Abstract
Objectives:
In summer 2021, the number of COVID-19–associated hospitalizations in the United States increased with the surge of the SARS-CoV-2 Delta variant. We assessed how COVID-19 vaccine initiation and dose completion changed during the Delta variant surge, based on jurisdictional vaccination coverage before the surge.
Methods:
We analyzed COVID-19 vaccination data reported to the Centers for Disease Control and Prevention. We classified jurisdictions (50 states and the District of Columbia) into quartiles ranging from high to low first-dose vaccination coverage among people aged ≥12 years as of June 30, 2021. We calculated first-dose vaccination coverage as of June 30 and October 31, 2021, and stratified coverage by quartile, age (12-17, 18-64, ≥65 years), and sex. We assessed dose completion among those who initiated a 2-dose vaccine series.
Results:
Of 51 jurisdictions, 15 reached at least 70% vaccination coverage before the Delta variant surge (ie, as of June 30, 2021), while 35 reached that goal as of October 31, 2021. Jurisdictions in the lowest quartile of vaccination coverage (44.9%-54.9%) had the greatest absolute (9.7%-17.9%) and relative (18.1%-39.8%) percentage increase in vaccination coverage during July 1–October 31, 2021. Of those who received the first dose during this period across all jurisdictions, nearly 1 in 5 missed the second dose.
Conclusions:
Although COVID-19 vaccination initiation increased during July 1–October 31, 2021, in jurisdictions in the lowest quartile of vaccination coverage, coverage remained below that of jurisdictions in the highest quartile of vaccination coverage before the Delta variant surge. Efforts are needed to improve access to and increase confidence in COVID-19 vaccines, especially in low-coverage areas.
The SARS-CoV-2 B.1.617.2 (Delta) variant was first identified in the United States in March 2021. By July 2021, the highly transmissible Delta variant had spread rapidly and become the predominant strain in the United States, causing nearly a million cases and >100 000 deaths.1,2 The Delta variant surge disproportionately affected certain geographic areas, with some regions experiencing an increase in incidence and severity of illness, hospitalization, and death driven largely by those who were unvaccinated. 3 During the surge, hospitalization rates among unvaccinated adolescents and adults were 10 and 17 times higher, respectively, than among those who were fully vaccinated.4,5
Prior to the Delta variant surge, COVID-19 vaccination coverage varied widely by geographic region,2,6,7 with some states meeting the White House goal of at least 70% of the eligible population receiving ≥1 dose of a COVID-19 vaccine. 8 Survey data found that the increase in cases due to the Delta variant was a substantial motivator to initiate vaccination, as was personally knowing someone who became seriously ill or died of COVID-19. 9 The objective of this analysis was to assess how COVID-19 vaccine initiation and dose completion changed overall vaccination coverage during the Delta variant surge, based on jurisdictional vaccination coverage before the surge, and to stratify vaccine recipients by age, sex, and jurisdiction (50 states and the District of Columbia).
Methods
To determine COVID-19 vaccine initiation during the Delta variant surge, we analyzed data on COVID-19 vaccine administration during December 14, 2020–October 31, 2021, in the United States as reported to the Centers for Disease Control and Prevention (CDC) by jurisdictions, pharmacies, and federal entities.6,7 To assess vaccination coverage prior to the Delta variant becoming the predominant strain in July 2021, 10 we analyzed jurisdiction-level COVID-19 vaccine administration data among people aged ≥12 years who received their first dose of the Pfizer-BioNTech or Moderna COVID-19 vaccine or a single dose of the Janssen COVID-19 vaccine (Johnson & Johnson) during December 14, 2020–June 30, 2021, in 51 US jurisdictions (50 states and the District of Columbia). We calculated vaccination coverage by dividing the population who received ≥1 dose of a COVID-19 vaccine by the total population eligible for vaccination living in the defined jurisdiction. 11 We classified jurisdictions into quartiles ranging from high to low vaccination coverage for the first dose of COVID-19 vaccine. The 4 coverage quartiles were as follows: high (n = 13 jurisdictions, ≥71.1% coverage), medium high (n = 13, 62.2%-71.0%), medium low (n = 13, 55.7%-61.9%), and low (n = 12, <55.7%).
