Abstract
Objectives
Widespread global transmission of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus causing coronavirus disease 2019 (COVID-19), continues. Many questions remain about asymptomatic or atypical infections and transmission dynamics. We used comprehensive contact tracing of the first 2 confirmed patients in Illinois with COVID-19 and serologic SARS-CoV-2 antibody testing to determine whether contacts had evidence of undetected COVID-19.
Methods
Contacts were eligible for serologic follow-up if previously tested for COVID-19 during an initial investigation or had greater-risk exposures. Contacts completed a standardized questionnaire during the initial investigation. We classified exposure risk as high, medium, or low based on interactions with 2 index patients and use of personal protective equipment (PPE). Serologic testing used a SARS-CoV-2 spike enzyme-linked immunosorbent assay on serum specimens collected from participants approximately 6 weeks after initial exposure to either index patient. The 2 index patients provided serum specimens throughout their illness. We collected data on demographic, exposure, and epidemiologic characteristics.
Results
Of 347 contacts, 110 were eligible for serologic follow-up; 59 (17% of all contacts) enrolled. Of these, 53 (90%) were health care personnel and 6 (10%) were community contacts. Seventeen (29%) reported high-risk exposures, 15 (25%) medium-risk, and 27 (46%) low-risk. No participant had evidence of SARS-CoV-2 antibodies. The 2 index patients had antibodies detected at dilutions >1:6400 within 4 weeks after symptom onset.
Conclusions
In serologic follow-up of the first 2 known patients in Illinois with COVID-19, we found no secondary transmission among tested contacts. Lack of seroconversion among these contacts adds to our understanding of conditions (ie, use of PPE) under which SARS-CoV-2 infections might not result in transmission and demonstrates that SARS-CoV-2 antibody testing is a useful tool to verify epidemiologic findings.
Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has become a global pandemic. 1 Transmission of SARS-CoV-2 has been documented in households, health care facilities, and community settings.2-4 Symptomatic infections as well as infections lacking typical symptoms (fever, cough, and difficulty breathing) are known to occur.3-5 However, questions remain about the proportion of infected people who lack typical symptoms or remain asymptomatic after exposure.
During January and February 2020, an extensive investigation of the first detected case of COVID-19 in Illinois identified secondary transmission between the first patient (Patient 1) and a single household contact (Patient 2). 6 Contacts of Patients 1 and 2 were actively monitored for symptoms (ie, fever, cough, or difficulty breathing) 7 for 14 days after their last interaction with these patients. Contacts who became symptomatic were tested for SARS-CoV-2 infection using real-time reverse transcription polymerase chain reaction (rRT-PCR) testing of nasopharyngeal specimens. In addition, a convenience sample of health care personnel contacts without symptoms and with varying types of interactions with Patient 1 or 2 were also screened by using rRT-PCR 7 to 14 days after exposure. 6 All contacts tested had negative test results, and no untested contacts developed fever, cough, or difficulty breathing during their monitoring period. This initial investigation might have missed infections in asymptomatic contacts or contacts lacking classic symptoms, because not all contacts were tested. Infections that were in the incubation period or developed after a single rRT-PCR test would have also been missed. Therefore, to determine whether contacts had unidentified COVID-19, we used SARS-CoV-2 antibody testing as part of serologic follow-up investigation of contacts who had rRT-PCR testing performed and of contacts not tested but who had greater-risk exposures to the 2 index patients.
Methods
The Centers for Disease Control and Prevention (CDC) and the Illinois Department of Public Health reviewed the protocol for this follow-up investigation and determined it to be nonresearch.
