Abstract
Objectives:
COVID-19 surveillance data are rarely collected or disaggregated by gender identity in the United States. We quantified COVID-19 testing experiences and SARS-CoV-2 infection history among transgender and gender-diverse (TGD) people to inform testing strategies and public health responses.
Methods:
From June 14 through December 16, 2021, TGD adults enrolled in a US nationwide online survey with optional SARS-CoV-2 antibody testing. We used multinomial regression analyses to identify correlates of suspected and confirmed SARS-CoV-2 infection (vs no known infection). We identified correlates of inability to access COVID-19 testing when needed using generalized linear models for binomial variables.
Results:
Participants (N = 2092) reported trans masculine (30.5%), trans feminine (27.3%), and nonbinary (42.2%) gender identities. Ten percent of respondents had a confirmed history of SARS-CoV-2 infection, and 29.8% had a history of suspected SARS-CoV-2 infection. Nonbinary gender (adjusted prevalence ratio [aPR] = 1.68; 95% CI, 1.12-2.53), experiencing homelessness (aPR = 1.65; 95% CI, 1.05-2.60), and food insecurity (aPR = 1.45; 95% CI, 1.03-2.04) were associated with confirmed SARS-CoV-2 infection. Food insecurity (aPR = 1.38; 95% CI, 1.10-1.72), chronic physical health condition (aPR = 1.44; 95% CI, 1.15-1.80), chronic mental health condition (aPR = 3.65; 95% CI, 2.40-5.56), and increased anticipated discrimination scores (aPR = 1.03; 95% CI, 1.01-1.05) were associated with suspected SARS-CoV-2 infection. Thirty-four percent (n = 694 of 2024) of participants reported an inability to access COVID-19 testing when needed, which was associated with Latinx or Hispanic ethnicity, inconsistent telephone access, homelessness, disability, and transportation limitations. The majority (79.4%) reported a complete COVID-19 vaccine course at the time of participation.
Conclusions:
Inclusion of TGD people in public health surveillance and tailored public health strategies to address TGD communities’ social and structural vulnerabilities may reduce barriers to COVID-19 testing.
Keywords
As of September 2022, 2.5 years after the World Health Organization’s declaration of the global COVID-19 pandemic, more than 96 million COVID-19 cases had been confirmed in the United States. 1 Early research and surveillance identified risk factors for severe disease and social conditions related to disparities in infection. Individuals with compromised immune systems and underlying health conditions were more vulnerable to severe illness and death.2,3 Epidemiologic trends in the United States highlighted the disproportionate number of SARS-CoV-2 infections and COVID-19–related deaths among Black and Latinx communities,2,3 and monitoring has highlighted disparities by sex. 4 The epidemiology of SARS-CoV-2 and COVID-19 disease revealed vulnerabilities and populations at greater-than-average risk, but data are rarely collected or disaggregated by gender identity. 5
Prior research demonstrated disparities in the prevalence and incidence of disease and health behaviors among transgender and gender-diverse (TGD) people in the United States, including cardiovascular disease and comorbidities 6 and elevated prevalence of HIV, 7 which may increase the risk for severe COVID-19 disease. The use of exogenous hormones in gender-affirming care has been hypothesized to increase the risk for or protect against SARS-CoV-2 infection when estrogens are suppressed or enhanced, respectively. 8 Multilevel stigma, discrimination, violence victimization, and material hardship 9 are high among TGD people and correspond to high levels of psychosocial morbidities including depression, anxiety, and posttraumatic stress disorder. 10 These experiences may indirectly confer COVID-19 vulnerability: stress may increase inflammatory processes to increase the risk of severe disease, unstable housing may prevent social distancing, essential jobs may also increase the risk of exposure to COVID-19 in high-density settings, and discrimination in health care may prohibit access to testing for people with COVID-19 disease. These effects may also be buffered by extensive community support and mutual aid that rapidly emerged during early phases of the pandemic. 11
Understanding the burden of COVID-19 among TGD people and identifying strategies to mitigate disparities in testing and health services is a matter of health equity. 12 The objective of this study was to quantify SARS-CoV-2 seroprevalence, COVID-19 testing experiences, and strategies to overcome testing barriers among TGD people in the United States.
Methods
We conducted a cross-sectional study, known as LITE Connect, to study COVID-19 via remote survey questionnaire and antibody testing in a nationwide sample of TGD people. This study leveraged the existing infrastructure of ongoing, technology-enhanced cohorts focused on transgender women in the eastern and southern United States.13-15 As with those cohorts, we designed this study to focus exclusively on the experiences of TGD people to gather data on the roles of psychosocial and contextual factors in COVID-19 outcomes and barriers to testing. This approach has been found to be highly informative for public health response and programming for populations in which health disparities have been previously documented. 16 The Johns Hopkins School of Medicine Institutional Review Board conducted the ethical review and approved the study.
Recruitment
We used community-based and digital recruitment strategies to reach and engage candidate participants. We conducted recruitment in collaboration with community-based organizations working across geographic locations and serving racially and ethnically diverse communities. Electronic recruitment materials with links to the study website were shared through the following strategies: by community partners via list-servs and social media sites, peer-to-peer referral, and email and text message invitations to participants of related cohorts. To mitigate the risk of fraudulent activity, we did not formally advertise on social media platforms, although participants and community-based organizations were able to share the study information via their own social media accounts and networks. The study website provided a link to the screening form, which transitioned to an online consent form and questionnaire for those who were eligible and consented, respectively. Recruitment continued until the target sample size of 2000 participants was achieved.
