Abstract
Objective
A recommendation in March 2020 to expand hepatitis C virus (HCV) screening to all adults in the United States will likely increase the need for HCV treatment programs and guidance on how to provide this service for diverse populations. We evaluated a pharmacist-led HCV treatment program within a routine screening program in an urban safety-net health system in Chicago, Illinois.
Methods
We collected data on all patient treatment applications submitted from January 1, 2017, through June 30, 2019, and assessed outcomes of and patient retention in the treatment cascade.
Results
During the study period, 203 HCV treatment applications were submitted for 187 patients (>1 application could be submitted per patient): 49% (n = 91) were aged 55-64, 62% (n = 116) were male, 67% (n = 125) were Black, and 15% (n = 28) were Hispanic. Of the 203 HCV treatment applications, 87% (n = 176) of patients were approved for treatment, 91% (n = 161) of whom completed treatment. Of the 161 patients who completed treatment, 81% (n = 131) attended their sustained virologic response (SVR) follow-up visit, 98% (n = 129) of whom reached SVR. The largest drop in the treatment cascade was the 19% decline from receipt of treatment to SVR follow-up visit.
Conclusion
The pharmacist-led model for HCV treatment was effective in navigating patients through the treatment cascade and achieving SVR. Widespread implementation of pharmacist-led HCV treatment models may help to hasten progress toward 2030 HCV elimination goals.
From 2010 to 2016, the number of hepatitis C virus (HCV) infections increased 3.5-fold in the United States, and HCV is now the most frequently reported blood-borne infection in the United States. 1 -3 This surge of new infections is largely due to the opioid crisis. 3,4 The prevalence of HCV infection varies in the United States, with some areas experiencing higher rates of HCV infection than other areas. 1,3 In Chicago, roughly 25 400 people were living with HCV infection in 2017, with communities on the west and southwest sides of the city having a higher prevalence compared with the city average. 5
Although HCV screening and diagnosis are important public health responses to the HCV epidemic, 6 a large drop-off occurs in the care cascade among people who achieve sustained virologic response (SVR) or cure, the metric for successful HCV treatment. 7 SVR is defined as an undetectable or unquantifiable HCV RNA ≥12 weeks after treatment completion. 8 Although no national surveillance data on SVR rates are available, a 2014 meta-analysis estimated that, of the 3.5 million people living with chronic HCV infection in the United States, only 9% have reached SVR. 7 In Chicago, the rate was similar (11%).5
Research focuses on assessing HCV screening programs and SVR outcomes; however, few studies have evaluated barriers to treatment retention in correlation with screening and achieving SVR. 7,9,10 Furthermore, few studies have outlined protocols for moving patients along the treatment cascade, especially after the revolutionary advent of HCV direct-acting antiviral (DAA) agents in 2014. 7,10,11 Because the HIV treatment cascade 12,13 has been used to identify gaps in the delivery of care 7 and has similarities to the HCV care continuum, 14 identification of treatment barriers and evaluation of approaches to navigation of the continuum of care should be applied to the HCV treatment cascade.
Lastly, a small number of studies implemented and evaluated universal HCV testing with treatment outcomes, 15 with most published data focusing only on groups at increased risk of HCV, including baby boomers, injection drug users, people with a history of illicit drug use, and/or people with HIV infection. 16
Pharmacists can play a critical role in treating HCV infection, 17 -21 reinforcing treatment adherence, and increasing SVR rates. 19,20,22 Pharmacists contribute to HCV treatment care by reviewing patient medical records to recommend appropriate treatment regimens, evaluating drug–drug interactions, acquiring medications through prior authorizations of health insurance, offering treatment education and medication adherence counseling, and monitoring and interpreting laboratory tests. 18,21,23 However, studies describing these collaborative practices did not evaluate retention at each step of the treatment cascade and, therefore, did not quantitatively assess the barriers commonly encountered in pharmacist-led treatment protocols. 18,22,24
Our study adds to the literature in several ways. First, we describe treatment outcomes at multiple points of the treatment cascade rather than conventionally reporting only SVR rates. 25 Second, a universal routine HCV screening program allowed us to measure treatment outcomes for all patients, regardless of birth year and hospital admission. Third, this study describes the collaborative practice and outcomes of a pharmacist-led treatment model in an urban safety-net health system. Our objective was to evaluate outcomes in the HCV treatment cascade for patients linked to care through an urban safety-net hospital’s HCV navigation model with a pharmacist-led treatment program.