We calculated vaccination coverage, absolute percentage-point increase, and relative percentage change before (ie, December 14, 2020–June 30, 2021) and after (ie, July 1–October 31, 2021) the Delta variant surge. We stratified vaccination coverage by jurisdiction, age (12-17, 18-64, and ≥65 years), and sex. We also calculated monthly vaccination rates for the month before (June) and the months during (July, August, September, October) the Delta variant surge. We assessed dose completion among those who initiated a 2-dose series and had adequate time to complete the series (ie, received the second dose within 42 days of the first dose). We obtained data on population size from the US Census 2020 population estimates. 12 We did not conduct tests of significance because these data reflect the US population and were not based on population samples. We conducted analyses using SAS version 9.4 (SAS Institute, Inc). CDC reviewed this activity and determined that it was conducted consistent with applicable federal law and CDC policy.
Results
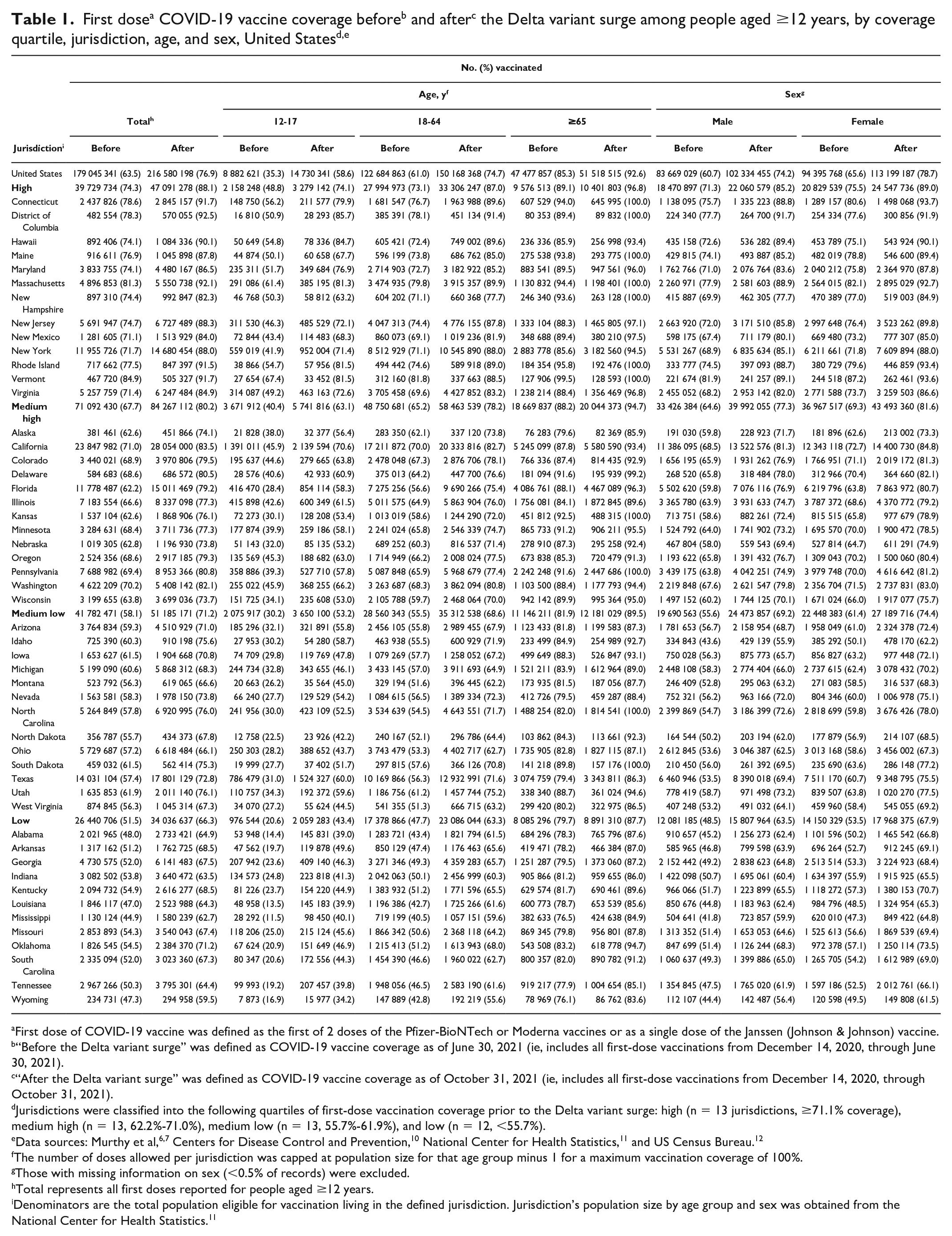
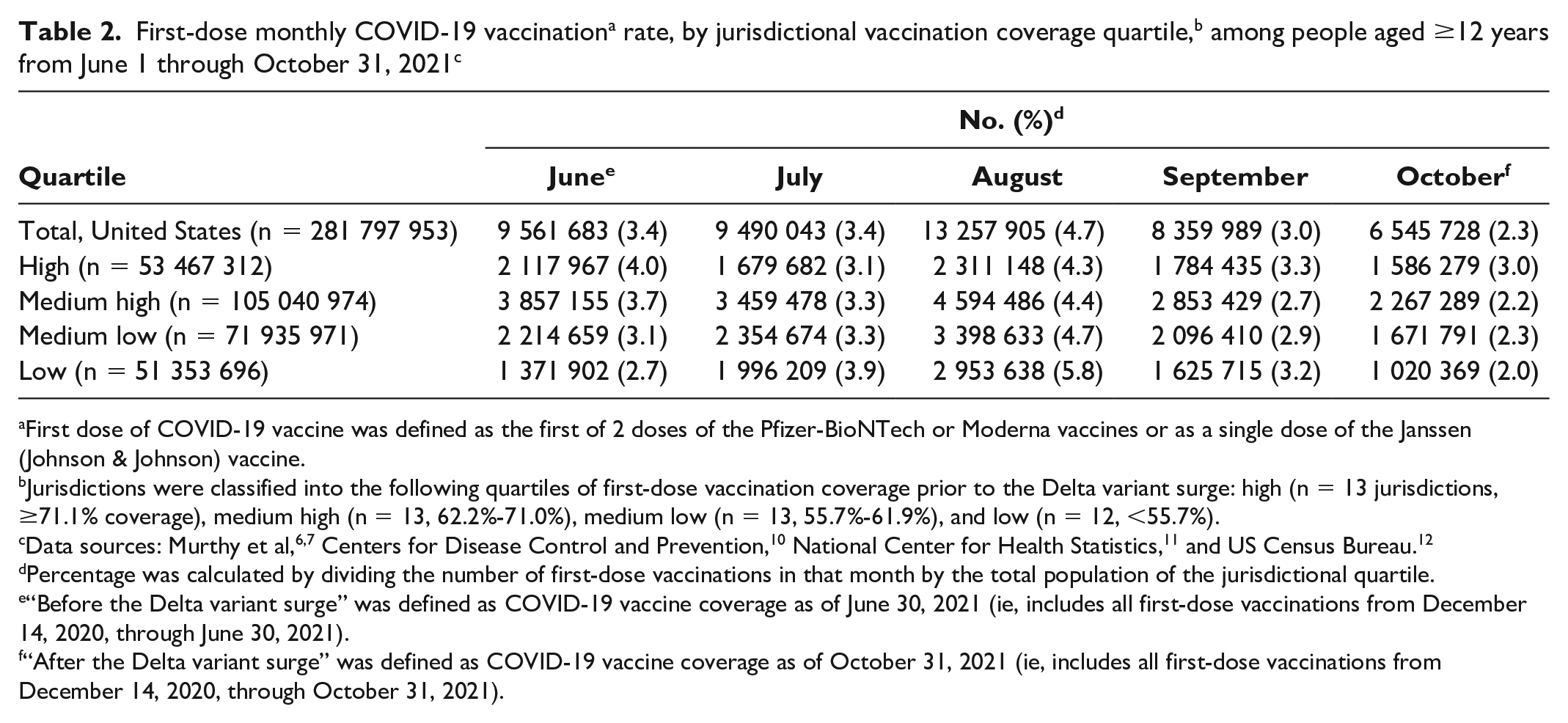
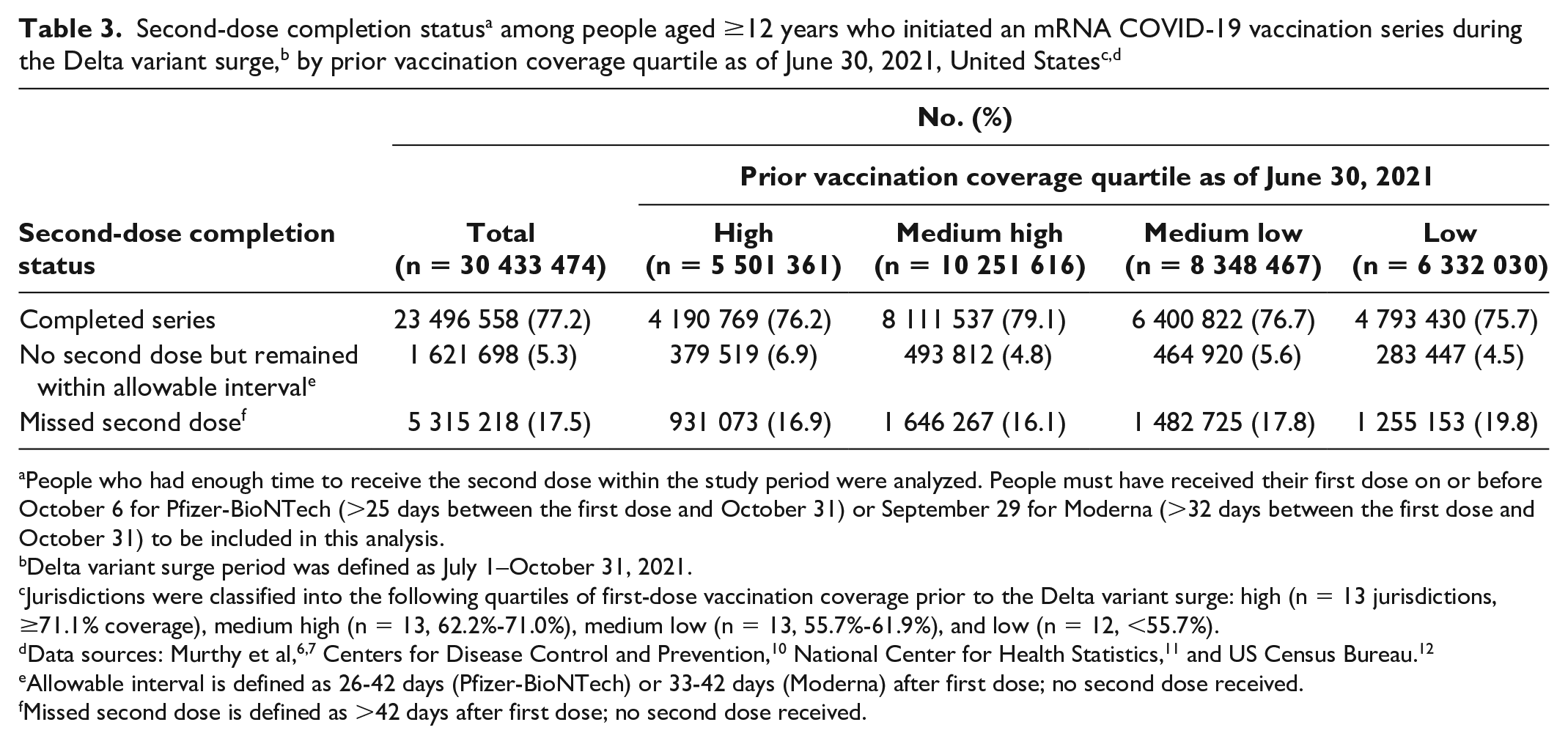
Of 51 jurisdictions, 15 reached at least 70% vaccination coverage before the Delta variant surge (ie, as of June 30, 2021), while 35 reached that goal as of October 31, 2021 (Table 1). All quartiles experienced an increase in COVID-19 vaccination coverage during the Delta variant surge (Figure 1). Several jurisdictions in the lowest quartile (Alabama, Arkansas, Louisiana, Mississippi) had the highest relative percentage change in first-dose vaccination coverage (39.8%, 36.7%, 35.2%, and 33.8%, respectively) across all 51 jurisdictions during July 1–October 31, 2021. The monthly vaccination rate was higher in August than in other months across all quartiles; however, we found the highest rate in jurisdictions in the lowest quartile in August (Table 2). Jurisdictions in the lowest quartile of vaccination coverage also had the greatest absolute percentage-point increase during July 1–October 31, 2021, but the lowest relative decrease in the unvaccinated population (Figure 2). First-dose vaccination coverage varied by age (ie, higher among older populations [ie, aged ≥65 years] than among adolescents [ie, aged 12-17 years]) but not by sex (before: males = 60.7%, females = 65.6%; after: males = 74.2%, females = 78.7%). Among those who initiated the 2-dose mRNA vaccines during July 1–October 31, 2021, 17.5% were overdue for receipt of the second dose (Table 3).
First dose of COVID-19 vaccine was defined as the first of 2 doses of the Pfizer-BioNTech or Moderna vaccines or as a single dose of the Janssen (Johnson & Johnson) vaccine.