Study Population
Participants were eligible if they were aged ≥18 years; identified as trans masculine, trans feminine, or nonbinary; resided in any US state or territory; and spoke English or Spanish. Gender identity was assessed during screening by using the 2-step method, which measures assigned sex and gender identity. 17 To validate participation and mitigate the risk of fraudulent participation, we used a 2-step verification procedure, we excluded voice-over-internet protocol telephone numbers in registration, we used internal algorithms to run real-time checks on duplicate information, and staff contacted participants when registration information was flagged.
We used strategies to foster inclusivity. Staff conducted interviewer-administered screening, obtained consent, and administered the questionnaire to maximize participation of people with low literacy levels and/or low digital literacy. All study materials were available in English and Spanish, and bilingual staff served on the study team.
Measures
The questionnaire included common data elements developed under the National Institutes of Health’s Rapid Acceleration of Diagnostics for Underserved Populations (RADx-UP) program, 18 with some modifications to reduce measurement error. Common data elements included demographic characteristics, livelihood and social impacts of the pandemic, history of COVID-19 vaccination, history of COVID-19 testing and diagnosis, and self-reported disability. We developed new measures of COVID-19 illness severity, duration, and recovery for those who reported a reactive test result. Given the lack of availability of and barriers to COVID-19 testing during various periods of the pandemic, we developed additional measures to capture data on (1) any inability to get a COVID-19 test when needed and (2) self-reported suspected, but untested, SARS-CoV-2 infection based on symptoms and exposure. We included existing measures of chronic illness 19 and the Intersectional Anticipated Discrimination Index. 20 We developed items to measure issues unique to the experience of TGD people, such as disruptions in gender-affirming care and increased morbidities. The study team, which included co-investigators from TGD communities, drafted the questionnaire in English and Spanish and pilot tested it for electronic self-administration.
Laboratory Testing and Provision of Results
Participants were offered the option of SARS-CoV-2 antibody testing using self-collected specimens. Because of high levels of mistrust of research in the TGD population, antibody testing was not required for participation. Laboratory orders were automatically sent from our electronic system to an external Clinical Laboratory Improvement Amendments–certified laboratory using their application programming interface. Participants received discreet self-collection kits by mail that were customized and branded to the study. Kits contained Whatman dried blood spot cards, alcohol swabs, lancets, bandages, and instructions in English and Spanish. Participants received an automated email to notify them when their kit was mailed and provide a link to an animated instructional video. The video was professionally developed for diverse TGD people with input from the study team. Participants returned the specimens to the laboratory using a self-enclosed, prepaid mailer.
Self-collected dried-blood spot specimens were tested at the laboratory to detect the presence of antibodies to SARS-CoV-2. Testing used Platelia SARS-CoV-2 Total Ab (Bio-Rad Laboratories), which detects anti–SARS-CoV-2 immunoglobulin M, immunoglobulin A, and immunoglobulin G to determine immune response to SARS-CoV-2 infection, with a reflex to an anti–SARS-CoV-2 enzyme-linked immunosorbent assay immunoglobulin G test (EUROIMMUN US Inc). This testing algorithm allows for the identification of naturally derived immunity. Both assays received Emergency Use Authorization by the US Food and Drug Administration. 21
Once tests were processed in the laboratory, participants received an automated email with results and interpretation. Staff members also called participants who had a reactive antibody test to inform them of the result, discuss what the result meant, and answer any questions. Notifications also included links to Centers for Disease Control and Prevention websites for further information and guidance.
Statistical Analysis
We used descriptive statistics to examine the study population, differences across gender identities, barriers to COVID-19 testing, and preferences for future testing options. Our primary interests were history of SARS-CoV-2 infection and access to testing. Given variations in availability of testing and frequency of asymptomatic cases, we created a composite categorical variable for confirmed, suspected, and no known history of SARS-CoV-2 infection (Table 1).
Analytic outcomes and operational definitions of history of SARS-CoV-2 infection and access to testing among a sample of transgender and gender-diverse people during the COVID-19 pandemic, United States

We used multinomial regression analyses with robust variance estimation to identify correlates and calculate prevalence ratios (PRs) of suspected and confirmed SARS-CoV-2 infection against the reference group of no known infection. We tested independent variables of interest, including known correlates of SARS-CoV-2 infection and severity and situated vulnerabilities well-described for TGD people. 22 We included an indicator for phase of participation that was categorized as participation from June 14 through August 31, 2021, and from September 1 through December 16, 2021, to account for surges in cases and test availability in the United States. We included variables that were significant at P < .05 in the unadjusted models in the final multivariable model.
We identified correlates of inability to access COVID-19 testing when needed by using generalized linear models for binomial variables with robust variance estimation to calculate PRs for variables of interest. We constructed multivariable models using the same process. Because models failed to converge with further addition of covariates, we constructed several models. All models adjusted for age, geographic region, and ethnicity.