Methods
Setting
Sinai Chicago serves the west and southwest sides of Chicago, communities with large racial/ethnic minority populations and populations with low socioeconomic status who are medically underserved. Sinai Chicago includes Mount Sinai Hospital and Holy Cross Hospital, both of which have emergency departments and offer a full range of inpatient and outpatient services.15 The Mount Sinai Hospital Infectious Disease Clinic (MSHIDC) is an outpatient hospital-based clinic that provides comprehensive infectious disease care with a staff of 3 infectious disease physicians, 1 nurse practitioner, 1 clinical pharmacist, 1 pharmacy liaison, and 1 HCV navigator. The Mount Sinai Hospital Institutional Review Board approved this project.
The Project
The first steps of the treatment cascade, Sinai Chicago’s routine universal screening program, and patient navigation services for linkage to first medical appointment, funded by Gilead’s Frontlines of Communities in the United States, are described elsewhere. 15 Briefly, this program screened more than 20 000 patients during a 2-year period with 6.3% HCV antibody positivity, 4.2% RNA positivity, and 23% of RNA-positive patients ultimately linked to a medical appointment. Sinai Chicago’s unique pharmacist-led HCV treatment program, funded by Sinai Chicago in part via the Medicaid 340B program, is described hereinafter.
Treatment Assessment Appointment
The infectious disease physician and/or nurse practitioner (infectious disease care provider) and the infectious disease pharmacist assess the patient’s eligibility for HCV treatment and determine the most appropriate treatment regimen based on clinical factors, drug–drug interactions, and health insurance formulary plans. The infectious disease pharmacist recommends baseline laboratory tests and hepatitis immunizations, and the infectious disease care provider orders a liver ultrasound to assess for hepatocellular carcinoma and FibroScan/elastography to assess the severity of liver fibrosis. Once the infectious disease care provider and patient sign an HCV letter of commitment, in which the patient agrees to adhere to medication and clinic appointment regimens and abstain from drug and alcohol use, the infectious disease pharmacist explains to the patient the treatment application and submission process and the guidelines for adhering to medication and medical visits.
Treatment Application Submission
The infectious disease pharmacist obtains pertinent laboratory results, imaging, progress notes, medication lists, signed HCV letter of commitment, and health insurance (public and private) prior authorization forms for the HCV treatment application. Once the application is complete and reviewed by the infectious disease care provider, the infectious disease pharmacist submits the treatment application directly to the patient’s health insurance plan for approval or denial of medication.
Treatment Approvals, Denials, and Appeals
After the advent of DAA agents, health insurance plans were approving only HCV treatment for patients with Metavir/fibrosis scores of F3 (severe fibrosis) and F4 (cirrhosis) because of the high cost of medications. On November 1, 2018, the Illinois Department of Healthcare and Family Services lifted this restriction and expanded coverage of DAA agents to all HCV patients regardless of Metavir/fibrosis score.
The treatment application decision from the health insurance company is received by the infectious disease pharmacist. If the HCV medication is approved, the infectious disease pharmacist notifies the patient and infectious disease care provider of the decision and verbally sends a prescription for the HCV medication to the health insurance company’s designated specialty pharmacy. If the treatment is denied, peer-to-peer reviews and letters of appeal to the health insurance plan are attempted by the infectious disease pharmacist until the medication is approved.
Medication Delivery
All HCV medication refills are delivered to the MSHIDC to ensure medication adherence and to avoid home delivery problems with an expensive medication. In general, appointments to initiate HCV treatment are not scheduled until after the first month’s medication refill is delivered to the clinic. The pharmacy liaison coordinates each monthly medication refill with the specialty pharmacy and tracks each medication delivery to the clinic. After treatment is started, the infectious disease pharmacist or pharmacy liaison contacts the patient each month to schedule a date and time for the patient to pick up a medication refill from the clinic. For optimal adherence, the infectious disease pharmacist schedules the patient’s second medication refill pick-up with the week 4 follow-up appointment with the infectious disease care provider.
Treatment Initiation Appointment
Before starting HCV treatment, the infectious disease pharmacist evaluates the potential for drug–drug interactions and assesses hepatitis serologies to ensure updated hepatitis A virus and hepatitis B virus vaccinations, which are communicated to the infectious disease care provider. During the appointment for initiation of HCV treatment, the infectious disease pharmacist provides the first month of medication along with counseling on the administration, storage, adverse effects of the medication, and what to do after a missed dose. One week after initiating therapy, the infectious disease pharmacist follows up with the patient by telephone to assess adverse effects, missed doses, and adherence problems.
Follow-up Appointments
The patient attends an appointment with the infectious disease care provider for follow-up laboratory tests at multiple points during the treatment process, including at week 4 of treatment, treatment completion, and SVR, which is ≥12 weeks after treatment completion.