“Before the Delta variant surge” was defined as COVID-19 vaccine coverage as of June 30, 2021 (ie, includes all first-dose vaccinations from December 14, 2020, through June 30, 2021).
“After the Delta variant surge” was defined as COVID-19 vaccine coverage as of October 31, 2021 (ie, includes all first-dose vaccinations from December 14, 2020, through October 31, 2021).
Jurisdictions were classified into the following quartiles of first-dose vaccination coverage prior to the Delta variant surge: high (n = 13 jurisdictions, ≥71.1% coverage), medium high (n = 13, 62.2%-71.0%), medium low (n = 13, 55.7%-61.9%), and low (n = 12, <55.7%).
Data sources: Murthy et al,6,7 Centers for Disease Control and Prevention, 10 National Center for Health Statistics, 11 and US Census Bureau. 12
The number of doses allowed per jurisdiction was capped at population size for that age group minus 1 for a maximum vaccination coverage of 100%.
Those with missing information on sex (<0.5% of records) were excluded.
Total represents all first doses reported for people aged ≥12 years.
Denominators are the total population eligible for vaccination living in the defined jurisdiction. Jurisdiction’s population size by age group and sex was obtained from the National Center for Health Statistics. 11
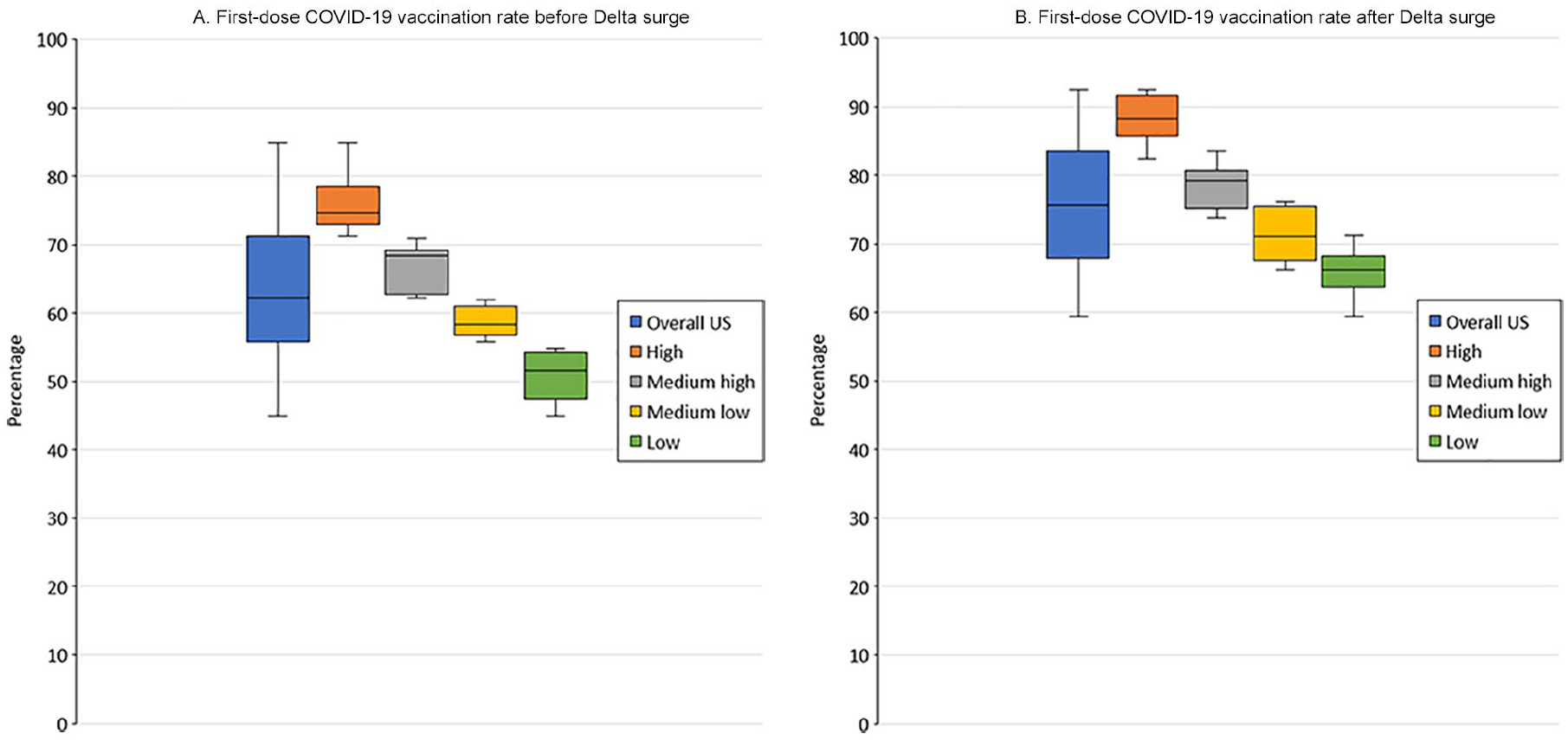
First-dose COVID-19 vaccination coverage among people aged ≥12 years before and after the Delta variant surge, by prior vaccination coverage quartile, as of June 30, 2021, United States. First dose of COVID-19 vaccine is defined as the first of 2 doses of the Pfizer-BioNTech or Moderna vaccines or as a single dose of the Janssen (Johnson & Johnson) vaccine. The Delta variant surge period is defined as July 1–October 31, 2021. Jurisdictions were classified into the following quartiles of first-dose vaccination coverage prior to the Delta variant surge: high (n = 13 jurisdictions, ≥71.1% coverage), medium high (n = 13, 62.2%-71.0%), medium low (n = 13, 55.7%-61.9%), and low (n = 12, <55.7%). Data sources: Murthy et al,6,7Centers for Disease Control and Prevention 10 National Center for Health Statistics, 11 and US Census Bureau. 12
First dose of COVID-19 vaccine was defined as the first of 2 doses of the Pfizer-BioNTech or Moderna vaccines or as a single dose of the Janssen (Johnson & Johnson) vaccine.
Jurisdictions were classified into the following quartiles of first-dose vaccination coverage prior to the Delta variant surge: high (n = 13 jurisdictions, ≥71.1% coverage), medium high (n = 13, 62.2%-71.0%), medium low (n = 13, 55.7%-61.9%), and low (n = 12, <55.7%).
Data sources: Murthy et al,6,7 Centers for Disease Control and Prevention 10 National Center for Health Statistics, 11 and US Census Bureau. 12