Study Population
An extensive investigation of Patients 1 and 2 and their contacts is documented elsewhere. 6 That investigation included 372 named contacts, and 347 (93%) had active symptom monitoring for 14 days. We invited contacts identified in the initial investigation to participate in serologic follow-up approximately 6 weeks after their exposure event (March 3-6, 2020) if (1) they were tested for COVID-19 by SARS-CoV-2–specific rRT-PCR of a nasopharyngeal specimen or (2) they were not previously tested but had a greater-risk exposure to the 2 patients. We defined “greater-risk” exposure according to a framework used in the initial investigation (Table 1) as a high-risk exposure (eg, health care personnel performing an aerosol-generating procedure without a respirator or having close contact with an unmasked patient with COVID-19 without a face mask or respirator) or a medium–high-risk exposure (eg, health care personnel or community contact with >15 minutes of exposure without appropriate personal protective equipment [PPE]). 6 Although the initial investigation named 372 contacts of the 2 index patients, we were unable to perform serologic testing for all because of limited logistical capacity. To increase the likelihood of identifying undetected COVID-19, we selected a study population that consisted of all contacts who developed fever, cough, or difficulty breathing while being monitored, as well as contacts who had greater-risk exposures. Community transmission had not been documented in Illinois at the time of initial investigation or serologic follow-up, and this approach assumed that no additional exposures to SARS-CoV-2 existed other than the contacts’ known exposure to the 2 index patients. We invited contacts to participate by email, with at least 1 follow-up telephone call for those who did not respond.
Risk classification of community contacts and health care personnel with potential exposure to COVID-19, Illinois, 2020 a
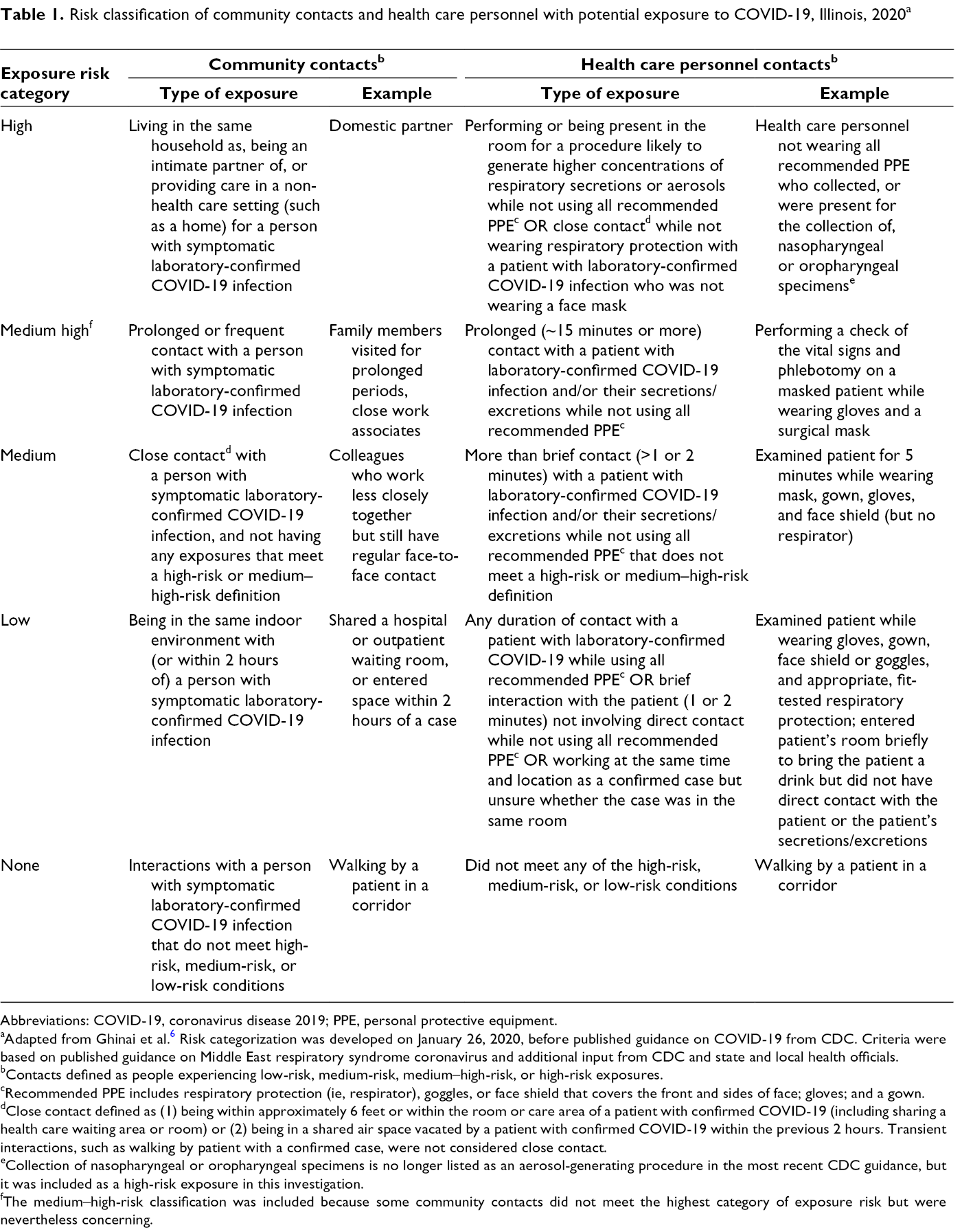
Abbreviations: COVID-19, coronavirus disease 2019; PPE, personal protective equipment.
