Abstract
Objectives
In 2012, onetime hepatitis C virus (HCV) screening was recommended for all baby boomers (people born during 1945-1965) in the United States, but only 4.0%-12.9% of baby boomers have ever had a screening ordered by a health care provider. This study examined the HCV screening prevalence among adult patients in a large academic health care system and assessed factors associated with the completion of screening when ordered for baby boomers.
Methods
We defined HCV screening completion as the completion of an HCV antibody test when it was ordered. We used electronic health records to examine HCV screening completion rates among adults (N = 106 630) from August 1, 2015, through July 31, 2020, by birth cohort. Among baby boomers whose health care provider ordered HCV screening, we examined frequency and percentages of HCV screening completion by sociodemographic and clinical characteristics. We conducted univariate and multivariable logistic regression analyses to assess factors associated with HCV screening completion among baby boomers.
Results
During the study period, 73.0% of baby boomers completed HCV screening when it was ordered. HCV completion did not differ by sex or race and ethnicity among baby boomers. Baby boomers with Medicare supplemental health insurance compared with commercial health insurance (adjusted odds ratio [aOR] = 1.87) and those seeing only advanced practice professionals compared with specialty care physicians (aOR = 2.24) were more likely to complete HCV screening when it was ordered.
Conclusions
Noncompletion of HCV screening is one of many barriers along the HCV treatment continuum. Our findings suggest a need for interventions targeting systems, health care providers, and patients to increase HCV screening rates in the United States.
Approximately half of patients infected with hepatitis C virus (HCV) develop chronic HCV infection. 1 Untreated chronic HCV infection is a main contributor to the development of liver cancer 2,3 and the need for liver transplantation. 1,4 The number of people infected with HCV in the United States is estimated to be at least 4.6 million. 5 An estimated 75% of people with HCV infection are baby boomers (people born from 1945 through 1965). 6 Given the increased risk to baby boomers for HCV infection, the Centers for Disease Control and Prevention (CDC) and the US Preventive Services Task Force (USPSTF) in 2012 and 2013 augmented risk-based HCV screening recommendations to include onetime HCV screening for all baby boomers. 6,7 In 2020, CDC and USPSTF updated their recommendations to include onetime universal screening for all adults. 8,9 One strategy to reverse rising US liver cancer incidence and mortality trends is to promote HCV screening and confirmatory testing and to connect people who have a positive test result to care.
Treatment for HCV infection has improved dramatically in recent years with the advent of direct-acting antiviral agents, which achieve cure rates >95% with minimal side effects. 10 Despite effective screening and treatment methods, HCV screening rates remain low. Studies conducted since the 2012 and 2013 screening recommendations report HCV screening rates of 1.4% to 30%. 11 -13 Furthermore, a recent study indicated that only 50% of people who received a positive result for HCV during antibody testing completed confirmatory RNA testing. 14 Given the availability of curative treatments, it is vital that both HCV antibody test order rates and completion rates are improved so that HCV infection can be detected and treated before the disease progresses.
Research by our study team found that from August 1, 2015, through July 31, 2017, a low rate (12.9%) of baby boomers in a large academic health care system had an HCV test ordered. 15 We did not determine, however, what percentage of these patients followed through in completing HCV antibody testing after their health care provider ordered it, nor did we ascertain what factors were associated with HCV antibody test completion. These data are needed to inform future interventions aimed at promoting HCV screening. The current study examined (1) HCV antibody completion percentages among all adult patients when ordered by a health care provider and (2) factors associated with HCV antibody test completion among baby boomers when ordered by a health care provider. Understanding HCV antibody test completion rates among all patients and factors associated with baby boomers’ completion of ordered tests can inform future intervention strategies.
Methods
A detailed description of study methods, electronic health records (EHRs), and data acquisition is available elsewhere. 15 Briefly, we analyzed EHR data collected from a large academic health care system during a 5-year period, from August 1, 2015, through July 31, 2020. We restricted data to visits with primary care providers (eg, family medicine, general internal medicine) or a relevant specialist (eg, gastroenterologist, infectious disease specialist) and removed any visit after screening was completed from the analyses, so that HCV screening completion would not be explained by a possible referral for a positive test result. The institutional review board at the University of South Florida and the scientific review committee at the Moffitt Cancer Center reviewed the protocol and granted exempt status.
