Abstract
Background
Estimates of the prevalence of hepatitis C virus (HCV) amongst people who inject drugs are in the range of 50%. Opioid Agonist Treatment (OAT) clinics have a large proportion of people who inject drugs. These clinics, therefore, present an excellent opportunity to identify and treat patients with HCV. Despite this, it remains unclear as to how well these clinics screen patients for HCV.
Aim
This study aims to quantify the rate of HCV screening of patients that enrolled into treatment at an OAT clinic attached to a large teaching hospital in NSW.
Methods
A retrospective audit of the electronic medical record of patients commencing OAT at the clinic was performed to determine the rate of HCV screening.
Findings
During the audit period, there were 99 episodes of care that involved the commencement of OAT at the clinic, for patients who had a history of injecting drugs. Successful screening for HCV occurred in 56% (55/99) of these episodes. Of those screened, 26% were ribonucleic acid positive. Analysis of demographics did not reveal a statistically significant difference between those that were screened and those that were not.
Discussion
This study revealed a suboptimal rate of HCV screening at the OAT clinic. To improve the screening rate, the clinic could consider providing dried blood spot testing at the initial nurse assessment or having a clinician capable of venepuncture at the clinic.
Introduction
The hepatitis C virus (HCV) places a large physical, social, financial and emotional burden on the affected individual and society. An estimated 71 million people are living with HCV worldwide and approximately 399,000 people died as a result of HCV in 2016 (World Health Organization, 2019). The majority of these deaths are attributed to liver cirrhosis and hepatocellular carcinoma.
According to the World Health Organization (WHO), the most common methods of transmission of HCV are injecting drug use, unsterile medical equipment, transfusion of unscreened blood products and sexual practices that lead to exposure to blood (World Health Organization, 2019). Within Australia, injecting drug use is believed to be the predominant mode of transmission (Razali et al., 2007).
Direct acting antiviral (DAA) medications (Spengler, 2018) for the treatment of HCV were listed on the Pharmaceutical Benefits Scheme (PBS) in Australia in March 2016. In the same year, the Australian Government endorsed WHO’s goal of elimination of viral hepatitis as a major public health threat by 2030 (Australian Government Department of Health, 2019). The number of Australians living with HCV in 2014 (pre-DAA) was estimated to be 230,470 (Hajarizadeh et al., 2017), whilst by the end of 2017, this number had reduced to 182,144 (Hepatitis Australia, 2019). Between March 2016 and December 2018, an estimated 70,260 Australians with HCV received DAA treatment (Hepatitis Australia, 2019). This illustrates the unique opportunity that currently exists with regard to largely eradicating HCV from the population.
In Australia, the prevalence rate of chronic hepatitis C amongst people who inject drugs (PWID) was estimated to be 50%, prior to the arrival of DAAs (Martin et al., 2013). Furthermore, through mathematical modelling, Scott et al. have predicted that despite the advent of DAAs, by 2030, there will still be 24,000 Australians living with HCV (Scott et al., 2017). It is predicted that the majority (74%) of those remaining untreated in 2030, will be undiagnosed and amongst PWID (Scott et al., 2017). Accordingly, priority settings for the treatment of HCV, identified by the Australian Government include drug and alcohol (D&A) services, needle and syringe programs, and custodial settings (Australian Government Department of Health, 2019).
The prevalence of HCV amongst those receiving Opioid Agonist Treatment (OAT) in Australia is not well established, but thought to be even higher than the prevalence of HCV amongst PWID (Shand et al., 2014). Therefore, within the priority setting of D&A services, OAT clinics potentially have the greatest prevalence of HCV positive patients. OAT clinics must therefore be seen as a priority for screening and treatment of HCV within D&A services. Larney et al. have shown that amongst PWID, those in OAT are more likely to undergo HCV screening than those that are not (Larney et al., 2015). However, the question remains as to how successful OAT clinics are at screening their patients, and whether their screening rate can be improved.