aAdapted from Ghinai et al. 6 Risk categorization was developed on January 26, 2020, before published guidance on COVID-19 from CDC. Criteria were based on published guidance on Middle East respiratory syndrome coronavirus and additional input from CDC and state and local health officials.
bContacts defined as people experiencing low-risk, medium-risk, medium–high-risk, or high-risk exposures.
cRecommended PPE includes respiratory protection (ie, respirator), goggles, or face shield that covers the front and sides of face; gloves; and a gown.
dClose contact defined as (1) being within approximately 6 feet or within the room or care area of a patient with confirmed COVID-19 (including sharing a health care waiting area or room) or (2) being in a shared air space vacated by a patient with confirmed COVID-19 within the previous 2 hours. Transient interactions, such as walking by patient with a confirmed case, were not considered close contact.
eCollection of nasopharyngeal or oropharyngeal specimens is no longer listed as an aerosol-generating procedure in the most recent CDC guidance, but it was included as a high-risk exposure in this investigation.
fThe medium–high-risk classification was included because some community contacts did not meet the highest category of exposure risk but were nevertheless concerning.
Data Collection and Exposure Risk Assessment
In the initial investigation, all contacts completed a standardized questionnaire asking for information on type and dates of contact with the 2 index patients, the use of PPE and other health care infection control practices (where applicable), and demographic characteristics. Contacts provided daily logs on symptoms and temperature to public health authorities during active monitoring until 14 days after their last exposure to the 2 index patients. The initial investigation classified exposure risk for contacts on January 26, 2020, before release of CDC’s initial guidance on exposure risk classification on February 3, 2020. 8 For the serologic follow-up investigation, we reclassified health care personnel contacts as high-risk (eg, interaction with an unmasked patient while not wearing an N95 respirator, other respirator, or surgical mask), medium-risk (eg, interaction with a masked patient while not wearing an N95 respirator, other respirator, or surgical mask), or low-risk (eg, wearing all recommended PPE) exposure based on CDC guidance as of March 7, 2020, which was released before widespread community transmission of COVID-19 (Table 2). 9 We also reclassified community contacts as high-risk (eg, household contacts), medium-risk (eg, close contact but not household member), or low-risk (eg, being in the same indoor environment but without close contact) based on CDC guidance as of March 7, 2020 (Table 3). 10 For contacts with multiple interactions with Patient 1 or 2, we used the greatest-risk exposure for analyses. At enrollment in serologic follow-up, participants reported medical comorbidities from a list provided: chronic lung disease or asthma, hypertension, diabetes, obesity (body mass index >30 kg/m2), neurologic conditions that impair cough, weakened immune system, chemotherapy or radiation for cancer in the previous 12 months, sickle cell anemia, congestive heart failure, coronary artery disease, end-stage renal disease requiring dialysis, cirrhosis of the liver, or asplenia.
Epidemiologic risk classification of health care personnel after exposure to patients with COVID-19 or their secretions/excretions in a health care setting a

Abbreviations: COVID-19, coronavirus disease 2019; PPE, personal protective equipment.