Statistical Analysis
We used clinical data from 1 601 252 unique patients, of whom 304 859 were aged ≥18, that were housed in a centralized data repository and available for health services research. After removing patients who did not meet the study inclusion criteria, the final dataset included 106 630 adult patients aged ≥18, of whom 35 859 were baby boomers who had clinical encounters in the University of South Florida Health System (Supplemental Figure). We examined the EHRs of patients who had an HCV antibody test ordered by a health care professional. The primary determination of this study was whether the patient completed testing when ordered. We included patients in the denominator of the analytic population if the EHR documented an order for HCV screening. If patients had a quantitative or qualitative (eg, “reactive,” “not detected”) HCV result in the EHR, we considered them to have completed HCV screening and included them in the numerator. Demographic variables examined as potential predictors of test completion were sex, race and ethnicity, language, payor (eg, health insurance coverage), type of health care visits (ie, primary care physicians only, specialty care physicians only, primary and specialty care physicians, advanced practice professionals only [ie, advanced registered nurse practitioner and physician assistant]), and HCV risk factors (ie, HIV positive, history of hepatitis, known exposure to HCV, and history of liver cancer). Types of health insurance were categorized as commercial, Medicaid, Medicare, Medicare Supplement, military, and “other” (ie, self-pay patients, behavioral health patients, and those for whom health insurance category could not be determined [eg, health insurance plan provides multiple categories of health insurance]).
To assess HCV antibody completion percentages for all patients, we examined the rates at which HCV antibody tests were ordered and completed (percentage and 95% CIs) stratified by birth cohort (ie, those born before 1945, those born from 1945 through 1965, those born from 1966 through 1985, and those born after 1985). We also examined test completion rates stratified by risk status (ie, average risk and high risk), because recommendations for non–baby boomer patients varied by risk status when we collected these data. We defined high risk as having ≥1 of the following characteristics: HIV positive, history of hepatitis, history of liver cancer, or known exposure to HCV. No information on intravenous drug use was reported in the EHR; thus, we could not classify patients by this risk factor. 1 We considered patients who did not have one of these risk factors reported in the EHR as average risk.
Because the recommendation for universal screening for baby boomers was in place during data collection, we investigated factors associated with completion of an HCV antibody test by baby boomers after it was ordered by a health care professional. We first described HCV completion for the baby boomer sample based on relevant sociodemographic and HCV risk factors. We then examined the association between baby boomers’ HCV screening completion rates and relevant covariates (ie, sociodemographic and HCV risk factors) using bivariate logistic regression, followed by an adjusted multivariable logistic regression analysis to assess independent factors associated with HCV antibody test completion among average-risk baby boomers. Finally, we conducted a post hoc analysis to examine whether HCV antibody test completion was comparable with completion of other routine laboratory tests, including complete blood count and comprehensive metabolic panel among all patients (including high-risk patients) using EHR data during August 1, 2015, through July 31, 2018. We used a significance level of .05. We conducted all statistical analyses using SAS version 9.4 (SAS Institute, Inc).
Results
The total analytic sample consisted of 106 630 unique patients. The average age was 48.1 (SD, 18.0), and most were female (n = 74 253; 69.6%). The largest proportion were baby boomers (n = 35 859; 33.6%) and non-Hispanic White patients (n = 63 641; 59.7%). Only 10.8% (n = 11 505) had ever had an HCV antibody test ordered, and only 8.0% (n = 8479) had completed the HCV antibody test (Table 1).
Sample description of the study population in a large academic health care system using the electronic health record, Tampa, Florida, August 1, 2015, through July 31, 2020 (N = 106 630) a
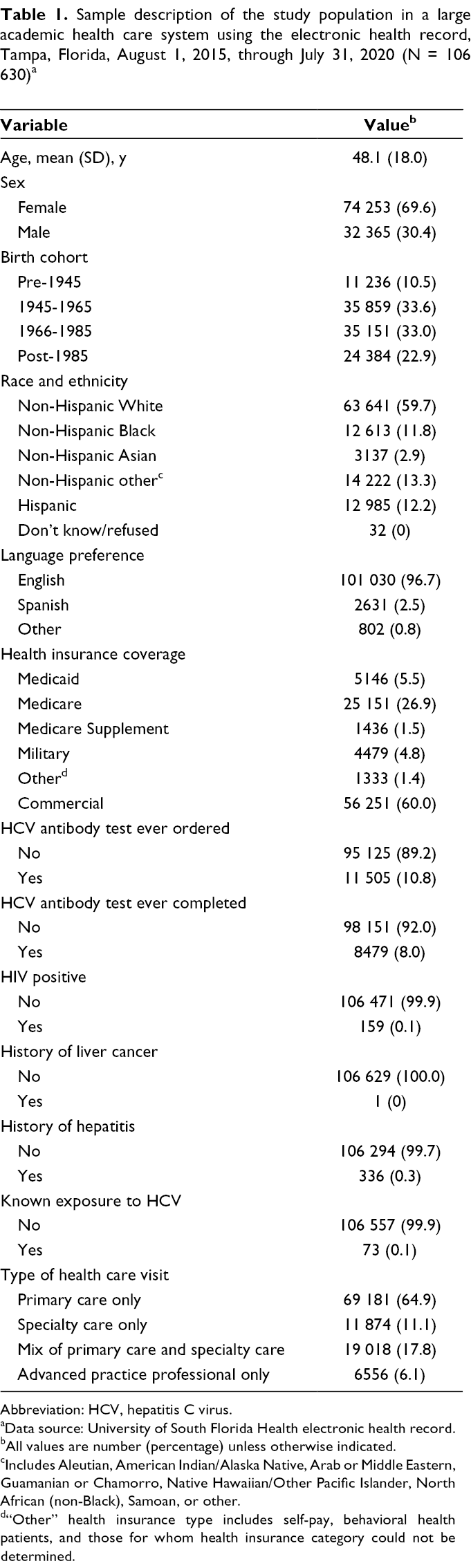
Abbreviation: HCV, hepatitis C virus.