The Gastroenterology Society of Australia recommends that those that have ongoing risk factors for HCV transmission should undergo HCV screening annually (Hepatitis C Virus Infection Consensus Statement Working Group, 2016). An HCV antibody test is the appropriate initial screening, and if positive, the patient should proceed to HCV Polymerase Chain Reaction (PCR) to detect the presence of viral ribonucleic acid (RNA). If the patient is known to be HCV antibody positive, then HCV PCR should be ordered as the initial test.
It is extremely common that patients entering an OAT program satisfy the criteria of requiring HCV screening due to recent intravenous drug use. An exception to this may be a prospective OAT patient who has never injected drugs, such as those that have developed Opioid Use Disorder due to oral prescription opioid medications. Within the OAT clinic involved in this audit, HCV is always discussed at initial assessment and screening is offered if it has not occurred within the previous 12 month period. Alternatively, if screening has occurred, then an attempt is made to retrieve a copy of the result for scanning into the patient’s Electronic Medical Record (EMR). For those patients who have never injected drugs, a decision on HCV screening is made after assessing for other transmission risk factors (Hepatitis C Virus Infection Consensus Statement Working Group, 2016).
The literature provides limited examples of the quantification of HCV screening for those on OAT in a particular setting. Brown et al. reported that 34.9% of patients in an OAT clinic in Midwestern USA received HCV screening in the last 12 months (Brown et al., 2017). However, this result was extracted from a self-reported patient survey (evidence of screening not assessed). Similarly, there are three other studies reporting screening rates in the range of 30–33% (Du et al., 2012; Ng et al., 2013; Walley et al., 2005). In all studies, screening rates are derived from patient questionnaires. The actual screening rate may well differ from that reported by the patient, due to the patient’s understanding of their result, and the patient’s memory. In a retrospective observational study, Harris et al. reported a screening rate of 97% (281/291) over a 2 year period at an OAT clinic in New York, USA (Harris et al., 2010). This screening rate is for HCV antibody testing only. A patient who is HCV antibody positive is not necessarily infected with HCV. Of those that were HCV antibody positive, 74% were successfully screened for HCV RNA. It should be noted that this high screening rate was achieved via a concerted effort upon co-locating HCV assessment and treatment services, with an OAT clinic.
The aim of the present audit was to determine how successful an OAT clinic attached to a large teaching hospital was in providing HCV screening to those that enrolled into OAT. The screening rate was determined using actual review of screening results on file, rather than the less accurate method of a patient survey. Also of interest was whether the rate of screening changed during the audit period. It was envisaged that the audit results might reveal demographic differences between those that were screened and those that were not, thereby guiding the modification of HCV screening procedures. Overall, by conducting this audit, we hoped to increase the future rate of HCV screening and treatment within the clinic.
Methods
A retrospective audit of EMR files was conducted for patients who were sequentially enrolled into OAT at an OAT clinic, attached to a teaching hospital in New South Wales, Australia. The clinic has onsite dosing of both methadone and buprenorphine. Some patients are transferred to a community pharmacy for dosing, but still see a clinician at the clinic periodically for routine review.
At this OAT clinic, HCV screening can be conducted on site, utilizing dried blood spot (DBS) testing, or by providing a pathology order form to the patient and asking them to have a blood test done. Venepuncture is not available on site. There are multiple pathology collection points in near vicinity of the clinic, including the local hospital approximately 250 m away. In this audit, a positive DBS result was recorded as ‘HCV RNA positive’.
The audit process
The audit period chosen was from 1 January 2018 to 30 June 2019. During this period, the use of DAAs for the treatment of HCV was already established.