Post-SVR Follow-up Appointment
The patient attends a final surveillance follow-up appointment with the infectious disease care provider after reaching SVR. This visit occurs at 3 months post-SVR for patients with Metavir/fibrosis scores of F3 (severe fibrosis) or F4 (cirrhosis) and 6 months post-SVR for patients with F0-F1 (absent or mild fibrosis) or F2 (significant fibrosis). After achieving SVR, the infectious disease pharmacist sends a treatment completion letter describing details of the patient’s HCV treatment course to the infectious disease care provider for review and then to the patient’s referring health care provider or primary care physician. Finally, the pharmacy liaison scans the treatment application and approval/denial letters into a shared drive.
Participants
We collected data on HCV treatment outcomes for patients who screened positive for HCV at Sinai Chicago, were successfully navigated to their first medical appointment by the HCV patient navigator, and had a treatment application submitted by the infectious disease pharmacist from January 1, 2017, through June 30, 2019. We chose this end date to allow time for completion of treatment regimens by the time of analysis.
Data Collection and Analysis
For this study, we collected data on patient demographic characteristics (age at time of treatment application submission, sex, race, ethnicity), HCV genotype, Metavir/fibrosis score, health insurance payor, treatment application outcome, HCV treatment regimen and duration, time from treatment application submission to approval, HCV treatment initiation date, HCV treatment completion date, time from MSHIDC receiving medication from specialty pharmacy to patient initiating treatment, time from treatment approval to treatment initiation, and attendance at treatment completion and SVR follow-up appointments. We used Pearson χ2 tests to analyze all variables, except time from treatment application submission to approval, for which we used the Kruskal–Wallis test.
The infectious disease pharmacist collected and entered all data points in a password-protected REDCap database that was kept on the hospital server and de-identified for analysis. We performed univariate analyses (counts, medians, Pearson χ2 tests, and Kruskal–Wallis tests) using Stata version 16 (StataCorp). We considered P < .05 to be significant.
Results
Demographic Characteristics
From January 1, 2017, through June 30, 2019, HCV treatment applications were submitted for 187 patients: 49% (n = 91) were aged 55-64, 62% (n = 116) were male, 67% (n = 125) were Black, and 15% (n = 28) were Hispanic/Latino. Most patients (80%; n = 149) were infected with HCV genotype 1, and 57% (n = 106) had a Metavir/fibrosis score <F3 (Table).
Demographic characteristics of hepatitis C virus (HCV)–positive patients at Sinai Chicago (n = 187), Chicago, Illinois, January 1, 2017–June 30, 2019

aFibrosis stage F0-F1 indicates absent or mild fibrosis, F2 indicates significant fibrosis, F3 indicates severe fibrosis, and F4 indicates severe liver disease or cirrhosis.
Treatment Approvals, Denials, and Appeals
The infectious disease pharmacist submitted 203 HCV treatment applications for 187 patients from January 1, 2017, through June 30, 2019. (Multiple applications could have been submitted if the patient relapsed [ie, did not reach SVR after completing treatment], underwent multiple appeals for treatment denials, or switched health insurance plans.) During the study period, 76, 78, and 49 treatment applications were submitted in 2017, 2018, and 2019 (through June 30), respectively, of which 61 (80%), 67 (86%), and 48 (98%), respectively, were approved. A significantly higher percentage of treatment applications was approved in 2019 than in 2017 (P = .004).
HCV treatment approvals varied by Metavir/fibrosis score, with higher approval rates among patients with scores of F3 or F4 compared with patients with scores of F0-F2 in 2017 and 2018 (Figure 1). Treatment denials among patients with low fibrosis stages decreased from 26% in 2017 to 0% in 2019 (P = .002).