Percentage was calculated by dividing the number of first-dose vaccinations in that month by the total population of the jurisdictional quartile.
“Before the Delta variant surge” was defined as COVID-19 vaccine coverage as of June 30, 2021 (ie, includes all first-dose vaccinations from December 14, 2020, through June 30, 2021).
“After the Delta variant surge” was defined as COVID-19 vaccine coverage as of October 31, 2021 (ie, includes all first-dose vaccinations from December 14, 2020, through October 31, 2021).
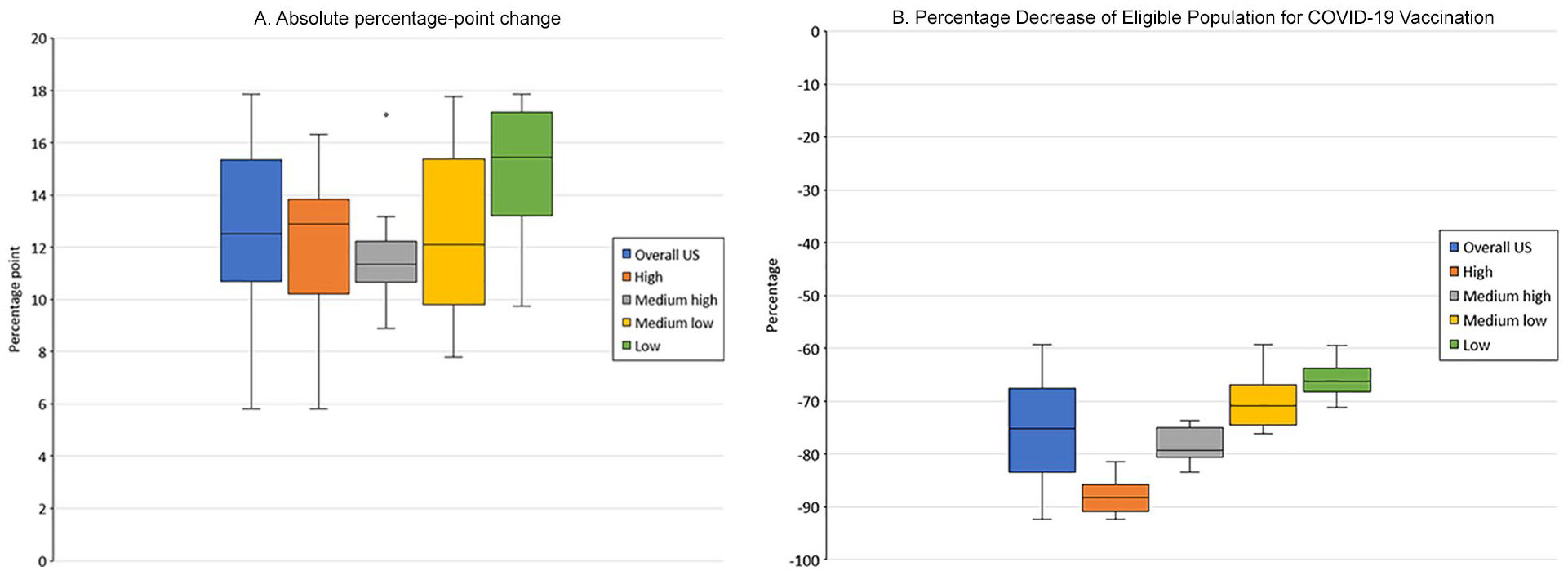
Absolute percentage-point change in first-dose COVID-19 vaccination coverage and relative percentage change among eligible unvaccinated people aged ≥12 years during the Delta variant surge, by prior vaccination coverage quartile, as of June 30, 2021, United States. First dose of COVID-19 vaccine is defined as the first of 2 doses of the Pfizer-BioNTech or Moderna vaccines or as a single dose of the Janssen (Johnson & Johnson) vaccine. The Delta variant surge period is defined as July 1–October 31, 2021. Jurisdictions were classified into the following quartiles of first-dose vaccination coverage prior to the Delta variant surge: high (n = 13 jurisdictions, ≥71.1% coverage), medium high (n = 13, 62.2%-71.0%), medium low (n = 13, 55.7%-61.9%), and low (n = 12, <55.7%). Data sources: Murthy et al, 6,7 Centers for Disease Control and Prevention, 10 National Center for Health Statistics, 11 and US Census Bureau. 12
People who had enough time to receive the second dose within the study period were analyzed. People must have received their first dose on or before October 6 for Pfizer-BioNTech (>25 days between the first dose and October 31) or September 29 for Moderna (>32 days between the first dose and October 31) to be included in this analysis.
Delta variant surge period was defined as July 1–October 31, 2021.
Jurisdictions were classified into the following quartiles of first-dose vaccination coverage prior to the Delta variant surge: high (n = 13 jurisdictions, ≥71.1% coverage), medium high (n = 13, 62.2%-71.0%), medium low (n = 13, 55.7%-61.9%), and low (n = 12, <55.7%).
Data sources: Murthy et al, 6,7 Centers for Disease Control and Prevention, 10 National Center for Health Statistics, 11 and US Census Bureau. 12
Allowable interval is defined as 26-42 days (Pfizer-BioNTech) or 33-42 days (Moderna) after first dose; no second dose received.
Missed second dose is defined as >42 days after first dose; no second dose received.
Discussion