An episode of care was considered to be the time from when the patient commenced OAT, until they were discharged from the service. Episodes of care were identified by conducting an automated search of EMR files for patients who had a ‘Drug and Alcohol Assessment’ form completed by a staff member at the OAT clinic, during the audit period. A Drug and Alcohol Assessment form is always completed when a new patient presents to the OAT clinic, seeking OAT. This assessment form is generally completed by a nurse. Other data automatically extracted at this point included gender, age, whether the patient identified as Aboriginal and/or Torres Strait islander (ATSI) and whether the patient had ever injected a drug (injecting status was also cross-checked manually by reviewing the doctor’s initial assessment). Automatic data extraction was conducted in October 2019.
If the same patient underwent more than one episode of care during the audit period, each episode was treated separately, as some of their baseline demographic data, and screening status may have changed on each occasion.
Once the episodes of care were identified via the automated search, the EMR files were then reviewed by the first author and the following information are extracted manually: date of doctor’s assessment, date of OAT commencement, discharge date (if discharge had occurred at the time of file review), type of OAT prescribed, date of HCV screening, type of screening and origin of patient (Justice Health or community). In order to maintain consistency, only the first author conducted manual extraction of data.
HCV screening was deemed to have occurred if a screening result appeared in the EMR file, during the episode of care (commencing after the doctor’s assessment, and ending at discharge), or if a result was on file in the 12 month period leading up to the date of the doctor’s assessment. The decision to utilize this threshold is guided by the recommendation that patients should undergo HCV screening every 12 months if engaged in ‘high risk activities’ (Hepatitis C Virus Infection Consensus Statement Working Group, 2016).
The HCV screening result was present on file in one of three forms: (1) a result in EMR pathology when the blood test had been conducted at a public hospital within the local health district, (2) a scanned document when the blood test was conducted by a private pathology company or (3) a scanned document when the blood test was conducted by Justice Health (prior to release from prison). If a patient informed staff that they had been screened elsewhere (by their GP), but this result was not retrieved and scanned into EMR, then the patient was deemed to have not been screened. Visual proof of screening was required.
Ethical considerations
Prior to commencement of the audit, ethics approval was sought. The Health and Medical Research Office of the hospital advised that the project would be considered a ‘quality audit’, and therefore, further ethical review was not required.
Data analysis
Data were entered into an Excel spreadsheet for analysis. IBM SPSS Statistics version 21 was used to conduct the analysis. When comparing the baseline demographic data, gender, ATSI status and type of OAT (methadone or buprenorphine) between the ‘screened’ and ‘non-screened’ groups, the Chi-squared test was performed. Age was compared using the Mann–Whitney U test. The difference in the duration of episode of care in the screened and non-screened groups was assessed using the Mann–Whitney U test. The difference in the number of patients remaining in an ongoing episode of care at the date of data extraction (1 October 2019), with respect to screened and non-screened groups, was determined using a Chi-squared test. Finally, whether there was a significant change in screening rates, during the 18 month audit period (when divided into 3×6 month periods), was determined using a Z test.