We determined all final models by model fit and evaluated them for collinearity. Because the missingness of the included variables was <10%, we did not perform additional imputation methods. We conducted the statistical analyses using Stata version 17 (StataCorp LLC).
Participants received a national resource guide with contact information for national trans-affirming and inclusive services for mental health, violence, substance use, COVID-19, and other health services. Study incentives included $40 for completion of the survey and $10 for antibody testing (Amazon gift cards or mailed debit cards).
Results
Sample Characteristics
From June 14 through December 16, 2021, a total of 2092 participants were enrolled in the study across all states; Washington, DC; and Puerto Rico, of whom 1002 (47.9%) opted in and 475 (22.7%) completed antibody testing (Table 2). Most (1639 of 2064, 79.4%) participants reported a complete COVID-19 vaccine course at the time of participation. Although the demographic characteristics, health, and vaccination history of participants differed by gender identity categories, we found no differences in COVID-19 testing or serology results or severity of illness. Gender categories with assigned sex are available in eTable 1 in the Supplement.
Demographic characteristics, health status, and COVID-19 history among transgender and gender-diverse participants (N = 2092) in a nationwide survey, stratified by gender identity, United States, June 14–December 16, 2021
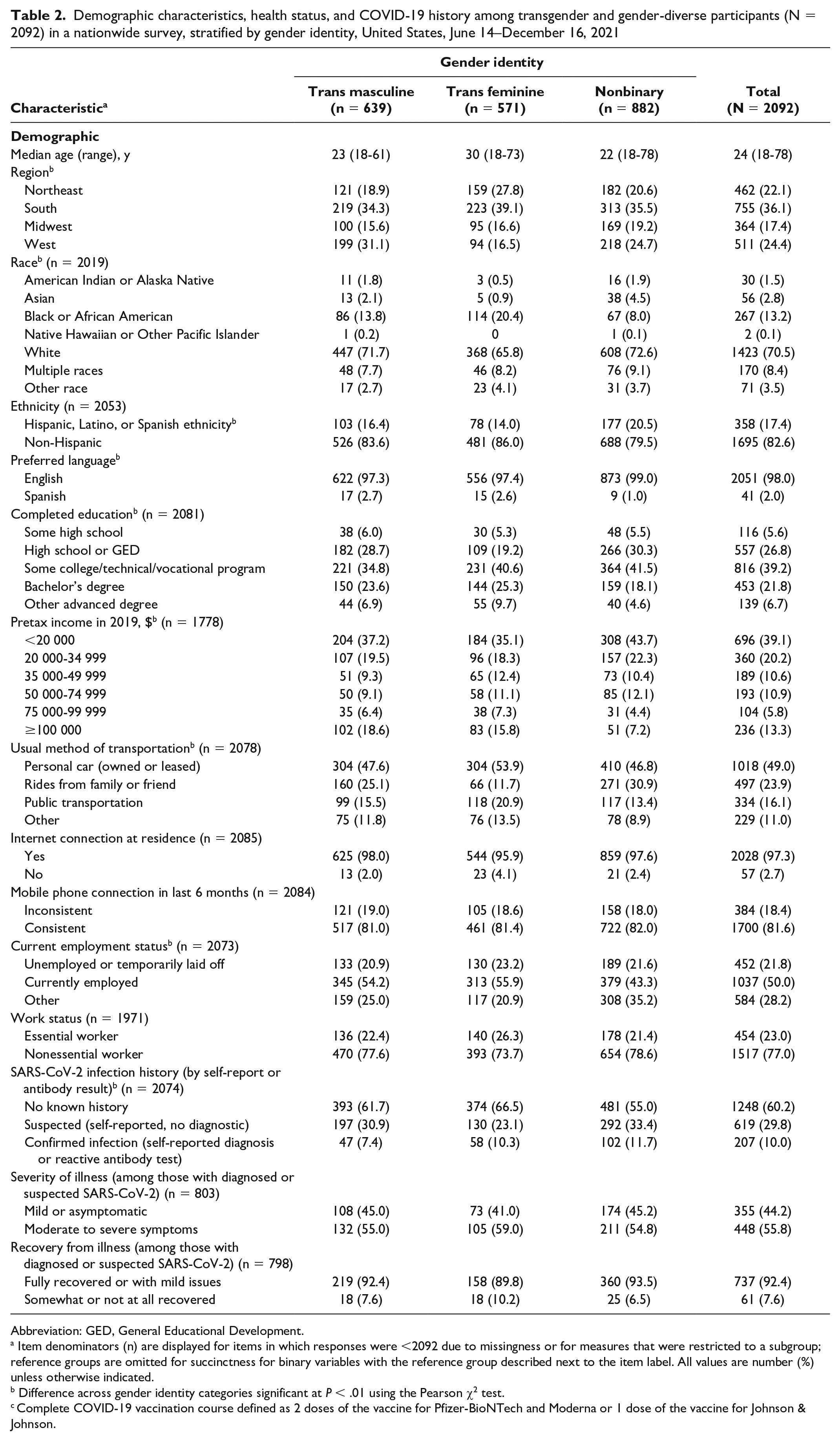
Abbreviation: GED, General Educational Development.