aData source: University of South Florida Health electronic health record.
bAll values are number (percentage) unless otherwise indicated.
cIncludes Aleutian, American Indian/Alaska Native, Arab or Middle Eastern, Guamanian or Chamorro, Native Hawaiian/Other Pacific Islander, North African (non-Black), Samoan, or other.
d“Other” health insurance type includes self-pay, behavioral health patients, and those for whom health insurance category could not be determined.
Among patients for whom the HCV antibody test was ordered, the group with the lowest rates of antibody test completion were average-risk patients born before 1945 (63.5% completion; Table 2). Among average-risk baby boomers, 73.0% completed an HCV antibody test when it was ordered, compared with 74.3% of average-risk patients in the 1966-1985 birth cohort. The highest HCV screening completion rates when HCV antibody testing was ordered (77.8%) were among average-risk patients born after 1985. Within birth cohorts, percentages fluctuated during the study period (Figure), with the greatest fluctuation among patients born before 1945 (53.7% in year 1 and 75.4% in year 3). Patients in the 1966-1985 birth cohort had the least fluctuation (71.5% in years 1 and 2 and 75.8% in year 4).
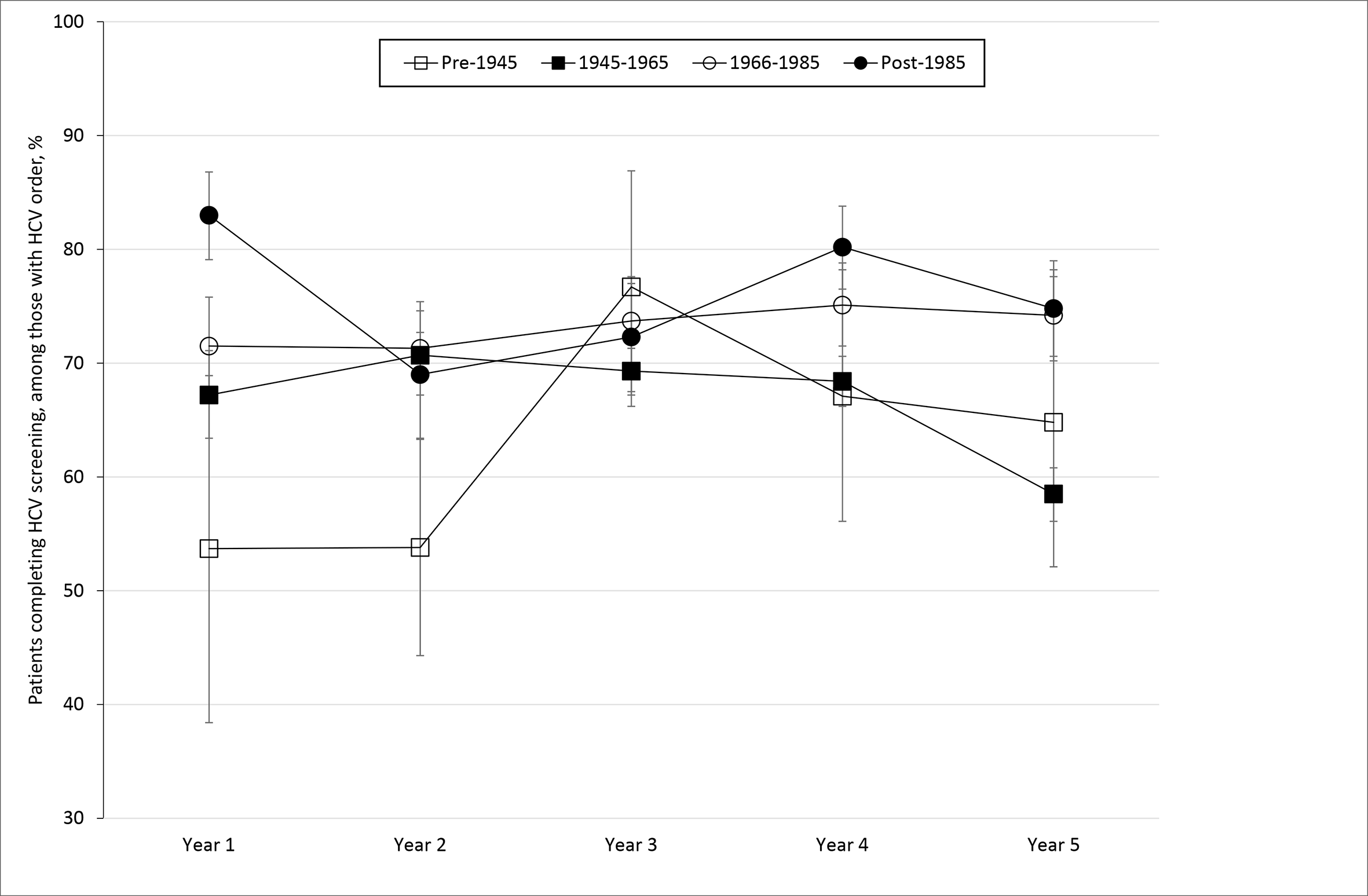
Percentage of patients who completed hepatitis C virus (HCV) antibody testing, by birth cohort and year of study, Tampa, Florida, August 1, 2015, through July 31, 2020. Error bars indicate 95% CIs.