Results
A total of 157 Drug and Alcohol Assessment forms were completed during the audit period between 1 January 2018 and 30 June 2019. Of these, 78% progressed to a doctor assessment, and 71% commenced OAT treatment. Of those who commenced treatment, 89% had a history of injecting drug use. Of these, 56% had HCV screening completed with 26% being HCV RNA positive (Figure 1). Audit process of patients enrolled in an OAT from 1 January 2018 to 30 June 2019.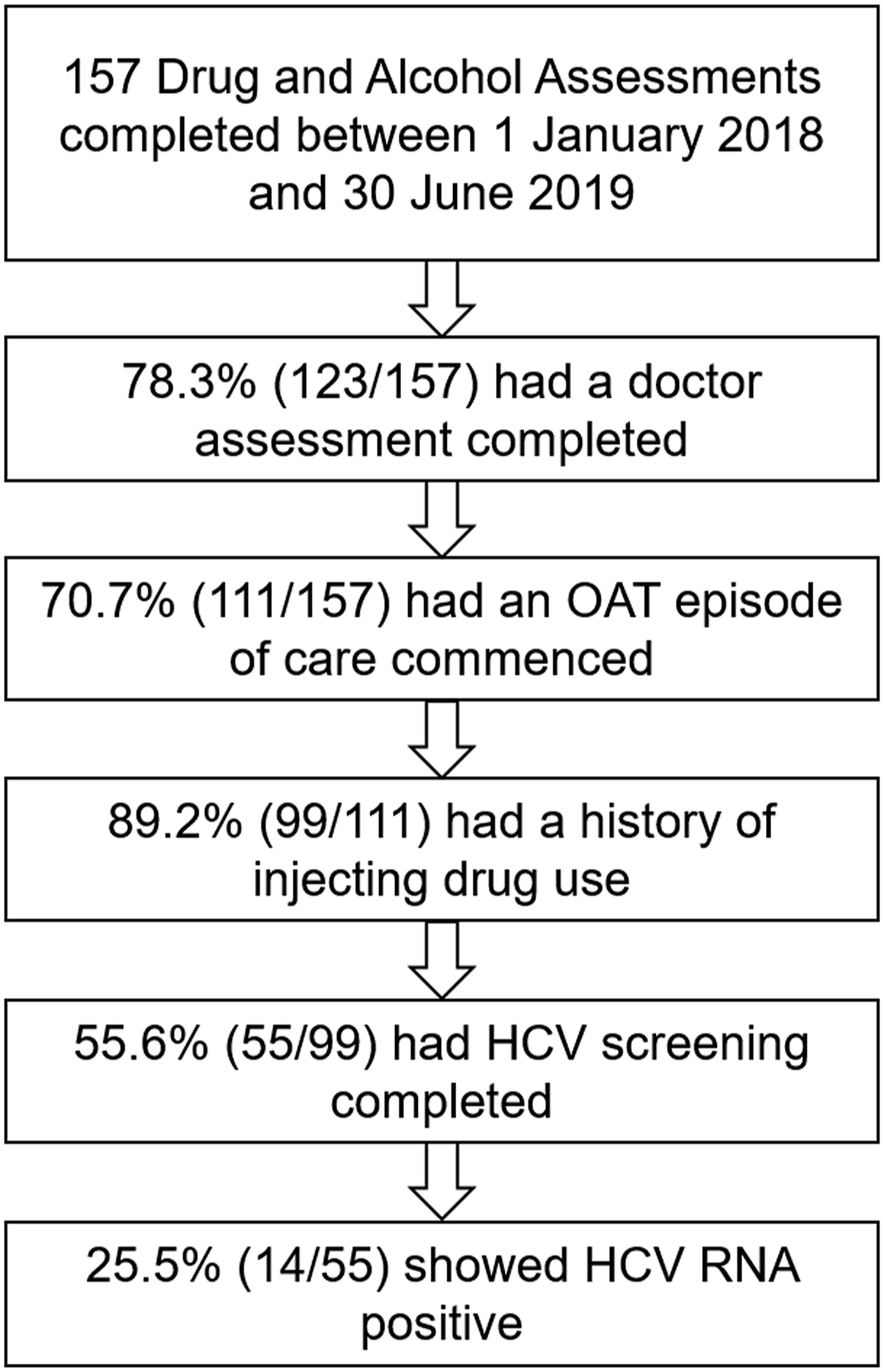
Characteristics of episodes of care incurred by patients with history of injecting drug use.