aAdapted from interim guidance of Centers for Disease Control and Prevention on exposures among health care personnel, published March 7, 2020. 9
bData are insufficient to precisely define the duration of time that constitutes a prolonged exposure. However, until more is known about transmission risks, it is reasonable to consider an exposure greater than a few minutes as a prolonged exposure. 9
cThe risk category for these rows would be elevated by 1 level if health care personnel had extensive body contact with a patient (eg, rolling a patient). 9
dThe risk category for these rows would be elevated by 1 level if health care personnel performed or were present for a procedure likely to generate higher concentrations of respiratory secretions or aerosols (eg, cardiopulmonary resuscitation, intubation, extubation, bronchoscopy, nebulizer therapy, sputum induction). For example, health care personnel who were wearing a gown, gloves, eye protection, and a face mask (instead of a respirator) during an aerosol-generating procedure would be considered to have a medium-risk exposure. 9
Epidemiologic risk classification of community contacts identified during contact investigations of patients with laboratory-confirmed COVID-19 a

Abbreviation: COVID-19, coronavirus disease 2019.
aAdapted from interim guidance of Centers for Disease Control and Prevention on exposures among community contacts, published March 7, 2020. 10
bClose contact is defined as being within approximately 6 feet (2 meters) of a patient with COVID-19 for a prolonged period of time; close contact can occur while caring for, living with, visiting, or sharing a health care waiting area or room with a patient with COVID-19. Close contact also includes having direct contact with infectious secretions of a patient with COVID-19 (eg, being coughed on). 10
cData are insufficient to precisely define the duration of time that constitutes a prolonged exposure. However, until more is known about transmission risks, it is reasonable to consider an exposure greater than a few minutes as a prolonged exposure. 10
Serum Collection and Testing
All contacts previously tested for SARS-CoV-2 with rRT-PCR of a nasopharyngeal specimen during the initial investigation also provided baseline serum specimens; therefore, paired serum specimens were available for a subset of participants. We collected follow-up serum from contacts approximately 6 weeks after their first exposure to Patient 1 or 2 to allow sufficient time for antibody development; we selected this period based on information on development and duration of SARS-CoV antibodies. 11 We collected 5-10 mL of whole venous blood from participants in serum-separator tubes. After centrifugation, we sent serum on ice packs to CDC for testing. The 2 index patients provided serum specimens throughout their illness.
CDC conducted serologic testing using a protein-based enzyme-linked immunosorbent assay (ELISA) newly developed to detect antibodies against SARS-CoV-2 spike protein in serum, with validation studies indicating a specificity of >99% and sensitivity of 96%. 12 ELISA plates were incubated with 4-fold dilutions of each serum sample from 1:100 to 1:6400. Antigen–antibody complexes were detected with goat antihuman immunoglobulin G, immunoglobulin M, immunoglobulin A, horseradish peroxidase-conjugated secondary antibody, and ABTS peroxidase substrate (LGC SeraCare). Optical density values were calculated at 405 nm with a 490 nm reference filter. An optimal cutoff optical value was set at 0.4, and specimens with titers ≥1:400 were considered seropositive.
Statistical Analysis
The primary outcome of this study was seropositivity, that is, detection of SARS-CoV-2 antibodies in follow-up serum specimens among all contacts and among contacts stratified by exposure risk category. To assess for nonresponse bias, we calculated descriptive statistics and corresponding P values for each variable to evaluate its association with the likelihood of enrollment among contacts who provided a follow-up serum specimen (participants) and contacts who declined to provide a follow-up serum specimen or were lost to follow-up (nonparticipants). We calculated medians for continuous variables and proportions for categorical variables. We used the Wilcoxon rank-sum test to compare continuous variables and the χ2 test of independence, Fisher exact test, or Cochran–Armitage test to compare categorical variables. We considered 2-sided statistical tests to be significant at P < .05. We used SAS version 9.4 for all analyses (SAS Institute Inc).
Results
Of 347 contacts who completed active monitoring in the initial investigation, 110 were eligible to participate in the follow-up serologic study; 75 (68%) provided baseline serum specimens during the initial investigation, and 35 (32%) did not provide baseline serum specimens but had greater-risk exposures. Of these 110, 59 (54% of eligible; 17% of all contacts) enrolled and provided follow-up serum specimens, 9 (8%) declined follow-up serum collection, and 42 (38%) were lost to follow-up (Figure). Of 59 participants, 41 (69%) had previous rRT-PCR testing and baseline serum specimens and 18 (31%) did not have rRT-PCR testing or baseline serum specimens but had greater-risk exposures. Participants were more likely than nonparticipants to be older (median age 39 vs 32; P < .001) (Table 4). Participants did not differ significantly from nonparticipants on any other characteristic. In addition, symptomatic participants were significantly more likely than nonparticipants to be older (median age 38 vs 32; P = .01) and to be health care personnel (56% vs 44%; P = .01).