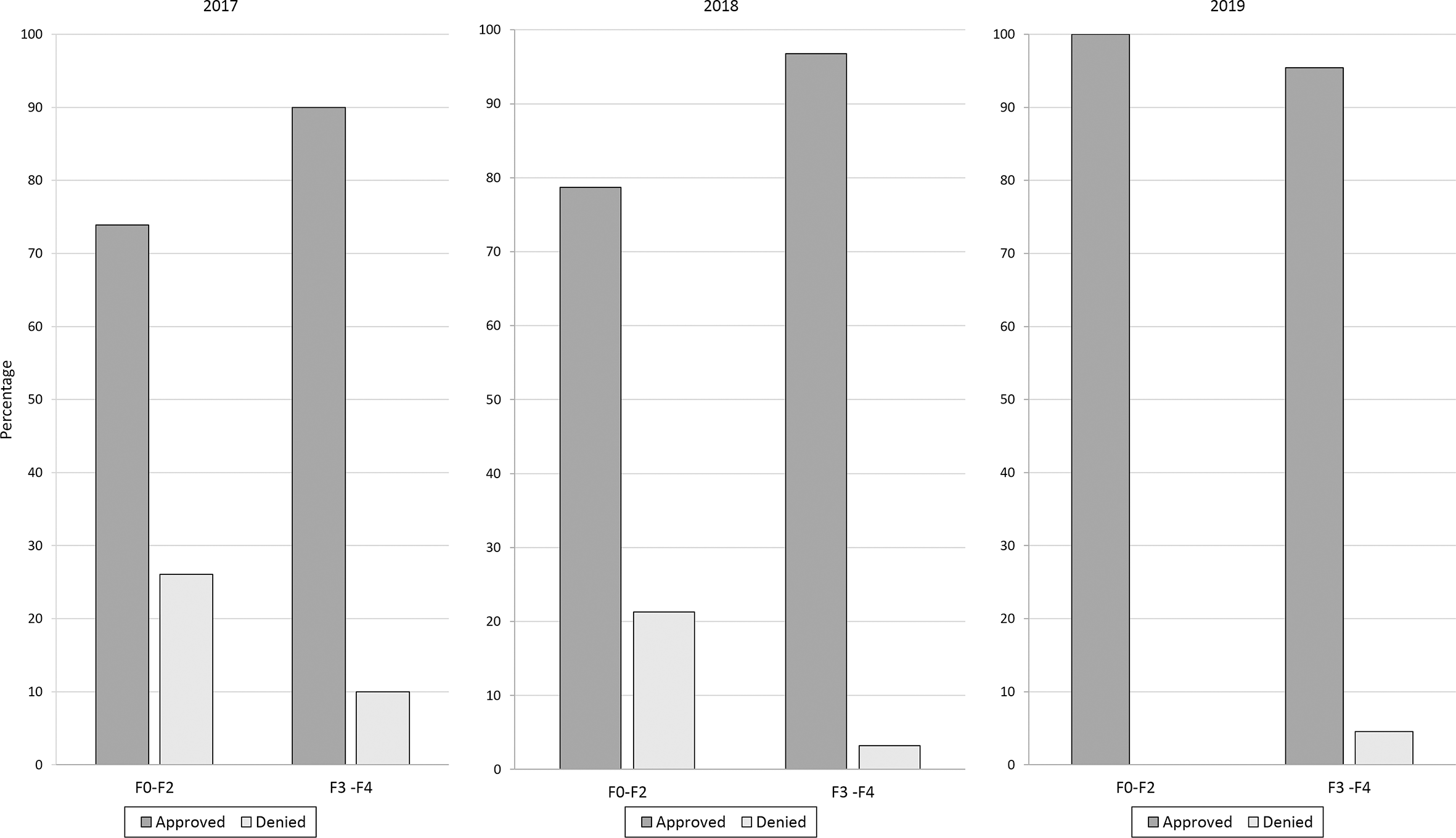
Hepatitis C virus treatment application approvals, by Metavir/fibrosis score and year, in an urban safety-net health system in Chicago, Illinois, January 1, 2017–June 30, 2019. A score of F0-F1 indicates absent or mild fibrosis, F2 indicates significant fibrosis, F3 indicates severe fibrosis, and F4 indicates severe liver disease or cirrhosis.
Treatment approvals for Medicare, private health insurance (health maintenance organization/preferred provider organization), and patient assistance programs were consistently >90% during the study period (Figure 2). However, from 2017 to 2019, the approval rate for Medicaid treatment applications increased from 59% to 97% (P < .001).