Order and completion rates for HCV antibody test among birth cohorts in a large academic health care system using electronic health records, Tampa, Florida, August 1, 2015, through July 31, 2020 a ,b
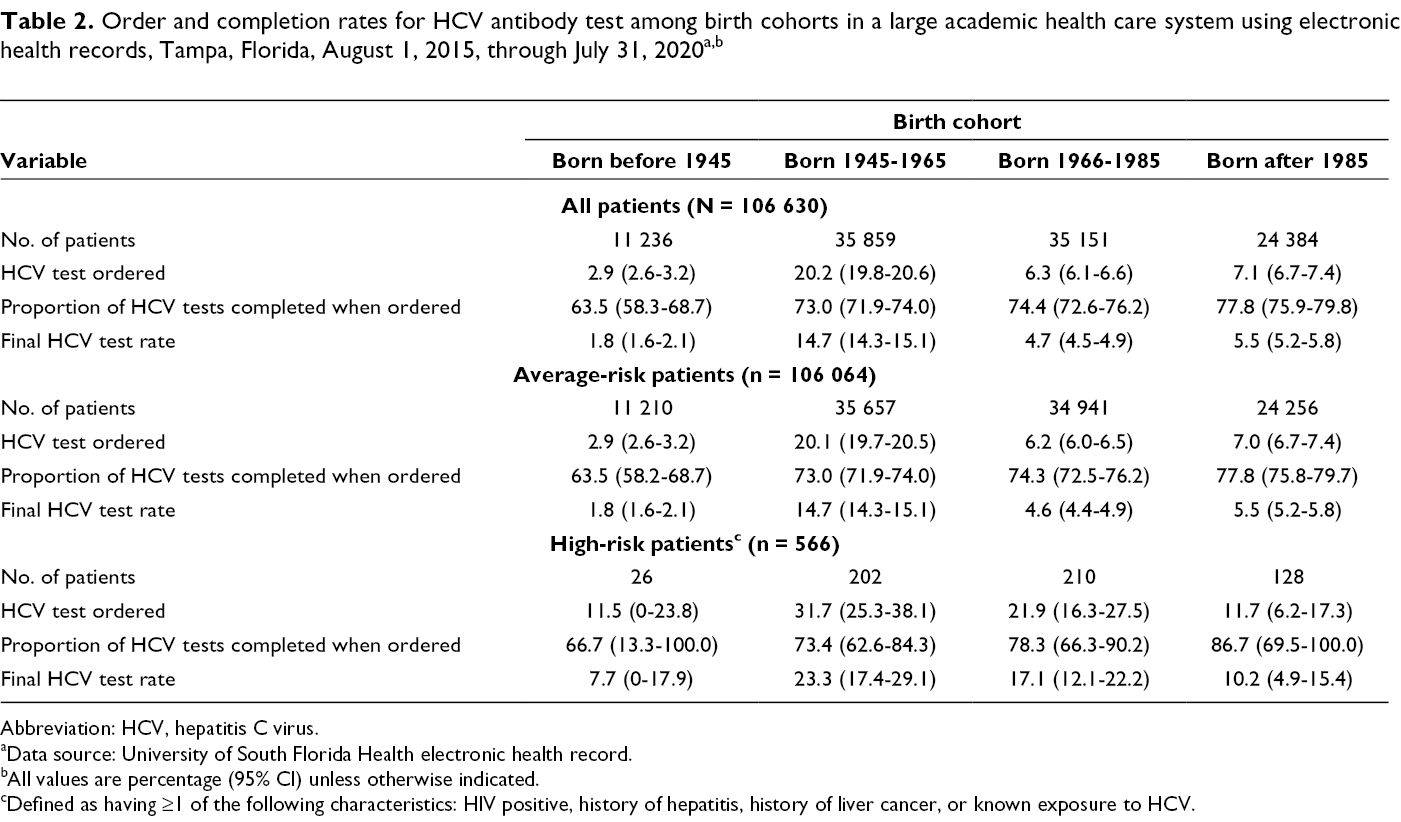
Abbreviation: HCV, hepatitis C virus.
aData source: University of South Florida Health electronic health record.
bAll values are percentage (95% CI) unless otherwise indicated.
cDefined as having ≥1 of the following characteristics: HIV positive, history of hepatitis, history of liver cancer, or known exposure to HCV.