To our knowledge, this analysis is the first attempt to assess changes in COVID-19 vaccine initiation and dose completion based on prior jurisdictional coverage as the Delta variant emerged and spread rapidly in the United States, leading to a nationwide surge in cases. Overall, jurisdictions in the lowest quartile of vaccination coverage as of June 30, 2021, had the highest percentage increase in vaccine initiation and dose completion and the smallest relative decrease in its unvaccinated population during July 1–October 31, 2021, with several jurisdictions in the lowest quartile of vaccination coverage (eg, Alabama, Arkansas, Louisiana, Mississippi) having the highest relative percentage increase in coverage. The reasons for differences in vaccination uptake in the jurisdictions in the lowest quartile as compared with jurisdictions in the highest quartile may reflect fear of contracting COVID-19 during the Delta variant surge, which is supported by research showing that rising numbers of local COVID-19 cases can lead to behavior change, wherein individuals voluntarily engage in protective behaviors such as social distancing.9,13 Residents of jurisdictions in the lowest quartile were more likely to experience the Delta variant surge firsthand, as many of these areas were hot spots during the surge. 14 When compared with prior variants, the Delta variant was more transmissible and caused more severe illness resulting in hospitalization and death. 15 Therefore, it is plausible that this Delta variant surge led to a sharp rise in vaccinations, especially during August, as people living in hot spots may have personally known someone who became seriously ill or died of COVID-19, thereby motivating them to initiate vaccination. 9
Many important events occurred in summer and fall 2021 that could have affected COVID-19 vaccination uptake across all 4 quartiles. For example, in May 2021, the US Food and Drug Administration authorized the Pfizer BioNTech vaccine for adolescents aged 12-15 years. 7 In July 2021, the White House announced vaccination requirements for federal employees and encouraged private sectors to also ensure that their workers are vaccinated. 16 Although enforcement of the federal vaccine mandate did not go into effect until November 2021, federal employees may have initiated COVID-19 vaccination in late summer or early fall in anticipation of the federal vaccine mandate. Additionally, the US Food and Drug Administration granted approval for a biologics license application for the Pfizer-BioNTech COVID-19 vaccine on August 23, 2021, which also may have affected vaccination uptake. 17 However, there is no indication that any of these events would have differentially affected certain jurisdictions over others, because the rollout of these events uniformly affected the entire nation.