Baseline demographic characteristics, exposure characteristics, and symptom development of contacts invited to participate in a serologic follow-up investigation after exposure to the first 2 confirmed patients with COVID-19 in Illinois, by enrollment status, 2020
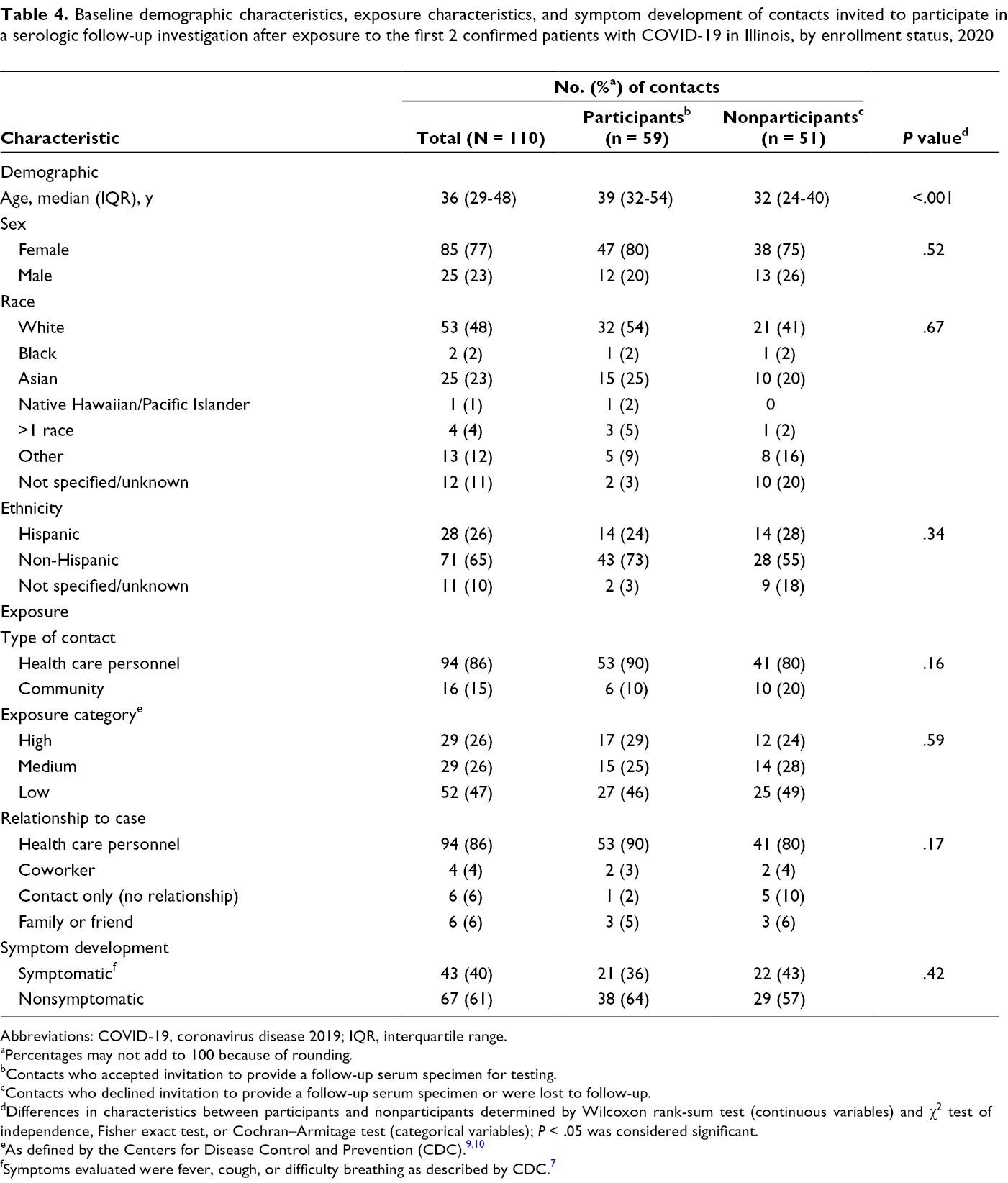
Abbreviations: COVID-19, coronavirus disease 2019; IQR, interquartile range.