HCV antibody test completion rates for baby boomers varied little by sociodemographic and clinical characteristics (Table 3). Non-Hispanic Black patients tended to have slightly higher completion rates (76.4%) than non-Hispanic White patients did (72.3%;
Prevalence of completion of HCV antibody test, by demographic characteristics, among patients born during 1945-1965, Tampa, Florida, August 1, 2015, through July 31, 2020 a
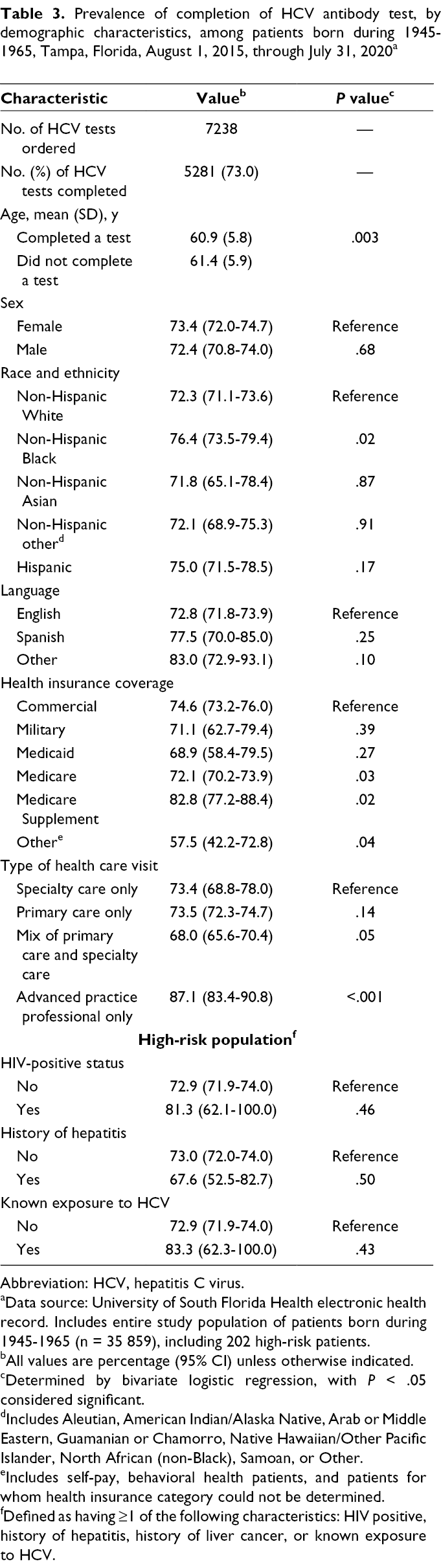
Abbreviation: HCV, hepatitis C virus.
aData source: University of South Florida Health electronic health record. Includes entire study population of patients born during 1945-1965 (n = 35 859), including 202 high-risk patients.
bAll values are percentage (95% CI) unless otherwise indicated.
cDetermined by bivariate logistic regression, with
dIncludes Aleutian, American Indian/Alaska Native, Arab or Middle Eastern, Guamanian or Chamorro, Native Hawaiian/Other Pacific Islander, North African (non-Black), Samoan, or Other.
eIncludes self-pay, behavioral health patients, and patients for whom health insurance category could not be determined.
fDefined as having ≥1 of the following characteristics: HIV positive, history of hepatitis, history of liver cancer, or known exposure to HCV.