Vaccination initiation during July 1–October 31, 2021, also varied by age, with initiation being highest among adolescents and lowest among adults aged ≥65 years. However, vaccination uptake among adolescents aged 12-17 years may have been affected by expanded eligibility of COVID-19 vaccines for those aged 12-15 years in May 2021. 7 Adults aged ≥65 years already had relatively high vaccination coverage in most jurisdictions by the end of June 2021, which may explain why this age group had the smallest absolute percentage-point increase in vaccinations during the Delta variant surge.
Among those who initiated the 2-dose mRNA vaccine series during the Delta variant surge, the proportion of people who did not complete their second dose within the allowable interval was higher in our analysis than in previous analyses on dose completion.7,18 An analysis on dose completion before the Delta variant surge showed that 88.0% of people initiating vaccination went on to complete the primary series 18 ; however, our analysis showed that only 77.2% of people who initiated vaccination during the Delta variant surge completed the primary series. If fear was a motivator to initiate vaccination during the Delta variant surge, then perhaps this perceived fear gradually diminished as the Delta variant surge passed, which could explain why those who initiated vaccination chose not to complete the 2-dose series. Nevertheless, these findings underscore the need for all people to complete the primary series and to stay up-to-date on all recommended vaccines (eg, boosters and additional doses) for maximal protection against COVID-19, especially given that staying up-to-date on all recommended doses is effective against serious illness and death. 19
Limitations
Our findings had 3 limitations. First, vaccination coverage was not analyzed at more granular levels (eg, by county), which could better identify geographic areas with low coverage. Second, people who (1) received COVID-19 vaccines from different entities that used different methods for submitting data (eg, if the first dose was administered at a mass vaccination site and the subsequent doses were given at a pharmacy) or (2) received vaccine doses in different jurisdictions might not have their first and subsequent doses linked, which could lead to an underestimation of the percentage of people who completed the vaccination series and the number of people who were unvaccinated. Lastly, if a person had inadvertently received a different recipient identification number when receiving their first and subsequent doses, those records would not be linked.
Conclusions
Jurisdictions in the lowest quartile of vaccination coverage prior to the Delta variant surge had the highest absolute and relative percentage increase in vaccination coverage during July 1–October 31, 2021. However, as of October 31, 2021, many of these jurisdictions in the lowest quartile of vaccination coverage, as well as some jurisdictions in the medium-low quartile, still did not reach first-dose vaccination coverage of 70%. 8 The highest quartile of vaccination coverage also had substantial vaccination uptake during the Delta variant surge, and lessons learned from these jurisdictions can help identify best practices to improve COVID-19 vaccination coverage that may be applied to jurisdictions in other quartiles. Furthermore, dose completion lagged among those who started the 2-dose mRNA vaccine series during this time, with almost 1 in 5 people missing their second dose. Strategic efforts should be made to identify and reduce barriers to receiving COVID-19 vaccines, including addressing vaccine hesitancy, especially in jurisdictions with low vaccination coverage to reduce COVID-19–associated morbidity and mortality. 20 Improving vaccination coverage nationally and globally is essential to protect populations and prevent the rise of more variants.
Footnotes
Acknowledgements
The authors acknowledge the COVID-19 Vaccine Task Force, the US Department of Defense, immunization program managers, immunization information system managers, and other staff members of the immunization programs in the 64 jurisdictions and 5 federal entities who provided these data.
Disclaimer
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the US government, including the Centers for Disease Control and Prevention and the US Department of State.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.