aPercentages may not add to 100 because of rounding.
bContacts who accepted invitation to provide a follow-up serum specimen for testing.
cContacts who declined invitation to provide a follow-up serum specimen or were lost to follow-up.
dDifferences in characteristics between participants and nonparticipants determined by Wilcoxon rank-sum test (continuous variables) and χ2 test of independence, Fisher exact test, or Cochran–Armitage test (categorical variables); P < .05 was considered significant.
fSymptoms evaluated were fever, cough, or difficulty breathing as described by CDC. 7
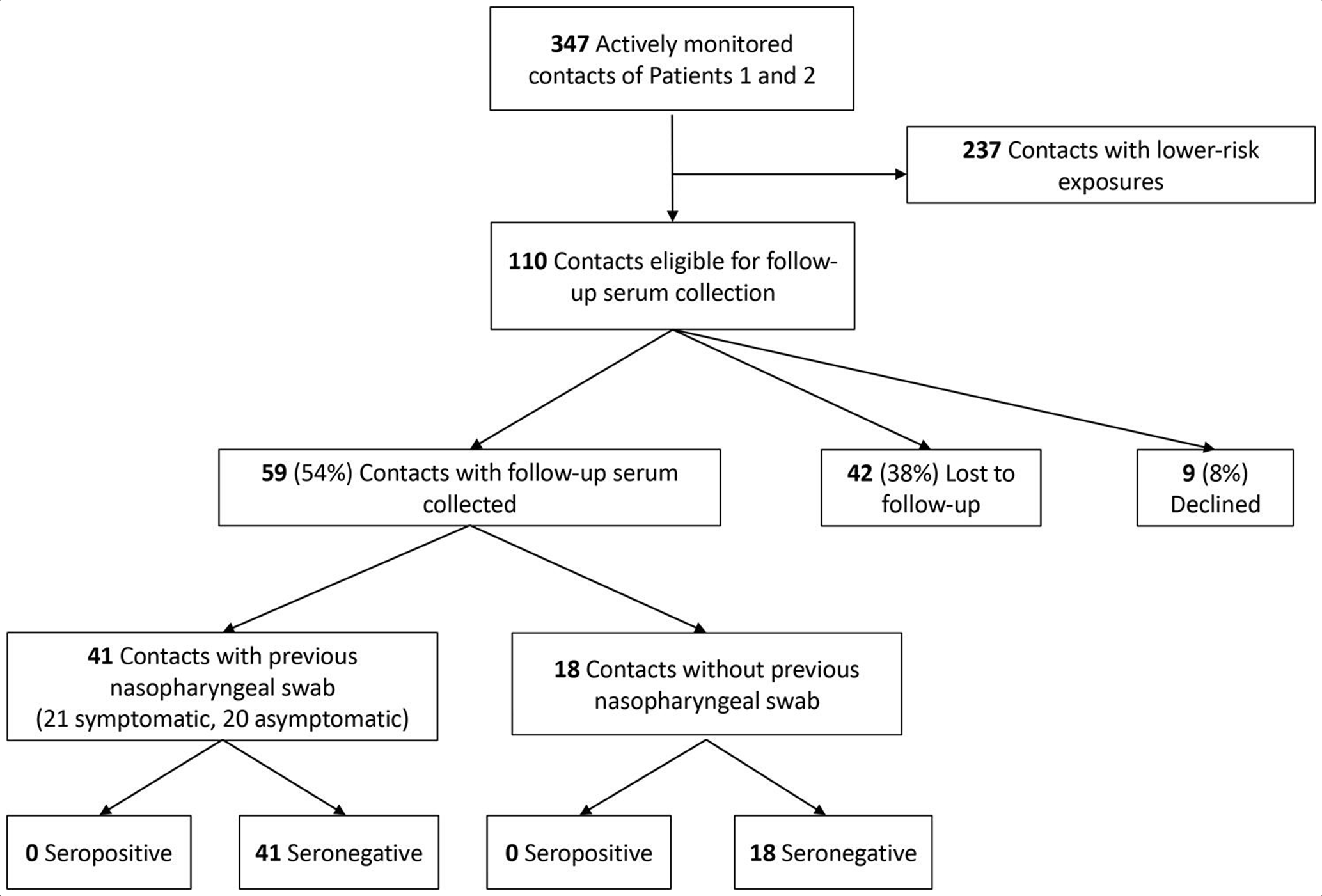
Flow of contacts in a serologic follow-up investigation among people exposed to the first 2 confirmed patients (Patients 1 and 2) with coronavirus disease 2019 (COVID-19) in Illinois, 2020. “Lower-risk” refers to medium or low risk as opposed to high risk or medium–high risk, according to framework developed by the authors in an initial investigation. 6 Symptoms evaluated included fever, cough, or difficulty breathing as described by the Centers for Disease Control and Prevention on January 17, 2020. 7
Of 59 participants, none demonstrated SARS-CoV-2 antibodies by ELISA, with all exhibiting titers of <1:100, including all participants with both baseline and follow-up serologic specimens (n = 41). Fifty-three (90%) were health care personnel and 6 (10%) were non-health care personnel. Median time from both first exposure and last exposure to the 2 index patients and the collection of follow-up serum was the same, at 43 days (range, 37-49 days and 34-49 days, respectively). Seventeen (29%) contacts had high-risk exposures to the 2 index patients, 15 (25%) had medium-risk, and 27 (46%) had low-risk.
All 17 contacts with high-risk exposures were health care personnel and involved interaction with an unmasked patient for >5 minutes while not wearing an N95 respirator, other respirator, or surgical mask. Among 15 contacts with medium-risk exposures, 5 were health care personnel who interacted with a masked patient while not wearing an N95 respirator, other respirator, or surgical mask; 5 were health care personnel who interacted with an unmasked patient while wearing a surgical mask but not wearing protective eyewear; and 5 were community contacts (family members or coworkers not wearing face masks while interacting with patient for >30 minutes). No aerosol-generating procedures were performed on patients without appropriate PPE (gown, gloves, eye protection, and respirators). Sixteen of 59 (27%) participants reported ≥1 medical comorbidity; 6 (10%) reported chronic lung disease or asthma, 6 (10%) reported hypertension, 5 (9%) reported diabetes, 5 (9%) reported obesity, and 1 (2%) had a neurological condition that impaired a cough.