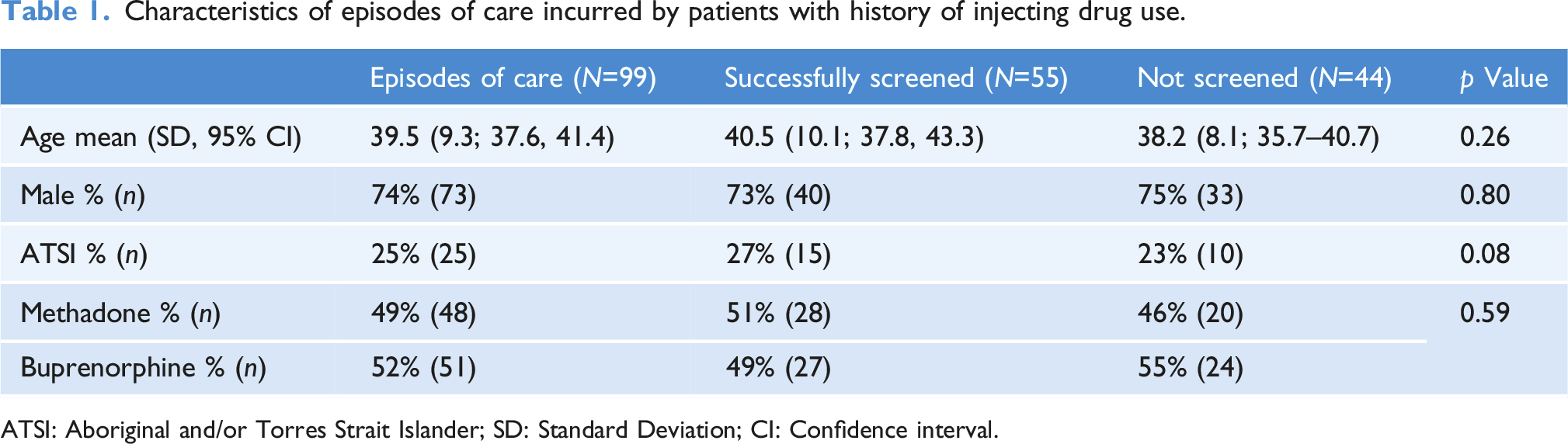
ATSI: Aboriginal and/or Torres Strait Islander; SD: Standard Deviation; CI: Confidence interval.
Of the 55 episodes of care that had HCV screening completed, 26% were HCV RNA positive, 73% were HCV RNA negative and 2% were HCV antibody negative. Of these 55 episodes, 49% (27) underwent screening after the doctor assessment and the other 51% had an HCV screening result that was less than 12 months old at the commencement of OAT. Of the 27 episodes that underwent screening after the doctor assessment, the average time from the assessment, at which a patient was provided with a pathology form, to the blood test being completed was 65.1 days (Standard Deviation [SD]=98.4, 95% Confidence Interval [CI]=27.4–103.0).
Comparison of duration of episodes of care between screened and unscreened groups.
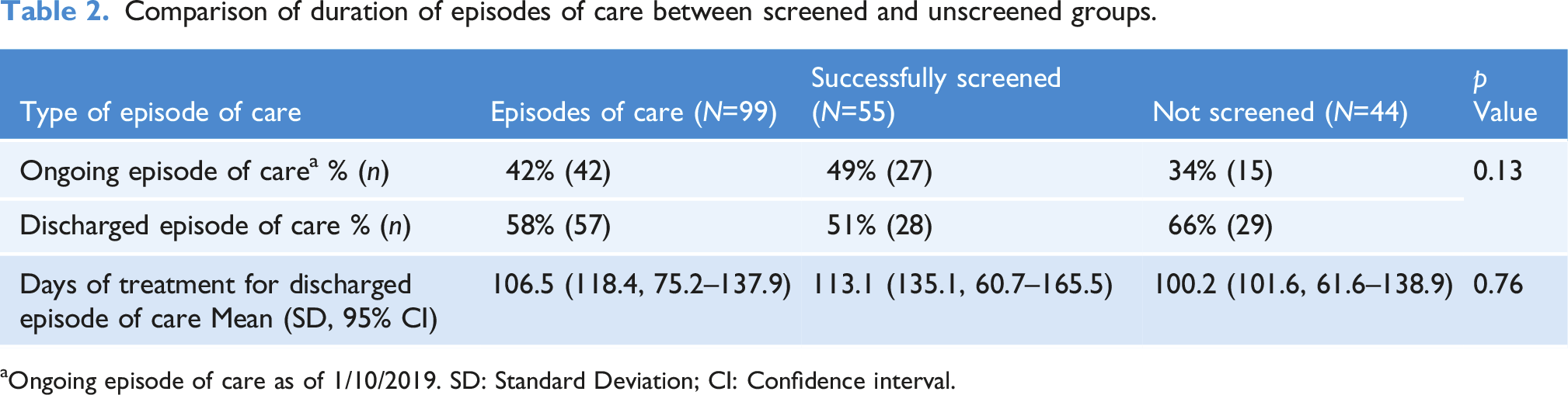
aOngoing episode of care as of 1/10/2019. SD: Standard Deviation; CI: Confidence interval.