Factors associated with completion of HCV antibody test among average-risk a patients born during 1945-1965, Tampa, Florida, August 1, 2015, through July 31, 2020 b
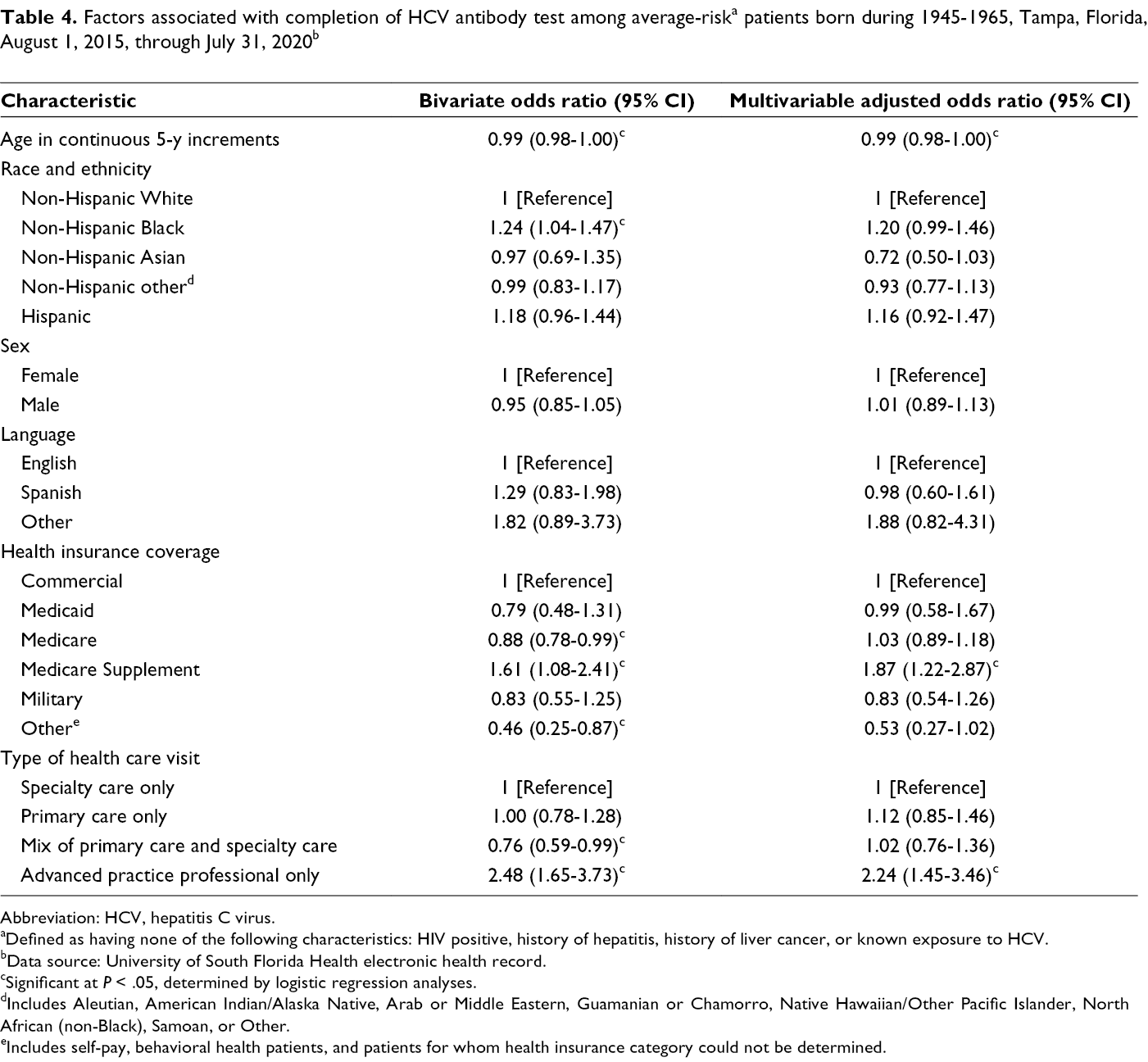
Abbreviation: HCV, hepatitis C virus.
aDefined as having none of the following characteristics: HIV positive, history of hepatitis, history of liver cancer, or known exposure to HCV.
bData source: University of South Florida Health electronic health record.
cSignificant at
dIncludes Aleutian, American Indian/Alaska Native, Arab or Middle Eastern, Guamanian or Chamorro, Native Hawaiian/Other Pacific Islander, North African (non-Black), Samoan, or Other.
eIncludes self-pay, behavioral health patients, and patients for whom health insurance category could not be determined.
HCV test completion was comparable with, if not greater than, completion of the complete blood count (59.5%; 95% CI, 59.1%-59.9%) and comprehensive metabolic panel (59.4%; 95% CI, 59.1%-59.8%) among all patients from August 1, 2015, through July 31, 2018.
Discussion
To our knowledge, our study is the first to examine completion rates of HCV antibody tests among patients who had an HCV antibody test ordered and to assess factors associated with the completion of an HCV antibody test among baby boomers using EHR-verified data and during multiple years. Although overall completion rates of HCV screening when ordered by a health care provider were relatively high (73.0%), about one-quarter (26.3%) of patients, regardless of birth cohort, did not complete an HCV antibody test when it was ordered by a health care provider. Previous research generally focused on HCV screening test order rates and factors associated with a health care provider ordering the test. 15 -18 Those studies found low rates of HCV test ordering by health care providers. Compounding the problem of low HCV test order rates, we found that some patients do not complete the screening test when it is ordered for them, regardless of birth cohort.