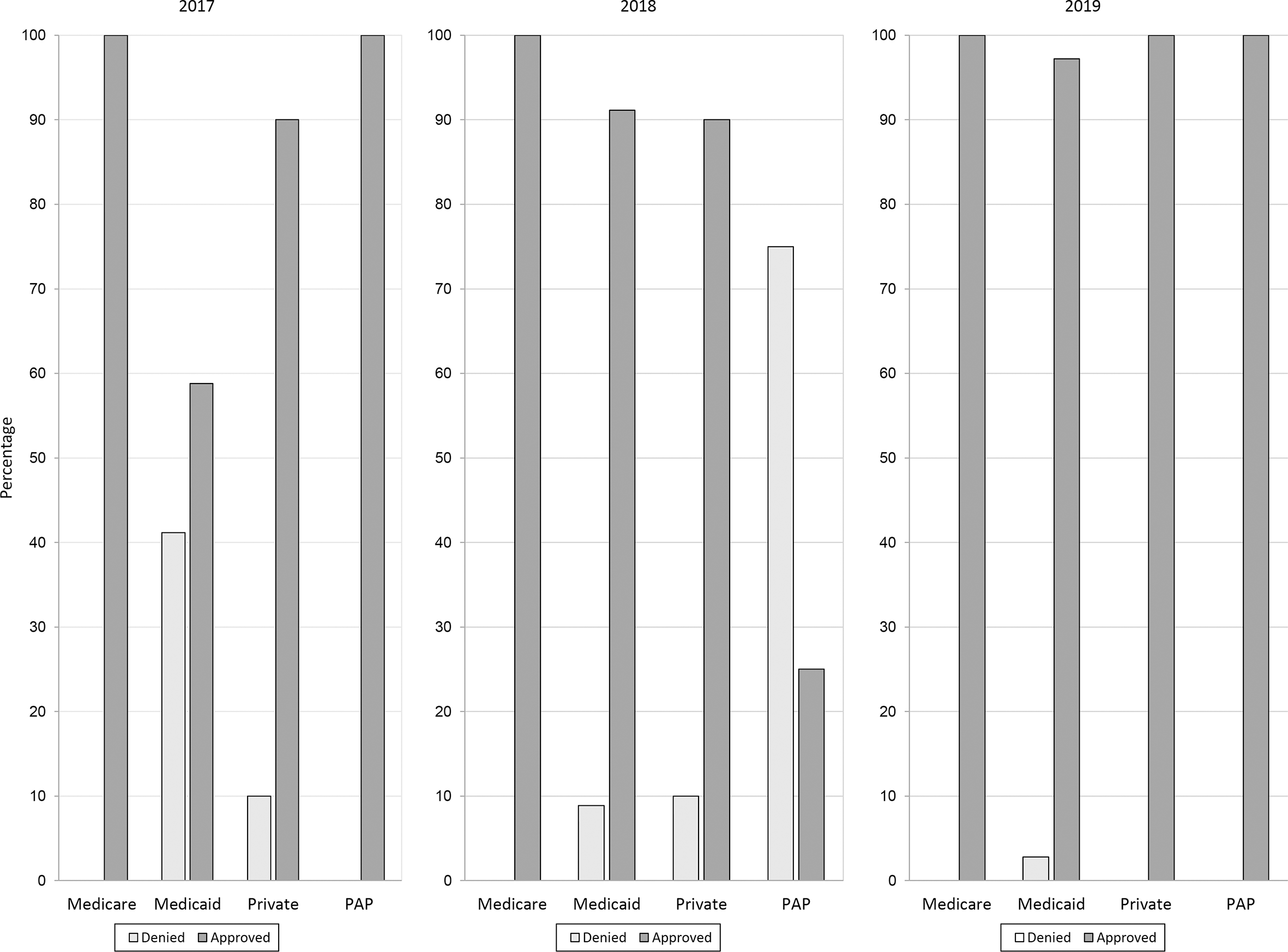
Hepatitis C virus treatment application denials, by health insurance type and year, in an urban safety-net health system in Chicago, Illinois, 2017-2019. Abbreviation: PAP, patient assistance program.
The percentage of applications approved on the first submission increased from 53% in 2017 to 82% in 2019 (P = .001). In addition, the number of treatment denials after an appeal decreased from 12% in 2017 to 0% in 2019 (P = .01). First submission approvals for Medicaid health insurance plans also increased significantly, from 12% in 2017 to 74% in 2019 (P < .001) (Figure 3).
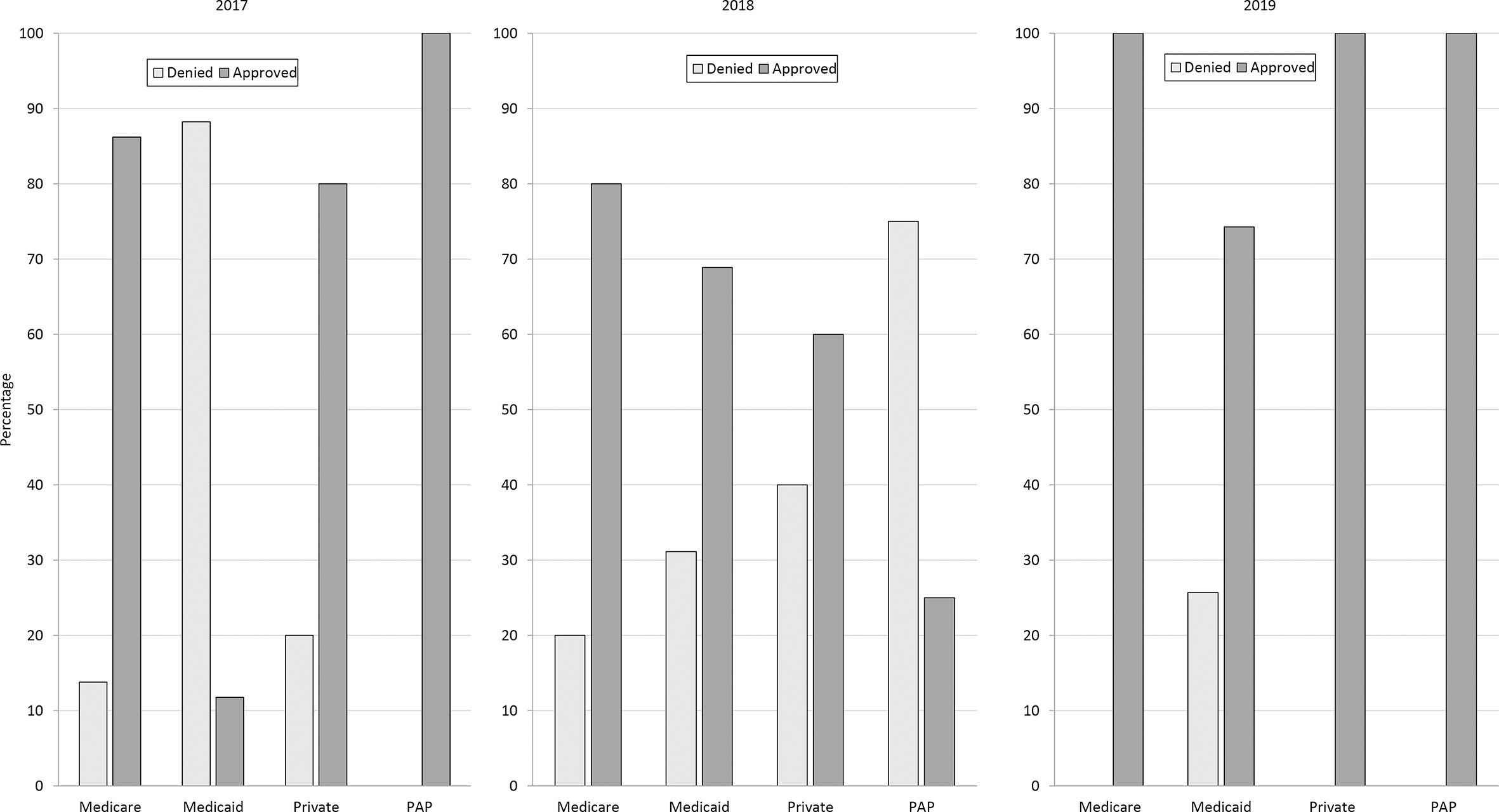
Initial hepatitis C virus treatment application outcomes, by health insurance type and year, in an urban safety-net health system in Chicago, Illinois, 2017-2019. Abbreviation: PAP, patient assistance program.
Time From Treatment Approval to Treatment Initiation
Of the 176 total applications approved during the study period, the median number of days from submission to approval was 2 (range, 0-22) for treatments approved on the first submission compared with 20 (range 1-77), 38 (range, 13-251), and 42 (range, 40-51) for approved applications requiring 1, 2, or 3 appeals, respectively (P = .001).
The median number of days from treatment approval to delivery of HCV medication to the MSHIDC by the specialty pharmacy was 4 days during each study year. The median number of days from receipt of HCV medication at the clinic to treatment initiation was 15, 20, and 29 days in 2017, 2018, and 2019, respectively (P = .002).