In comparison, although neither of the 2 index patients had antibodies against SARS-CoV-2 S protein (titers <1:100) at day 7 after symptom onset, Patient 1 demonstrated a titer >1:6400 on day 23 after symptom onset and Patient 2 had a titer of >1:6400 on day 11 after symptom onset.
Discussion
This article describes the serologic follow-up of the first 2 confirmed cases of COVID-19 in Illinois and a subset of their health care personnel and community contacts. Both Patients 1 and 2 demonstrated strong SARS-CoV-2–specific antibody responses fewer than 4 weeks after symptom onset. However, none of the contacts had evidence of antibodies specific to SARS-CoV-2 approximately 6 weeks after these exposures. These data support the findings of the initial epidemiologic investigation, which suggested that, among contacts of the 2 index patients, no further transmission of SARS-CoV-2 occurred. 6 In contrast to the findings of our study, transmission to health care personnel and community contacts has been well-documented elsewhere, even with limited direct contact. 3
Multiple factors likely contribute to SARS-CoV-2 transmission, including factors related to the infected patient’s severity of disease and overall viral shedding. Currently, it is unclear if illness severity is associated with infectivity, as seen in other viral diseases.13,14 Both index patients required hospitalization for clinical reasons but neither required intensive care, indicating they did not experience the most severe spectrum of disease. 15 In addition, without procedures such as intubation, the number of aerosol-generating procedures would have been reduced, therefore decreasing the amount of viral particles in the environment. Patient 1 had SARS-CoV-2 rRT-PCR cycle threshold (Ct) values as low as 23-25 (lower values indicate a higher viral load) on lower respiratory tract specimens on day 9 after symptom onset, whereas Patient 2 had lowest Ct values of 30-31 on lower respiratory specimens on day 16 after symptom onset. 6 This difference in Ct values might indicate that Patient 1 was more infectious than Patient 2; however, interpretation of infectiousness based on Ct values is limited, as neither patient had specimens collected during their first week of illness and no viral cultures were performed to assess virus viability.