Although few previous studies reported HCV test completion rates among average-risk patients in primary care, some research examined completion of HCV screening in other settings. One study prospectively examined all adults accessing outpatient endoscopy from July 1, 2015, to July 1, 2016, at an urban safety-net hospital in California and found a 63.3% (318 of 502) completion rate among patients who had an order for HCV antibody screening. 19 Other studies have reported the number of eligible patients and the number of patients who completed HCV antibody tests but did not cite the number of HCV antibody tests ordered, precluding the ability to ascertain whether patients drop off in that step of the HCV continuum of care. 18,20
We examined predictors of HCV screening completion among the baby boomer population. When we collected our data, universal screening was recommended only for baby boomers. 6,7 However, our team was aware that CDC and USPSTF were drafting updated recommendations to include onetime universal screening for all adults for release in spring 2020. Therefore, we examined screening completion among all cohorts to provide baseline screening rates, with a more thorough analysis of the predictors of screening among the baby boomer cohort. Among baby boomers, the type of health insurance coverage and seeing an advanced practice professional were significantly associated with HCV antibody test completion. Specifically, seeing only an advanced practice professional was associated with higher odds of completing an HCV antibody test. This finding contradicts our previous research, which found no association between seeing an advanced practice professional and the likelihood of having an HCV antibody test ordered. 15 Given the differences in these findings, it is possible that patients who see only an advanced practice professional have fewer comorbidities than patients seeing physicians. Patients with few comorbidities may be able to focus on uptake of preventive screenings because they do not have competing demands on their time to manage other chronic conditions. This possibility is consistent with previous research on colorectal cancer screening, which found that comorbidities were negatively associated with follow-up after a positive screening result. 21 However, further research is necessary to better understand the relationship between these variables.
We also found that being uninsured was associated with lower odds of completing the HCV antibody test, and we found no significant differences between having commercial health insurance and public health insurance. Research on cohorts of patients who have completed HCV antibody testing generally shows that patients with public health insurance have higher rates of test completion than do patients with commercial health insurance. For example, an examination of HCV infection rates by health insurance among 1123 baby boomers found that, of the people who completed an HCV antibody test, a greater percentage had public (58.7%) versus commercial (41.3%) health insurance. 22 Another study reported that among 1529 baby boomers who completed an HCV antibody test, the largest percentage of patients had Medicare (31.7%), followed by commercial health insurance (24.7%), no health insurance (21.3%), public health insurance/Medicaid (18.7%), and other health insurance (2.9%). 23 However, in both studies, the cohort comprised patients who had already received an HCV antibody test, thereby limiting the ability to determine whether health insurance status is a barrier to completing the screening.
Our previous work demonstrated that non-Hispanic Black baby boomer patients had higher odds of having a health care provider order the HCV antibody test for them than baby boomer patients of other races and ethnicities (aOR = 1.36; 95% CI, 1.19-1.55). 15 Our current study demonstrated that non-Hispanic Black baby boomer patients also had higher odds of completing HCV antibody testing when ordered compared with other birth cohorts and people of other races and ethnicities. This finding is in contrast to research conducted during 2013-2015, in which non-Hispanic Black participants reported lower odds of having ever been screened for HCV than participants in other racial and ethnic groups. 13 However, a 2016 update to national data showed that the difference between non-Hispanic Black participants and non-Hispanic White participants was no longer significant. 24 This disparity warrants further evaluation in future studies, particularly given research showing that non-Hispanic Black people are more likely than non-Hispanic White people to have a current HCV infection. 25
Our post hoc analyses examining HCV test completion and completion of other laboratory tests showed completion rates similar to other blood-based laboratory tests. These results suggest that the process of blood draw may be a common barrier to multiple blood-based screening tests ordered by health care providers, and this barrier may not be specific to HCV screening in this population. This finding may indicate that barriers to HCV screening are more likely to be at the systems level (eg, barriers to getting blood drawn) as opposed to barriers at the patient or health care provider level (eg, patient concern about stigma, low self-efficacy among health care providers).
Despite a onetime universal HCV screening recommendation for baby boomers that has been in place for nearly a decade, the rates of self-reported HCV screening uptake in this birth cohort remain low. 13,24 Little research has examined EHR-verified screening rates, and these studies typically examine either the health care provider having ordered the test 15 or results from the screening tests, 22,23 but none examined whether a barrier exists to completing the test when ordered, as our study does. Previous intervention studies 11,17,26,27 showed that adding EHR alerts and improving health care provider knowledge and awareness about HCV birth cohort screening can lead to increases in HCV screening ordered for baby boomers (37%-254% improvements). However, our current study indicates that interventions may need to target both the patient and the health care provider to effectively increase HCV screening test completion rates, including patient and health care provider education or opt-out screening strategies.