Item denominators (n) are displayed for items in which responses were <2092 due to missingness or for measures that were restricted to a subgroup; reference groups are omitted for succinctness for binary variables with the reference group described next to the item label. All values are number (%) unless otherwise indicated.
Difference across gender identity categories significant at P < .01 using the Pearson χ2 test.
Complete COVID-19 vaccination course defined as 2 doses of the vaccine for Pfizer-BioNTech and Moderna or 1 dose of the vaccine for Johnson & Johnson.
SARS-CoV-2 Infection
Among 2074 participants with self-reported laboratory results and/or serology results, 207 (10.0%) had a confirmed infection and 619 (29.8%) had a suspected infection (Table 2). Residence in the South versus the Northeast (aPR = 1.63; 95% CI, 1.22-2.18), food insecurity versus food security (aPR = 1.38; 95% CI, 1.10-1.72), having a chronic physical health condition versus not (aPR = 1.44; 95% CI, 1.15-1.80), having a chronic mental health condition versus not (aPR = 3.65; 95% CI, 2.40-5.56), and increased Intersectional Anticipated Discrimination Index scores (aPR = 1.03; 95% CI, 1.01-1.05) were significantly associated with suspected SARS-CoV-2 infection (Table 3). Participation during September–December 2021 versus June–August 2021 (aPR = 2.25; 95% CI, 1.28-3.97), residence in the South versus the Northeast (aPR = 1.95; 95% CI, 1.24-3.07), nonbinary gender versus trans masculine gender (aPR = 1.68; 95% CI, 1.12-2.53), experiencing homelessness during the pandemic versus not (aPR = 1.65; 95% CI, 1.05-2.60), and experiencing food insecurity versus food security (aPR = 1.45; 95% CI, 1.03-2.04) were significantly associated with confirmed infection.
Multinomial regression models to identify correlates of suspected and confirmed SARS-CoV-2 infection among transgender and gender-diverse people who participated in a nationwide survey (N = 2092), June 14–December 16, 2021
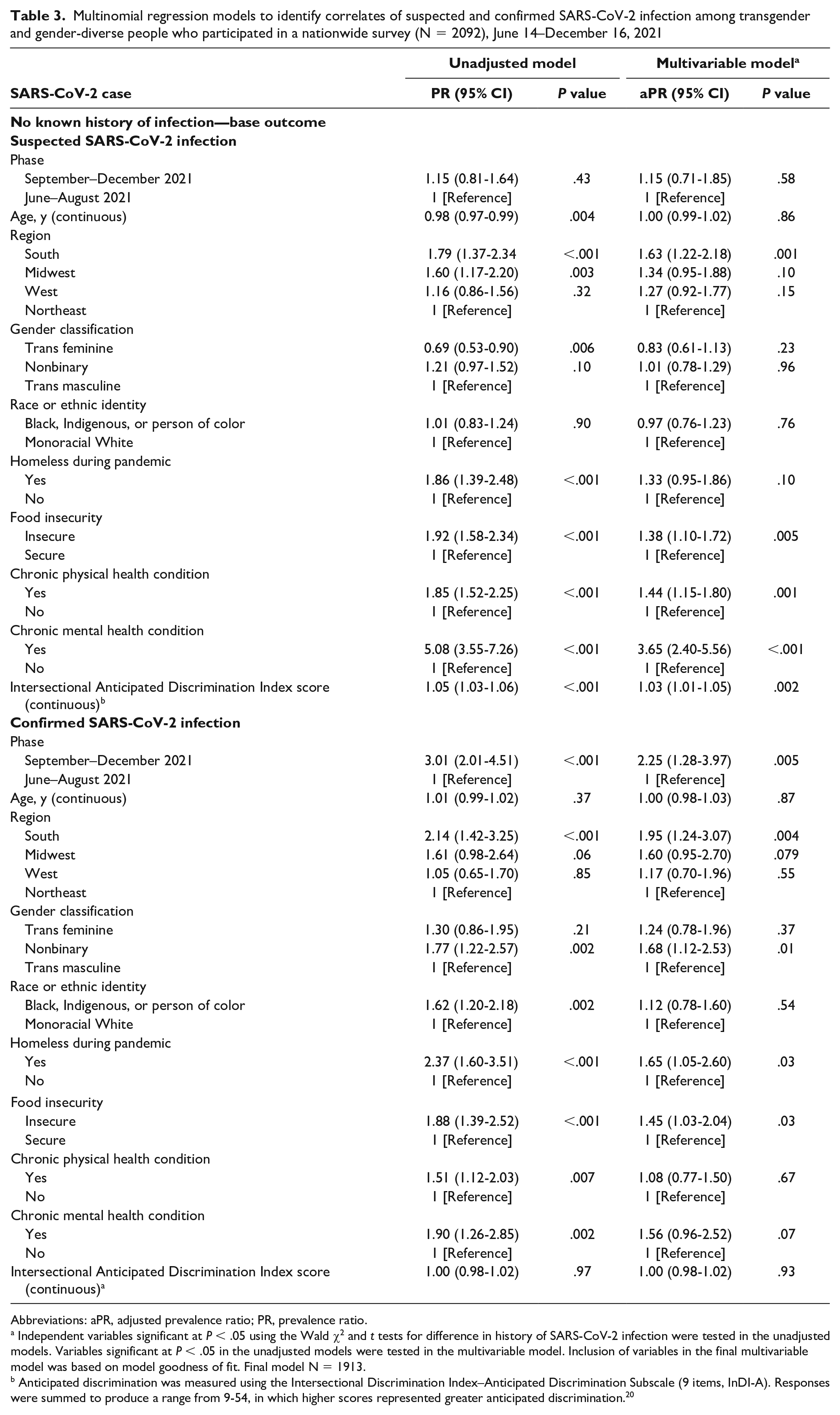
Abbreviations: aPR, adjusted prevalence ratio; PR, prevalence ratio.