Change of screening rate over the audit period, by trimesters.
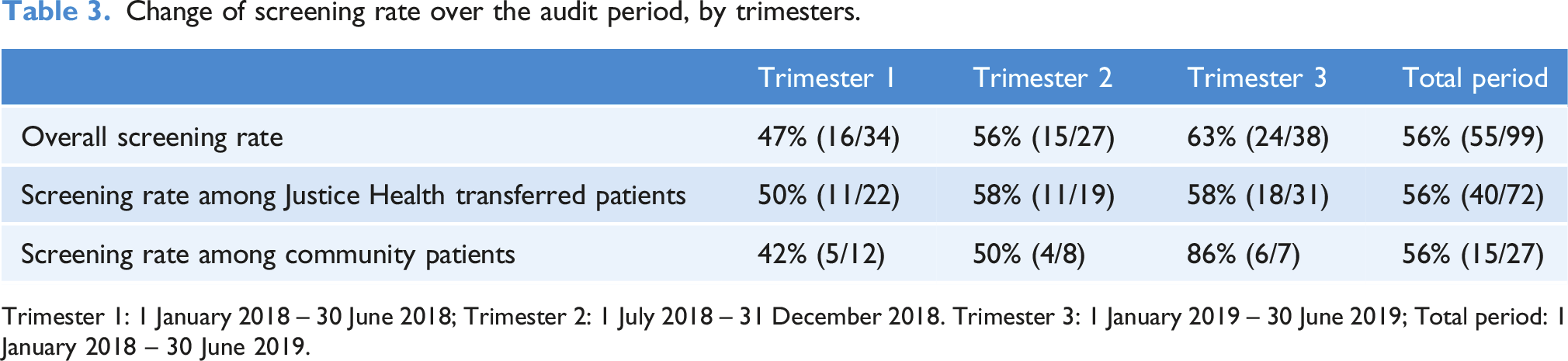
Trimester 1: 1 January 2018 – 30 June 2018; Trimester 2: 1 July 2018 – 31 December 2018. Trimester 3: 1 January 2019 – 30 June 2019; Total period: 1 January 2018 – 30 June 2019.
Discussion
Successful screening for HCV was achieved in 56% of the audited episodes in this study. Clearly, the ideal screening rate would be 100%. It would be difficult to achieve such a rate as some patients only engage in the treatment very briefly. A literature search provided limited comparative data. As discussed in the introduction, some OAT clinics have reported HCV screening rates in the range of 30%–35% (Brown et al., 2017; Du et al., 2012; Ng et al., 2013; Walley et al., 2005). But these figures have been derived from patient questionnaires, rather than review of results on file. In a retrospective review of files, Harris et al. reported that of those that tested HCV antibody positive, 74% proceeded to have an HCV RNA test (Harris et al., 2010). A concerted effort was made to achieve this high screening result, in particular, by co-locating HCV assessment and treatment services at the OAT clinic. The OAT clinic subject to the current audit has not screened at a rate as high as that reported by Harris et al. All HCV assessment services are not co-located at the OAT clinic subject to this audit. Venepuncture is not available at the clinic.