In addition, transmission events likely depend on many environmental and patient-related factors, as has been demonstrated with similar coronaviruses, 16 and these factors have not yet been well-defined for SARS-CoV-2. For example, after diagnosis of COVID-19, both index patients were hospitalized and treated in airborne infection isolation rooms, and health care personnel wore N95 or other respirators even when not conducting aerosol-generating procedures. Before diagnosis of COVID-19, most health care personnel used surgical face masks, gowns, and gloves, consistent with CDC guidance published on April 13, 2020, which indicated the use of a face mask if an N95 or other respirator is not available. 17 Although some health care personnel reported high-risk exposures, none occurred while performing an aerosol-generating procedure. Exposures described in this study mostly occurred in a health care environment and differ from exposures in reports describing transmission at close-contact community events 18 and reports of health care personnel becoming infected after performing high-risk procedures without appropriate PPE. 19 As such, the use of recommended PPE and other environmental controls (eg, airborne infection isolation rooms) likely contributed to the lack of transmission in this investigation.
Limitations
Our study had several limitations. First, the initial investigation identified more than 300 health care and community contacts of the 2 index patients, 6 but we were unable to perform serologic testing for all contacts. Contacts eligible for this study included all actively monitored contacts with initial testing for COVID-19 and contacts with greater-risk exposures (mainly health care personnel); we selected this population to maximize the opportunity to identify undetected infections. It is possible that asymptomatic transmission or infections lacking fever, cough, or difficulty breathing were undetected in contacts who were not included. Second, health care personnel and community contacts might not have accurately reported all details of exposure history. However, direct observation of PPE and infection control practices was implemented at the time of COVID-19 diagnosis in Patient 1, allowing us to confirm accuracy of self-report for health care personnel. Third, because many contacts had repeated exposures to Patient 1 or 2, and because the date of symptom onset for Patient 2 was unclear, 6 we were unable to assess the temporal relationship between symptom onset and contact exposure, which has been proposed as a transmission risk factor. 20 Fourth, among contacts who were eligible for the follow-up study, only a subset participated; this subset was not meaningfully different from the subset that did not participate in demographic characteristics, exposure risk, or relationship to patients. We were unable to obtain complete comorbidity data for those who did not participate and, therefore, could not determine differences in comorbidities between the 2 subsets. Fifth, the serologic techniques used were novel and may be refined in the future. However, validation of this serologic assay indicates excellent specificity and sensitivity. 12 Finally, most contacts in this investigation were health care personnel. Although we included other nonhousehold community contacts, they do not represent the diversity of all types of community exposure, such as exposures occurring on airplanes or cruise ships.
Conclusions
This serologic investigation supports the findings, provided by comprehensive contact tracing and rRT-PCR testing, of a lack of transmission beyond a single household contact of Patient 1. To our knowledge, this study also presents some of the earliest data related to SARS-CoV-2 antibody response in patients with COVID-19 in the United States and demonstrates the potential to use antibody testing in epidemiologic investigations in combination with comprehensive contact tracing and rRT-PCR testing. Although this study provides details on a single investigation and must be interpreted in the context of widespread transmission of SARS-CoV-2, it adds to our understanding of conditions under which SARS-CoV-2 infections might not result in transmission, specifically related to PPE and infection control practices. Continued research on transmission dynamics will help inform containment efforts.
Footnotes
Acknowledgments
The authors thank all participants for their time and interest in this investigation; Barney Graham, MD, PhD, and Kizzmekia Corbett, PhD, for the SARS-CoV-2 expression plasmid for assay use; Allison Arwady, MD, Erin Conners, PhD, and Michael Hernandez, MD, for logistical support; and Stefany Moye, BS, for laboratory support. We also acknowledge numerous associates and staff members at AMITA Health St. Alexius Medical Center, Cook County Department of Public Health, Chicago Department of Public Health, DuPage County Health Department, Illinois Department of Public Health, and members of the Emergency Operations Center and Division of Viral Diseases in the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention.
Author’s Note
The findings and conclusions in this article are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention. Company and product names are used for identification purposes only and do not imply endorsement.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Centers for Disease Control and Prevention.