Limitations and Strengths
Our study had several limitations, which could limit generalizability. First, a history of injection drug use, a common behavioral risk factor for HCV infection, was not routinely captured in the EHR. Therefore, some high-risk patients may have been included in the average-risk group, which would result in an underreporting of high-risk groups by this behavioral factor. Second, if a patient completed testing through a laboratory that was not linked to the University of South Florida Health EHR, then results may not be included in the patient’s University of South Florida–based record. Third, we were unable to access data before 2015 (2012-2014) to assess whether CDC and USPSTF recommendations affected screening rates; however, national rates of HCV screening were low during these years, which may mitigate this limitation somewhat. 28 Fourth, we assessed the ordering and completion of HCV antibody tests during a set period (August 1, 2015–July 31, 2020); some ordered tests may have been completed after July 31, 2020. Fifth, although our large sample size (N = 106 945) gave us adequate statistical power, it also resulted in significant but small effect sizes. For example, the difference in completion between non-Hispanic Black patients (76.4%) and non-Hispanic white patients (72.3%) is significant, but it may not be meaningful in a broad public health perspective.
Sixth, our study population was approximately 70% female and, therefore, might not be representative of the overall US adult population. The greater proportion of female patients could be due to the inclusion of obstetricians/gynecologists as primary care providers or the higher rate of primary care service use among women than among men. 29 -31 Finally, the COVID-19 pandemic began during our study period. 32 The pandemic likely affected screening in the last 6 months of data collection (compared with the first 4.5 years of data collection) and influenced the results. However, because we were examining screening completion when ordered, patients in our study were accessing health care at this time, and it was important to include them in the study to determine whether HCV screening completion was different, even during a time of pandemic restrictions and low levels of access to care. Furthermore, we examined HCV screening completion rates before and after March 2020, and we found no differences between patients born before 1945 and patients born after 1985. The completion rate among people born during 1945-1965 was 55% (95% CI, 49%-61%) and among people born during 1966-1985 was 67% (95% CI, 58%-77%); both rates are slightly lower than prepandemic rates. Future research is needed to determine to what extent, if any, the COVID-19 pandemic affected HCV screening rates.
Our study also had several strengths. First, our study included a large sample size and multiple years of data, allowing us to compare groups and include more variables in the regression model than in previous studies. Our study also examined completion rates when the HCV antibody test was ordered, providing a novel look at this step in the HCV continuum of care and allowing us to determine whether barriers to completion exist other than a lack of an antibody test order from a health care provider.
Conclusions
Our results indicate that more than one-quarter of patients who receive an order for an HCV antibody test from their health care provider do not complete the test, regardless of birth cohort. These data highlight the need for targeted interventions that not only promote HCV antibody test ordering and education for health care providers but also consider broader systems-level interventions to support patient completion of testing to improve uptake. Future directions include expanding HCV screening research to multiple health care providers and different settings to determine whether similar findings are observed and examining whether screening completion rates vary according to patient health status, including liver function. Interventions should target systems, health care providers, and patients in primary care settings. Efforts to improve HCV testing rates and linkage to care can reduce incidence and mortality related to chronic HCV infection.
Supplemental Material
Figure S1 - Supplemental material for Hepatitis C Virus Screening: Factors Associated With Test Completion in a Large Academic Health Care System
Supplemental material, Figure S1, for Hepatitis C Virus Screening: Factors Associated With Test Completion in a Large Academic Health Care System by Monica L. Kasting, Shannon M. Christy, Richard R. Reich, Julie A. Rathwell, Richard G. Roetzheim, Susan T. Vadaparampil and Anna R. Giuliano in Public Health Reports
Footnotes
Acknowledgments
The authors thank William Stewart, MHA, for assisting with data extraction and management of the electronic health records system and Linh Duong, PhD, for her early work conceptualizing the study.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Giuliano is a member of the Merck Advisory Board and currently receives funding through the Merck Investigator Studies Program. Dr Vadaparampil currently receives funding through a Gilead investigator-initiated studies program, HCV CHIME program: Conquering Hepatitis via Micro-Elimination.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported, in part, by a 2016 Moffitt Cancer Center Team Science Award (principal investigators: A.R.G., S.T.V.) and by the Biostatistics and Bioinformatics Shared Resource at the H. Lee Moffitt Cancer Center & Research Institute, a National Cancer Institute (NCI)–designated Comprehensive Cancer Center (P30 CA076292; principal investigator: John L. Cleveland, Moffitt Cancer Center). Dr Giuliano is supported, in part, by the NCI-funded Center for Immunization and Infection Research in Cancer, as was Dr Kasting while the work on this project was being completed (K05 CA181320; principal investigator: A.R.G.). Dr Kasting began this study while she was a postdoctoral fellow and was funded by NCI (R25 CA090314; principal investigator: Thomas H. Brandon, Moffitt Cancer Center). Dr Kasting is supported by the Indiana Clinical and Translational Sciences Institute (KL2 Program), grant numbers KL2TR002530 (principal investigator: Brownsyne Tucker Edmonds, Indiana University) and UL1TR002529 (co-principal investigators: Sharon Moe and Sarah Wiehe, Indiana University) from the National Institutes of Health’s National Center for Advancing Translational Sciences, Clinical and Translational Sciences Award. The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of NCI or the National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