Independent variables significant at P < .05 using the Wald χ2 and t tests for difference in history of SARS-CoV-2 infection were tested in the unadjusted models. Variables significant at P < .05 in the unadjusted models were tested in the multivariable model. Inclusion of variables in the final multivariable model was based on model goodness of fit. Final model N = 1913.
Anticipated discrimination was measured using the Intersectional Discrimination Index–Anticipated Discrimination Subscale (9 items, InDI-A). Responses were summed to produce a range from 9-54, in which higher scores represented greater anticipated discrimination. 20
Access to COVID-19 Testing
Overall, 696 of 2026 (34.4%) participants reported an inability to access COVID-19 testing when needed (Table 2). We found no difference in the inability to access COVID-19 testing by phase of participation (34.1% in June–August 2021 vs 37.2% in September–December 2021). Compared with non–Latinx/Hispanic participants, those with Hispanic or Latinx ethnicity were significantly more likely (model 1; P = .03) or more likely (models 2 [P = .08] and 3 [P = .09]) to experience inability to access testing (Table 4). Having inconsistent telephone access in the past 6 months versus consistent telephone access (models 1 and 3), experiencing homelessness during the pandemic versus not (models 1 and 2), using public transportation (models 2 and 3) or another mode of transportation (Models 2 and 3) versus having one’s own car, and having a self-reported disability versus no reported disability (models 2 and 3) were significantly associated with increased likelihood of experienced inability to access a COVID-19 test when needed.
Correlates of experienced inability to access a COVID-19 test when needed in a nationwide sample of transgender and gender-diverse people in the United States (N = 2092), June 14–December 16, 2021 a
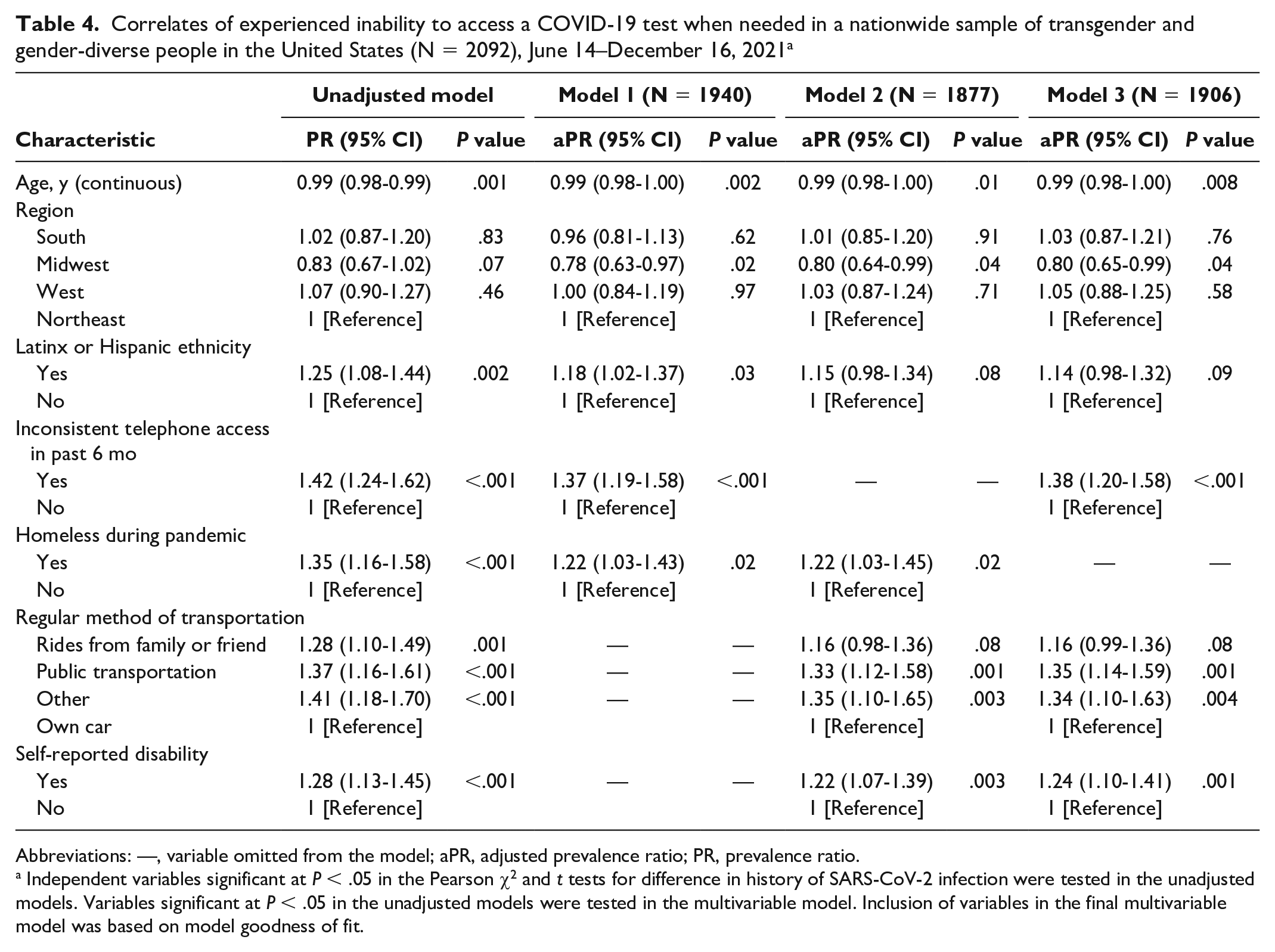
Abbreviations: —, variable omitted from the model; aPR, adjusted prevalence ratio; PR, prevalence ratio.