Treatment Regimens and Duration
Ledipasvir/sofosbuvir and elbasvir/grazoprevir were the most commonly prescribed treatment regimens in 2017 (40% and 26%, respectively), whereas glecaprevir/pibrentasvir and sofosbuvir/velpatasvir were the most commonly prescribed treatment regimens in 2019 (58% and 23%, respectively). Twelve-week treatment durations were most common in 2017 and 2018 (67% and 47%, respectively), whereas 8-week treatment durations were commonly seen in 2019 (57%). Commonly seen treatment durations correlated with the most commonly prescribed treatment regimens during that study year.
Attendance at Follow-up Appointment
Attendance rates at the SVR follow-up visit (≥12 weeks after treatment completion) were significantly lower in 2019 (67%) than in 2017 and 2018 (88% and 86%, respectively) (P = .02).
Patient Retention Throughout Treatment Cascade and SVR Outcomes
Of the 203 HCV treatment applications, 87% (n = 176) were approved for treatment, 91% (n = 161) of whom completed treatment. Of the 161 patients who completed treatment, 81% (n = 131) attended their SVR follow-up visit, 98% (n = 129) of whom reached SVR (Figure 4). The intent-to-treat SVR rate was 77% (129 of 167). Nine percent (n = 15) were approved but did not complete treatment, and 19% (n = 30) did not attend their SVR follow-up visit. Only 1 of the 187 patients did not reach SVR and did not undergo retreatment. This patient had HCV genotype 1, had Metavir/fibrosis score F0-F1, and received 12 weeks of ledipasvir/sofosbuvir.