It is well known that females tend to engage in healthcare to a greater degree compared to males (Green and Pope, 1999) and that increasing age leads to greater executive function and forward planning, perhaps facilitating a greater desire of curing an illness that may cause harm in the future. It would appear obvious that a longer episode of care allows more opportunities to educate the patient about HCV, and encourage screening and treatment.
The fact that the ‘screened’ group versus ‘non-screened’ contained a greater proportion of ATSI was unexpected. During the audit period, an Aboriginal Health Worker facilitated a visit from the hepatology nurse, to the ‘Koori Smart’ meetings, where HCV screening was conducted on the spot using DBS testing. This may have been one of the contributors to the observed result. It is uncertain as to why the ‘screened’ group contained a greater proportion of methadone versus buprenorphine patients, relative to the ‘non-screened’ group.
Since DAAs became available on the Australian PBS in March 2016, there has been a rapidly increasing awareness amongst both patients and health practitioners, of the availability of these treatments and the relatively simple manner in which HCV can now be treated. Accordingly, it was suspected that screening rates may not be stable during the audit period, but rather, have increased. In addition, there had been the anecdotal observation at the clinic that as the audit period progressed, it became more common for patients from Justice Health to present to the clinic with Justice Health discharge paperwork that contained HCV screening results. This audit revealed that there was a trend of increased screening (Table 3). Interestingly, the greatest increase in screening rate was achieved amongst community enrolled patients, not Justice Health patients. However, the numbers in this analysis are quite small; therefore, one should be cautious in drawing conclusions from these results.
In this audit, it was discovered that of those screened, 26% were HCV RNA positive. In 2014, Shand et al. reported that amongst a cohort receiving OAT in NSW, Australia, 55% were HCV RNA positive (Shand et al., 2014). The likely explanation for this difference is the fact that the current study has been conducted after the introduction of DAAs, whilst the study of Shand et al. was conducted prior. This observed difference probably demonstrates the great impact that DAAs have made.
This study found that it took patients an average of 65.1 days to have their blood test done after being provided with a pathology form. Our anecdotal experience at the OAT clinic is that providing a pathology form to a patient for screening often results in the form being ‘lost’. As positive effect of ‘on-site testing’ on HCV screening has been noted in the literature (Bajis et al., 2017; Harris et al., 2010), it would be helpful to have a clinician capable of venepuncture at the clinic at all times, so that a fleeting moment of interest in HCV screening might be seized.
In relation to non-patient related barriers to screening, it was noted that whilst DBS testing is available at the OAT clinic, it is used infrequently. Its main use within the local health district is at the needle and syringe programs which are at a different location to the OAT clinic. DBS or ‘point of care’ testing has been discussed in the literature with regard to HCV and has been found to be acceptable and preferable by patients (Bajis et al., 2018; Hayes et al., 2014). Therefore, consideration is being given as to whether DBS testing should be encouraged at the nurse’s initial Drug and Alcohol Assessment. The advantage of this is that it could be integrated and normalized as a routine part of the initial assessment. It may also capture some positive HCV results in patients who do not commence OAT, or only remain in OAT for a very brief time, and therefore never receive the opportunity for HCV screening and treatment. Likewise, it is suspected that the rate of screening could be improved by enabling venepuncture to be available at all times at the OAT clinic (currently not available at all). If and when the above-mentioned measures are introduced, this audit should be repeated to assess the effect on screening.
As discussed in the Methods section, screening was only assumed to have occurred if the pathology result appeared in the EMR file. It is possible that some patients were screened by other healthcare providers, such as their GPs, and that this result was not requested by, or not made available to the OAT clinic, thereby leading to an underestimation of screening in this audit.
Conclusion
This audit found that just over half of the OAT patients with a history of injecting drug use were successfully screened for HCV. Of those screened, just over a quarter were found to be HCV RNA positive. Although not statistically significant, the screening rate was found to increase with time during the audit period.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