Independent variables significant at P < .05 in the Pearson χ2 and t tests for difference in history of SARS-CoV-2 infection were tested in the unadjusted models. Variables significant at P < .05 in the unadjusted models were tested in the multivariable model. Inclusion of variables in the final multivariable model was based on model goodness of fit.
Barriers to Testing
Twenty-two percent (436 of 2026) of respondents reported no barrier to accessing COVID-19 testing (Figure). Among those reporting barriers, common testing barriers included difficulty scheduling, not knowing where to go for a test, and concern about being exposed to others with COVID-19 at the testing site. Almost 30% of participants reported fear of mistreatment or misgendering at testing facilities as a barrier. All barriers to testing were significantly higher among participants who reported being unable to access COVID-19 testing when needed compared with those who were able to test. Barriers to testing stratified by gender identity are available in eFigure 1 in the Supplement. Barriers to testing were significantly different by gender identity at P < .05 for all reported barriers except for cost or financial concerns and other barriers. Participants who identified as nonbinary assigned female sex at birth had the highest levels of barriers to testing across all categories except for fear of mistreatment or misgendering at the testing site, which was commonly reported by trans masculine participants.
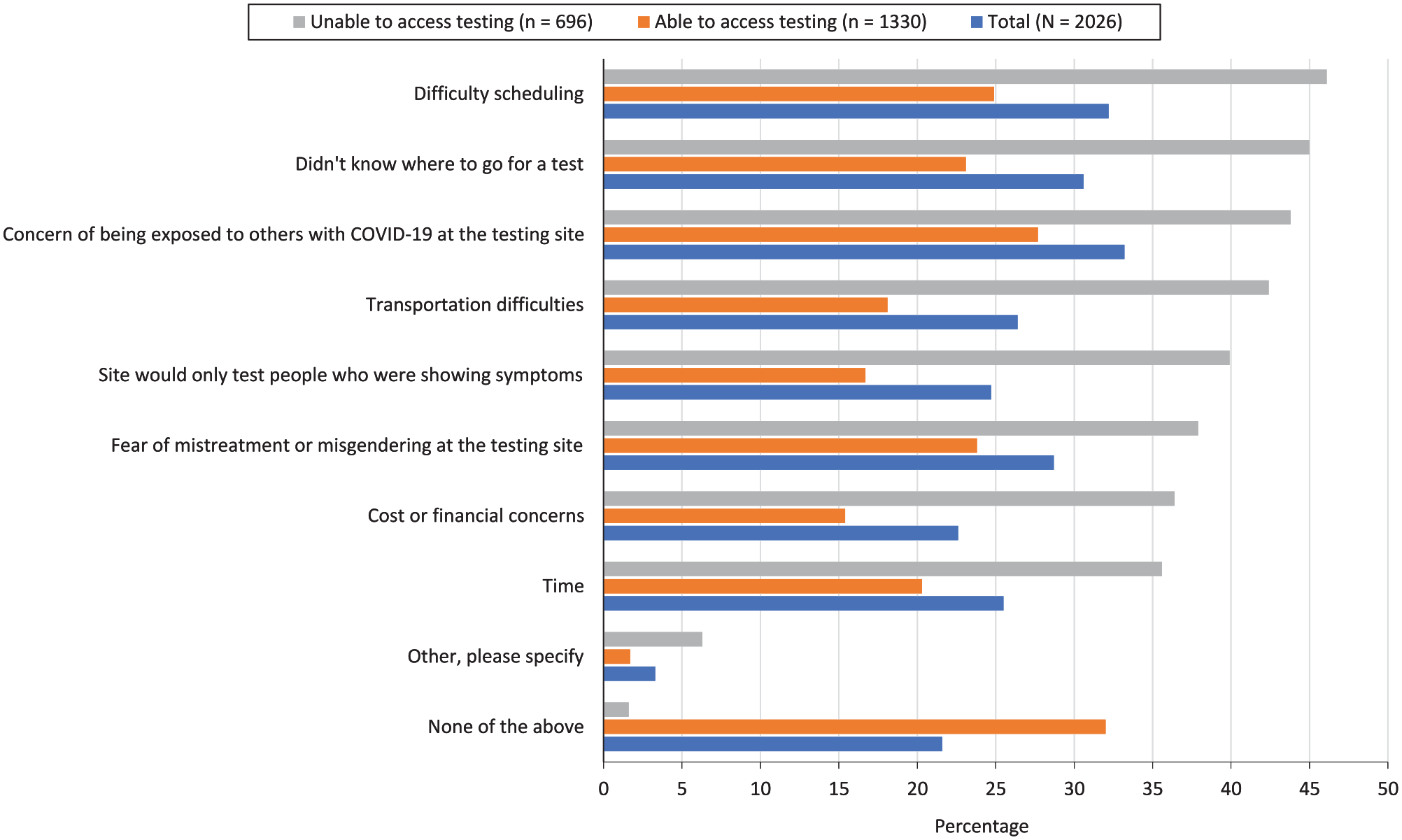
Reported barriers to accessing COVID-19 testing among transgender and gender-diverse participants in a nationwide sample (N = 2092), stratified by inability to access a COVID-19 test when needed, June 14–December 16, 2021. All differences were significant at P < .05; determined by Pearson χ2 test.
