional information about their work. These materials have not been edited or formatted by Public Health Reports’s scientific editors and, thus, may not conform to the guidelines of the AMA Manual of Style, 11th Edition.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.