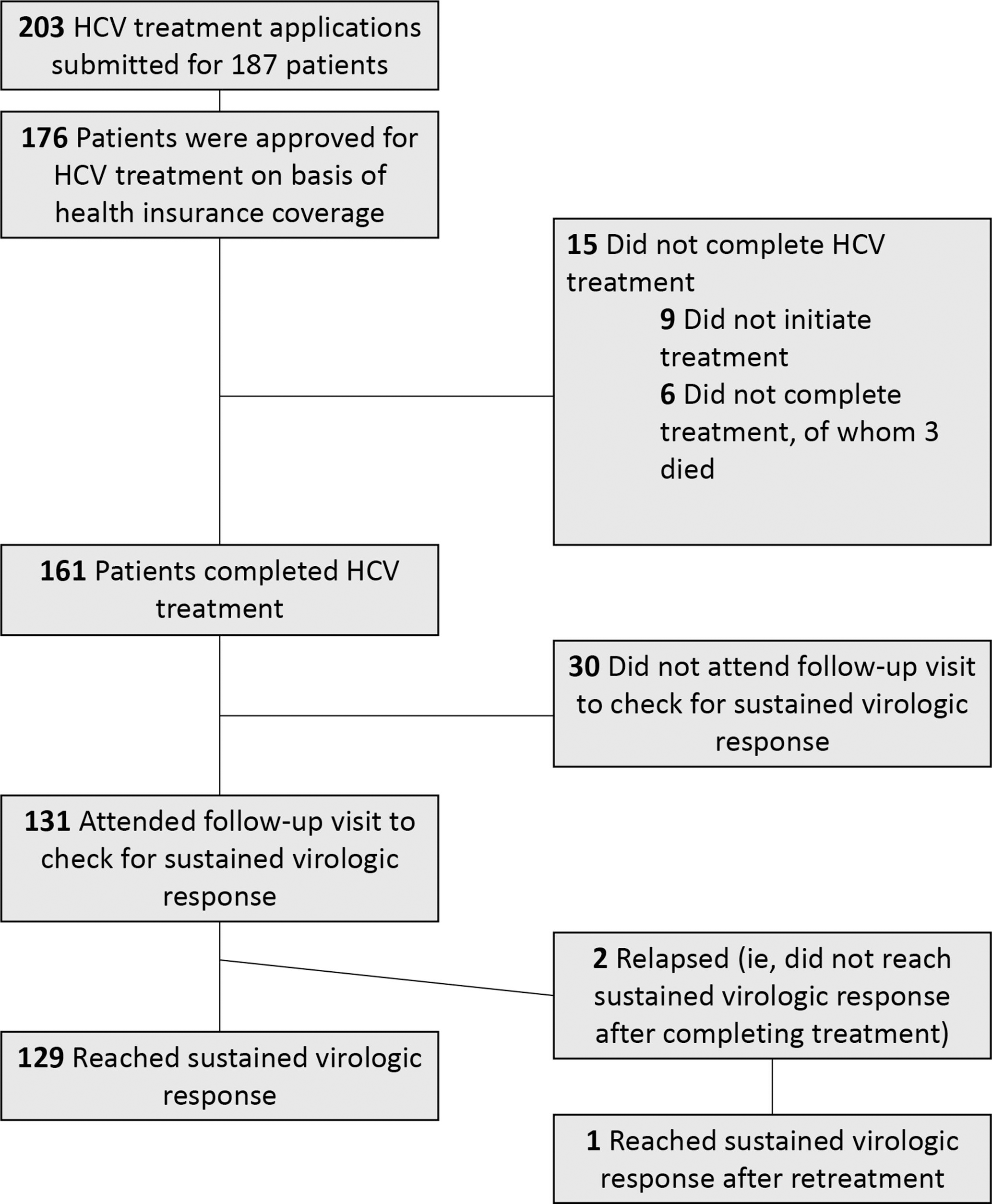
HCV treatment cascade among patients (n = 187) at an urban safety-net health system in Chicago, Illinois, 2017-2019. Abbreviation: HCV, hepatitis C virus.
During 2017-2019, the largest patient drop-off in the HCV treatment cascade was the 19% decline from receipt of treatment (n = 161) to attending the SVR follow-up appointment (n = 131).
Discussion
In the first 2 full years of treating HCV infection with DAA agents, our program created a strong infrastructure that was successful in obtaining health insurance approvals for costly HCV medications and achieving high SVR rates in a patient population who is medically underserved and has low socioeconomic status. Of the 131 patients who completed HCV treatment and attended their SVR follow-up visit, 98% achieved SVR. The pharmacist played a critical role in moving patients along the treatment cascade by managing and submitting treatment applications, appealing treatment denials with health insurance companies, coordinating medication delivery and pick-up, and providing counseling on medication adherence.