COVID-19 Testing Preferences
Testing preferences varied by prior (in)ability to access testing (eTable 2 in the Supplement). The most preferred location for testing was a health facility (81% overall), but this location was less commonly reported by participants who indicated inability to access testing when needed compared with those who had been able to access testing (76.9% vs 83.4%). Participants who reported prior inability to access testing (compared with those who had been able to access testing) were more likely to prefer testing at home (50.6% vs 44.7%) and less likely to prefer drive-through (54.0% vs 59.9%) or laboratory (45.3% vs 55.2%) testing options. Most (1405 of 1917, 73.3%) participants indicated interest in peer-provided tests if available. Twenty-three percent of participants opted in and completed self-collected specimens for serology testing with no difference by prior (in)ability to access testing.
Discussion
To our knowledge, this study provides the first estimates of SARS-CoV-2 infection and testing from TGD people across the United States. Among this diverse population of 2092 TGD people sampled from June through December 2021, 10% had a history of confirmed infection and an additional 30% reported suspected infection without accessing testing. Confirmed SARS-CoV-2 infection, either by self-reported diagnosis or serology results, more than doubled among those who participated from September through December 2021 versus June through August 2021, reflecting both national trends of infection and increased availability of testing. Seroprevalence and self-reported results in our study were slightly lower than national estimates from blood donor seroprevalence surveys, which estimated a national seroprevalence of 20% to 25% from June to December 2021. 23 When considering suspected infections alongside confirmed cases (40% cumulatively), the percentage of people who reported having a history of SARS-CoV-2 infection may be higher than national estimates. Notably, and despite evident barriers to testing that could translate to vaccination uptake, our finding of complete vaccination coverage at 79% was slightly higher than national estimates of 75% by March 2022. 24
Correlates of suspected and confirmed SARS-CoV-2 infection highlight unique factors that are likely associated with increased opportunity for exposure to COVID-19. Participants who reported experiencing food insecurity (vs food security) were almost 1.5 times more likely to have a suspected or confirmed infection, and those experiencing homelessness (vs not) were 1.7 times more likely to have confirmed infection. These associations may be explained by an inability to socially distance when in need of shelter and by prioritization of work for basic needs and survival. Such associations are not unique to TGD people; however, both food insecurity and homelessness have been historically disproportionately higher for TGD people in the United States, particularly for TGD people of color, and are likely exacerbated by the pandemic.25,26 Race and ethnicity were not associated with suspected or confirmed SARS-CoV-2 infection in our study; however, given described disparities in food insecurity, the effect may have been attenuated by the inclusion of food insecurity in the model. Ultimately, 60% of participants reported that they and/or someone in their household lost employment income during the pandemic, suggesting that preexisting disparities presumably worsened during the pandemic and had a compounding effect on exposure and access to testing for COVID-19.
Suspected infections may also reflect factors that prevented TGD people from testing even if symptomatic and/or known to have an exposure. Chronic physical and mental health conditions reported by participants were associated with 1.4-fold and 3.7-fold increases in suspected SARS-CoV-2 infection, respectively; neither was associated with confirmed infection. While chronic health conditions are associated with severe COVID-19 disease, associations between chronic physical and mental health conditions and infection may be attributed to exposure from more time spent in medical facilities. These associations with suspected infection may also reflect restricted mobility that prevented access to testing locations when needed and, thus, prevented confirmation of infection. Almost half of participants reported a chronic physical health condition, and more than 80% reported a chronic mental or behavioral health condition, highlighting the importance of strategies to ensure access to testing and care for people with chronic illness. Provision of low-barrier access to testing through multiple channels, including mail-in, self-administered, and peer-based approaches, as a complement to traditional testing facilities may also address structural barriers related to consistent telephone access and transportation.