Recent developments in HCV testing and treatment guidelines underscore the need for a pharmacist-led program. First, the March 2020 recommendation by the US Preventive Services Task Force expands HCV testing to all adults, 26 which will result in more people getting tested and likely more diagnoses. In addition, in November 2019, the American Association for the Study of Liver Diseases, in collaboration with other Clinton Health Access Initiative hepatology societies, released a call to action 27 to simplify HCV testing and treatment, including guidance to make treatment available at point of care. Thus, it is likely that both new and existing health care providers will be seeking guidance to build and/or improve treatment navigation programs. The pharmacist-led model has demonstrated success, and we attempted to clearly delineate the responsibilities so that this protocol may be replicated.
The main barriers to treatment identified in this study were health insurance approvals and attendance at the SVR follow-up visit. From 2017 to 2019, the health insurance approval rate for Medicaid treatment applications increased from 59% to 97%. This shift was in large part due to a policy change at the Illinois state level in November 2018. After the advent of DAA agents, Illinois Medicaid payors did not cover HCV treatment for patients with a Metavir/fibrosis score <F3. In response to threats of litigation, the Illinois Department of Healthcare and Family Services removed restrictive criteria on DAA agents, thus expanding coverage to all patients living with HCV infection regardless of Metavir/fibrosis score. 28 Although health insurance approval has thus become less of a barrier in Illinois, this expanded coverage has not reached all states 29 and may continue to pose a barrier to treatment for patients who have not reached an advanced fibrosis stage.
The second barrier is the attendance rate at the SVR follow-up visit, which pertains more to measuring the success of treatment than to actual treatment itself. In our study, 19% of patients who completed HCV treatment did not attend their follow-up visit to check for SVR. Thus, it is possible that more patients achieved SVR, but these data are unknown. One possible reason for the lack of follow-up could be that patients perceive being “cured” after completing their medication and having a preliminary undetectable HCV RNA at their week 4 laboratory visit. Future directions to improve adherence with follow-up visits are to emphasize early and consistently throughout the treatment process that this final SVR follow-up visit is important for determining whether the patient has truly been cured.
Another reason for the lack of follow-up at the SVR visit is that our patient population faces unique challenges related to the social determinants of health. Our patients reside on the west and southwest sides of Chicago in communities that are medically underserved and that face unemployment, homelessness, substance use, chronic violence, segregation, and disinvestment at the city level. 30,31 These structural issues mean that seemingly simple activities such as attending a medical appointment are challenged by lack of transportation, health insurance, citizenship/documentation, and other resources necessary to facilitate attendance. These barriers affect the entire care continuum from attendance at first medical appointment through the SVR follow-up visit.
Staff members on this project were consistently engaged with the study throughout the study period. Therefore, changes in health insurance approvals and attendance at SVR follow-up visits would not have been due to any changes in staffing. Although staff members may have become more proficient at completing health insurance applications, potentially contributing to the increase in approval rates, the protocol for completing applications did not change, and this increase was likely due to the policy change at the state level. The observed decrease in SVR follow-up attendance is less well understood. Staff messaging to patients about the importance of attending this visit may have changed over time; to that end, program staff members may have emphasized this messaging over time. Multiple factors contributed to delays in the time between receiving the medication from the specialty pharmacy and initiating treatment, ranging from patient preference to start later after a holiday, clinic inability to contact the patient to schedule an appointment, and appointment availability with an infectious disease care provider. It is unclear why these 3 factors would have changed.
Limitations
This study had several limitations. First, the study setting is unique in that it is an urban safety-net hospital with a routine screening program; therefore, our treatment population was not based on birth cohort or risk factors. In addition, the study setting may make results less generalizable to dissimilar settings. The change in Illinois policy that removed restrictions on access to DAA agents greatly affected our treatment approval rates; however, in other states where restrictions have not been removed, health insurance restrictions will continue to pose a barrier to treatment.
Conclusion
The pharmacist-led model for HCV treatment was effective in navigating patients through the treatment cascade and achieving SVR. Our data show that all treatments were effective in eradicating HCV infection, and many of the barriers to care were due to the restrictions imposed by health insurance companies. Now that HCV treatment access in Illinois has expanded, we can engage and link more patients into care for treatment sooner. More broadly, widespread implementation of pharmacist-led HCV treatment models may help to hasten progress toward 2030 HCV elimination goals. 32
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: FOCUS funding supports HIV, hepatitis C virus, and hepatitis B virus screening and linkage to the first medical appointment after diagnosis; FOCUS funding does not support any activities beyond the first medical appointment and does not specify how FOCUS partners should handle subsequent patient care and treatment.