Intersectional anticipated discrimination was also associated with suspected infection, wherein each unit increase of the discrimination score was associated with a 3% increased likelihood of suspected infection. This finding suggests that participants with suspected infection may have avoided seeking testing due to concerns about discrimination at testing facilities. Fear of mistreatment and misgendering as a barrier to testing was almost as common as other well-known barriers associated with testing availability in the United States, such as low availability of locations and restriction of testing to symptomatic patients. Intersectional anticipated and experienced discrimination may also partially explain why Hispanic/Latinx participants were more likely than non-Hispanic participants to report an inability to access testing. 27 These findings are echoed by recent qualitative research that described historical and ongoing medical trauma, including misgendering, and perceived emotional violence as significant barriers to COVID-19 vaccination. 28 Historical health care discrimination of TGD people continues today and has been highly publicized through a 2020 decision by the US Department of Health and Human Services to exclude gender identity from prohibitions against sex discrimination in health care as well as ongoing court cases debating support for gender-affirming care for transgender young people.29,30 As a result, TGD people may face discrimination or delay seeking testing for COVID-19, particularly when asymptomatic or experiencing mild symptoms.
Confirmed SARS-COV-2 infection was also higher for nonbinary relative to trans masculine participants, and we found no difference in suspected infection by gender identity, despite participants who were nonbinary assigned female sex at birth reporting more barriers to testing than other TGD participants. Nonbinary participants were younger than the other gender identity groups in our sample; thus, they may have been in settings for young people, such as college dormitories, where social distancing and other preventive strategies were difficult to implement but where regular testing was often required. This may explain the higher proportion of confirmed cases among these participants. 31 Overall, our study is consistent with a prior global study of 536 TGD adults wherein 8% self-reported a positive COVID-19 test result, of whom 69% were nonbinary and 75% were aged 18-29 years. 32 Our findings underscore the importance of gender identity data collection, including nonbinary gender identity, in understanding and monitoring SARS-COV-2 infection. 5
Limitations and Strengths
This study had several strengths and limitations. First, the online survey allowed us to reach individuals in areas outside of common research catchment areas; however, we may not have reached people with limited technological access despite substantial efforts to ensure inclusion. Forty percent of participants identified as Black, Indigenous, or people of color, although the study sample may insufficiently represent the experiences of diverse racial and ethnic groups. Antibody testing was optional in an effort to build trust with participants; however, the participation rate was low, which may have been due to a lack of perceived need. As a cross-sectional study, we were unable to establish nor assess trends over time in the sample. Another strength of this study was that it had a large, diverse sample of TGD adults in the United States that permitted characterization and in-depth exploration of factors associated with SARS-CoV-2 infection and testing experiences for people often excluded from national estimates. Furthermore, we were able to evaluate SARS-CoV-2 seroprevalence via self-collected biospecimens from participants, providing information on acceptability of this method and suggested strategies to support equitable testing programs.
Conclusion
Our findings highlight the importance of collecting and reporting gender identity data in public health surveillance for SARS-CoV-2 infection. The history of SARS-CoV-2 infection among TGD people in the United States was similar to or potentially higher than estimates for the US adult population. Common testing challenges were clear barriers for many but were exacerbated by anticipated discrimination in health care. Disproportionately high levels of housing and food insecurity and mental health conditions that were associated with COVID-19 and testing access underscore the need for inclusive and diverse pathways to access COVID-19 testing, public health services, and support that mitigate the effects of the social determinants of health.
Supplemental Material
sj-docx-1-phr-10.1177_00333549221138853 – Supplemental material for SARS-CoV-2 Infection and Testing Experiences in a Nationwide Sample of Transgender and Gender-Diverse Adults, June–December 2021
Supplemental material, sj-docx-1-phr-10.1177_00333549221138853 for SARS-CoV-2 Infection and Testing Experiences in a Nationwide Sample of Transgender and Gender-Diverse Adults, June–December 2021 by Andrea L. Wirtz, Dee Adams, Tonia C. Poteat, S. Wilson Beckham, Marissa Miller, Carter Brown and Sari L. Reisner in Public Health Reports
Footnotes
Acknowledgements
The authors thank the participants of this study, without whose involvement and shared experienced this study would not be possible. We are grateful for the contributions of James Case, MBI; Nicole Tayrien, MSN, RN; Theo Schall, MSW, MBE; Javier Perez de Acha Chavez, MD, MPH; and Arianna’s Center. We acknowledge the support of the LITE Study co-investigators and the LITE Community Advisory Board.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Poteat serves as a research consultant for studies conducted by ViiV Healthcare and Merck & Co, Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by the National Institute of Allergy and Infectious Diseases, the National Institute of Mental Health, and the National Institute of Child Health and Human Development of the National Institutes of Health through a ![]() from the Rapid Acceleration of Diagnostics–Underserved Populations (RADx-UP) initiative under award no. UH3AI133669 (ALW, SLR). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The LITE Connect study is also appreciative of support from the Centers for AIDS Research at Johns Hopkins University (P30AI094189), Harvard University (P30AI060354), and the University of North Carolina Chapel Hill (P30 AI050410).
from the Rapid Acceleration of Diagnostics–Underserved Populations (RADx-UP) initiative under award no. UH3AI133669 (ALW, SLR). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The LITE Connect study is also appreciative of support from the Centers for AIDS Research at Johns Hopkins University (P30AI094189), Harvard University (P30AI060354), and the University of North Carolina Chapel Hill (P30 AI050410).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
